Abstract
Background and Objectives
Sex hormone concentrations and telomere length are age related responses of human body, while whether there is a direct relation between sex hormone and telomere length is uncertain. Therefore, we used the data of National Health and Nutrition Examination Survey (NHANES) to quantify their direct association.
Research Design and Methods
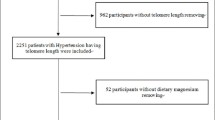
A total of 710 women aged 35–60 years and 539 men aged 20–85 years were included from two cycles of the NHANES (1999–2002). Telomere length relative to standard reference DNA (T/S ratio) was measured using quantitative polymerase chain reaction method. Seven hormones in serum (5 in men and 2 in women) were assayed. Logistic regressions were used to calculate the odds ratios to evaluate the telomere length-sex hormones association.
Results
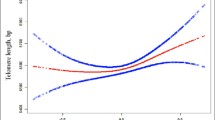
Men with vigorous physical activity (71.1%) and without history of cardiovascular diseases, diabetes, and lipid-lowering drugs using tended to have a longer telomere length (all P-values < 0.05); while women with longer sedentary time, smaller pregnant or live birth, and with older ages of firth/last birth were likely with longer telomere length (all P-values < 0.05). After adjusted for potential confounders, only anti-Mullerian hormone was positively and stably associated with short leukocytes telomere length in men (OR: 1.098; 95% CI: 1.034, 1.165). We did not detect any significant association of short telomere length with sex hormones in men and women.
Discussion and Implications
Serum anti-Mullerian hormone in men was positively and stably associated with telomere length. More large-scaled and well-designed prospective studies are warranted to reconfirm our conclusions.
Similar content being viewed by others
References
Shay JW, Wright WE: Senescence and immortalization: role of telomeres and telomerase. Carcinogenesis 2005, 26(5):867–874. doi:https://doi.org/10.1093/carcin/bgh296
Okuda K, Bardeguez A, Gardner JP, Rodriguez P, Ganesh V, Kimura M, Skurnick J, Awad G, Aviv A: Telomere length in the newborn. Pediatric Research 2002, 52(3):377–381. doi: https://doi.org/10.1203/01.PDR.0000022341.72856.72
Mayer S, Bruderlein S, Perner S, Waibel I, Holdenried A, Ciloglu N, Hasel C, Mattfeldt T, Nielsen KV, Moller P: Sex-specific telomere length profiles and age-dependent erosion dynamics of individual chromosome arms in humans. Cytogenetic and Genome Research 2006, 112(3–4):194–201. doi:https://doi.org/10.1159/000089870
Correia-Melo C, Hewitt G, Passos JF: Telomeres, oxidative stress and inflammatory factors: partners in cellular senescence? Longevity & healthspan 2014, 3(1):1–1. doi:https://doi.org/10.1186/2046-2395-3-1
Mann V, Huber C, Kogianni G, Collins F, Noble B: The antioxidant effect of estrogen and Selective Estrogen Receptor Modulators in the inhibition of osteocyte apoptosis in vitro. Bone 2007, 40(3):674–684. doi:https://doi.org/10.1016/j.bone.2006.10.014
Cha Y, Kwon SJ, Seol W, Park K-S: Estrogen Receptor-alpha Mediates the Effects of Estradiol on Telomerase Activity in Human Mesenchymal Stem Cells. Molecules and Cells 2008, 26(5):454–458.
Song Y, Cho M, Brennan KM, Chen BH, Song Y, Manson JE, Hevener AL, You NY, Butch AW, Liu S: Relationships of sex hormone levels with leukocyte telomere length in Black, Hispanic, and Asian/Pacific Islander postmenopausal women. Journal of diabetes 2018, 10(6):502–511. doi:https://doi.org/10.1111/1753-0407.12577
Coburn SB, Graubard BI, Trabert B, McGlynn KA, Cook MB: Associations between circulating sex steroid hormones and leukocyte telomere length in men in the National Health and Nutrition Examination Survey. Andrology 2018, 6(4):542–546. doi: https://doi.org/10.1111/andr.12494
Yeap BB, Knuiman MW, Divitini ML, Hui J, Arscott GM, Handelsman DJ, McLennan SV, Twigg SM, McQuillan B, Hung J et al: Epidemiological and Mendelian Randomization Studies of Dihydrotestosterone and Estradiol and Leukocyte Telomere Length in Men. Journal of Clinical Endocrinology & Metabolism 2016, 101(3):1299–1306. doi: https://doi.org/10.1210/jc.2015-4139
National Health and Nutrition Examination Survey 1999–2000 Data Documentation, Codebook, and Frequencies [https://wwwn.cdc.gov/Nchs/Nhanes/1999-2000/SSCHL_A.htm]
Needham BL, Adler N, Gregorich S, Rehkopf D, Lin J, Blackburn EH, Epel ES: Socioeconomic status, health behavior, and leukocyte telomere length in the National Health and Nutrition Examination Survey, 1999–2002. Social Science & Medicine 2013, 85:1–8. doi: https://doi.org/10.1016/j.socscimed.2013.02.023
Cawthon RM: Telomere measurement by quantitative PCR. Nucleic Acids Research 2002, 30(10).doi:https://doi.org/10.1093/nar/30.10.e47
National Health and Nutrition Examination Survey1999–2000 Data Documentation, Codebook, and Frequencies [https://wwwn.cdc.gov/Nchs/Nhanes/1999-2000/TELO_A.htm#TeloMean]
Calvert PA, Liew T-V, Gorenne I, Clarke M, Costopoulos C, Obaid DR, O’Sullivan M, Shapiro LM, McNab DC, Densem CG et al: Leukocyte Telomere Length Is Associated With High-Risk Plaques on Virtual Histology Intravascular Ultrasound and Increased Proinflammatory Activity. Arteriosclerosis Thrombosis and Vascular Biology 2011, 31(9):2157–U2580. doi: https://doi.org/10.1161/ATVBAHA.111.229237
Haffner SM: Sex hormone-binding protein, hyperinsulinemia, insulin resistance and noninsulin-dependent diabetes. Hormone Research 1996, 45(3–5):233–237.doi: https://doi.org/10.1159/000184794
Ding EL, Song YQ, Malik VS, Liu SM: Sex differences of endogenous sex hormones and risk of type 2 diabetes — A systematic review and meta-analysis. Jama-Journal of the American Medical Association 2006, 295(11):1288–1299. doi: https://doi.org/10.1001/jama.295.11.1288
Lindstedt G, Lundberg PA, Lapidus L, Lundgren H, Bengtsson C, Bjorntorp P: Low sex-hormone binding globulin conxentration as independent risk factor for development of NIDDM-12-yr follow-up of population study of women in gothenbugs, sweden. Diabetes 1991, 40(1):123–128. doi: https://doi.org/10.2337/diab.40.1.123
Golden SH, Dobs AS, Vaidya D, Szklo M, Gapstur S, Kopp P, Liu K, Ouyang P: Endogenous sex hormones and glucose tolerance status in postmenopausal women. Journal of Clinical Endocrinology & Metabolism 2007, 92(4):1289–1295. doi: https://doi.org/10.1210/jc.2006-1895
Ding EL, Song Y, Manson JE, Rifai N, Buring JE, Liu S: Plasma sex steroid hormones and risk of developing type 2 diabetes in women: a prospective study. Diabetologia 2007, 50(10):2076–2084. doi:https://doi.org/10.1007/s00125-007-0785-y
Fortunati N, Becchis M, Catalano MG, Comba A, Ferrera P, Raineri M, Berta L, Frairia R: Sex hormone-binding globulin, its membrane receptor, and breast cancer: a new approach to the modulation of estradiol action in neoplastic cells. Journal of Steroid Biochemistry and Molecular Biology 1999, 69(1–6):473–479. doi:https://doi.org/10.1016/s0960-0760(99)00068-0
Shander D, Anderson LD, Barraclough CA: Follicle-stimulating hormone and luteinizing hormone affect the endogenous release of pituitary follicle-stimulating hormone and the ovarian secretion of inhibin in rats. Endocrinology 1980, 106(4):1047–1053. doi: https://doi.org/10.1210/endo-106-4-1047
Wang N, Zhang K, Han B, Li Q, Chen Y, Zhu C, Chen Y, Xia F, Zhai H, Jiang B et al: Follicle stimulating hormone, its novel association with sex hormone binding globulin in men and postmenopausal women. Endocrine 2017, 56(3):649–657. doi: https://doi.org/10.1007/s12020-017-1272-y
Ibrahim E, Aballa TC, Roudebush WE, Lynne CM, Brackett NL: Inhibin B is lower and anti-Mullerian hormone is similar in serum of men with spinal cord injuries compared to controls. Systems biology in reproductive medicine 2015, 61(2):72–77. doi: https://doi.org/10.3109/19396368.2014.1002583
Grunewald S, Glander HJ, Paasch U, Kratzsch J: Age-dependent inhibin B concentration in relation to FSH and semen sample qualities: a study in 2448 men. Reproduction (Cambridge, England) 2013, 145(3):237–244. doi: https://doi.org/10.1530/REP-12-0415
Acknowledgements
The project described was supported by NNSFC (National Natural Science Foundation of China, No.: 81703238) (Dr. Zhang and Dan-Tong Gu).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest: The authors have no conflict of interest to report
Ethical standard: The data was from National Health and Nutrition Examination Survey (NHANES), and all protocols were approved by the National Center for Health Statistics Research ethics review board. All participants have singed informed consent.
Electronic supplementary material
12603_2019_1291_MOESM1_ESM.docx
Supplemental table 1. Sensitivity analyses of relation between sex hormone concentrations and telomere length in women, OR 95% CI
Rights and permissions
About this article
Cite this article
Gu, D., Li, J., Little, J. et al. Associations between Serum Sex Hormone Concentrations and Telomere Length among U.S. Adults, 1999–2002. J Nutr Health Aging 24, 48–54 (2020). https://doi.org/10.1007/s12603-019-1291-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12603-019-1291-x