Abstract
Vigor and selected physiological parameters (content of phenolic compounds, soluble sugars, chlorophyll a and b, and carotenoids) of eight naked and two husked oat cultivars harvested at 15% moisture content were determined. Oat seeds were threshed using two rotational speeds of the threshing drum: 1.6 m s−1 (LS) and 2.4 m s−1 (HS). They were then inoculated with a medium pathogenicity strain of Fusarium culmorum, strain IPO 348–01. In naked cultivars, the use of HS resulted in more severe mechanical damage; in consequence, seedling vigor decreased by 16%. In naked cultivars chlorophyll a and b and carotenoids content were significantly reduced—by more than 64%—when the HS was used. The inoculation caused over a 100% increase of carbohydrates in roots at LS but only a slight increase at HS. Phenolic compound content was twice as high in roots than in leaves after inoculation for both LS and HS. Area of microdamage and reduction of root fresh weight (f.wt.) are significantly correlated with biochemical parameters. Smaller microdamage area and root f.wt. reduction are connected with higher physiological parameters, which confirms lower seedling susceptibility to pathogen infection.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Genetically conditioned, naked oat cultivars still arouse interest, which is reflected by the number of research works published on this topic, as well as the increasing number of these cultivars being entered into the Polish National List and the Common Catalogue (CCA 2010; LORR 2010). The lack of husks, which is favorable from the technological point of view, may be a cause of increased susceptibility of naked oat seeds to biotic and abiotic factors affecting the development of seeds with high vigor parameters. A problem that is especially difficult to avoid is connected with the dynamic loading of seed during harvest that causes, among other things, increased susceptibility to secondary infections by Fusarium culmorum, one of the most frequently occurring facultative cereal pathogens causing seedling blight and root rot (Grey and Mathre 1988; Inch and Gilbert 2003; Kiecana et al. 2002; Magan et al. 2002). This species also causes Fusarium head blight (FHB), which is a devastating disease to most small-grained cereals. For example, FHB epidemics of barley were reported in central parts of the USA in 1993–1996 (Salas et al. 1997). Fusaria cause quantitative losses of cereal yield and reduce grain quality due to reduced kernel weight and size; moreover, secondary metabolites produced by pathogenic species of Fusarium spp. are accumulated (Jones and Mirocha 1999; Ruckenbauer et al. 2001; Wiśniewska and Kowalczyk 2005). The literature data show that the presence of Fusarium spp. may contribute to the occurrence of diseases, which, during the vegetative season, lead to deterioration in the yield structure (Suchowilska et al. 2007), decrease in seed weight (Kiecana et al. 2005), or a decrease in vigor and germinability, which in turn affects seedling and subsequent plant developmental stages (Bechtel et al. 1985). Moreover, seeds intended for consumption and for the production of feedstuff, if contaminated with mycotoxins, are a hazard to humans and animals (Hussein and Brasel 2001; Soroka et al. 2008; Torp and Nirenberg 2004). In Europe (Germany), research on the presence of toxins in cereals revealed a high percentage of contamination; in oat samples, 71% were contaminated with DON (deoxynivalenol) mycotoxin (Schollenberger et al. 2006).
The danger of F. culmorum infections is increased by the high share of cereals in crop rotation, reaching 56% in the EU and 73% in Poland (GUS 2009; EUROSTAT 2008), making it difficult to limit the occurrence of the diseases caused by this pathogen. The importance of the problem is stressed by the constantly revised standards concerning the maximum admissible levels of mycotoxins in food products in the European Union (European Commission 2006a, b, c, 2007). The secondary influence of the infections by the pathogens on reproductivity also seems to be of significance because, according to TeKrony and Egli (1991), epidemics of Fusarium wilt diseases are a serious problem for crop and sowable material producers. Considering the climate of Central and Eastern Europe, it is of significance because, according to Agarwal and Sinclair (1997), rainfall and high temperatures during the period of spike development have a significant effect on microflora development. This is exacerbated by the morphological structure of cereal seeds, which are particularly susceptible to the accumulation of fungal spores on their surface, favoring their tendency to become infected (Trojanowska 2002). It is of special importance in the case of naked oat cultivars, the seeds of which are covered with hairs, whereas in traditional cultivars the husk seems to provide a barrier that protects seeds against the influence of external factors, including the settlement of spores directly on their surface.
According to Nicholson and Hammerschmidt (1992), the natural defensive reaction of a plant organism to infection by a pathogen shows itself, among other things, in the release of phenolic compounds, the components of the cell walls, and their intensive collection and synthesis at the infection site. Phenolic compounds exert a toxic action on the pathogen or, by participation in lignification of the cell walls and formation of structural barriers, prevent penetration of the pathogen into the cells adjacent to the infection site (Appel 1993; Lattanzio et al. 2006; Puupponen-Pimia et al. 2005). Ferulic acid plays a fundamental role in the resistance of wheat cultivars to Fusarium; its concentration in mature, well-developed seeds is similar in both susceptible and resistant forms, however, from the beginning of flowering until approximately the 20th day. Resistant cultivars show higher ferulic acid values (McKeehen et al. 1999). Many phenolic compounds occurring in plants also have the properties of signal particles; as phytoanticipins, phytoalexins, modulators of pathogenesis, and activators of plant disease resistance genes, they play a varied role in resistance processes in plants (Dakora 1996; Mauch-Mani and Métraux 1998; Nicholson and Hammerschmidt 1992; Stachel et al. 1986). Oxidation of phenolic compounds, as a frequent phenomenon stimulated by infection, leads to the formation of chinones and free radicals which may block enzymes, the main weapon of pathogens (Appel 1993; Patil and Dimond 1967). Additionally, oxidized chinones can be directly engaged in stopping the development of pathogens (Urs and Dunleavy 1975).
Since oats can be harvested with various drum speeds, it is important for crop producers to know how the speed might affect the infection of the grain by fungal pathogens.
The objective of this research was to determine the susceptibility of oat to F. culmorum by using selected physiological parameters after varying dynamic loading procedures during harvest.
Materials and methods
Plant material
The plant material used included seeds of naked oat cultivars: ‘Abel’ (Selgen a.s., Czech Republic), ‘Avenuda’ (Selgen a.s.u. Prestic, Czech Republic), ‘Bullion’ (IGER, U.K.), ‘Cacko’ (HR Strzelce sp. z o.o., Poland), ‘Izak’ (Selgen a.s., Czech Republic), ‘Pikant’ (Toft Planteforaedling, Roslev, Denmark), ‘Polar’ (HR Strzelce sp. z o.o., Poland), ‘Saul’ (Selgen a.s., Czech Republic), as well as seeds of husked oat cultivars: ‘Cwał’ (Danko HR sp. z o.o., Poland) and ‘Stoper’ (HR Strzelce sp. z o.o., Poland). In the year 2009 the material was multiplied in a field experiment conducted at the Experiment Station of the Department of Plant Breeding and Seed Science located in Prusy near Krakow. The harvest was done manually when the moisture level of the seeds averaged 15%. After cutting off the panicles, the threshing was done using a laboratory threshing machine, the gear of which enabled regulation of the rotary speed to set it at 1.6 m s−1 (low speed—LS) or 2.4 m s−1 (high speed—HS).
Microdamage measurement
The evaluation of microdamage to seeds of naked oat cultivars was done on the basis of computer image analysis using the Multiscan 12.07 program on 50 seeds with four replications, after dyeing starch with Lugol’s solution (2%) (Zieliński et al. 2007). The total projected area of seeds and the projected area of seed microdamage were determined.
Evaluation of vigor
This was done on the basis of the percentage of normally developed seedlings over a period of 10 days in Szmal’s germinators on 50-seed samples with three replications.
Plate assay for resistance
Inoculation of semi-germinated seeds was performed with IPO348-01, an isolate of F. culmorum (Plant Breeding Institute, Wageningen, the Netherlands) cultured over a period of 7 days in petri dishes on PDA medium (potato dextrose agar—Sigma) at a temperature of 22°C without access to light, in a microbiological incubator (B 6060—Heraeus, USA). The seeds were surface disinfected for 15 min with 20% Domestos solution (commercial bleach, with sodium hypochlorite as the active ingredient). They were then washed three times in sterile water and placed on blotting paper for 24 h for germination. After that time, the semi-germinated seeds were transferred onto PDA medium discs (ø 4 mm) overgrown with F. culmorum mycelium. Seeds placed on medium discs free of the pathogen served as the control. The assay for resistance was carried out in an air-conditioned chamber over a period of 7 days at 22°C / 20°C, with 130 μE·m−2·s−1 lighting, 12 hL/12 hD photoperiod and 100% r.h. To determine the effect of infection on seedling development, direct assessment using a ranking scale (Chełkowski and Mańka 1983) and determination of the weight of leaves and roots of inoculated and control seedlings in three replications were done. The reduction in the weight of these organs was determined in accordance with the following formula:
where R is the reduction in the fresh weight (f.wt.) caused by inoculation, MI is the f.wt. of the inoculated combination, and MK is the f.wt. of the control combination.
Biochemical analyses
The chlorophyll a and b and carotenoid contents were determined spectrophotometrically using the modified method developed by Lichtenthaler and Wellburn (1983). The total soluble sugar content was determined according to Dubois et al. (1956). The phenolic compound content was determined by the method developed by Folin and Ciocalteu (Singleton and Rossi 1965). All the spectrophotometric determinations were done in three replications, using the Lambda Bio 20 spectrophotometer (Perkin Elmer, USA) with plant tissue obtained from seedlings after a period of 7 days when the plate assay for resistance was completed.
Statistical analysis
For the examined parameters, two- and three-factor variance analyses using the independent system were done. The distinguished sources of variability were tested using the fixed model. The results of the observations, expressed as percentages, were transformed into angle values according to the formula \( y = \arcsin \sqrt {x} \). To assess the share of the distinguished sources of variability in the total variability of the examined characteristics, the components of variance were estimated (Steel and Torrie 1980) and their percentages were given. The evaluation of the correlations between characteristics was performed on the basis of the Pearson linear correlation coefficient.
Results and discussion
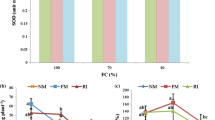
The results of the vigor, blight score, and seedling f.wt. reduction presented in Table 1 show that the symptoms of the disease caused by the pathogen were considerably more serious on the roots of the seedlings than on the leaves, irrespective of the mechanical loading to which the oat seeds were subjected during the threshing. When assessing the infection by Fusarium, a significant interaction of cultivar and rotational speed was observed. Less intense symptoms of the disease, both on leaves and on roots, were found in husked Stoper and Cwał, which at the same time were characterized by the highest vigor parameters. The greatest sensitivity to an increase in the rotational speed of the threshing drum was found for the cultivar Pikant, in which a significant increase in the leaves and roots infection intensity was observed. The values obtained for the weight of seedlings infected by Fusarium indicate that the change in the threshing speed results in an increase in the susceptibility of oat cultivars to Fusarium seedling blight. In naked cultivars, the weight of leaves and roots was decreased by 20% and 10%, respectively, as compared with husked cultivars; however, this was only for seed samples for which the LS of the threshing drum was used. In the case of the husked form, the results of evaluation using the rating scale (no visual symptoms of the disease on seedling leaves) as well as seedling weight values may suggest their greater resistance to F. culmorum. However, when HS was used, the differences in the average reduction in the weight of leaves and roots between husked and naked cultivars were not significant. In spite of the lack of symptoms of infection in husked oat seedlings, this is an indication of certain development disturbances caused by inoculation with Fusarium of seeds threshed with HS. It is worth noting that the husked cultivar Stoper, in both variants of mechanical loading, was the only one which showed the smallest decrease in the seedling weight. The values obtained for the weight of seedling roots, which decreased more than twice as much as the weight of leaves, confirm the results of evaluation using the rating scale, showing the occurrence of intense symptoms of the disease both in husked cultivars and in naked cultivars. As Grey and Mathre (1988), as well as Wojciechowski et al. (1997) show, the destruction caused by Fusarium seedling blight is much greater in the case of the root system, and that is why the root infection score is considered more reliable than the leaf infection score. In consequence of the root system damage, physiological processes—those connected with uptake and transport of water and mineral salts, as well as distribution of assimilates—become disrupted, which some time later may have a negative effect on the development of plants.
The analysis of variance revealed that inoculation, threshing speed, and cultivar were significant for all measured biochemical parameters (Table 2). Pigment content after inoculation in husked cultivars is lower at LS, and in naked cultivars at HS (Table 3). The content of soluble carbohydrates in leaves is similar independent of inoculation and threshing speed, whereas in roots inoculation caused a drastic increase, over 100% at LS. Also, phenolic compounds increased after inoculation for both LS and HS. Their concentration was twice as high in roots as in leaves. Gavrilova et al. (2008) shows that in traditional oat cultivars husks may, during the vegetative season, significantly limit infections caused by Fusarium, as well as unfavorable consequences of such diseases. According to Kurowski and Wysocka (2009), in comparison with husked seed, naked oat seeds may show lower susceptibility to the accumulation of spores on their surface. Many fungal pathogens colonizing plant tissues produce enzymes which decompose polysaccharides in cell walls. These sugars are a source of carbon for the mycelium, and are the signal for initiating defense reactions in plants. Sugars also limit the spread of the pathogen by isolating the infected cells, and protect the tissues against water loss (Herbers et al. 1996; Streuter et al. 1989). Soluble sugars directly and indirectly play a significant role in resistance processes. A high concentration of soluble sugars may directly limit the pathogen colonization of the cells as a result of increased osmotic potential (Farrar 1989). Fungal pathogens of plants have a definite range of tolerance of water potential necessary for optimal growth and development, and accumulation of sugars and other osmotically active substances decreases water potential in the host cells and may limit the pathogen development (Cook and Papendick 1978). This is how the resistance of grasses to a complex of pathogens causing pink snow mold was explained; additionally, the cultivars resistant to that complex of pathogens were characterized by low water content (Tronsmo 1986; Yoshida et al. 1997). Indirectly, sugars may affect plant disease resistance genes; hexoses induce the expression of many genes by hexokinase signal transduction, e.g. by activating the genes responsible for the production of peroxidase and pathogenesis-related proteins. Hexoses can also be a source of defensive compound precursors (Herbers et al. 1996; Streuter et al. 1989).
Correlations between studied biochemical parameters and root f.wt. reduction, as well as area of microdamage, were insignificant in the case of husked genotypes. Results presented in Tables 3 and 4 suggested a strong relation between the area of microdamage, vigor, roots f.wt. reduction, and selected physiological parameters (pigments, soluble carbohydrates, phenolic compounds) in the case of naked cultivars. A significant negative correlation between the area of microdamage, roots f.wt. reduction, and selected physiological parameters clearly reveals that higher contents of chlorophyll a, b, carotenoids, carbohydrates and phenolic compounds are related to lower root f.wt. reduction after infection, which means that the examined genotypes are more resistant. Therefore, the above parameters can be utilized for indirectly assessing susceptibility to F. culmorum infection for naked oat genotypes. A plate assay for resistance takes at least 14 days and requires fungi production, inoculation processes, and strictly controlled conditions, whereas measurements of the biochemical parameters described above can be conducted 1 day after seed germination and green seedling formation.
References
Agarwal, V. K., & Sinclair, J. B. (1997). Principles of seed pathology (2nd ed.). Boca Raton, FL, USA: CRC Press Inc. / Lewis.
Appel, H. M. (1993). Phenolics in ecological interactions: The importance of oxidation. Journal of Chemical Ecology, 19, 1521–1552.
Bechtel, D. B., Kaleikau, L. A., Gaines, R. L., & Seitz, L. M. (1985). The effects of Fusarium graminearum on wheat kernels. Cereal Chemistry, 62, 191–197.
CCA (2010). [Common catalogue of varieties of agricultural plant species.] Wspólnotowy Katalog Odmian Roślin Rolniczych. Dz. U. UE 29, 2010/C 337, A/01, vol. 53, pp. 263–271, Polish edition. Official journal of the European Union.
Chełkowski, J., & Mańka, M. (1983). The ability of Fusaria pathogenic to wheat, barley and corn to produce zearalenone. Phytopathologie Zeitung, 106, 354–359.
Cook, R. J., & Papendick, R. I. (1978). Role of water potential in microbial growth and development of plant disease with special reference to postharvest pathology. Hortscience, 13, 559–564.
Dakora, F. D. (1996). Diverse functions of isoflavonoids in legumes transcend anti-microbial definitions of phytoalexins. Physiological and Molecular Plant Pathology, 49, 1–20.
Dubois, M., Gilles, K. A., Hamilton, J. K., Rebers, P. A., & Smith, F. (1956). Colorimetric method for determination of sugars and related substances. Analytical Chemistry, 28, 350–356.
European Commission (2006a). Commission regulation of 23 February 2006 laying down the methods of sampling and analysis for the official control of the levels of mycotoxins in foodstuffs, 2006/401/EC. In: Official journal, L 70, 09/03/2006, (pp. 12–34).
European Commission (2006b). Commission recommendation of 17 August 2006 on the presence of deoxynivalenol, zearalenone, ochratoxin A, T-2 and HT-2 and fumonisins in products intended for animal feeding, 2006/576/EC. In: Official journal, L 229, 23/08/2006, (pp. 7–9).
European Commission (2006c). Commission Regulation of 19 December 2006 setting maximum levels for certain contaminants in foodstuffs sets maximum levels for Fusarium toxins in certain foodstuffs, 2006/1881/EC. In: Official journal, L 364, 20/12/2006, (pp. 5–24).
European Commission (2007). Commission Regulation No. 2007/1126/EC of 28 September 2007 amending Regulation No. 2006/1881/EC setting maximum levels for certain contaminants in foodstuffs as regards Fusarium toxins in maize and maize products. In: Official journal, L 255, 29/09/2007, (pp. 14–17).
EUROSTAT (2008). Agricultural statistics. Main results—2006–2007. http://epp.eurostat.ec.europa.eu.
Farrar, J. F. (1989). Fluxes and turnover of sucrose and fructans in healthy and diseased plants. Journal of Plant Physiology, 134, 137–140.
Gavrilova, O., Gagkaeva, T., Burkin, A., Kononenko, G., & Loskutov, I. (2008). Susceptibility of oat germplasm to Fusarium infection and mycotoxin accumulation in grains. In: Proceedings of the 8th International Oat Conference (pp. 7–16). Minneapolis, MN, USA.
Grey, W., & Mathre, D. E. (1988). Evaluation of spring barley for reaction to Fusarium seedling blight and root rot. Canadian Journal of Plant Science, 68, 23–30.
GUS (2009). Statistical yearbook of agriculture. http://www.stat.gov.pl.
Herbers, K., Meuwley, P., Frommer, W. B., Metraux, J.-P., & Sonnewald, U. (1996). Systemic acquired resistance mediated by ecotopic expression of invertase: Possible hexose sensing in the secretory pathway. The Plant Cell, 8, 793–803.
Hussein, H. S., & Brasel, J. M. (2001). Toxicity, metabolism and impact of mycotoxins on humans and animals. Toxicology, 167, 101–134.
Inch, S. A., & Gilbert, J. (2003). Survival of Gibberella zeae in Fusarium-damaged wheat kernels. Plant Disease, 87, 282–287.
Jones, R. K., & Mirocha, C. J. (1999). Quality parameters in small grains from Minnesota affected by Fusarium head blight. Plant Disease, 83, 506–511.
Kiecana, I., Mielniczuk, E., Kaczmarek, Z., Kostecki, M., & Goliński, P. (2002). Scab response and Moniliformin accumulation in kernels of oat genotypes inoculated with Fusarium avenaceum in Poland. European Journal of Plant Pathology, 108, 245–251.
Kiecana, I., Mielniczuk, E., Perkowski, J., & Goliński, P. (2005). Porażenie wiech przez Fusarium poae (Peck) Wollenw. oraz zawartość mikotoksyn w ziarnie owsa. [Infection of panicles with Fusarium poae (Peck) Wollenw. and mycotoxin content in oat grain.]. Acta Agrobotanica, 58, 91–102.
Kurowski, T. P., & Wysocka, U. (2009). Fungal communities colonizing grain of hulled and naked oat grown under organic farming system. Phytopathologia, 54, 53–59.
Lattanzio, V., Lattanzio, V. M. T., & Cardinali, A. (2006). Role of phenolics in the resistance mechanisms of plants against fungal pathogens and insects. In F. Imperato (Ed.), Phytochemistry: Advances in research (pp. 23–67). Kerala, India: Research Signpost.
Lichtenthaler, H. K., & Wellburn, A. R. (1983). Determinations of total carotenoids and chlorophylls a and b of leaf extracts in different solvents. Biochemical Society Transactions, 11, 591–592.
LORR (2010). Lista Odmian Roślin Rolniczych. [Polish National List of Agricultural Plant Varieties] COBORU [The Research Centre for Cultivar Testing] Slupia Wielka, Poland pp. 68–69.
Magan, N., Hope, R., Colleate, A., & Baxter, E. S. (2002). Relationship between growth and mycotoxin production by Fusarium species, biocides and environment. European Journal of Plant Pathology, 108, 685–690.
Mauch-Mani, B., & Métraux, J.-P. (1998). Salicylic acid and systemic acquired resistance to pathogen attack. Annals of Botany, 82, 535–540.
McKeehen, J. D., Busch, R. H., & Fulcher, R. G. (1999). Evaluation of wheat (Triticum aestivum L.) phenolic acids during grain development and their contribution to Fusarium resistance. Journal of Agricultural and Food Chemistry, 47, 1476–1482.
Nicholson, R. L., & Hammerschmidt, R. (1992). Phenolic compounds and their role in disease resistance. Annual Review of Phytopathology, 30, 369–389.
Patil, S. S., & Dimond, A. E. (1967). Inhibition of Verticillium polygalacturonase by oxidation products of polyphenols. Phytopathology, 57, 492–496.
Puupponen-Pimia, R., Nohynek, L., Hartmann-Schmidlin, S., Kahkonen, M., Heinonen, M., Maatta-Riihinen, K., et al. (2005). Berry phenolics selectively inhibit the growth of intestinal pathogens. Journal of Applied Microbiology, 98, 991–1000.
Ruckenbauer, P., Buerstmayr, H., & Lemmens, M. (2001). Present strategies in resistance breeding against scab (Fusarium spp.). Euphytica, 119, 121–127.
Salas, B., Steffenson, B. J., Casper, H. H., & Prom, L. K. (1997). Fusarium species pathogenic to barley and their associated toxins. Cereal Research Communications, 25, 483–487.
Schollenberger, M., Müller, H. M., Rüfle, M., Suchy, S., Plank, S., & Drochner, W. (2006). Natural occurrence of 16 Fusarium toxins in grains and feedstuffs of plant origin from Germany. Mycopathologia, 161, 43–52.
Singleton, U. L., & Rossi, J. (1965). Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagent. American Journal of Enology and Viticulture, 16, 144.
Soroka, P. M., Cyprowski, M., & Szadkowska-Stańczyk, I. (2008). [Occupational exposure to mycotoxins in various branches of industry.]. Medycyna Pracy, 59, 333–345.
Stachel, S. E., Nester, E. W., & Zambryski, P. C. (1986). A plant cell factor induces Agrobacterium tumefaciens vir gene expression. Proceedings of the National Academy of Sciences of the USA, 83, 379–383.
Steel, R. G. D., & Torrie, J. H. (1980). Principles and procedures of statistics (pp. 355–361). New York, NY: McGraw–Hill Book Company.
Streuter, N., Moerschbacher, B. M., Fischer, Y., Noll, U., & Reisener, H. J. (1989). Fructose-2,6-bisphosphate in wheat leaves infected with stem rust. Journal of Plant Physiology, 134, 254–257.
Suchowilska, E., Wiwart, M., & Borusiewicz, A. (2007). The reaction of the selected Triticum aestivum, Triticum spelta and Triticum dicoccum genotypes to spike infection by Fusarium culmorum. Proceedings of conference “Organic farming 2007” (Prague, Czech Republic), pp. 172–174.
TeKrony, D. M., & Egli, D. B. (1991). Relationship of seed vigor to crop yield: A review. Crop Science, 31, 816–822.
Torp, M., & Nirenberg, H. I. (2004). Fusarium langsethiae sp. nov. on cereals in Europe. International Journal of Food Microbiology, 95, 247–256.
Trojanowska, K. (2002). Zagrożenia ze strony mikroflory występującej na ziarnie zbożowym i jego przetworach. [Hazards caused by microflora occurring on cereal grain and its derivative products.]. Przegląd Zbożowo-Młynarski [Cereals and Milling Review], 2, 9–12. in Polish.
Tronsmo, A. M. (1986). Host water potentials may restrict development of snow mold fungi in low temperature hardened grasses. Physiologia Plantarum, 68, 175–179.
Urs, N. V. R., & Dunleavy, J. M. (1975). Enhancement of bactericidal activity of a peroxidase system by phenolic compounds. Phytopathology, 65, 686–690.
Wiśniewska, H., & Kowalczyk, K. (2005). Resistance of cultivars and breeding lines of spring wheat to Fusarium culmorum and powdery mildew. Journal of Applied Genetics, 46, 35–40.
Wojciechowski, S., Chełkowski, J., Ponitka, A., & Ślusarkiewicz-Jarzina, A. (1997). Evaluation of spring and winter wheat reaction to Fusarium culmorum and Fusarium avenaceum. Journal of Phytopathology, 145, 99–103.
Yoshida, M., Abe, J., Moriyama, M., Shimokawa, S., & Nakamura, Y. (1997). Seasonal changes in the physical state of crown water associated with freezing tolerance in winter wheat. Physiologia Plantarum, 99, 363–370.
Zieliński, A., Moś, M., & Wójtowicz, T. (2007). Effect of grain moisture content and threshing cylinder speed on mechanical damage in naked oat cultivars. Electronic Journal of Polish Agricultural Universities (EJPAU), 10(4), #12, http://www.ejpau.media.pl/volume10/issue4/art-12.html.
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Warzecha, T., Zieliński, A., Skrzypek, E. et al. Effect of mechanical damage on vigor, physiological parameters, and susceptibility of oat (Avena sativa) to Fusarium culmorum infection. Phytoparasitica 40, 29–36 (2012). https://doi.org/10.1007/s12600-011-0196-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12600-011-0196-y