Abstract
The oxidation of SiO and FO by LH, ER, NER and TER mechanisms on metal doped nanotubes is investigated in order to find the catalytic abilities of metal doped nanotubes. The SiO and FO molecules are joined on metal doped nanotube-O2* and metal doped nanotube-O* and calculated barrier energies are lower than metal based catalysts. Results shown that Fe and V doped BNNT have higher potential than metal doped CNT to adsorb the O2, SiO2 and FO2. The metal doped nanotube-OSiOO* and metal doped nanotube-OFOO* intermediates in ER are permanent than corresponding intermediate. Result shown that in NER mechanism the OOSiSiOO* and OOFFOO* formation is rate limiting step and the creation of surface-OSiOOSiO* and surface-OFOOFO* is the rate-limiting steps of TER mechanisms. The metal doped nanotubes (V-BNNT, V-CNT, Fe-BNNT and Fe-CNT) can catalyze the processes of SiO and FO oxidation by LH, ER, NER and TER mechanisms.
Similar content being viewed by others

Data Availability
Not applicable.
Code Availability
Not applicable.
References
Zhang Q, Tang C (2018) J Phys Chem C 122:22838–22848
Zhang Q, Tang C, Ling Fu (2019) Appl Surf Sci 497:143723
Wang Y, Zhang K, Yang Z (2019) Chem Phys Lett 734:136733
Guo G-C, Wang R-Z, Zhang M (2019) Appl Sur Sci 475:102–108
Petnikota S (2019) ChemElectroChem 6:493–503
Ravaei I, Haghighat M, Azami SM (2019) Appl Sur Sci 469:103–112
Oku T, Kuno M (2001) Kitahara. Int J Inorg Mater 3:597–612
Oku T, Nishiwaki A, Narita I (2004) Sci Technol Adv Mater 5:635–638
Esrafili MD, Nurazar R (2014) Surf Sci 626:44–48
Munsif S, Ayub K (2018) J Mol Liq 259:249–259
Tsierkezos NG, Ritter U (2015) Chem Phys Lett 639:217–224
Tsierkezos NG, Szroeder P (2015) J Solid State Electrochem 19:891–905
Altalhi T, Mezni A (2016) Chem Phys Lett 658:92–96
Chen M, Shao L-L (2016) Chem Eng J 304:303–312
Srivastava D, Susi T (2014) RSC Adv 4:15225–15235
George V, Papamokos J (2004) Phys Chem A 108:7291–7300
Allam O, Cho BW (2018) RSC Adv 8:39414–39420
Valadbeigi Y (2016) Comput Theor Chem 1091:169–175
Stenlid JH (2017) J Phys Chem C 121:27483–27492
Tavakol H, Shahabi D (2015) J Phys Chem C 119:6502–6510
Smith DGA, Patkowski K (2015) J Phys Chem C 119:4934–4948
Smith DGA, Patkowski K (2014) J Phys Chem C 118:544–550
Zhao J-Y (2013) J Phys Chem A 117:12519–12528
Cunha R, Elías AL (2018) Carbon 127:312–319
Kravchyk KV (2020) ACS Energy Lett 5:545–549
Kumar S, Bhauriyal P (2019) J Phys Chem C 123:23863–23871
Zhu Y, Peng W (2019) J Mater Chem A 7:23577–23603
Li Z, Gao C, Yang L (2019) J Alloys Compo 798:500–506
Bhauriyal P, Pathak B (2019) Chem An Asian J 520
Li W, Kim U-H (2017) ACS Nano 11:5853–5863
Zhang Y, Pan Z, Yang J, Chen J, Chen K, Yan K, He M (2022) Powder Technol 399:1. https://doi.org/10.1016/j.powtec.2022.117193
Xiong Q, Chen Z, Huang J, Zhang M, Song H, Hou X, Feng Z (2020) Rare Met 39:589–596. https://doi.org/10.1007/s12598-020-01385-6
Lu T, Yan W, Feng G, Luo X, Hu Y, Guo J, Ding S (2022). Green Chem. https://doi.org/10.1039/D2GC00869F
Li Z, Teng M, Yang R, Lin F, Fu Y, Lin W, Liao Y (2022) Sens Actua B Chem 361:131691. https://doi.org/10.1016/j.snb.2022.131691
Yu F, Zhu Z, Li C, Li W, Liang R, Yu S, Zhang Z (2022) Appl Cataly B Environ 314:121467. https://doi.org/10.1016/j.apcatb.2022.121467
Hu X, Zhang P, Wang D, Jiang J, Chen X, Liu Y, Li P (2021) Biosens Bioelectron 182:113188
Zhuo Z, Wan Y, Guan D, Ni S, Wang L, Zhang Z, Zhang BT (2020) Adv Sci 7:1903451. https://doi.org/10.1002/advs.201903451
Feng Z, Li G, Wang X, Gómez-García CJ, Xin J, Ma H, Gao K (2022) Chem Eng J 445:136797. https://doi.org/10.1016/j.cej.2022.136797
Liu C, Ying P (2022) Chin Phys B 31:26201
Li H, Zhu H, Zhao R (2022). New J Chem. https://doi.org/10.1039/D2NJ01702D
Huang Z, Cao S, Yu J, Tang X, Guo Y, Guo Y, Zhan W (2022) Environ Sci Technol 56:9661–9671
Yang Y, Wang SQ, Wen H, Ye T, Chen J, Li CP, Du M (2019) Angew Chem 58:15362–15366
Bai B, Rao D, Chang T, Guo ZJ (2019) Hydrology 578:124080
Wang Y, Wu X, Liu J, Zhai Z, Yang Z, Xia J, Zhang QJ (2022) Environ Chem Eng 10:107091
Liu P, Li S, Zhang L, Yin X, Ma Y (2022). Catal Sci Technol. https://doi.org/10.1039/d2cy00474g
Lakshman MJ (2022) Synthetic Chem 1:48–51
Alizadeh T, Jahani R (2015) Mater Chem Phys 168:180–186
Hashemi SA, Farhangdoost K, Ma W, Moghadam DG, Nejad RM, Berto F (2022) Theor Appl Fract Mech 122:103573
Liu R, Hu Y, Xu J, Cai A, Wu A, Chen L, Wang F (2021) Oncologie 23:303–320. https://doi.org/10.32604/oncologie.2021.018514
Zhao G, Hooman M, Yarigarravesh M, Algarni M, Opulencia MJC, Alsaikhan F, Sarjadi MS (2022) Arab J Chem 15:104115
Kadhim MM, Sead FF, Jalil AT, Taban TZ, Rheima AM, Almashhadani HA, Hamel S (2022) Monatsh Chem 153:589–596
Budi HS, Davidyants A, Rudiansyah M, Ansari MJ, Suksatan W, Sultan MQ, Kazemnejadi M (2022) Mater Today Commun 32:104108
Salahdin OD, Sayadi H, Solanki R, Parra RMR, Al-Thamir M, Jalil AT, Kianfar E (2022) Appl Phys A 128:1–23
Jasim SA, Hadi JM, Jalil AT, Opulencia MJC, Hammid AT, Tohidimoghadam M, Moghaddam-Manesh M (2022) Front Chem 10:1–10
Honarvari B, Karimifard S, Akhtari N, Mehrarya M, Moghaddam ZS, Ansari MJ, Chiani M (2022) Molecules 27:4634
Zhang Y, Li C, Ji H, Yang X, Yang M, Jia D, Zhang X, Li R, Wang J (2017) Int J Mach Tools Manuf 122:81–97
Yang M, Li C, Zhang Y, Jia D, Zhang X, Hou Y, Li R, Wang J (2017) Int J Mach Tools Manuf 122:55–65
Wang X, Li C, Zhang Y, Said Z, Debnath S, Sharma S, Yang M, Gao T (2022) Int J Adv Manuf Technol 119:631–646
Yang YY, Gong YD, Li CH, Wen XL, Sun JY (2021) J Mater Process Technol 291:117023
Gao T, Zhang Y, Li C, Wang Y, Chen Y, An Q, Zhang S, Li HN, Cao H, Muhammad Ali H, Zhou Z, Sharma S (2022) Front Mech Eng 17:24
Xu W, Li Ch, Zhang Y, Muhammad Ali H, Sharma S, Li R, Yang M, Gao T, Liu M, Wang X, Said Z, Liu X, Zou Z (2022). Int J Extrem Manuf. https://doi.org/10.1088/2631-7990/ac9652
Tang Y, Chen W (2021) Chem Phys Chem 22:606–618
Jiang Q, Zhang J (2020) J Mater Chem A 8:287–295
Xu G, Wang R (2017) Carbon 118:35–42
Velez RP, Bentrup U (2017) Top Catal 60:1641–1652
Acknowledgements
We thank our university for computational help.
Author information
Authors and Affiliations
Contributions
Ahmed B. Mahdi (Funding acquisition, Investigation, Methodology), Rathab Abbass (Project administration, Resources, Software), Gufran Abd (Supervision, Validation, Visualization), Ashraq Mohammed Kadim (Conceptualization, Data curation, Formal analysis), Munthir Mohammed Radhy AL Kubaisy (Validation, Formal analysis, Methodology), Samar Emad Izzat (Funding acquisition, Methodology, Data curation), Qiao (Conceptualization, Data curation, Formal analysis).
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Consent to Participate
I confirmed.
Consent for Publication
I confirmed.
Ethical Approval
All procedures performed in studies involving human participants are in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Conflicts of Interest/Competing Interests
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
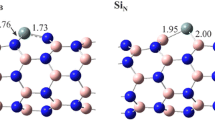
In this paper, the structures of complexes of FO, SiO, O2, FO2and SiO2 with Fe-CNT, V-CNT, Fe-BNT and V-BNNT are presented in Figures 1S and 2S. The oxidation pathways and structures of studied complexes for SiO and FO oxidation are presented in Figures 3S and 4S. The calculated energies of complexes for SiO and FO oxidation on nanotube surfaces are presented in Table 1S.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mahdi, A.B., Abbass, R., Abd, G. et al. Investigation of SiO and FO Oxidation Mechanisms by Carbon and Boron Nitride as Acceptable Nano-catalysts. Silicon 15, 3485–3495 (2023). https://doi.org/10.1007/s12633-022-02271-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12633-022-02271-9