Abstract
Purpose
The study was conducted to improve the productivity of the multi-component cellulolytic enzymes using thermophilic Aspergilus terreus strain and sweet sorghum bagasse as substrate. One of the major objectives was to study the interactions between different operating parameters and appraise the potential of the optimized process for validation studies.
Methods
Response surface methodology (RSM) based on central composite design (CCD) was used to optimize the process parameters for cellulase production by thermophilic Aspergillus terreus via a solid-state fermentation (SSF) process. A set of 50 experiments in triplicate with five factors (moisture content, inoculum level, pH, temperature and incubation period), three levels with six axial points (α ± 1.68) and five replications at the central point were conducted in this study with filter paper (FP) cellulase and β-glucosidase as output parameters.
Results
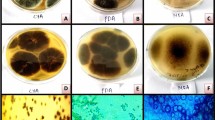
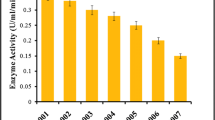
Micrographs and scanning electron microscopy (SEM) of A. terreus RWY revealed a chain of conidia in a columnar arrangement with an average size of conidium being 2.12 µ. Statistical process optimization suggested temperature of 45 °C, pH of 5.8, incubation time of 72 h, inoculum concentration of 10% and initial moisture content of 80% (w/w) as optimum for conducting validation studies. Validation studies showed comparable FP and β-glucosidase activities as predicted by the model equations. In addition to FP and β-glucosidase, A. terreus RWY also produced endoglucanase (EG), β-xylosidase, α-l-arabinofuranosidase, CBHI, xylanase and xylan esterase of 149.54, 26.94, 183.16, 17.52, 1264.47 and 1106.46 U/gds, respectively during the validation process. Response surface optimization also led to a nearly two-fold increase in FP and β-glucosidase activities.
Conclusion
Coupled with the use of thermophilic strains which confer specific benefits during industrial applications, statistical process optimization holds potential for scale-up studies for cellulase production using the optimized parameters, SSB as substrate and thermophilic A. terreus RWY.






Similar content being viewed by others
References
Kafy, A., Kim, H.C., Zhai, L., Kim, J.W., Hai, L.V., Kang, T.J., Kim, J.: Cellulose long fibres fabricated from cellulose nanofibers and its strong and tough characteristics. Sci. Rep. 7(1), 17683 (2017)
Juturu, V., Wu, J.C.: Microbial cellulases: engineering, production and applications. Renew. Sust. Energy Rev. 33, 188–203 (2014)
Walia, A., Guleria, S., Mehta, P., Chauhan, A., Prakash, J.: Microbial xylanases and their industrial application in pulp and paper biobleaching: a review. 3 Biotech 7(1), 11 (2017). https://doi.org/10.1007/s13205-016-0584-6
Gupta, R., Mehta, G., Deswal, D., Sharma, S., Jain, K.K., Kuhad, R.C., Singh, A.: Cellulases and their biotechnological applications. Chapter in Biotechnology for Environmental Management and Resource Recovery, pp. 89–106. Springer, New Delhi (2013)
Menendez, E., Garcia-Fraile, P., Rivas, R.: Review biotechnological applications of bacterial cellulases. AIMS Bioeng. 2(3), 163–182 (2015). https://doi.org/10.3934/bioeng.2015.3.163
Klein-Marcuschamer, D., Oleskowicz-Popiel, P., Simmons, B.A., Blanch, H.W.: The challenge of enzyme cost in the production of lignocellulosic biofuels. Biotechnol. Bioeng. (2012). https://doi.org/10.1002/bit.24370
Oberoi, H.S., Rawat, R., Chadha, B.S.: Response surface optimization for enhanced production of cellulases with improved functional characteristics by newly isolated Aspergillus niger HN-2. Antonie van Leeuwen. 105, 119–134 (2014). https://doi.org/10.1007/s10482-013-0060-9
Baig, M.M.V., Baig, M.L.B., Baig, M.I.A., Yasmeen, M.: Saccharification of banana agro-waste by cellulolytic enzymes. Afr. J. Biotechnol. 3, 447–450 (2004)
Milala, M.A., Shugaba, A., Gidado, A., Ene, A.C., Wafer, J.A.: Studies on the use of agricultural wastes for cellulase enzyme production by Aspergillus niger. Res. J. Agric. Biol. Sci. 1, 325–328 (2005)
Oberoi, H.S., Chavan, Y., Bansal, S., Dhillon, G.S.: Production of cellulases through solid state fermentation using kinnow pulp as a major substrate. Food Bioproc. Technol. 3, 528–536 (2010)
Ratnavathi, C.V., Chakravarthy, K., Komala, V.V., Chavan, U.D., Patil, J.V.: Sweet sorghum as feedstock for biofuel production: a review. Sugar Tech. 13, 399–407 (2011)
Sajith, S., Sreedevi, S., Priji, P., Unni, K.N., Benjamin, S.: Production and partial purification of cellulase from a novel fungus, Aspergillus flavus BS1. Ann. Microbiol. 64, 763–771 (2014)
Shen, X.L., Xia, L.M.: Production and immobilization of cellobiase from Aspergillusniger ZU-07. Proc. Biochem. 39, 1363–1367 (2004)
Rawat, R., Srivastava, N., Chadha, B.S., Oberoi, H.S.: Generating fermentable sugars from rice straw using functionally active cellulolytic enzymes from Aspergillus niger HO. Energy Fuels 28, 5067–5075 (2014). https://doi.org/10.1021/ef500891g
Viikari, L., Alapuranen, M., Puranen, T., Vehmaanper, J., Siikaaho, M.: Thermostable enzymes in lignocellulose hydrolysis. Adv. Biochem. Eng. Biotechnol. 108, 121–145 (2007)
Szijarto, N., Siika-Aho, M., Tenkanen, M., Alapuranen, M., Vehmaanpera, J., Reczey, K., Viikari, L.: Hydrolysis of amorphous and crystalline cellulose by heterologously produced cellulases of Melanocarpusalbomyces. J. Biotechnol. 136, 140–147 (2008)
Du, R., Wang, Yu., Zhao, F., Qiao, X., Song, Q., Li, S., Kim, R.-C., Ye, H., Xiao, H., Zhou, Z.: Production, optimization and partial characterization of bacterial cellulose from Gluconacetobacterxylinus TJU-D2. Waste Biomass Valor. (2018). https://doi.org/10.1007/s12649-018-0440-5
Jaramillo, L., Santos, D., Borges, E., Dias, D., Periera, N.: Low-cost effective culture medium optimization for d-lactic acid production by Lactobacillus coryniformis subsp. torquens under oxygen-deprived condition. Ann. Microbiol. 68(9), 547–555 (2018). https://doi.org/10.1007/s13213-018-1362-y
Mubarak, M., Shailja, A., Suchithra, T.V.: Cost effective approach for production of Chlorella pyrenoidosa: a RSM based study. Waste Biomass Valor. (2018). https://doi.org/10.1007/s12649-018-0330-x
Pereira, J.C., Marques, N.P., Rodrigues, A., de Oliveira, T.B., Boscolo, M., da Silva, R., Gomes, E., Bocchini, D.A.: Thermophilic fungi as new sources for production of cellulases and xylanases with potential use in sugarcane bagasse saccharification. J. Appl. Microbiol. 118, 928–939 (2015)
Maheshwari, R., Bharadwaj, G., Bhat, M.K.: Thermophilic fungi: their physiology and enzymes. Microbiol. Mol. Biol. Rev. 64, 461–488 (2000)
Rawat, R., Kumar, S., Chadha, B.S., Kumar, D., Oberoi, H.S.: An acidothermophilic functionally active novel GH12 family endoglucanase from Aspergillus niger HO: purification, characterization and molecular interaction studies. Antonie Van Leeuwenhoek 107, 103–117 (2015). https://doi.org/10.1007/s10482-014-0308-z
Bizukojc, M., Ledakowicz, S.: The morphological and physiological evolution of Aspergillus terreus mycelium in the submerged culture and its relation to the formation of secondary metabolites. World J. Microbiol. Biotechnol. 26, 41–54 (2010). https://doi.org/10.1007/s11274-009-0140-1
Kumar, A.K., Parikh, B.S.: Cellulose-degrading enzymes from Aspergillus terreus D34 and enzymatic saccharification of mild-alkali and dilute-acid pretreated lignocellulosic biomass residues. Bioresour. Bioproc. 2, 7 (2015). https://doi.org/10.1186/s40643-015-0038-8
Sharma, R., Kocher, G.S., Bhogal, R.S., Oberoi, H.S.: Cellulolytic and xylanolytic enzymes from thermophilic Aspergillus terreus RWY. J. Basic Microbiol. 54, 1–11 (2014)
Kasana, R.C., Salwan, R., Dhar, H., Dutt, S.: Arapid and easy method for the detection of microbialcellulases on agar plates using Gram’s Iodine. Curr. Microbiol. 57, 503–507 (2008)
Alves, M.S., Pozza, E.A.: Scanning electron microscopy detection of seed-borne fungi in blotter test. Curr. Adv. Sci. Technol. 24, 231–238 (2012)
Moretti, M.M.S., Bocchini-Martins, D.A., da-Silva, R., Rodrigues, A., Sette, L.D., Gomes, E.: Selection of thermophilic and thermotolerant fungi for the production of cellulases and xylanases under solid state fermentation. Braz. J. Microbiol. 24, 1062–1071 (2012)
Rai, P., Tiwari, S., Gaur, R.: Optimization of process parameters for cellulase production by novel thermotolerant yeast. Bioresour. 7, 5401–5414 (2012)
Brijwani, K., Oberoi, H.S., Vadlani, P.V.: Production of a cellulolytic enzyme system in mixed-culture solid-state fermentation of soybean hulls supplemented with wheat bran. Proc. Biochem. 45, 120–128 (2010)
Dhillon, G.S., Oberoi, H.S., Kaur, S., Bansal, S., Brar, S.K.: Value addition of agricultural wastes for augmented cellulase and xylanase production through solid-state tray fermentation employing mixed-culture of fungi. Ind. Crops Prod. 34, 1160–1167 (2011)
Ghose, T.K.: Measurement of cellulase activities. Pure Appl. Chem. 59, 257–268 (1987)
Ghose, T.K., Bisaria, V.S.: Measurement of hemicellulase activities. Part 1: xylanases. Pure Appl. Chem. 59, 1739–1752 (1987)
Bailey, M.J., Tahtiharju, J.: Efficient cellulose production by Trichodermareesei in continuous cultivation on lactose medium with a computer-controlled feeding strategy. Appl. Microbiol. Biotechnol. 62, 156–162 (2003)
Poutanen, K., Ratto, M., Puls, J., Viikari, L.: Evaluation of different microbial xylanolytic systems. J. Biotechnol. 6, 49–60 (1987)
Bradford, M.M.: A rapid and sensitive method for thequantification of microgram quantities of protein utilizingthe principle of protein dye binding. Anal. Biochem. 72, 248–254 (1976)
Gao, J., Weng, H., Zhu, D., Yuan, M., Guan, F., Yu, X.: Production and characterization of cellulolytic enzymes from the thermoacidophilic fungal Aspergillus terreus M11 under solid-state cultivation of corn stover. Bioresour. Technol. 99, 7623–7629 (2008)
Khot, M., Kamat, S., Zinjarde, S., Pant, A., Chopade, B., Kumar, R.A.: Single cell oil of oleaginous fungi from the tropical mangrove wetlands as a potential feedstock for biodiesel. Microb. Cell Fact. 11, 71 (2012)
Aiba, S., Humphrey, A.E., Millis, N.F.: Kinetics. Biochemical engineering, 2nd edn, pp. 92–127. Academic Press, New York (1973)
Issac, G.S., Abu-Tahon, M.A.: Enhanced alkaline cellulase production by the thermohalophilic Aspergillus terreus AUMC 10138 mutated by physical and chemical mutagens using corn stover as substrate. Braz. J. Microbiol. 46, 1269–1277 (2015)
Vu, V.H., Pham, T.A., Kim, K.: Improvement of fungal cellulases production by mutation and optimization of solid state fermentation. Mycobiol. 39, 20–25 (2011)
Mekala, N.K., Singhania, R.R., Sukumaran, R.K., Pandey, A.: Cellulase production under solid state fermentation by Trichodermareesei RUT-30: statistical optimization of process parameters. Appl. Biochem. Biotechnol. 151, 122–131 (2008)
Ilyas, U., Ahmed, S., Majeed, A., Nadeem, M.: Biohydrolysis of Saccharumspontaneum for cellulase production by Aspergillus terreus. Afr. J. Biotechnol. 11(21), 4914–4920 (2012). https://doi.org/10.5897/AJB11.1194
Ali, S., Sayed, A., Saker, R.I., Alam, R.: Factors affecting cellulose production by Aspergillus terreus using water hyacinth. J. Microbiol. Biotechnol. 7, 62–66 (1991)
Pushalkar, S., Rao, K.K., Menon, K.: Production of beta-glucosidase by Aspergillusterrus. Curr. Microbiol. 30, 255–258 (1995)
da Silva, V.C.T., de Souza, A.L.C., Souza, R.C., Bertoldi, M., Neves, S., Gomes, E., Bonilla-Rodriguez, G.O.: Effect of pH, temperature, and chemicals on the endoglucanases and β-glucosidases from the thermophilic fungusMyceliophthoraheterothallica F. 2. 1. 4. obtained by solid-state and submerged cultivation. Biochem. Res. Int. (2016). https://doi.org/10.1155/2016/9781216
Acharya, P.B., Acharya, D.K., Modi, H.A.: Optimization for cellulase production by Aspergillus niger using saw dust as substrate. Afr. J. Biotechnol. 7, 4147–4152 (2008)
Narra, M., Dixit, G., Divecha, J., Madamwar, D., Shah, A.M.: Production of cellulases by solid state fermentation with Aspergillus terreus and enzymatic hydrolysis of mild alkali-treated rice straw. Bioresour. Technol. 121, 355–361 (2012)
Hemansi, G.R., Kuhad, R.C., Saini, J.K.: Cost effective production of complete cellulase system by newly isolated Aspergillus niger RCKH-3 for efficient enzymatic saccharification: medium engineering by overall evaluation criteria approach (OEC). Biochem. Eng. J. 132, 182–190 (2018)
Saida, L., Oberoi, H.S., Narasu, M.L.: Studies on cellulase production by solid state fermentation using sweet sorghum bagasse. Helix 1, 261–266 (2013)
Gao, D., Chundawat, S., Liu, T., Hermanson, S., Gowda, K., Brumm, P., Dale, B., Balan, V.: Strategy for identification of novel fungal and bacterial glycosyl hydrolase hybrid mixtures that can efficiently saccharify pretreated lignocellulosic biomass. Bioenergy Res. 3, 67–81 (2010)
Jahromi, M.F., Liang, J.B., Rosfarizan, M., Goh, Y.M., Shokryazdan, P., Ho, Y.W.: Efficiency of rice straw lignocelluloses degradability by Aspergillus terreus ATCC 74135 in solid state fermentation. Afr. J. Biotechnol. 10(21), 4428–4435 (2011)
Ilyas, U., Gohar, F., Saeed, S., Bukhari, Z., Ilyas, H.: Screening of locally isolated Aspergillus species for their cellulolytic potential and their optimization on Vigna mungo in solid state fermentation. British Biotechnol. J. 3(3), 350–358 (2013)
El-Naggar, N.E., Haroun, S.A., Owis, E.A., Sherief, A.A.: Optimization of β-glucosidase production by Aspergillus terreus Strain EMOO 6-4 using response surface methodology under solid-state fermentation. J. Prep. Biochem. Biotechnol. 45(6), 568–587 (2015)
Saritha, M., Tiwari, R., Singh, S., Nain, P.K.S., Rana, S., Adak, A., Arora, A., Nain, L.: Glycoside hydrolase production by Aspergillus terreus CM20 using mixed design approach for enhanced enzymatic saccharification of alkali pretreated paddy straw. Ind. J. Exp. Biol. 54, 518–524 (2016)
Kulkarni, N., Vaidya, T., Rathi, G.: Optimization of cellulase production by Aspergillus species under solid state fermentation. Pharma Inn. J. 7(1), 193–196 (2018)
Funding
Authors thankfully acknowledge the financial support received under the project (BT/PR8488/PBD/26/68/2006) funded by the Department of Biotechnology (DBT), Government of India for conducting this study. Authors Sharma and Kocher also thankfully acknowledge the support received from the Punjab Agricultural University, Ludhiana, India.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Informed Consent
Informed consent was obtained from all individual participants included in the study.
Research Involving Human Participants and/or Animals
This study did not involve the use of human participants and/or animals.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sharma, R., Kocher, G.S., Rao, S.S. et al. Improved Production of Multi-component Cellulolytic Enzymes Using Sweet Sorghum Bagasse and Thermophilic Aspergillus terreus RWY Through Statistical Process Optimization. Waste Biomass Valor 11, 3355–3369 (2020). https://doi.org/10.1007/s12649-019-00670-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12649-019-00670-5