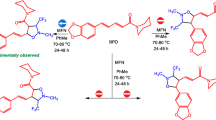
Abstract
Following our interest in exotic silylenes, here we compare and contrast 20 novel five-membered cyclic silylenes, including saturated (sila)0–4 cyclopentasilylenes (1–10) and unsaturated (sila)0-4cyclopentasilylenes-3-ene (1′-10′), reached at B3LYP/6–311++G** level of theory. Arduengo-type silasilylenes 1′-10′ turn out more stable than their corresponding 1–10, for showing higher singlet-triplet energy gap (ΔEs-t), with positive Gibbs free energy of hydrogenation (ΔGover). The band gaps (ΔEH-L) decrease by increasing the number of silicon atoms. Except for non-planar 10′, 6′, and 9′, every 1–10 turns out more nucleophilic than its corresponding 1′-10′. The highest nucleophilicity (N), proton affinity (PA), chemical potential (μ), dihedral angle (D̂), reactivity, and the lowest NBO charge on the divalent Si atom is exhibited by Si-saturated 10′.

Increasing silicons in 1–10 (and 1′-10′) decreases singlet-triplet energy gap (ΔEs-t). Every unsaturated 1′-10′ turns out more stable than its corresponding saturated 1–10 analog. As the most non-planar silylene, 10′ exhibits the highest nucleophilicity (N), proton affinity (PA), chemical potential (μ), dihedral angle \( \left(\hat{\mathrm{D}}\right), \) reactivity, and the lowest NBO charge on divalent Si atom.
Similar content being viewed by others
References
Becerra R, Walsh R (2010) Kinetic studies of reactions of organosilylenes: what have they taught us? Dalt Trans 39:9217–9228
Mizuhata Y, Sasamori T, Tokitoh N (2009) Stable heavier carbene analogues. Chem Rev 109:3479–3511
Tokitoh N, Okazaki R (2000) Recent topics in the chemistry of heavier congeners of carbenes. Coord Chem Rev 210:251–277
Cote DR, Van Nguyen S, Stamper AK, Armbrust DS, Tobben D, Conti RA, Lee GY (1999) Plasma-assisted chemical vapor deposition of dielectric thin films for ULSI semiconductor circuits. IBM J Res Dev 43:5–38
Heaven MW, Metha GF, Buntine MA (2001) Reaction pathways of singlet silylene and singlet germylene with water, methanol, ethanol, dimethyl ether, and trifluoromethanol: an ab initio molecular orbital study. J Phys Chem A 105:1185–1196
Tamao K, Kobayashi M, Matsuo T, Furukawa S, Tsuji H (2012) The first observation of electroluminescence from di (2-naphthyl) disilene, an SiSi double bond-containing π-conjugated compound. Chem Commun 48:1030–1032
Nefedov OM, Egorov MP, Ioffe AI, Menchikov LG, Zuev PS, Minkin VI, Simkin BY, Glukhovstev MN (1992) Critical compilation of physical properties of short-lived intermediates: Carbenes and carbene analogues (technical report). Pure Appl Chem 64:265–314
Okazaki M, Tobita H, Ogino H (2003) Reactivity of silylene complexes. Dalt Trans:493–506
Driess M, Yao S, Brym M, van Wüllen C, Lentz D (2006) A new type of N-heterocyclic silylene with ambivalent reactivity. J Am Chem Soc 128:9628–9629
Haaf M, Schmiedl A, Schmedake TA, Powell DR, Millevolte AJ, Denk M, West R (1998) Synthesis and reactivity of a stable silylene. J Am Chem Soc 120:12714–12719
Denk M, Lennon R, Hayashi R, West R, Belyakov AV, Verne HP, Haaland A, Wagner M, Metzler N (1994) Synthesis and structure of a stable silylene. J Am Chem Soc 116:2691–2692
Kassaee MZ, Musavi SM, Ghambarian M (2006) A quest for triplet silylenes XHSi3 at ab initio and DFT levels (X= H, F, Cl and Br). J Organomet Chem 691:1845–1856
Vessally E, Nikoorazm M, Esmaili F, Fereyduni E (2011) Substitution effects at α-position of divalent five-membered ring XC 4 H 3 M (M= C, Si and Ge). J Organomet Chem 696:932–939
Kassaee MZ, Najafi Z, Shakib FA, Momeni MR (2011) Stable silylenes with acyclic, cyclic, and unsaturated cyclic structures: effects of heteroatoms and cyclopropyl α-substituents at DFT. J Organomet Chem 696:2059–2064
Ayoubi-Chianeh M, Kassaee MZ, Ashenagar S, Cummings PT (2019). Nucleophilicity of cyclic conjugated silylenes using DFT method J Phys Org Chem:e3956
Ayoubi-Chianeh M, Kassaee MZ (2019) Novel silicon super bases at DFT level of theory: effects of fused benzene rings on the basicity of 2, 4, 6-cycloheptatrienesilylene. Res Chem Intermed 1–15
Kassaee MZ, Koohi M, Mohammadi R, Ghavami M (2013) 2, 2, 9, 9-Tetramethylcyclonona-3, 5, 7-trienylidene vs. its heterocyclic analogues: a quest for stable carbenes at DFT. J Phys Org Chem 26:908–916
Koohi M, Kassaee MZ, Haerizade BN, Ghavami M, Ashenagar S (2015) Substituent effects on cyclonona-3, 5, 7-trienylidenes: a quest for stable carbenes at density functional theory level. J Phys Org Chem 28:514–526
Kassaee MZ, Koohi M (2013) Breathing viability into cyclonona-3, 5, 7-trienylidenes via α-dimethyl and ά-moieties at DFT. J Phys Org Chem 26:540–550
Arduengo III AJ, Harlow RL, Kline M (1991) A stable crystalline carbene. J Am Chem Soc 113:361–363
West R, Denk M (1996) Stable silylenes: synthesis, structure, reactions. Pure Appl Chem 68:785–788
Schmedake TA, Haaf M, Apeloig Y, Müller T, Bukalov S, West R (1999) Reversible transformation between a diaminosilylene and a novel disilene. J Am Chem Soc 121:9479–9480
Choi S-B, Boudjouk P (2000) Synthesis and characterization of dibenzannulated silole dianions. The 1, 1-dilithiosilafluorene and 1, 1′-dilithiobis (silafluorene) dianions. Tetrahedron Lett 41:6685–6688
Dhiman A, Müller T, West R, Becker JY (2004) Electrochemistry and computations of stable silylenes and germylenes. Organometallics 23:5689–5693
Schwartz RL, Davico GE, Ramond TM, Lineberger WC (1999) Singlet− triplet Splittings in CX2 (X= F, Cl, Br, I) Dihalocarbenes via negative ion photoelectron spectroscopy. J Phys Chem A 103:8213–8221
Holthausen MC, Koch W, Apeloig Y (1999) Theory predicts triplet ground-state organic silylenes. J Am Chem Soc 121:2623–2624
Kassaee MZ, Zandi H (2012) P-Heterocyclic silylenes: a survey of stability with density functional theory. J Phys Org Chem 25:50–57
Luke BT, Pople JA, Krogh-Jespersen M-B, Apeloig Y, Karni M, Chandrasekhar J, Schleyer PVR (1986) A theoretical survey of unsaturated or multiply bonded and divalent silicon compounds. Comparison with carbon analogs. J Am Chem Soc 108:270–284
Kalcher J, Sax AF (1992) Singlet-triplet splittings and electron affinities of some substituted silylenes. J Mol Struct THEOCHEM 253:287–302
Krogh-Jespersen K (1985) Structural and energetic features of fully substituted silylenes, disilenes, and silylsilylenes (SiX2, X2SiSiX2, and XSiSiX3; X= lithium, methyl, and fluorine). J Am Chem Soc 107:537–543
Yoshida M, Tamaoki N (2002) DFT study on triplet ground state silylenes revisited: the quest for the triplet silylene must go on. Organometallics 21:2587–2589
Inoue S, Ichinohe M, Sekiguchi A (2008) Isolable alkali-metal-substituted Silyl radicals (t Bu2MeSi) 2SiM (M= Li, Na, K): electronically and Sterically accessible planar Silyl radicals. Organometallics 27:1358–1360
Sekiguchi A, Tanaka T, Ichinohe M, Akiyama K, Tero-Kubota S (2003) Bis (tri-tert-butylsilyl) silylene: triplet ground state silylene. J Am Chem Soc 125:4962–4963
West R, Fink MJ, Michl J (1981) Tetramesityldisilene, a stable compound containing a silicon-silicon double bond. Science 214(80):1343–1344
Ayoubi-Chianeh M, Kassaee MZ (2019) Toward triplet disilavinylidenes: A Hammett electronic survey for substituent effects on singlet-triplet energy gaps of silylenes by DFT. J Phys Org Chem e3988
Maier G, Reisenauer HP, Glatthaar J (2002) Reactions of silicon atoms with methane and Silane in solid argon: a matrix-spectroscopic study. Chem Eur J 8:4383–4391
Bogey M, Bolvin H, Demuynck C, Destombes JL (1991) Nonclassical double-bridged structure in silicon-containing molecules: experimental evidence in Si 2 H 2 from its submillimeter-wave spectrum. Phys Rev Lett 66:413
Bogey M, Bolvin H, Cordonnier M, Demuynck C, Destombes JL, Császárs AG (1994) Millimeter-and submillimeter-wave spectroscopy of dibridged Si2H2 isotopomers: experimental and theoretical structure. J Chem Phys 100:8614–8624
Andrews L, Wang X (2002) Infrared spectra of the novel Si2H2 and Si2H4 species and the SiH1, 2, 3 intermediates in solid neon, argon, and deuterium. J Phys Chem A 106:7696–7702
Yang T, Dangi BB, Kaiser RI, Chao K, Sun B, Chang AHH, Nguyen TL, Stanton JF (2017) Gas-phase formation of the Disilavinylidene (H2SiSi) transient. Angew Chemie Int Ed 56:1264–1268
Becke AD (1988) Density-functional exchange-energy approximation with correct asymptotic behavior. Phys Rev A 38:3098
Becke AD, Becke AD (1993) Density-functional thermochemistry. III. The role of exact exchange. J Chem Phys 98:5648–5652. https://doi.org/10.1063/1.464913
Lee C, Yang W, Parr RG (1988) Development of the Colle-Salvetti correlation-energy formula into a functional of the electron density. Phys Rev B 37:785–789. https://doi.org/10.1103/PhysRevB.37.785
Nemukhin AV, Grigorenko BL, Granovsky AA (2004) Molecular modeling by using the PC GAMESS program: from diatomic molecules to enzymes. Mosc Univ Chem Bull 45:75
Fletcher GD, Schmidt MW, Bode BM, Gordon MS (2000) The distributed data interface in GAMESS. Comput Phys Commun 128:190–200
Bode BM, Gordon MS (1998) Macmolplt: a graphical user interface for GAMESS. J Mol Graph Model 16:133–138
Umeda H, Koseki S, Nagashima U, Schmidt MW (2001) Parallelization of multireference perturbation calculations with GAMESS. J Comput Chem 22:1243–1251
Domingo LR, Chamorro E, Pérez P (2008) Understanding the reactivity of captodative ethylenes in polar cycloaddition reactions. A theoretical study. J Org Chem 73:4615–4624
Parr RG, Szentpály L, Liu S (1999) Electrophilicity index. J Am Chem Soc 121:1922–1924
Yang W, Parr RG (1985) Hardness, softness, and the Fukui function in the electronic theory of metals and catalysis. Proc Natl Acad Sci 82:6723–6726
Sheela NR, Muthu S, Sampathkrishnan S (2014) Molecular orbital studies (hardness, chemical potential and electrophilicity), vibrational investigation and theoretical NBO analysis of 4-4′-(1H-1, 2, 4-triazol-1-yl methylene) dibenzonitrile based on abinitio and DFT methods. Spectrochim Acta Part A Mol Biomol Spectrosc 120:237–251
Yang W, Pan Y, Zheng F, Cho H, Tai H-H, Zhan C-G (2009) Free-energy perturbation simulation on transition states and redesign of butyrylcholinesterase. Biophys J 96:1931–1938
Takeuchi K, Ichinohe M, Sekiguchi A (2012) A new Disilene with π-accepting groups from the reaction of Disilyne RSi□ SiR (R= Si i Pr [CH (SiMe3) 2]) with isocyanides. J Am Chem Soc 134:2954–2957
Acknowledgements
We gratefully appreciate Tarbiat Modares University for financial support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 41.9 kb)
Rights and permissions
About this article
Cite this article
Ayoubi-Chianeh, M., Kassaee, M.Z. A Quest for (sila)0-4cyclopentasilylenes and their Arduengo Analogs by DFT. Silicon 13, 939–960 (2021). https://doi.org/10.1007/s12633-020-00441-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12633-020-00441-1