Abstract
Objectives
This study investigates whether a citrus and pomegranate complex (CPC) improves physical fitness, mental well-being, and blood biomarkers for oxidative stress and endothelial function in healthy elderly.
Design
A randomized placebo-controlled cross-over trial.
Participants
The study included 36 healthy elderly aged 60–75 years old.
Intervention and Measurements
Participants received four weeks of CPC supplementation and performed the handgrip strength and senior fitness test. Quality of life (QOL) was assessed and blood samples were analyzed for oxidative stress and endothelial function markers.
Results
After four weeks of CPC supplementation, handgrip strength significantly improved (p=0.019), compared to placebo. Moreover, the thinking, memory, learning, and concentration facets were improved (p=0.042), compared to placebo, and plasma malondialdehyde decreased, compared to placebo (p=0.033). The intervention did not affect senior fitness and the other QOL domains and blood parameters.
Conclusion
Four weeks of daily CPC supplementation significantly improves handgrip strength and self-evaluated measures of psychological function in healthy older adults. Further research should focus on mechanisms associated with physical performance.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The life expectancy in Western countries is increasing rapidly. The World Health Organization has predicted that by 2050, 16% of the total world’s population will be aged 65 years and older (1). As ageing is considered a major risk factor for a variety of diseases such as cardiovascular disease, cognitive decline, and decreased physical function, an increasing number of individuals is dealing with a variety of age-related diseases (2, 3). Consequently, it is of importance to prevent the development of these diseases by maintaining physiological function and well-being at an older age, referred to as healthy ageing. Daskalopoulou et al. (4) reported in a systematic review that physical activity is positively associated with healthy ageing. Moreover, other modifiable factors related to a healthy lifestyle, such as smoking and drinking status, and leisure time activities, have also been shown to contribute to improved mental well-being and quality of life in elderly (5, 6). Therefore, maintaining a healthy lifestyle could be an important strategy to support healthy ageing and prevent these age-related pathologies (7).
Next to physical activity, nutritional status is an important lifestyle-related factor that has a substantial impact on healthy ageing (8). For example, diets high in fruits, vegetables, and fish are associated with longevity (9). Dietary supplementation with polyphenols has been suggested as a promising approach to prevent the negative consequences of ageing and to sustain a healthy lifestyle, before the onset of age-related comorbidities (10, 11). Moreover, four weeks of a high polyphenol diet resulted in improved physical- as well as mental health in hypertensive middle-aged adults (12). Hesperidin and punicalagins are polyphenols that are mainly found in citrus fruits and pomegranates, recognized for anti-inflammatory and antioxidant effects among others (13, 14). In previous randomized controlled trials, polyphenol supplementation has shown to decrease malondialdehyde (MDA) concentrations (15, 16), and to increase trolox equivalent antioxidative capacity (TEAC) (17), as well as nitrate levels, a marker for endothelial function (18). Moreover, beneficial effects of supplementation with these polyphenols on exercise performance have been observed in healthy adults and athletes (19, 20). However, to our knowledge, intervention studies using a combination of a citrus and pomegranate complex (CPC) in untrained older individuals have not been performed before. Therefore, the primary objective of this study was to investigate whether four weeks of supplementation with CPC improves physical fitness in healthy elderly, with secondary objectives to assess the effects of CPC on mental well-being and on systemic parameters of oxidative stress and endothelial function.
Materials and Methods
The study was approved by the Medical Ethics Committee of the Maastricht University Medical Centre+ and was conducted from June 2018 to January 2019 in full accordance with the principles of the Declaration of Helsinki as amended in 2013 (Fortaleza, Brazil), and the Dutch Regulations on Medical Research involving Human Subjects (1998). All participants provided written informed consent before participation. This trial was registered at ClinicalTrials.gov under NCT03781999.
Participants
Healthy adults aged 60 – 75y, and with a BMI between 18 and 28 kg/m2 were recruited through local media advertisements. Key exclusion criteria were: smoking; allergy to test product or citrus fruits; recent muscle injury before the start of the study; administration of dietary products containing citrus flavonoids or metabolites; supplement use; inability to perform the fitness tests; high blood pressure (systolic ≥ 140 mmHg, diastolic ≥ 90 mmHg); use of beta-blockers; abuse of alcohol (>20 alcoholic U/wk) or recreational drugs; and presence of medical conditions such as cardiovascular disease, and type 2 diabetes mellitus.
Study Design and Protocol
The study was designed as a randomized, double-blind, placebo-controlled, cross-over trial to investigate the effects of a daily dose of citrus and pomegranate complex (CPC) versus placebo on physical fitness and quality of life in elderly. The study consisted of two randomized intervention periods (CPC and placebo) of four weeks, with a wash-out period of at least four weeks (with a maximum of eight weeks) (median: 4 weeks) in between. Before the start of the study, a familiarization test was conducted. Subsequently, test days took place before and after each four week intervention period. Test days comprised anthropometric measurements, venous blood collection after an overnight fast, a standardized breakfast, the WHO-QOL-100 questionnaire, the handgrip strength test, and the senior fitness test as detailed below. A randomization list was generated by an independent researcher with Research Randomizer (http://randomizer.org). Researchers involved in the execution of the study were independently Good Clinical Practice (GCP) certified and received additional training where necessary.
Participants were instructed to abstain from high intensity resistance exercise and consumption of caffeine and alcohol 24 hours prior to each test day. During the supplementation period, participants were instructed to refrain from foods high in citrus flavonoids (oranges, lemons, pomegranate and grapefruit) and to maintain their weekly training schedule and habitual diet.
Physical Fitness Tests
Each test day, handgrip strength was measured using the Hydraulic Hand Dynamometer, SH5001 (Saehan, Gyeongsangnam-do, S. Korea). The participants were requested to sit on a chair with their elbow by the side of their body, unsupported and flexed in an angle of 90 degrees with the wrist in a natural position (21). Subsequently, subjects squeezed the handle as hard as possible for five seconds. Handgrip strength was measured in triplicate for the dominant hand in kg, using the best attempt for further analysis.
Physical fitness was assessed using the senior fitness test, a validated test battery for older adults. This test comprises six measurements: (1) Chair Stand Test for lower body strength; (2) Biceps Curl Test for upper body strength; (3) Six-Minute Walk Test (6-MWT) for aerobic capacity; (4) Chair Sit and Reach Test for lower body flexibility; (5) Back Scratch Test for upper body flexibility and (6) Up and Go Test for agility (22). In the Chair Stand Test, participants were instructed to stand up from a chair and sit down again with arms folded on their chest, as many times as possible during a time frame of 30s. The Biceps Curl Test was performed using a weight of 2.5 kg for women and 4.0 kg for men. The amount of completed curls in 30s was registered for the dominant hand. For the 6-MWT, participants were instructed to continuously walk back and forth in a 20m corridor for six minutes, after which total walking distance (m) was calculated. In the Chair Sit and Reach test, participants were instructed to sit at the edge of a chair with their dominant leg extended and to reach for their toes. Distance between their fingers and toes was determined (cm) in triplicate. For the Back Scratch Test, participants had to bring their hands together behind their back by reaching over the shoulder with one hand, and under the shoulder with the other hand. The distance between the middle fingers was determined in triplicate. In the Up and Go Test, participants were instructed to stand up from a chair, walk 2.45m, and return to the seated position as fast as possible. For all tests, best attempts were used for analysis.
WHO-QOL-100 Questionnaire
The WHO-QOL-100 questionnaire was used to determine quality of life of the participants on each test day. The questionnaire contained 100 questions, covering 24 facets. (23). From these facet scores, the domain scores of physical capacity, psychological capacity, social relationships, environment, independence and spirituality were calculated and used for further analysis. Furthermore, facets were evaluated only if results on the domains showed a trend or a significant time*treatment interaction.
Blood marker analysis
Blood samples were taken in a fasting state on each test day. Blood samples were collected in Heparin S-Monovette tubes (Sarstedt, Nümbrecht, Germany). Tubes were immediately centrifuged at 3000 rpm at 4 °C for plasma samples and at room temperature for serum samples. Hereafter, aliquots were snap-frozen in liquid nitrogen and stored at −80 °C for subsequent analysis.
Plasma MDA concentrations were determined as oxidative stress biomarker in blood plasma using the Thiobarbituric Acid Reactive Substances (TBARS) assay (TBARS Assay Kit — Malondialdehyde Quantification, Bioquochem, Spain) (24). TEAC was measured in blood plasma using the Cupric Reducing Antioxidant Capacity (CUPRAC) assay kit (CUPRAC Antioxidant Capacity Assay Kit, Bioquochem, Spain) as a measure for antioxidant capacity (25). Plasma nitrate (NO3) concentrations, which reflect endothelial function, were measured by assessing reduction of NO3 to gas using the gas-phase chemiluminescence technique as previously described (26). The reaction of reduced NO with ozone results in luminescence which was detected by an NOanalyzer.
Study product
Participants ingested two gelatin capsules per day containing 350 mg CPC (Actiful®, BioActor BV, Maastricht, The Netherlands), resulting in a total daily intake of 500 mg of a citrus sinensis extract (90% hesperidin, 450 mg), combined with 200 mg of a pomegranate fruit concentrate. This dose translates to approximately 0.9L of orange juice per day (27). Maltodextrin capsules (Gonmisol, Barcelona, Spain) were used as placebo and were identical to the study product in appearance and flavor due to the use of opaque capsules. Preparation of the capsules was performed by an independent researcher to ensure blinding. The capsules were consumed before breakfast with 200 mL of water. The daily dose of CPC was chosen based on previous studies showing a positive effect on exercise performance (20, 28).
Statistical Analysis
Sample size was calculated using a significance level of α = 0.05 and a power of β = 0.80, with an effect size of 0.39 based on the Six-Minute Walk Test (29). In total, 36 participants were required to obtain sufficient power. Statistical analysis was performed using IBM SPSS Statistics (version 26.0, IBM Corporation, Armonk, NY, USA). Data were reported as estimated mean ± standard error of the mean (SEM). Intention to treat linear mixed model analyses were carried out using period, treatment, and time as fixed factors, and time*treatment as the interaction term. If the interaction term was not significant, it was removed from the model. For all analyses, two sided p-values < 0.05 were considered to be statistically significant.
Results
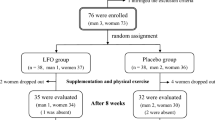
In total, 42 participants were assessed for eligibility, of which 37 were randomized. Five participants were excluded due to use of blood-pressure lowering medication. One participant dropped-out before receiving the allocated intervention, due to personal reasons. All remaining 36 participants completed the study and were included in the analyses. The mean age of the participants that finished the study was 66 ± 1 years, and consisted of 9 males and 27 females. Their average BMI was 25.3 ± 0.3 kg/m2, with a waist-hip-ratio of 0.90 ± 0.01. The CONSORT flow diagram is displayed in Figure 1.
Physical Fitness
Outcomes of the physical fitness tests are reported in Table 1. A significant time*treatment interaction was observed for handgrip strength (p = 0.019). After four weeks of CPC supplementation, a significant improvement was observed on handgrip strength, compared to placebo.
For the other physical fitness tests, no time*treatment interaction was observed. For the Chair Stand Test, Biceps Curl Test, and Up and Go test, a significant effect of time was observed (Chair Stand Test: Δ = 0.3 repeats, p = 0.034; Biceps Curl Test: Δ = 0.6 repeats, p = 0.009; Up and Go test: Δ = 0.1 s, p = 0.006), with both outcomes improving over time. No significant time or treatment effect were observed for the remaining physical fitness parameters.
Quality of Life Questionnaire
Results from the WHO-QOL-100 questionnaire were reported in Table 2. For the overall score and the six domain scores, no significant time*treatment effects were observed. However, a trend for a time*treatment interaction on the psychological function domain was found (p = 0.061). To further investigate the origin of this effect, the five facets that make up psychological function were analyzed separately: positive feelings; thinking, learning, memory, and concentration; self-esteem; bodily image and appearance; and negative feelings. Results can be found in Table 3. A significant time*treatment effect (p = 0.042) was observed for thinking, learning, memory, and concentration, showing improved scores after supplementation with CPC, compared to placebo. For the other facets, no time*treatment interaction or main effect of time or treatment was observed.
Blood biomarkers
Levels of the blood biomarkers before and after four weeks of supplementation with CPC or placebo were reported in Table 4. The time*treatment interaction for MDA was significant (p = 0.033), showing decreased levels of MDA after CPC supplementation, compared to placebo. For TEAC and NO3, no significant interaction was observed. A significant effect of time was found for TEAC (p = 0.017).
Discussion
In this study, the effects of CPC on physical fitness and mental wellbeing were evaluated in older adults. After four weeks of supplementation with CPC, handgrip strength significantly improved, as compared to placebo, which deteriorated. Moreover, self-evaluated psychological function showed a trend for improvement after CPC supplementation. This was supported by a significantly higher score for the CPC group in the thinking, memory, learning, and concentration facet, as compared to the placebo group. Lastly, a significant decrease in MDA was observed after CPC supplementation. No significant differences for the senior fitness test, the other QOL domains and blood parameters were observed between intervention and placebo.
This study was the first to determine the effects of a combined citrus and pomegranate complex on physical fitness in an older study population. Outcomes related to physical fitness included the handgrip strength test and the senior fitness test battery. Improved handgrip strength has previously been associated with reduced all-cause mortality, as well as with increased cognitive function, mobility, and functional status (30, 31). Moreover, in a cross-sectional study including over 68,000 participants, handgrip strength was positively associated with consumption of various food items in both men and women, and specifically fruit and vegetables in women (32). The values for handgrip strength observed in this study were comparable to normative values of the UK biobank, as well as a cohort from the United States within the same age group (33, 34). Only two previous studies have investigated the effects of a polyphenol-rich intervention on handgrip strength, both of which were performed in male adults (35, 36). Supplementation with grape juice in male volleyball athletes did not result in improved handgrip strength (35), while a significant improvement in handgrip strength 24 hours after intense resistance exercise was observed in male adults (36). Similar to our study, several polyphenol-rich intervention studies using different isometric strength tests have observed improvements in recreationally active males and older subjects (37–39). Here, we observed a slight increase in the intervention group as well as a decrease in the placebo group. Mitchell et al. (40) have shown that ageing results in muscle strength loss of 2.5–4% per year. However, the decrease in muscle strength observed in the placebo group is higher than expected based on ageing alone. Therefore, other underlying lifestyle factors such as diet and exercise may have been involved in this process. During the study, changes in these factors were not recorded, and participants were asked to refrain from citrus flavonoids. Consequently, the observed deterioration of muscle strength in the control group may have been the result of changes in lifestyle factors, which was attenuated by the CPC intervention.
In contrast, we did not observe significant improvements on the senior fitness test battery in the present study. Several studies have reported significant improvements in the 6MWT and the up and go test in (pre)frail and sarcopenic older adults and patients with peripheral artery disease (38, 41). The difference in health status of the participants (i.e. healthy older adults vs. (pre)frail older adults) could explain the contrasting results. Moreover, in an individualized nutritional intervention in (pre)frail older adults, 10m gait speed was significantly improved after a six month intervention of individualized nutrition, but not after only one month (42). Therefore, the senior fitness test might not be sensitive enough to observe changes in a four week dietary intervention study.
A potential mechanism that might be associated with the improvement in handgrip strength is improved mitochondrial function. With age, the number of mitochondria reduces and functionality decreases, which results in reduced ATP production, mitochondrial biogenesis, imbalanced fusion/fission, impaired mitophagy, and increased ROS production (43). This mitochondrial dysfunction has been shown to be a main contributor to the development of sarcopenia (44, 45). Recently, Migliavacca et al. (47) have provided evidence for a fundamental role of mitochondrial metabolism in skeletal muscle mass and function in elderly. However, based on this mechanism, an effect on walking distance would also be expected. For several dietary polyphenols, a role for mitochondrial metabolism and biogenesis in human health has been established [48]. For hesperetin, the aglycone of hesperidin, increased intracellular ATP production and mitochondrial spare capacity has been observed in vitro (49). Furthermore, neohesperidin, a glycoside of hesperetin, has shown to improve mitochondrial biogenesis in high fat diet fed mice (50). However, to our knowledge, this has not yet been confirmed in humans in vivo.
In this study, facets within the self-evaluated quality of life domain psychological function were improved after supplementation. As quality of life is an important factor of successful ageing, improved psychological functioning is of increasing relevance (51). In line with these findings, in various animal experiments, beneficial effects of hesperidin on depressive disorders, anxiety disorders, and stress were observed (52–54). Moreover, a review by Hajialyani et al. (55) has reported beneficial effects of hesperidin on neurodegenerative diseases both in animals and human studies. More specifically, a human study evaluating effects of a flavanone-rich orange juice in healthy older adults reported significant effects on global cognitive function (56).
The improvement in hand grip strength and quality of life might be the result of antioxidant properties and improvement of endothelial function. An improvement in antioxidant status during ageing could prevent the negative age-related effects of oxidative stress (57), and a link between polyphenol supplementation, exercise performance, and oxidative stress has already been established (58). Nitrate is considered to be an important signaling molecule involved in endothelial function by causing vasodilation in smooth muscle cells, thereby improving blood flow (59). Consequently, we analyzed TEAC and MDA as measures for oxidative stress, and NO3 as a measure for endothelial function. For TEAC, we did not observe a significant effect of CPC, while MDA decreased after CPC supplementation compared to placebo. In line with this observation, previous studies showed decreased MDA levels after cocoa flavonoid supplementation in older subjects (38) and after hesperidin supplementation in type 2 diabetes patients (60), indicating lowered oxidative stress. Lastly, we observed a slight increase in NO3, even though this did not reach significance. A higher potential to produce nitrite could contribute to enhanced blood flow and improved exercise capacity (61). In vitro studies have shown that exposure to hesperetin increases nitric oxide production in endothelial cells (62), and human in vivo studies have shown that hesperidin and punicalagin supplementation improve endothelial function (63–65).
A limitation of this study is that no skeletal muscle biopsies were collected. Consequently, effects of CPC supplementation on mitochondrial function could not be investigated. Moreover, the included study population was relatively healthy (BMI 18–28kg/m2, normal blood pressure, no cardiovascular disease, non-smokers), which might not be an accurate representation of this age group, as no significant effects on the senior fitness test were observed. For this population, the exercise battery may not have been strenuous enough to sufficiently challenge the participants, in order to detect small dietary intervention-induced improvements in performance capacity. Alternatively, it could be possible that the significant effect on hand grip strength was due to chance, as the other parameters were not significantly different between groups. Lastly, the study was not powered to determine differences between men and women, which could be of interest for future research as gender differences might affect physical activity levels. For a follow up study design, we suggest including physical fitness tests that are considered to be more challenging and collecting muscle biopsies to study potential underlying mechanisms.
In conclusion, this study shows that four weeks of daily CPC supplementation can significantly improve handgrip strength in healthy older adults. Furthermore, self-evaluated measures within the domain of psychological function (QOL) were significantly higher in the CPC group compared to placebo. Finally, blood levels of MDA were significantly decreased after CPC supplementation. Further research should focus on the mechanisms associated with the improvement in physical performance.
References
United Nations Department of Economic and Social Affairs Population Division. World Population Ageing 2020 Highlights: Living arrangements of older persons. (ST/ESA/SER.A/451) 2020.
Costantino, S.; Paneni, F.; Cosentino, F. Ageing, metabolism and cardiovascular disease. The Journal of Physiology 2016, 594, 2061–2073, doi:https://doi.org/10.1113/JP270538.
Garatachea, N.; Lucia, A. Genes, physical fitness and ageing. Ageing Res Rev 2013, 12, 90–102, doi:https://doi.org/10.1016/j.arr.2012.09.003.
Daskalopoulou, C.; Stubbs, B.; Kralj, C.; Koukounari, A.; Prince, M.; Prina, A.M. Physical activity and healthy ageing: A systematic review and meta-analysis of longitudinal cohort studies. Ageing Res Rev 2017, 38, 6–17, doi:https://doi.org/10.1016/j.arr.2017.06.003.
Zhang, X.; Xia, R.; Wang, S.; Xue, W.; Yang, J.; Sun, S.; Zhuang, G. Relative Contributions of Different Lifestyle Factors to Health-Related Quality of Life in the Elderly. Int J Environ Res Public Health 2018, 15, 256, doi:https://doi.org/10.3390/ijerph15020256.
Govindaraju, T.; Sahle, B.W.; McCaffrey, T.A.; McNeil, J.J.; Owen, A.J. Dietary Patterns and Quality of Life in Older Adults: A Systematic Review. Nutrients 2018, 10, doi:https://doi.org/10.3390/nu10080971.
Rudnicka, E.; Napierała, P.; Podfigurna, A.; Męczekalski, B.; Smolarczyk, R.; Grymowicz, M. The World Health Organization (WHO) approach to healthy ageing. Maturitas 2020, 139, 6–11, doi:https://doi.org/10.1016/j.maturitas.2020.05.018.
Wickramasinghe, K.; Mathers, J.C.; Wopereis, S.; Marsman, D.S.; Griffiths, J.C. From lifespan to healthspan: the role of nutrition in healthy ageing. J Nutr Sci 2020, 9, e33, doi:https://doi.org/10.1017/jns.2020.26.
Kiefte-de Jong, J.C.; Mathers, J.C.; Franco, O.H. Nutrition and healthy ageing: the key ingredients. Proc Nutr Soc 2014, 73, 249–259, doi:https://doi.org/10.1017/s0029665113003881.
Moore, K.; Hughes, C.F.; Ward, M.; Hoey, L.; McNulty, H. Diet, nutrition and the ageing brain: current evidence and new directions. Proc Nutr Soc 2018, 77, 152–163, doi:https://doi.org/10.1017/s0029665117004177.
Arora, I.; Sharma, M.; Sun, L.Y.; Tollefsbol, T.O. The Epigenetic Link between Polyphenols, Aging and Age-Related Diseases. Genes (Basel) 2020, 11, doi:https://doi.org/10.3390/genes11091094.
Kontogianni, M.D.; Vijayakumar, A.; Rooney, C.; Noad, R.L.; Appleton, K.M.; McCarthy, D.; Donnelly, M.; Young, I.S.; McKinley, M.C.; McKeown, P.P.; et al. A High Polyphenol Diet Improves Psychological Well-Being: The Polyphenol Intervention Trial (PPhIT). Nutrients 2020, 12, doi:https://doi.org/10.3390/nu12082445.
Li, C.; Schluesener, H. Health-promoting effects of the citrus flavanone hesperidin. Crit Rev Food Sci Nutr 2017, 57, 613–631, doi:https://doi.org/10.1080/10408398.2014.906382.
Venusova, E.; Kolesarova, A.; Horky, P.; Slama, P. Physiological and Immune Functions of Punicalagin. Nutrients 2021, 13, doi:https://doi.org/10.3390/nu13072150.
Panahi, Y.; Hosseini, M.S.; Khalili, N.; Naimi, E.; Majeed, M.; Sahebkar, A. Antioxidant and anti-inflammatory effects of curcuminoid-piperine combination in subjects with metabolic syndrome: A randomized controlled trial and an updated meta-analysis. Clin Nutr 2015, 34, 1101–1108, doi:https://doi.org/10.1016/j.clnu.2014.12.019.
Noronha, N.Y.; Pinhel, M.A.S.; Nicoletti, C.F.; Quinhoneiro, D.C.G.; Pinhanelli, V.C.; Oliveira, B.A.P.; Cortes-Oliveira, C.; Delfino, H.B.P.; Wolf, L.S.; Frantz, F.G.; et al. Green tea supplementation improves oxidative stress biomarkers and modulates IL-6 circulating levels in obese women. Nutr Hosp 2019, 36, 583–588, doi:https://doi.org/10.20960/nh.2159.
Decroix, L.; Tonoli, C.; Soares, D.D.; Descat, A.; Drittij-Reijnders, M.J.; Weseler, A.R.; Bast, A.; Stahl, W.; Heyman, E.; Meeusen, R. Acute cocoa Flavanols intake has minimal effects on exercise-induced oxidative stress and nitric oxide production in healthy cyclists: a randomized controlled trial. J Int Soc Sports Nutr 2017, 14, 28, doi:https://doi.org/10.1186/s12970-017-0186-7.
Moreno-Luna, R.; Muñoz-Hernandez, R.; Miranda, M.L.; Costa, A.F.; Jimenez-Jimenez, L.; Vallejo-Vaz, A.J.; Muriana, F.J.; Villar, J.; Stiefel, P. Olive oil polyphenols decrease blood pressure and improve endothelial function in young women with mild hypertension. Am J Hypertens 2012, 25, 1299–1304, doi:https://doi.org/10.1038/ajh.2012.128.
Ammar, A.; Bailey, S.J.; Chtourou, H.; Trabelsi, K.; Turki, M.; Hökelmann, A.; Souissi, N. Effects of pomegranate supplementation on exercise performance and post-exercise recovery in healthy adults: a systematic review. Br J Nutr 2018, 120, 1201–1216, doi:https://doi.org/10.1017/s0007114518002696.
Overdevest, E.; Wouters, J.A.; Wolfs, K.H.M.; van Leeuwen, J.J.M.; Possemiers, S. Citrus Flavonoid Supplementation Improves Exercise Performance in Trained Athletes. J Sports Sci Med 2018, 17, 24–30.
Massy-Westropp, N.M.; Gill, T.K.; Taylor, A.W.; Bohannon, R.W.; Hill, C.L. Hand Grip Strength: age and gender stratified normative data in a population-based study. BMC research notes 2011, 4, 127, doi:https://doi.org/10.1186/1756-0500-4-127.
Langhammer, B.; Stanghelle, J. The Senior Fitness Test. Journal of Physiotherapy 2015, 61, doi:https://doi.org/10.1016/j.jphys.2015.04.001.
Masthoff, E.D.; Trompenaars, F.J.; Van Heck, G.L.; Hodiamont, P.P.; De Vries, J. Validation of the WHO Quality of Life assessment instrument (WHOQOL-100) in a population of Dutch adult psychiatric outpatients. Eur Psychiatry 2005, 20, 465–473, doi:https://doi.org/10.1016/j.eurpsy.2004.09.012.
Tsikas, D. Assessment of lipid peroxidation by measuring malondialdehyde (MDA) and relatives in biological samples: Analytical and biological challenges. Anal Biochem 2017, 524, 13–30, doi:https://doi.org/10.1016/j.ab.2016.10.021.
Apak, R.; Guclu, K.; Demirata, B.; Ozyurek, M.; Celik, S.E.; Bektasoglu, B.; Berker, K.I.; Ozyurt, D. Comparative evaluation of various total antioxidant capacity assays applied to phenolic compounds with the CUPRAC assay. Molecules (Basel, Switzerland) 2007, 12, 1496–1547, doi:https://doi.org/10.3390/12071496.
Cermak, N.M.; Hansen, D.; Kouw, I.W.; van Dijk, J.W.; Blackwell, J.R.; Jones, A.M.; Gibala, M.J.; van Loon, L.J. A single dose of sodium nitrate does not improve oral glucose tolerance in patients with type 2 diabetes mellitus. Nutr Res 2015, 35, 674–680, doi:https://doi.org/10.1016/j.nutres.2015.05.017.
Gattuso, G.; Barreca, D.; Gargiulli, C.; Leuzzi, U.; Caristi, C. Flavonoid composition of Citrus juices. Molecules (Basel, Switzerland) 2007, 12, 1641–1673, doi:https://doi.org/10.3390/12081641.
van Iersel, L.E.; Stevens, Y.R.; Conchillo, J.M.; Troost, F.J. The effect of citrus flavonoid extract supplementation on anaerobic capacity in moderately trained athletes: a randomized controlled trial. Journal of the International Society of Sports Nutrition 2021, 18, 2, doi:https://doi.org/10.1186/s12970-020-00399-w.
Duncan, M.J.; Clarke, N.D.; Tallis, J.; Guimarães-Ferreira, L.; Leddington Wright, S. The effect of caffeine ingestion on functional performance in older adults. J Nutr Health Aging 2014, 18, 883–887, doi:https://doi.org/10.1007/s12603-014-0474-8.
Bae, E.-J.; Park, N.-J.; Sohn, H.-S.; Kim, Y.-H. Handgrip Strength and All-Cause Mortality in Middle-Aged and Older Koreans. Int J Environ Res Public Health 2019, 16, 740, doi:https://doi.org/10.3390/ijerph16050740.
Rijk, J.M.; Roos, P.R.; Deckx, L.; van den Akker, M.; Buntinx, F. Prognostic value of handgrip strength in people aged 60 years and older: A systematic review and meta-analysis. Geriatr Gerontol Int 2016, 16, 5–20, doi:https://doi.org/10.1111/ggi.12508.
Gedmantaite, A.; Celis-Morales, C.A.; Ho, F.; Pell, J.P.; Ratkevicius, A.; Gray, S.R. Associations between diet and handgrip strength: a cross-sectional study from UK Biobank. Mech Ageing Dev 2020, 189, 111269, doi:https://doi.org/10.1016/j.mad.2020.111269.
Spruit, M.A.; Sillen, M.J.; Groenen, M.T.; Wouters, E.F.; Franssen, F.M. New normative values for handgrip strength: results from the UK Biobank. J Am Med Dir Assoc 2013, 14, 775.e775-711, doi:https://doi.org/10.1016/j.jamda.2013.06.013.
Wang, Y.C.; Bohannon, R.W.; Li, X.; Sindhu, B.; Kapellusch, J. Hand-Grip Strength: Normative Reference Values and Equations for Individuals 18 to 85 Years of Age Residing in the United States. J Orthop Sports Phys Ther 2018, 48, 685–693, doi:https://doi.org/10.2519/jospt.2018.7851.
Martins, N.C.; Dorneles, G.P.; Blembeel, A.S.; Marinho, J.P.; Proença, I.C.T.; da Cunha Goulart, M.J.V.; Moller, G.B.; Marques, E.P.; Pochmann, D.; Salvador, M.; et al. Effects of grape juice consumption on oxidative stress and inflammation in male volleyball players: A randomized, double-blind, placebo-controlled clinical trial. Complement Ther Med 2020, 54, 102570, doi:https://doi.org/10.1016/j.ctim.2020.102570.
Hooper, D.R.; Orange, T.; Gruber, M.T.; Darakjian, A.A.; Conway, K.L.; Hausenblas, H.A. Broad Spectrum Polyphenol Supplementation from Tart Cherry Extract on Markers of Recovery from Intense Resistance Exercise. J Int Soc Sports Nutr 2021, 18, 47, doi:https://doi.org/10.1186/s12970-021-00449-x.
Trombold, J.R.; Barnes, J.N.; Critchley, L.; Coyle, E.F. Ellagitannin consumption improves strength recovery 2–3 d after eccentric exercise. Med Sci Sports Exerc 2010, 42, 493–498, doi:https://doi.org/10.1249/MSS.0b013e3181b64edd.
Munguia, L.; Rubio-Gayosso, I.; Ramirez-Sanchez, I.; Ortiz, A.; Hidalgo, I.; Gonzalez, C.; Meaney, E.; Villarreal, F.; Najera, N.; Ceballos, G. High Flavonoid Cocoa Supplement Ameliorates Plasma Oxidative Stress and Inflammation Levels While Improving Mobility and Quality of Life in Older Subjects: A Double-Blind Randomized Clinical Trial. The Journals of Gerontology: Series A 2019, 74, 1620–1627, doi:https://doi.org/10.1093/gerona/glz107.
Trombold, J.R.; Reinfeld, A.S.; Casler, J.R.; Coyle, E.F. The effect of pomegranate juice supplementation on strength and soreness after eccentric exercise. J Strength Cond Res 2011, 25, 1782–1788, doi:https://doi.org/10.1519/JSC.0b013e318220d992.
Mitchell, W.; Atherton, P.; Williams, J.; Larvin, M.; Lund, J.; Narici, M. Sarcopenia, Dynapenia, and the Impact of Advancing Age on Human Skeletal Muscle Size and Strength; a Quantitative Review. Frontiers in Physiology 2012, 3, doi:https://doi.org/10.3389/fphys.2012.00260.
McDermott, M.M.; Criqui, M.H.; Domanchuk, K.; Ferrucci, L.; Guralnik, J.M.; Kibbe, M.R.; Kosmac, K.; Kramer, C.M.; Leeuwenburgh, C.; Li, L.; et al. Cocoa to Improve Walking Performance in Older People With Peripheral Artery Disease. Circulation Research 2020, 126, 589–599, doi:doi:https://doi.org/10.1161/CIRCRESAHA.119.315600.
Hsieh, T.-J.; Su, S.-C.; Chen, C.-W.; Kang, Y.-W.; Hu, M.-H.; Hsu, L.-L.; Wu, S.-Y.; Chen, L.; Chang, H.-Y.; Chuang, S.-Y.; et al. Individualized home-based exercise and nutrition interventions improve frailty in older adults: a randomized controlled trial. International Journal of Behavioral Nutrition and Physical Activity 2019, 16, 119, doi:https://doi.org/10.1186/s12966-019-0855-9.
Boengler, K.; Kosiol, M.; Mayr, M.; Schulz, R.; Rohrbach, S. Mitochondria and ageing: role in heart, skeletal muscle and adipose tissue. J Cachexia Sarcopenia Muscle 2017, 8, 349–369, doi:https://doi.org/10.1002/jcsm.12178.
Marzetti, E.; Calvani, R.; Tosato, M.; Cesari, M.; Di Bari, M.; Cherubini, A.; Collamati, A.; D’Angelo, E.; Pahor, M.; Bernabei, R.; et al. Sarcopenia: an overview. Aging Clin Exp Res 2017, 29, 11–17, doi:https://doi.org/10.1007/s40520-016-0704-5.
Ferri, E.; Marzetti, E.; Calvani, R.; Picca, A.; Cesari, M.; Arosio, B. Role of Age-Related Mitochondrial Dysfunction in Sarcopenia. International Journal of Molecular Sciences 2020, 21, 5236.
Zane, A.C.; Reiter, D.A.; Shardell, M.; Cameron, D.; Simonsick, E.M.; Fishbein, K.W.; Studenski, S.A.; Spencer, R.G.; Ferrucci, L. Muscle strength mediates the relationship between mitochondrial energetics and walking performance. Aging Cell 2017, 16, 461–468, doi:10.1111/acel.12568.
Migliavacca, E.; Tay, S.K.H.; Patel, H.P.; Sonntag, T.; Civiletto, G.; McFarlane, C.; Forrester, T.; Barton, S.J.; Leow, M.K.; Antoun, E.; et al. Mitochondrial oxidative capacity and NAD(+) biosynthesis are reduced in human sarcopenia across ethnicities. Nat Commun 2019, 10, 5808–5808, doi:https://doi.org/10.1038/s41467-019-13694-1.
Teixeira, J.; Chavarria, D.; Borges, F.; Wojtczak, L.; Wieckowski, M.R.; Karkucinska-Wieckowska, A.; Oliveira, P.J. Dietary Polyphenols and Mitochondrial Function: Role in Health and Disease. Curr Med Chem 2019, 26, 3376–3406, doi:https://doi.org/10.2174/0929867324666170529101810.
Biesemann, N.; Ried, J.S.; Ding-Pfennigdorff, D.; Dietrich, A.; Rudolph, C.; Hahn, S.; Hennerici, W.; Asbrand, C.; Leeuw, T.; Strübing, C. High throughput screening of mitochondrial bioenergetics in human differentiated myotubes identifies novel enhancers of muscle performance in aged mice. Sci Rep 2018, 8, 9408, doi:https://doi.org/10.1038/s41598-018-27614-8.
Wang, S.W.; Sheng, H.; Bai, Y.F.; Weng, Y.Y.; Fan, X.Y.; Lou, L.J.; Zhang, F. Neohesperidin enhances PGC-1α-mediated mitochondrial biogenesis and alleviates hepatic steatosis in high fat diet fed mice. Nutr Diabetes 2020, 10, 27, doi:https://doi.org/10.1038/s41387-020-00130-3.
Choi, M.; Lee, M.; Lee, M.J.; Jung, D. Physical activity, quality of life and successful ageing among community-dwelling older adults. Int Nurs Rev 2017, 64, 396–404, doi:https://doi.org/10.1111/inr.12397.
Souza, L.C.; de Gomes, M.G.; Goes, A.T.; Del Fabbro, L.; Carlos Filho, B.; Boeira, S.P.; Jesse, C.R. Evidence for the involvement of the serotonergic 5-HT1A receptors in the antidepressant-like effect caused by hesperidin in mice. Progress in Neuro-Psychopharmacology and Biological Psychiatry 2013, 40, 103–109.
Wasowski, C.; Loscalzo, L.M.; Higgs, J.; Marder, M. Chronic intraperitoneal and oral treatments with hesperidin induce central nervous system effects in mice. Phytotherapy Research 2012, 26, 308–312.
Viswanatha, G.; Shylaja, H.; Sandeep Rao, K.; Santhosh Kumar, V.; Jagadeesh, M. Hesperidin ameliorates immobilization-stress-induced behavioral and biochemical alterations and mitochondrial dysfunction in mice by modulating nitrergic pathway. International Scholarly Research Notices 2012, 2012.
Hajialyani, M.; Hosein Farzaei, M.; Echeverría, J.; Nabavi, S.M.; Uriarte, E.; Sobarzo-Sánchez, E. Hesperidin as a Neuroprotective Agent: A Review of Animal and Clinical Evidence. Molecules 2019, 24, doi:10.3390/molecules24030648.
Kean, R.J.; Lamport, D.J.; Dodd, G.F.; Freeman, J.E.; Williams, C.M.; Ellis, J.A.; Butler, L.T.; Spencer, J.P. Chronic consumption of flavanone-rich orange juice is associated with cognitive benefits: an 8-wk, randomized, double-blind, placebo-controlled trial in healthy older adults. Am J Clin Nutr 2015, 101, 506–514, doi:https://doi.org/10.3945/ajcn.114.088518.
Simioni, C.; Zauli, G.; Martelli, A.M.; Vitale, M.; Sacchetti, G.; Gonelli, A.; Neri, L.M. Oxidative stress: role of physical exercise and antioxidant nutraceuticals in adulthood and aging. Oncotarget 2018, 9, 17181–17198, doi:https://doi.org/10.18632/oncotarget.24729.
Myburgh, K.H. Polyphenol supplementation: benefits for exercise performance or oxidative stress? Sports Med 2014, 44 Suppl 1, S57–70, doi:https://doi.org/10.1007/s40279-014-0151-4.
Hobbs, D.A.; George, T.W.; Lovegrove, J.A. The effects of dietary nitrate on blood pressure and endothelial function: a review of human intervention studies. Nutr Res Rev 2013, 26, 210–222, doi:https://doi.org/10.1017/s0954422413000188.
Homayouni, F.; Haidari, F.; Hedayati, M.; Zakerkish, M.; Ahmadi, K. Hesperidin Supplementation Alleviates Oxidative DNA Damage and Lipid Peroxidation in Type 2 Diabetes: A Randomized Double-Blind Placebo-Controlled Clinical Trial. Phytother Res 2017, 31, 1539–1545, doi:https://doi.org/10.1002/ptr.5881.
Jones, A.M. Dietary nitrate supplementation and exercise performance. Sports medicine (Auckland, N.Z.) 2014, 44 Suppl 1, S35–S45, doi:https://doi.org/10.1007/s40279-014-0149-y.
Liu, L.; Xu, D.M.; Cheng, Y.Y. Distinct effects of naringenin and hesperetin on nitric oxide production from endothelial cells. J Agric Food Chem 2008, 56, 824–829, doi:https://doi.org/10.1021/jf0723007.
Rizza, S.; Muniyappa, R.; Iantorno, M.; Kim, J.A.; Chen, H.; Pullikotil, P.; Senese, N.; Tesauro, M.; Lauro, D.; Cardillo, C.; et al. Citrus polyphenol hesperidin stimulates production of nitric oxide in endothelial cells while improving endothelial function and reducing inflammatory markers in patients with metabolic syndrome. J Clin Endocrinol Metab 2011, 96, E782–792, doi:https://doi.org/10.1210/jc.2010-2879.
Amiot, M.J.; Riva, C.; Vinet, A. Effects of dietary polyphenols on metabolic syndrome features in humans: a systematic review. Obes Rev 2016, 17, 573–586, doi:https://doi.org/10.1111/obr.12409.
Liu, X.; Cao, K.; Lv, W.; Feng, Z.; Liu, J.; Gao, J.; Li, H.; Zang, W.; Liu, J. Punicalagin attenuates endothelial dysfunction by activating FoxO1, a pivotal regulating switch of mitochondrial biogenesis. Free Radic Biol Med 2019, 135, 251–260, doi:https://doi.org/10.1016/j.freeradbiomed.2019.03.011.
Funding
Funding: Authors IC and FT are supported by the Province of Limburg, The Netherlands [grant number HEFI-2]. This research project was supported by BioActor B.V.
Author information
Authors and Affiliations
Contributions
Author contributions: Conceptualization: FJT. Formal analysis: SA. Original draft preparation: SA. Critical revision: IC, FH, FJT. The authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest: S.A. is an employee of BioActor BV. F.H. is a sports medicine consultant and owner of Sports Medicine Center Maastricht*Parkstad. All other authors declare no conflict of interest. The funders had no role in the design of the study, in the preparation of the manuscript, or in the decision to publish the results.
Ethical standard: The experiments carried out complied with the law in the Netherlands, at the time of study execution.
Rights and permissions
Open Access: This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits use, duplication, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license and indicate if changes were made.
About this article
Cite this article
Ahles, S., Cuijpers, I., Hartgens, F. et al. The Effect of a Citrus and Pomegranate Complex on Physical Fitness and Mental Well-Being in Healthy Elderly: A Randomized Placebo-Controlled Trial. J Nutr Health Aging 26, 839–846 (2022). https://doi.org/10.1007/s12603-022-1834-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12603-022-1834-4