Abstract
White spot syndrome virus (WSSV) has been one of the most devastating pathogens affecting the global shrimp industry since its initial outbreaks in Asia in the early 1990s. In this study, we recovered 13 complete metagenome-assembled genomes (MAGs) of Japanese WSSV isolates and 30 draft WSSV MAGs recovered from publicly available sequencing data, to investigate the genomic evolution of WSSV. Phylogenetic analysis revealed two major phylotypes, designated phylotypes I and II. Bayesian divergence time estimates placed the divergence time of the two phylotypes between 1970 and the early 1980s, with an estimated substitution rate of 1.1 × 10–5 substitutions per site per year, implying the existence of pre-pandemic genetic diversity of WSSV in Asia. Based on this scenario, phylotype I was responsible for the 1990s pandemic and spread worldwide, whereas phylotype II was localized in Asia and infiltrated Australia. Two cross-phylotype recombinant lineages were identified, which demonstrate the role of genomic recombination in generating the genetic diversity of WSSV. These results provide important insights into the evolution of WSSV and may help uncover the ultimate origins of this devastating pathogen.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Penaeid shrimp aquaculture has experienced rapid growth since the latter half of the twentieth century, but it has been constantly threatened by various infections by bacteria, fungi, parasites, and viruses (Lightner et al. 2012; Momoyama and Muroga 2005; Stentiford et al. 2012). One particularly lethal virus for penaeid shrimps is white spot syndrome virus (WSSV), which is a large, double-stranded DNA virus that infects a wide range of decapod crustaceans (H.-C. Wang et al. 2019). WSSV has a bacilliform, enveloped virion which contains more than 40 different structural proteins (H.-C. Wang et al. 2019) and replicates in the cell nuclei of tissues of ectodermal and mesodermal origins (Chou et al. 1995). The WSSV genome is a circular, double-stranded DNA ranging 280–315 kbp in length, encoding 150 to 180 predicted protein-coding genes (Li et al. 2017; Tsai et al. 2004; van Hulten et al. 2001; Yang et al. 2001).
The earliest known outbreak of WSSV occurred in Fujian Province, China in July 1992 (Cai and Su 1993; Lo et al. 2005; Su et al. 1995), and it has since spread rapidly to other regions through human activities, affecting all shrimp-farming regions in the world (Lightner et al. 2012; Oakey and Smith 2018; Onihary et al. 2021; Tang et al. 2013). In Japan, the first recorded incidence of WSSV occurred in Hiroshima Prefecture in 1993 via the introduction of infected kuruma shrimp Penaeus japonicus seeds (Inouye et al. 1994; Momoyama et al. 1994; Nakano et al. 1994). WSSV rapidly spread to other regions in Japan, causing massive damage to shrimp production. By the end of the 1990s, WSSV established itself in wild Japanese crustacean populations (Maeda et al. 1998; Okamoto and Suzuki 1999; Fukuzumi and Chikushi 2003; Izumikawa 2013) and still continues to affect the kuruma shrimp industry.
Understanding the origins and evolution of infectious diseases has various socioeconomic implications such as the development of biosecurity policies. Genomic information is essential for understanding the pathogenesis, epidemiology, and evolution of a virus. Dozens of WSSV genomes have been published from various parts of the world (Cruz-Flores et al. 2020; Han et al. 2017; Kooloth Valappil et al. 2021; Oakey and Smith 2018; Parrilla-Taylor et al. 2018; Rodriguez-Anaya et al. 2016; van Hulten et al. 2001; Yang et al. 2001), but there have been few attempts to decipher the evolution of WSSV at the genome level. In this study, we aimed to dissect the genomic evolution of WSSV by leveraging existing genomic assemblies and newly generated WSSV genome assemblies through de novo sequencing and exploration of publicly available datasets.
Materials and methods
WSSV specimens
We sequenced a total of 12 specimens collected from seven Prefectures across Japan (Table 1). JP01 was derived from diseased P. japonicus in Miyazaki Prefecture between 1995 and 2000 and had been maintained as an infectious virus in the Laboratory of Genome Science, Tokyo University of Marine Science and Technology, Tokyo. JP02 was derived from diseased P. japonicus in Yamaguchi Prefecture between 1995 and 2000 and had been maintained as an infectious virus at the National Fisheries University, Yamaguchi Prefecture. JP03 and JP04 were identified from the shotgun sequencing data of Metapenaeopsis lamellata and Trachysalambria curvirostris samples, respectively, which were accidentally found to contain WSSV sequences. Both samples had been fixed with ethanol and therefore infectious viruses could not be recovered. S14, E1, E2, and 1–4 were recovered from diagnostic samples submitted to the Okinawa Prefectural Government and tested positive for WSSV by PCR. Sample 79 was collected during an epidemiological survey conducted by the Deepsea Water Research Center, Kumejima Island, Okinawa Prefecture. P. japonicus samples infected with isolates 0722–1 and Miyako2021 were provided by commercial farmers in Okinawa Island and Miyako Island, respectively. Pc2020 was derived from a naturally infected red swamp crayfish Procambarus clarkii originating from Chiba Prefecture. All Japanese WSSV genomes were regarded as MAGs because the sample preparation procedures did not involve the purification of virions.
DNA extraction
For JP01 and JP02 (Fig. 1), genomic DNA was extracted from shrimp homogenate prepared as a viral inoculum. We first concentrated the homogenate using Amicon Ultracells (Merck) and extracted DNA by phenol–chloroform-isoamyl alcohol extraction. The DNA was further concentrated using Amicon Ultracells. For JP03 and JP04, genomic DNA was extracted from the muscle tissue of the ethanol-preserved specimens by phenol–chloroform-isoamyl alcohol extraction. We used a swimming leg from specimen 79 for DNA extraction by phenol–chloroform-isoamyl alcohol extraction. For Pc2020, we extracted genomic DNA from the gills of a dead crayfish using a MagAttract HMW DNA Kit (Qiagen).
Genome diagram of white spot syndrome virus JP02. Outer track: protein-coding genes and their transcriptional orientations (blue). Middle track: GC skew of 100 bp sliding windows with 10-bp increments (positive: emerald, negative: purple). Inner track: deviation of GC contents from the average, 100 bp sliding windows with 10-bp increments
Multiple displacement amplification
Before Oxford Nanopore Technologies (ONT) library preparation, the genomic DNA of JP01, JP02, 79, S14, E1, E2, and 1–4 was amplified by multiple displacement amplification (MDA). Amplification was performed using the REPLI-g Mini Kit (150023, Qiagen), and the amplicons were purified using Agencourt AMPure XP beads (Beckman Coulter) and quantified using a Qubit dsDNA BR Assay kit (Thermo Fischer Scientific). Approximately 1.5 µg of the amplicon was then digested with T7 Endonuclease I (M0302, New England Biolabs), purified again using Agencourt AMPure XP beads and quantified using the Qubit dsDNA BR Assay kit. Approximately 750 ng of the purified DNA was used for ONT library preparation.
Sequencing
Paired-end libraries for Illumina sequencing were prepared using the Nextera XT DNA Library Preparation Kit (Illumina) and were sequenced using the MiSeq Reagent kit v2 (2 × 150 cycle) or MiSeq Reagent Kit v3 (2 × 300 cycle). ONT libraries were prepared using the Ligation Sequencing Kit (SQK-LSK109, Oxford Nanopore Technologies) and NEBNext Companion Module for Oxford Nanopore Technologies Ligation Sequencing (E7180, New England Biolabs). The enzymatic reaction steps were extended to twice the suggested duration. The libraries were sequenced on R9.4.1 flow cells on a MinION or a GridION platform. The fast5 files were base-called using Guppy v. 5.0.11 in super-accuracy mode.
De novo assembly and assembly curation
The ONT reads were filtered by length using SeqKit (Shen et al. 2016) and aligned to the CN01 genome (RefSeq Accession no. NC_003225.3) with Minimap2 (Li 2018, p. 2). The mapped reads were extracted and de novo assembled using Flye v. 2.9 (Kolmogorov et al. 2019) or Canu v. 2.2 (Koren et al. 2017). Canu was particularly useful for removing chimeric reads arising from MDA, which improved the assembly quality. The resulting assemblies were polished with POLCA v. 3.4.2 (Zimin and Salzberg 2020) using Illumina reads trimmed using Fastp v. 0.20.1 (Chen et al. 2018). JP01 was found to be a mixture of two closely related haplotypes, and therefore two genotypes were resolved using FreeBayes v. 1.3.6 (Garrison and Marth 2012) and WhatsHap v. 1.6 (Patterson et al. 2015). Haplotype-specific reads were reassembled using Canu v. 2.2 and polished with POLCA v. 3.4.2.
WSSV genome reconstruction from publicly available sequence data
The Sequence Read Archive (SRA) entries that contained signatures of WSSV were identified using BigQuery (last accessed December 2022), and the corresponding entries were downloaded from the DDBJ/NCBI/ENA database. The downloaded entries included the genomes of WSSV and host crustaceans, shotgun metagenomes, and transcriptomes. The sampling dates were determined based on the corresponding BioSample metadata and literature. The reads were trimmed using Fastp v. 0.20.1, and the trimmed reads were mapped to the CN01 genome with Minimap2. The mapped reads were extracted and depth-normalized using BBnorm (Bushnell et al. 2017). The normalized reads were assembled using SPAdes v. 3.15.5 (Nurk et al. 2013), and the contigs were scaffolded by Chromosomer v. 0.1.4a (Tamazian et al. 2016). The scaffolds were polished using Pilon v. 1.24 (Walker et al. 2014) and were manually curated.
Annotation
Open reading frames (ORFs) were predicted using a custom script (https://github.com/satoshikawato/bio_small_scripts/blob/main/orfind.py), and the genomic coordinates were adjusted to the start codon of the wsv001 (collagen-like protein) gene. We then queried the predicted protein sequences by BLASTP against the WSSV proteome, and the annotation files were produced with custom scripts. The final DDBJ flat files were produced using GFF3-to-DDBJ v0.4.0 (https://github.com/yamaton/gff3toddbj) and FFconv (https://www.ddbj.nig.ac.jp/ddbj/mss-tool.html). The WSSV MAGs generated in this study are available as Online Resource 1 (Online_Resource_1 _MAGs.zip).
Phylogenetic analysis and divergence time estimation
Publicly available WSSV genomes were downloaded from the NCBI database (last accessed February 2023). Protein-coding genes in some of the publicly available WSSV genomes were highly fragmented. The presence of extensive frameshift mutations was likely due to the use of particular sequencing platforms (IonTorrent PGM or ONT), which have been known to struggle with homopolymers. The fragmentation of core genes such as the major capsid protein (wsv360) and DNA polymerase (wsv514) (Kawato et al. 2019) strongly suggested that the majority of mutations in these assemblies did not originate biologically, and therefore these assemblies were excluded from the analysis.
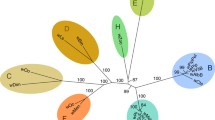
We used Snippy (https://github.com/tseemann/snippy) to construct a core genome alignment of 61 WSSV genomes by mapping simulated short reads generated from each assembly to the CN01 genome (clean.full.aln in Online Resource 2, Online_Resource_2_whole_genome_alignments.zip). A maximum-likelihood phylogenetic tree was built (clean.full.aln.treefile in Online Resource 2, Online_Resource_2_whole_genome_alignments.zip) using IQ-TREE 2.2.2.3 (Minh et al. 2020), which revealed two major phylotypes, I and II (Fig. 2). Possible recombinants (lineages 1601 and Qingdao; see “Identification of recombination breakpoints” Section) and genomes with unknown sampling dates were excluded from the downstream analyses.
Maximum-likelihood phylogenetic tree of WSSV genomes. Japanese WSSV MAGs sequenced in this study are highlighted with a filled circle (●); WSSV MAGs recovered from publicly available high-throughput sequencing data are highlighted with a triangle (▲). Sampling years (if known) are indicated after an @ sign, followed by the NCBI accession numbers, if they exist. The sampling years for JP01A, JP01B, and JP02, which range from 1995 to 2000, due to uncertainty, were arbitrarily indicated as 1998. Numbers beside nodes indicate the SH-aLRT support (%)/ultrafast bootstrap support (%). Phylotypes I and II are shaded with light blue and pink, respectively. Cross-phylotype recombinant lineages 1601 and Qingdao are shaded with orange and light green, respectively
We then built a maximum likelihood phylogenetic tree with IQ-TREE v. 2.2.2.3 from the recombinant-free, dated whole genome alignment containing 48 sequences (clean2.full.aln.treefile and clean2.full.aln in Online Resource 2, Online_Resource_2_whole_genome_alignments.zip) and inspected the tree with Tempest v. 1.5.3 (Rambaut et al. 2016). Rooting, based on heuristic residual mean squares, placed the root between phylotypes I and II (R2 = 0.5056; function: heuristic residual mean squared). The root position was independently supported by the minimum variance method implemented in FastRoot v. 1.5 (Mai et al. 2017).
Bayesian phylogenetic analysis was performed with BEAST v. 2.7.4 (Bouckaert et al. 2014). Preliminary analysis using 48 genomes placed WSSV-TH or WSSV-TW as ancestral strains, but this was considered implausible since it drastically deviated from the most likely root position found in the Tempest and FastRoot analyses. Tempest analysis found WSSV-TH and WSSV-TW had large root-to-tip genetic distance residuals, which indicated that the two genomes contained disproportionately large numbers of unique nucleotide variations relative to their sampling dates. We suspect that the observed large residuals were sequencing artifacts arising from the Sanger-based shotgun sequencing strategy. Therefore, we excluded from the dataset WSSV-TW, WSSV-TH, and six other sequences with the absolute values of root-to-tip genetic distance residuals larger than 1.30 × 10–4 (WSSV-LS, POMZ4, POMZ1, Pc2020, PG1, WSSV-Peru).
The final recombinant-free whole genome alignment contained 40 genomes (clean3.full.aln in Online Resource 2, Online_Resource_2_whole_genome_alignments.zip). The maximum-likelihood phylogenetic tree built with IQ-TREE v. 2.2.2.3 (clean3.full.aln.treefile in Online Resource 2, Online_Resource_2_whole_genome_alignments.zip) yielded a Tempest R2 value of 0.8049 (function: heuristic residual mean squared). Bayesian divergence time estimation was performed using BEAST v. 2.7.4 (Bouckaert et al. 2014). To account for the ambiguity in the sampling dates of JP01A, JP01B, and JP02, we set a prior distribution of the three tips as a normal distribution (σ = 1.0) with the mean at 1998. Fifty million iterations were performed, which were sampled every 10,000 steps after a 10% burn-in. We used Tracer v. 1.7.2 (Rambaut et al. 2018) to monitor the progress of the run and to ensure that the effective sampling sizes of all parameters were larger than 200, except the posterior of run “coalescent constant, GTR,” which was 175.3. Three population models (constant, exponential, and Bayesian skyline) and two substitution models (HKY and GTR) were used to assess the impact of model selection. A maximum clade credibility tree was generated for each run with TreeAnnotator (Drummond and Rambaut 2007), which was visualized with FigTree v. 1.4.4 (http://tree.bio.ed.ac.uk/software/figtree/). We arbitrarily selected the estimate under the HKY model assuming a constant population size for presentation in Fig. 3, as all estimates converged on similar tree topologies and estimated divergence dates. BEAST XML files and resulting trees are available as Online Resource 3 (Online_Resource_3_BEAST_trees.zip).
Identification of recombination breakpoints
Possible recombinants (lineages 1601 and Qingdao) were detected by inspecting neighbor-net (Bryant and Moulton 2004) phylogenetic networks constructed from the whole genome alignment of 61 WSSV genomes using SplitsTree4 v. 4.19.0 (Huson 1998). We used RDP4 v. 4.101 (Martin et al. 2015) to analyze the recombination breakpoints of the cross-phylotype lineages. Subsampling of WSSV genomes was necessary to complete RDP4 analysis. Recombination sites of lineage 1601 were identified by analyzing the following six genomes: phylotype I, CN01 and PC; phylotype II, CN03 and WSSV-AU; lineage 1601, 1601 and GCF7. Recombination sites of lineage Qingdao were identified by analyzing the following six genomes: phylotype I, CN01 and PC; phylotype II, CN03 and WSSV-AU; lineage Qingdao, Qingdao2019 and Qingdao2020. Recombination breakpoints were plotted against the CN01 genome, and corresponding regions of genome alignment were extracted with a custom script (https://github.com/satoshikawato/bio_small_scripts/blob/main/crop_alignment.py). Maximum-likelihood phylogenetic trees based on the subgenomic alignments were constructed with IQ-TREE 2.2.2.3. Subgenomic alignments and maximum-likelihood phylogenetic trees are available as Online Resource 4 (Online_Resource_4_subgenomic_alignments.zip).
Results
Sequencing and assembly of Japanese WSSV genomes
We sequenced a total of 12 specimens from farmed and wild crustaceans collected in Japan (Table 1), resulting in 0.35 to 3.74 Gb of Illumina reads per specimen and 69 Mb to 14.9 Gb of ONT reads per specimen (Online Resource, Table S1). A total of 13 genomes were recovered, ranging in size from 288,190 bp (Miyako2021) to 311,562 bp (JP02), with the number of protein-coding genes ranging from 160–180 (Table 2).
JP01A (299,976 bp) and JP01B (293,923 bp) originate from diseased P. japonicus in Miyazaki Prefecture between 1995 and 2000. Haplotype phasing resolved two closely related genotypes present in the viral inoculum. Isolate JP02 (Fig. 1), originating from Yamaguchi Prefecture between 1995 and 2000, had a genome size of 311,562 bp — the largest complete WSSV genome sequenced to date and approaching the estimated genome size of TH-96-II (312 kbp) (Marks et al. 2005). The coding regions were overall very similar to those of CN01 (309,286 bp; NCBI RefSeq Accession no. NC_003225) (Li et al. 2017). The difference in genome size between JP02 and CN01 was mainly due to variations in repeat sequence lengths, including homologous repeats and variable number tandem repeats in ORFs.
Isolates JP03 and JP04, obtained from Mikawa Bay in Aichi Prefecture and Lake Hamana in Shizuoka Prefecture, respectively, shared (i) the translocation of wsv486 gene (Online Resource, Fig. S1), (ii) a 3381-bp deletion around the ORF14/15 region (Online Resource, Fig. S2), and (iii) a 7057-bp deletion around the ORF23/24 region (Online Resource, Fig. S3). The translocation of wsv486 has previously been reported in CN03 (GenBank Accession no. KT995471.1) (Li et al. 2017). The similarity of WSSV genomes from Mikawa Bay and Lake Hamana suggests that the two isolates share a common origin, which is consistent with the fact that wild P. japonicus populations in the two prefectures share a common spawning ground, the Sea of Enshu (Suitoh et al. 2014). Isolate 79 was identified during an epidemiological survey of WSSV in P. japonicus, while Pc2020 was identified in a wild Procambarus clarkii from Chiba Prefecture. Collectively, WSSV genomes derived from wild crustaceans provide further evidence that WSSV has become established in the wild crustacean populations in Japan.
Isolates from Okinawa (S14, E1, E2, and 0722–1) and Miyako (1–4 and Miyako2021) islands shared a 2771-bp deletion, found in wsv237, wsv238, and wsv242, suggesting a shared ancestry of WSSV isolates derived from different islands in Okinawa Prefecture (Online Resource, Fig. S4). This deletion is also shared by Mexican (Parrilla-Taylor et al. 2018) and Ecuadorian (WSSV-EC-15098; MH090824.1) (Restrepo et al. 2018) isolates. ORF14/15 and ORF23/24, in contrast, were variable between the Okinawa and Miyako isolates (Online Resource, Figs. S2 and S3), and therefore appear to have independently undergone genome shrinkage around these regions. Multiple sequence alignments of partial genome sequences covering the genomic deletions are available as a supplementary file of the manuscript (Online Resource 5, Online_Resource_5_genomic_deletions.zip).
Draft WSSV genomes recovered from publicly available sequencing data
A total of 30 WSSV draft genomes were recovered from publicly available sequencing datasets (Table 3). Nineteen MAGs were recovered from samples derived from China, followed by four MAGs from India and two each from Peru and Mexico. We successfully recovered multiple draft MAGs from not only metagenome but transcriptome datasets, likely due to the pervasive bi-stranded transcription that takes place throughout the WSSV genome; however, uneven coverages across the genome required in silico normalization prior to de novo assembly. The accuracy of the assembled draft genomes was confirmed by the fact that genome sequences CN_95_DFPE (genome; MN840357.1) (Cruz-Flores et al. 2020) and UAZ 00-173B (transcriptome) (Millard et al. 2021), which originated from the same isolate, were almost identical to each other. The length of the UAZ 00-173B assembly (310,944 bp) was longer than that of CN_95_DFPE (305,094 bp), which likely reflects differences in assembly methods. We discovered WSSV sequences in non-crustacean datasets, such as a human metagenome (EU129; SRR14509867) (Leung et al. 2022), a warthog genome (MES-753; SRR17256726), and a rhesus macaque feces metagenome (RNA-D9-1; SRR9701085) (Li et al. 2019). Some of these WSSV reads may represent contaminants, but they still provide valuable information about the evolution of WSSV. Overall, these results indicate that WSSV draft genomes can be recovered from various high-throughput sequencing data. The WSSV MAGs generated in this study are available as Online Resource 1 (Online_Resource_1 _MAGs.zip).
Two major phylotypes
The maximum-likelihood phylogenomic tree revealed two phylotypes (Fig. 2). Rooting between the two phylotypes was supported by heuristic residual mean squares, implemented in Tempest, and the minimum variance method implemented in FastRoot. We identified two cross-phylotype recombinant lineages (1601, shaded in orange in Fig. 2; Qingdao, light green), which will be discussed later (see “Emergence of recombinant strains”).
Divergence time estimates
We next estimated the divergence time of the two phylotypes by Bayesian phylogenetic analysis. For the analysis, we removed from the dataset (i) sequences with unknown sampling dates, (ii) cross-lineage recombinants, and (iii) WSSV genomes that had exceptionally large or small root-to-tip divergences in the maximum-likelihood phylogenetic tree. The Bayesian phylogenetic analyses using 40 WSSV genomes yielded 95% confidence intervals for the divergence between phylotypes I and II occurring around 1973–1981 (median in Fig. 3: 1977), suggesting that the two phylotypes had diverged prior to the 1990s pandemic. The estimate was robust to the choice of priors including population dynamics (coalescent constant, coalescent exponential, and Bayesian skyline) and substitution models (HKY and GTR).
Bayesian divergence time estimate of WSSV. A total of 40 WSSV genomes were used in the analysis. Cross-phylotype recombinants (lineages 1601 and Qingdao), genomes with unknown isolation dates, and genomes with extremely high or low mutation rates (as inferred from Tempest analysis) were excluded from the analysis. Phylotypes I and II are shaded with light blue and pink, respectively. Japanese WSSV MAGs sequenced in this study are highlighted with a filled circle (●); WSSV MAGs recovered from publicly available high-throughput sequencing data are highlighted with a triangle (▲). Sampling years are indicated after an @ sign. The sampling years for JP01A, JP01B, and JP02, which range from 1995 to 2000 due to uncertainty, were arbitrarily indicated as 1998. Blue bars at each node indicate the 95% confidence intervals of the divergence dates
The Bayesian phylogenetic analyses converged to median estimated mutation rates of 1.11 × 10–5 to 1.15 × 10–5 substitutions/site/year (Online Resource, Table S2). While this may be higher than estimated mutation rates of other dsDNA viruses (Brennan Greg et al. 2022; Firth et al. 2010; Guellil et al. 2022; Morga et al. 2021), it can be reasonably derived from an experimentally derived mutation rate of a baculovirus (1 × 10−7 mutations/site/replication) (Boezen et al. 2022) assuming 100 replications/year, both of which are reasonable assumptions considering the biology of WSSV. Although we should be cautious with interpreting this value since estimated virus mutation rates vary substantially between short and long terms (Duchêne et al. 2014; Ghafari et al. 2021), we believe that our estimate of the WSSV mutation rate is reasonable for analyzing the genomic evolution of WSSV in the past few decades.
Phylotype I was responsible for the 1990s pandemic
Phylotype I contained all strains isolated during the 1990s pandemic that originated in Asia. All Japanese WSSV genomes belonged to clade I. The lack of resolution among the phylotype I members in the tree is consistent with the rapid spread of the virus, which did not allow enough time for WSSV genomes to accumulate mutations to enable tracking its geographic spread.
Marks et al. (2004) proposed the existence of an ancestral strain with the largest genome, which subsequently shed redundant segments to become smaller genomes (Marks et al. 2004). Isolate TH-96-II, with an estimated genome size of 312 kbp, is believed to be a close representation of the ancestral WSSV genome, although the whole genome of this isolate has not been published (Marks et al. 2005). JP02 and CN01, both belonging to phylotype I, retained the intact ORF13/14 and ORF24/25 regions and are therefore likely to closely resemble TH-96-II (Online Resource, Figs. S2 and S3). The short branch lengths of JP02 and CN01 also suggest that they are close representations of the common ancestor(s) of phylotype I (Fig. 1).
A small branch which forms a sister clade to all other phylotype I members was found (Figs. 2 and 3). The estimated divergence time between this clade and the other members, including CN01 and JP02, predates the 1990s, suggesting that the two lineages had diverged before the pandemic. This might represent another line of evidence that WSSV had pre-pandemic genetic diversity. Alternatively, it is possible that there have been small cross-phylotype recombination events that could not be detected by our analysis, but which contributed phylotype II-like phylogenetic signals to these sequences.
Taken together, these findings suggest that phylotype I was responsible for the 1990s pandemic and that JP02, CN01, and TH-96-II represent the ancestral genotype(s) of phylotype I that emerged in the pandemic epicenter in the 1990s in East Asia (Zwart et al. 2010).
Phylotype II is localized in Asia
Phylotype II contained isolates originating from China (e.g., CN03 and CN04) (Li et al. 2017), Australia (WSSV-AU) (Oakey and Smith 2018), India (e.g., CWG3, DBA1182, and WSSV-LS) (Kooloth Valappil et al. 2021), and Bangladesh (SS304) and is characterized by smaller genome sizes, which probably reflect ancestral deletions. WSSV-AU clusters together with CN03 and other phylotype II members from China, confirming its Asian origin (Knibb et al. 2018).
Phylotype II is characterized by a 5949-bp deletion in the variable region ORF13/14 (relative to TH-96-II; Online Resource, Fig. S2). Based on this specific genomic deletion, it is likely that WSSV genotypes from Madagascar and Saudi Arabia belong to phylotype II (Onihary et al. 2021; Tang et al. 2013), although it is possible that other parts of their genomes have originated from other phylotype(s) due to recombination. ORF23/24 is more variable, with Indian isolates (CWG3, DBA1182, and WSSV-LS) sharing a 11,273-bp deletion (relative to CN01; Online Resource, Fig. S3), while Chinese (CN03 and CN04) and Bangladeshi (SS304) isolates share a 10,893-bp deletion (relative to CN01; Online Resource, Fig. S3). The ORF14/15 and ORF23/24 regions in WSSV-AU appear to have accumulated additional sequences.
Collectively, the distribution of phylotype II appears to be localized in Asia and have a distinct origin from that of phylotype I. This suggests that phylotype II represents a WSSV lineage already present in Asia prior to the 1990s pandemic.
Emergence of recombinant strains
Virus genomes can recombine, sometimes leading to a more complex evolutionary history with reticulate rather than bifurcating branches (Brennan Greg et al. 2022; Kolb et al. 2017). To investigate the possibility of genome recombination events in WSSV, we generated a neighbor-net phylogenetic network, which visualizes conflicting phylogenetic signals present in the whole genome alignment (Fig. 4a). By visually inspecting the phylogenetic network and comparing the topology with that of the maximum-likelihood phylogenetic tree in Fig. 2, we identified two possible cross-phylotype recombinant lineages, which we named 1601 and Qingdao.
Cross-phylotype recombinant WSSV lineages. a Neighbor-net phylogenetic network of 61 WSSV genomes using uncorrected distances (“Uncorrected_P”) ignoring constant sites. Phylotypes I and II are shaded with light blue and pink, respectively. Cross-phylotype recombinant lineages 1601 and Qingdao are shaded with orange and light green, respectively. Lineage 1601 clusters with phylotype II despite its phylogenetic position within phylotype I in the maximum-likelihood phylogenetic tree in Fig. 1. Lineage Qingdao is linked to Weifang2018 (phylotype I) with a reticulation, indicative of conflicting phylogenetic signals and suggesting their close relationships. b Coordinates of recombination breakpoints in lineages 1601 and Qingdao. The upper track shows the genome diagram of WSSV CN01. The middle and bottom tracks indicate the inferred origins of genome segments in lineages 1601 and Qingdao. The breakpoint coordinates are those of the CN01 genome. The gray boxes between the middle and bottom tracks denote the recombination-free segments used for the phylogenetic analyses in c to e. c Maximum-likelihood phylogenetic tree of recombination-free segment corresponding to nucleotides 23,315–111,734 in the CN01 genome. Phylotypes I and II are shaded with light blue and pink, respectively. Cross-phylotype recombinant lineages 1601 and Qingdao are shaded with orange and light green, respectively. Numbers beside nodes indicate the SH-aLRT support (%)/ultrafast bootstrap support (%). d Maximum-likelihood phylogenetic tree of recombination-free segment corresponding to nucleotides 111,735–190,858 in the CN01 genome. e Maximum-likelihood phylogenetic tree of recombination-free segment corresponding to nucleotides 205,155–297,825 in the CN01 genome
Lineage 1601 consists of isolate 1601 (MH663976.1; referred to as “Procambarus clarkii virus” and “WSSV-Cc” by the authors of the original publication) (Ke et al. 2021) and four related MAGs (Laibin2019, GCF7, Jangsu2019, and Sichuan2020). This cluster was placed within phylotype I in the maximum-likelihood phylogenetic tree in Fig. 2, whereas it fell into phylotype II in the neighbor-net network (Fig. 4a). The discrepancy in the phylogenetic positions implies the presence of conflicting phylogenetic signals in the genomes of lineage 1601 members.
Lineage Qingdao is represented by MAGs Qingdao2019 and Qingdao2020, which were identified from RNA-seq data of P. japonicus sampled in Qingdao, Shandong, China, in 2019 and 2020. Qingdao2019 and Qingdao2020 formed an early branching clade in phylotype II in the maximum-likelihood phylogenetic tree (Fig. 1). However, the phylogenetic network linked the Qingdao lineage to a phylotype I representative (Weifang2018) with a reticulation, suggesting a cross-phylotype recombination.
We hypothesized that these conflicting phylogenetic signals result from genomic recombination between two phylotypes, leading to distinct parts of the genome originating from different phylotypes. To test this, we used RDP4 to define potential recombination breakpoints within the two recombinant lineages (Fig. 4b). The analysis revealed two recombination events in the 1601 lineage and one in the Qingdao lineage (Online Resource, Table S3).
The predicted recombination breakpoints tell us which parts of the genome originate from which phylotype, and we expected that a phylogenetic analysis of selected genome segments should corroborate this. We selected three regions in the genome that showed no signs of recombination in either recombinant lineage (coordinates 23,315–111,734, 111,735–190,858, and 205,155–297,825; Fig. 4b). The maximum-likelihood phylogenetic trees constructed from these regions supported the presence of two major phylotypes. As expected, lineages 1601 and Qingdao were classified under different phylotypes depending on the genome segment (Fig. 4c–e). These results demonstrate that cross-phylotype recombination events gave rise to the 1601 and Qingdao lineages.
Our phylogenetic analysis of the selected segments mostly agreed with the two-phylotype classification scheme, with a few exceptions. For instance, in subtree 23,315–111,734, four Indian isolates from phylotype II (Minjur2018, WSSV-NS, wssv_ciba_003, and DBA1182) were grouped with phylotype I (Fig. 4c). Similarly, Nanning2017 (phylotype II) was classified under phylotype I in subtree 111,735–190,858 (Fig. 4d), and Jintang (phylotype I) grouped with the 1601 sequences within phylotype II in subtree 205,155–297825 (Fig. 4e). These sequences might be additional cross-phylotype recombinants, warranting further investigation.
In conclusion, these observations indicate that WSSV genomes have experienced cross-phylotype recombination events, resulting in the emergence of at least two chimeric strains.
Discussion
In this study, we aimed to understand the evolution of WSSV using whole genome sequences. Phylogenetic analysis indicated the presence of two major phylotypes. The overall topology of the phylogenetic tree is consistent with the explosive spread of phylotype I during the 1990s pandemic. The common ancestor of phylotype I, the probable pandemic strain, likely had the largest genome of over 310 kbp in size and spread worldwide, and the genome shrank independently in various geographic regions (Zwart et al. 2010). Phylotype II, in contrast, seems to have a distinct origin in Asia and spread to Australia. A divergence time estimate pointed to the most recent common ancestor of the two phylotypes existing between 1970 and the early 1980s. This suggests that there was a preexisting diversity of WSSV genotypes in Asia before the 1990s pandemic.
WSSV classification based on partial genomic segments should be interpreted with caution, as it only reflects the origin of given specific genomic segments, rather than the entire genome. The traditional molecular markers are useful in analyzing epidemiology within a country or a province, as clearly stated by the original authors (Dieu et al. 2004). Our results also indicate that WSSV genomes do recombine, further complicating the evolutionary history of the whole viral genome. In this regard, it may be difficult to classify WSSV isolates based on a handful of markers and discuss the origins of WSSV.
We identified two cross-phylotype recombinant lineages, 1601 and Qingdao. It is possible that there are recombinant lineages that have been missed out in our analysis, as suggested by the inconsistent placement of some isolates in the phylogenetic trees constructed from subgenomes. We have found that detecting recombination in WSSV genomes is a complex task which requires careful attention to various factors, such as the construction of a reliable whole genome alignment, selection of confidently non-recombinant reference sequences, and consideration of structural variations in accessory genes and repetitive sequences. We also explored the possibility that recombinant MAGs were artifacts resulting from mixed infection, but this was considered unlikely because highly similar sequences were identified from multiple datasets sampled at different localities and timepoints.
The hidden genetic diversity of WSSV in Asia has been suggested by Oakey et al. (Oakey et al. 2019) and Zeng (Zeng 2021) who assessed the diversity of variable numbers of tandem repeats. It is possible that a thorough epidemiological survey in Asia could reveal a yet unknown genotypic diversity of WSSV. Direct sampling of WSSV in diverse geographic locations is difficult for various reasons. However, we may be able to discover ancestral WSSV strains through bioinformatic analyses (Kawasaki et al. 2021), as we have successfully recovered multiple WSSV genomes from publicly available sequence data, including datasets that do not necessarily target WSSV.
Availability of data and materials
The raw read data generated in this study are available in DDBJ/ENA/NCBI under BioProject PRJDB13885. The accession numbers for the complete Japanese WSSV MAGs are summarized in Table 2. The following Online Resources are available on FigShare (https://doi.org/10.6084/m9.figshare.22178099); Online Resource 1 (Online_Resouce_1_MAGs.zip): draft WSSV MAGs generated from publicly available sequencing datasets. Online Resource 2 (Online_Resource_2_whole_genome_alignments.zip): whole genome alignments and maximum-likelihood phylogenetic trees. Online Resource 3 (Online_Resource_3_BEAST_trees.zip): BEAST2 XML files and Nexus-format phylogenetic tree for Bayesian divergence time estimation assuming different population dynamics. Online Resource 4 (Online_Resource_4_subgenomic_alignments.zip): subgenomic alignments used in the recombination analyses. Online Resource 5 (Online_Resource_5_genomic_deletions.zip): multiple sequence alignments of genomic deletions.
References
Bateman KS, Munro J, Uglow B, Small HJ, Stentiford GD (2012) Susceptibility of juvenile European lobster Homarus gammarus to shrimp products infected with high and low doses of white spot syndrome virus. Dis Aquat Organ 100:169–184
Bloomfield SJ, Zomer AL, O’Grady J, Kay GL, Wain J, Janecko N, Palau R, Mather AE (2023) Determination and quantification of microbial communities and antimicrobial resistance on food through host DNA-depleted metagenomics. Food Microbiol 110:104162. https://doi.org/10.1016/j.fm.2022.104162
Boezen D, Ali G, Wang M, Wang X, van der Werf W, Vlak JM, Zwart MP (2022) Empirical estimates of the mutation rate for an alphabaculovirus. PLOS Genet 18:e1009806. https://doi.org/10.1371/journal.pgen.1009806
Bouckaert R, Heled J, Kühnert D, Vaughan T, Wu C-H, Xie D, Suchard MA, Rambaut A, Drummond AJ (2014) BEAST 2: a software platform for Bayesian evolutionary analysis. PLOS Comput Biol 10:e1003537. https://doi.org/10.1371/journal.pcbi.1003537
Bryant D, Moulton V (2004) Neighbor-net: an agglomerative method for the construction of phylogenetic networks. Mol Biol Evol 21:255–265. https://doi.org/10.1093/molbev/msh018
Bushnell B, Rood J, Singer E (2017) BBMerge–accurate paired shotgun read merging via overlap. PLoS ONE 12:e0185056. https://doi.org/10.1371/journal.pone.0185056
Cai X, Su Y (1993) The causes and symptoms, prevalent of shrimp diseases and its countermeasure in Fujian in 1992. Mod Fish Inf 8:11–18
Chen S, Zhou Y, Chen Y, Gu J (2018) fastp: an ultra-fast all-in-one FASTQ preprocessor. Bioinformatics 34:i884–i890. https://doi.org/10.1093/bioinformatics/bty560
Chen X, Fan L, Qiu L, Dong X, Wang Q, Hu G, Meng S, Li D, Chen J (2021) Metagenomics analysis reveals compositional and functional differences in the gut microbiota of red swamp crayfish, Procambarus clarkii, grown on two different culture environments. Front Microbiol 12:735190
Chen D, Lu L, Pei Q, Chen D, Yang L, Zhang X, Zhang X, Ouyang P, Geng Y, Li Z, Li L, Huang X (2022) Transcriptome analysis of the immunomodulatory effects of Salvia miltiorrhiza polysaccharide on hemocyte immune response in Procambarus clarkii. Fish Shellfish Immunol 131:697–706. https://doi.org/10.1016/j.fsi.2022.10.054
Chou H, Huang C, Wang C, Chiang H, Lo C (1995) Pathogenicity of a baculovirus infection causing white spot syndrome in cultured penaeid shrimp in Taiwan. Dis Aquat Organ 23:165–173. https://doi.org/10.3354/dao023165
Cruz-Flores R, Mai HN, Kanrar S, Aranguren Caro LF, Dhar AK (2020) Genome reconstruction of white spot syndrome virus (WSSV) from archival Davidson’s-fixed paraffin embedded shrimp (Penaeus vannamei) tissue. Sci Rep 10:13425. https://doi.org/10.1038/s41598-020-70435-x
Dieu BTM, Marks H, Siebenga JJ, Goldbach RW, Zuidema D, Duong TP, Vlak JM (2004) Molecular epidemiology of white spot syndrome virus within Vietnam. J Gen Virol 85:3607
Drummond AJ, Rambaut A (2007) BEAST: Bayesian evolutionary analysis by sampling trees. BMC Evol Biol 7:214. https://doi.org/10.1186/1471-2148-7-214
Duchêne S, Holmes EC, Ho SYW (2014) Analyses of evolutionary dynamics in viruses are hindered by a time-dependent bias in rate estimates. Proc r Soc B Biol Sci 281:20140732. https://doi.org/10.1098/rspb.2014.0732
Feng W, Zhou L, Zhao P, Du H, Diao C, Zhang Y, Liu Z, Jin W, Yu J, Han J, Okoth E, Morode R, Liu J-F (2021) Genome assemblies of the warthog and Kenyan domestic pig provide insights into Suidae evolution and candidate genes for African swine fever tolerance. bioRxiv. https://doi.org/10.1101/2021.12.17.473133
Firth C, Kitchen A, Shapiro B, Suchard MA, Holmes EC, Rambaut A (2010) Using time-structured data to estimate evolutionary rates of double-stranded DNA viruses. Mol Biol Evol 27:2038–2051. https://doi.org/10.1093/molbev/msq088
Fukuzumi K, Chikushi Y (2003) Retention of PRDV in wild-caught kuruma prawn. Bull Fukuoka Fish Mar Technol Res Cent 13:13–19
Gao Y, Zhang X, Wei J, Sun X, Yuan J, Li F, Xiang J (2015) Whole transcriptome analysis provides insights into molecular mechanisms for molting in Litopenaeus vannamei. PLoS ONE 10:e0144350. https://doi.org/10.1371/journal.pone.0144350
Garrison E, Marth G (2012) Haplotype-based variant detection from short-read sequencing. arXiv. https://doi.org/10.48550/ARXIV.1207.3907
Ghafari M, Simmonds P, Pybus OG, Katzourakis A (2021) A mechanistic evolutionary model explains the time-dependent pattern of substitution rates in viruses. Curr Biol 31:4689-4696.e5. https://doi.org/10.1016/j.cub.2021.08.020
Greg B, Stoian Ana MM, Huibin Yu, Julhasur RM, Shefali B, Stroup JN, Chorong P, Loubna T, Stefan R (2022) Molecular mechanisms of poxvirus evolution. Bio. https://doi.org/10.1128/mbio.01526-22
Guellil M, van Dorp L, Inskip SA, Dittmar JM, Saag L, Tambets K, Hui R, Rose A, D’Atanasio E, Kriiska A, Varul L, Koekkelkoren AMHC, Goldina RD, Cessford C, Solnik A, Metspalu M, Krause J, Herbig A, Robb JE, Houldcroft CJ, Scheib CL (2022) Ancient herpes simplex 1 genomes reveal recent viral structure in Eurasia. Sci Adv 8:4435. https://doi.org/10.1126/sciadv.abo4435
Han Y, Li F, Xu L, Yang F (2017) A VP24-truncated isolate of white spot syndrome virus is inefficient in per os infection. Vet Res 48:87. https://doi.org/10.1186/s13567-017-0492-8
Hernández-Montiel Á, Giffard-Mena I, Weidmann M, Bekaert M, Ulrich K, Benkaroun J (2022) Virulence and genetic differences among white spot syndrome virus isolates inoculated in Penaeus vannamei. Dis Aquat Organ 152:85–98
Huson DH (1998) SplitsTree: analyzing and visualizing evolutionary data. Bioinformatics 14:68–73. https://doi.org/10.1093/bioinformatics/14.1.68
Inouye K, Miwa S, Oseko N, Nakano H, Kimura T, Momoyama K, Hiraoka M (1994) Mass mortalities of cultured kuruma shrimp Penaeus japonicus, in Japan in 1993: electron microscopic evidence of the causative virus. Fish Pathol 29:149–158. https://doi.org/10.3147/jsfp.29.149
Izumikawa K (2013) Retention of PRDV of some wild-caught Penaeidae in Northwestern Harima-nada. Bull Okayama Prefect Technol Cent Agric Fish Res Inst Fish Sci 28:50–54. https://www.pref.okayama.jp/uploaded/attachment/184888.pdfhttps://www.pref.okayama.jp/page/detail-59743.html
Kawasaki J, Kojima S, Tomonaga K, Horie M (2021) Hidden viral sequences in public sequencing data and warning for future emerging diseases. Mbio 12:e0163821. https://doi.org/10.1128/mBio.01638-21
Kawato S, Shitara A, Wang Y, Nozaki R, Kondo H, Hirono I (2019) Crustacean genome exploration reveals the evolutionary origin of white spot syndrome virus. J Virol 93:e01144-e1218. https://doi.org/10.1128/JVI.01144-18
Ke F, Gui L, Li T, Li F-H, Zhao X, Chen Z-Y, Lei C-K, Zhang Q-Y (2021) The features of an emerging whispovirus isolate from freshwater crayfish. Aquac Rep 20:100728. https://doi.org/10.1016/j.aqrep.2021.100728
Knibb W, Le C, Katouli M, Bar I, Lloyd C (2018) Assessment of the origin of white spot syndrome virus DNA sequences in farmed Penaeus monodon in Australia. Aquaculture 494:26–29. https://doi.org/10.1016/j.aquaculture.2018.05.018
Kolb AW, Lewin AC, Moeller Trane R, McLellan GJ, Brandt CR (2017) Phylogenetic and recombination analysis of the herpesvirus genus Varicellovirus. BMC Genom 18:887. https://doi.org/10.1186/s12864-017-4283-4
Kolmogorov M, Yuan J, Lin Y, Pevzner PA (2019) Assembly of long, error-prone reads using repeat graphs. Nat Biotechnol 37:540–546. https://doi.org/10.1038/s41587-019-0072-8
Kooloth Valappil R, Anand D, Kulkarni A, Mahapatra M, Kumar SH, Bedekar MK, Kollanoor RJ, Mulloorpeedikayil RG, Mohideenpitchai MM, Muthumariappan S, Panchavarnam S, Devaraj K, Bass D, van Aerle R (2021) Three draft genome sequences of white spot syndrome virus from India. Microbiol Resour Announc 10:e0057921–e0057921. https://doi.org/10.1128/MRA.00579-21
Koren S, Walenz BP, Berlin K, Miller JR, Bergman NH, Phillippy AM (2017) Canu: scalable and accurate long-read assembly via adaptive k-mer weighting and repeat separation. Genome Res 27:722–736. https://doi.org/10.1101/gr.215087.116
Leung H, Long X, Ni Y, Qian L, Nychas E, Siliceo SL, Pohl D, Hanhineva K, Liu Y, Xu A, Nielsen HB, Belda E, Clément K, Loomba R, Li H, Jia W, Panagiotou G (2022) Risk assessment with gut microbiome and metabolite markers in NAFLD development. Sci Transl Med 14:eabk0855. https://doi.org/10.1126/scitranslmed.abk0855
Li H (2018) Minimap2: pairwise alignment for nucleotide sequences. Bioinformatics 34:3094–3100. https://doi.org/10.1093/bioinformatics/bty191
Li F, Gao M, Xu L, Yang F (2017) Comparative genomic analysis of three white spot syndrome virus isolates of different virulence. Virus Genes 53:249–258. https://doi.org/10.1007/s11262-016-1421-z
Li H, Li H, Wang J, Guo L, Fan H, Zheng H, Yang Z, Huang X, Chu M, Yang F, He Z, Li N, Yang J, Wu Q, Shi H, Liu L (2019) The altered gut virome community in rhesus monkeys is correlated with the gut bacterial microbiome and associated metabolites. Virol J 16:105. https://doi.org/10.1186/s12985-019-1211-z
Li X, Luan S, Luo K, Cao B, Chen B, Kong J, Meng X (2022) Comparative transcriptomic analysis of Chinese shrimp Fenneropenaeus chinensis infected with white spot syndrome virus. Aquac Rep 22:100986. https://doi.org/10.1016/j.aqrep.2021.100986
Lightner DV, Redman RM, Pantoja CR, Tang KFJ, Noble BL, Schofield P, Mohney LL, Nunan LM, Navarro SA (2012) Historic emergence, impact and current status of shrimp pathogens in the Americas. Dis. Aquat Crustac Probl Solut Glob Food Secur 110:174–183. https://doi.org/10.1016/j.jip.2012.03.006
Liu P-P, Wei Z, Cheng Z-H, Wang X-W (2022) Small immune effectors coordinate peptidoglycan-derived immunity to regulate intestinal bacteria in shrimp. PLOS Pathog 18:e1010967. https://doi.org/10.1371/journal.ppat.1010967
Lo C, Peng S, Chang Y, Kou G-H (2005) White spot syndrome—what we have learned about the virus and the disease. Dis Asian Aquac. https://doi.org/10.1051/vetres/2010015
Luo K, Chen Y, Wang F (2022) Shrimp plasma MANF works as an invertebrate anti-inflammatory factor via a conserved receptor tyrosine phosphatase. J Immunol 208:1214–1223. https://doi.org/10.4049/jimmunol.2100595
Maeda M, Itami T, Furumoto A, Hennig O, Imamura T, Kondo M, Hirono I, Aoki T, Takahashi Y (1998) Detection of penaeid rod-shaped DNA virus (PRDV) in wild-caught shrimp and other crustaceans. Fish Pathol 33:373–380. https://doi.org/10.3147/jsfp.33.373
Mai U, Sayyari E, Mirarab S (2017) Minimum variance rooting of phylogenetic trees and implications for species tree reconstruction. PloS ONE 12:e0182238. https://doi.org/10.1371/journal.pone.0182238
Marks H, Goldbach RW, Vlak JM, van Hulten MCW (2004) Genetic variation among isolates of white spot syndrome virus. Arch Virol 149:673–697. https://doi.org/10.1007/s00705-003-0248-9
Marks H, van Duijse JJA, Zuidema D, van Hulten MCW, Vlak JM (2005) Fitness and virulence of an ancestral white spot syndrome virus isolate from shrimp. Virus Res 110:9–20. https://doi.org/10.1016/j.virusres.2005.01.002
Martin DP, Murrell B, Golden M, Khoosal A, Muhire B (2015) RDP4: detection and analysis of recombination patterns in virus genomes. Virus Evol 1:003. https://doi.org/10.1093/ve/vev003
Millard RS, Bickley LK, Bateman KS, Farbos A, Minardi D, Moore K, Ross SH, Stentiford GD, Tyler CR, van Aerle R, Santos EM (2021) Global mRNA and miRNA analysis reveal key processes in the initial response to infection with WSSV in the Pacific whiteleg shrimp. Viruses. https://doi.org/10.3390/v13061140
Minh BQ, Schmidt HA, Chernomor O, Schrempf D, Woodhams MD, von Haeseler A, Lanfear R (2020) IQ-TREE 2: new models and efficient methods for phylogenetic inference in the genomic era. Mol Biol Evol 37:1530–1534. https://doi.org/10.1093/molbev/msaa015
Momoyama K, Muroga K (2005) Diseases of cultured kuruma shrimp in Japan: a review. Fish Pathol 40:1–14. https://doi.org/10.3147/jsfp.40.1
Momoyama K, Hiraoka M, Nakano H, Koube H, Inouye K, Oseko N (1994) Mass mortalities of cultured kuruma shrimp, Penaeus japonicus, in Japan in 1993: histopathological study. Fish Pathol 29:141–148. https://doi.org/10.3147/jsfp.29.141
Morga B, Jacquot M, Pelletier C, Chevignon G, Dégremont L, Biétry A, Pepin J-F, Heurtebise S, Escoubas J-M, Bean TP, Rosani U, Bai C-M, Renault T, Lamy J-B (2021) Genomic diversity of the ostreid herpesvirus type 1 across time and location and among host species. Front Microbiol. https://doi.org/10.3389/fmicb.2021.711377
Nakano H, Koube H, Umezawa S, Momoyama K, Hiraoka M, Inouye K, Oseko N (1994) Mass mortalities of cultured kuruma shrimp, Penaeus japonicus, in Japan in 1993: epizootiological survey and infection trials. Fish Pathol 29:135–139. https://doi.org/10.3147/jsfp.29.135
Nurk S, Bankevich A, Antipov D, Gurevich A, Korobeynikov A, Lapidus A, Prjibelsky A, Pyshkin A, Sirotkin A, Sirotkin Y, Stepanauskas R, McLean J, Lasken R, Clingenpeel SR, Woyke T, Tesler G, Alekseyev MA, Pevzner PA (2013) Assembling genomes and mini-metagenomes from highly chimeric reads. In: Deng M, Jiang R, Sun F, Zhang X (eds) Research in Computational Molecular Biology. Springer, Berlin Heidelberg, Berlin, Heidelberg, pp 158–170
Oakey HJ, Smith CS (2018) Complete genome sequence of a white spot syndrome virus associated with a disease incursion in Australia. Aquaculture 484:152–159. https://doi.org/10.1016/j.aquaculture.2017.11.009
Oakey J, Smith C, Underwood D, Afsharnasab M, Alday-Sanz V, Dhar A, Sivakumar S, Sahul Hameed AS, Beattie K, Crook A (2019) Global distribution of white spot syndrome virus genotypes determined using a novel genotyping assay. Arch Virol 164:2061–2082. https://doi.org/10.1007/s00705-019-04265-2
Okamoto K, Suzuki M (1999) Detection of penaeid rod-shaped DNA virus (PRDV) in wild kuruma prawn, Marsupenaeus japonicus, in Lake Hamana and Ensyunada. Aquac Sci 47:299–302. https://doi.org/10.11233/aquaculturesci1953.47.299
Onihary AM, Razanajatovo IM, Rabetafika L, Bastaraud A, Heraud J-M, Rasolofo V (2021) Genotype diversity and spread of white spot syndrome virus (WSSV) in Madagascar (2012–2016). Viruses. https://doi.org/10.3390/v13091713
Parrilla-Taylor DP, Vibanco-Pérez N, Durán-Avelar MD, Gomez-Gil B, Llera-Herrera R, Vazquez-Juarez R (2018) Molecular variability and genetic structure of white spot syndrome virus strains from northwest Mexico based on the analysis of genomes. FEMS Microbiol Lett. https://doi.org/10.1093/femsle/fny216
Patterson M, Marschall T, Pisanti N, van Iersel L, Stougie L, Klau GW, Schönhuth A (2015) WhatsHap: weighted haplotype assembly for future-generation sequencing reads. J Comput Biol 22:498–509. https://doi.org/10.1089/cmb.2014.0157
Peruzza L, Thamizhvanan S, Vimal S, Vinaya Kumar K, Shekhar MS, Smith VJ, Hauton C, Vijayan KK, Sahul Hameed AS (2020) A comparative synthesis of transcriptomic analyses reveals major differences between WSSV-susceptible Litopenaeus vannamei and WSSV-refractory Macrobrachium rosenbergii. Dev Comp Immunol 104:103564. https://doi.org/10.1016/j.dci.2019.103564
Rambaut A, Lam TT, Max Carvalho L, Pybus OG (2016) Exploring the temporal structure of heterochronous sequences using TempEst (formerly Path-O-Gen). Virus Evol 2:vew07. https://doi.org/10.1093/ve/vew007
Rambaut A, Drummond AJ, Xie D, Baele G, Suchard MA (2018) Posterior summarization in Bayesian phylogenetics using tracer 1.7. Syst Biol 67:901–904. https://doi.org/10.1093/sysbio/syy032
Restrepo L, Reyes A, Bajaña L, Betancourt I, Bayot B (2018) Draft genome sequence of a white spot syndrome virus isolate obtained in Ecuador. Genome Announc 6:e00605–18. https://doi.org/10.1128/genomea.00605-18
Rodriguez-Anaya LZ, Gonzalez-Galaviz JR, Casillas-Hernandez R, Lares-Villa F, Estrada K, Ibarra-Gamez JC, Sanchez-Flores A (2016) Draft genome sequence of white spot syndrome virus isolated from cultured Litopenaeus vannamei in Mexico. Genome Announc 4:e01674-e1715. https://doi.org/10.1128/genomeA.01674-15
Shen W, Le S, Li Y, Hu F (2016) SeqKit: a cross-platform and ultrafast toolkit for FASTA/Q File manipulation. PLoS ONE 11:e0163962. https://doi.org/10.1371/journal.pone.0163962
Shen G, Zhang X, Gong J, Wang Y, Huang P, Shui Y, Xu Z, Shen H (2020) Transcriptomic analysis of Procambarus clarkii affected by “Black May” disease. Sci Rep 10:21225. https://doi.org/10.1038/s41598-020-78191-8
Sivakumar S, Vimal S, Abdul Majeed S, Santhosh Kumar S, Taju G, Madan N, Rajkumar T, Thamizhvanan S, Shamsudheen KV, Scaria V, Sivasubbu S, Sahul Hameed AS (2018) A new strain of white spot syndrome virus affecting Litopenaeus vannamei in Indian shrimp farms. J Fish Dis 41:1129–1146. https://doi.org/10.1111/jfd.12811
Stentiford GD, Neil DM, Peeler EJ, Shields JD, Small HJ, Flegel TW, Vlak JM, Jones B, Morado F, Moss S, Lotz J, Bartholomay L, Behringer DC, Hauton C, Lightner DV (2012) Disease will limit future food supply from the global crustacean fishery and aquaculture sectors. Dis. Aquat Crustac Probl Solut Glob Food Secur 110:141–157. https://doi.org/10.1016/j.jip.2012.03.013
Su Y, Cai X, Wang J (1995) Serious epidemic diseases of cultured penaeid in southern Fujian during 1992–1993. Mar Sci 19:1–4
Suitoh K, Okumura T, Yamane F, Tsuge A, Ogura Y, Yamano K (2014) Initiation and termination of spawning season of female kuruma prawn Marsupenaeus japonicus in Western Enshu-nada, Japan. Aquac Sci 62:295–305. https://doi.org/10.11233/aquaculturesci.62.295
Tamazian G, Dobrynin P, Krasheninnikova K, Komissarov A, Koepfli K-P, O’Brien SJ (2016) Chromosomer: a reference-based genome arrangement tool for producing draft chromosome sequences. GigaScience 5:38. https://doi.org/10.1186/s13742-016-0141-6
Tang K, Le Groumellec M, Lightner D (2013) Novel, closely related, white spot syndrome virus (WSSV) genotypes from Madagascar, Mozambique and the Kingdom of Saudi Arabia. Dis Aquat Organ 106:1–6. https://doi.org/10.3354/dao02645
Tsai J-M, Wang H-C, Leu J-H, Hsiao H-H, Wang AH-J, Kou G-H, Lo C-F (2004) Genomic and proteomic analysis of thirty-nine structural proteins of shrimp white spot syndrome virus. J Virol 78:11360–11370. https://doi.org/10.1128/JVI.78.20.11360-11370.2004
van Hulten MCW, Witteveldt J, Peters S, Kloosterboer N, Tarchini R, Fiers M, Sandbrink H, Lankhorst RK, Vlak JM (2001) The white spot syndrome virus DNA genome sequence. Virology 286:7–22. https://doi.org/10.1006/viro.2001.1002
Walker BJ, Abeel T, Shea T, Priest M, Abouelliel A, Sakthikumar S, Cuomo CA, Zeng Q, Wortman J, Young SK, Earl AM (2014) Pilon: an integrated tool for comprehensive microbial variant detection and genome assembly improvement. PLoS ONE 9:e112963. https://doi.org/10.1371/journal.pone.0112963
Wang H-C, Hirono I, Maningas MBB, Somboonwiwat K, Stentiford G, Consortium, I.R (2019) ICTV virus taxonomy profile: Nimaviridae. J Gen Virol 100:1053–1054
Wang Q, He Y, Li J (2019) Conjoint analysis of SMRT- and Illumina-based RNA-sequencing data of Fenneropenaeus chinensis provides insight into sex-biased expression genes involved in sexual dimorphism. Front Genet 10:1175
Woehle C, Roy A-S, Glock N, Michels J, Wein T, Weissenbach J, Romero D, Hiebenthal C, Gorb SN, Schönfeld J, Dagan T (2022) Denitrification in foraminifera has an ancient origin and is complemented by associated bacteria. Proc Natl Acad Sci 119:e2200198119. https://doi.org/10.1073/pnas.2200198119
Yang F, He J, Lin X, Li Q, Pan D, Zhang X, Xu X (2001) Complete genome sequence of the shrimp white spot bacilliform virus. J Virol 75:11811–11820. https://doi.org/10.1128/JVI.75.23.11811-11820.2001
Yu D, Zhai Y, He P, Jia R (2022) Comprehensive transcriptomic and metabolomic analysis of the Litopenaeus vannamei hepatopancreas after WSSV Challenge. Front Immunol 13:826914
Zeng Y (2021) Molecular epidemiology of white spot syndrome virus in the world. Aquaculture 537:736509. https://doi.org/10.1016/j.aquaculture.2021.736509
Zhang L, Zhou Y, Song Z, Liang H, Zhong S, Yu Y, Liu T, Sha H, He L, Gan J (2022a) Mercury induced tissue damage, redox metabolism, ion transport, apoptosis, and intestinal microbiota change in red swamp crayfish (Procambarus clarkii): application of multi-omics analysis in risk assessment of Hg. Antioxidants. https://doi.org/10.3390/antiox11101944
Zhang X, Shen G, Wang Y, Huang P, Shui Y, Xu Z, Shen H (2022b) Genome survey and large-scale isolation of microsatellites through Illumina sequencing from the red swamp crayfish Procambarus clarkii. PJZ. https://doi.org/10.17582/journal.pjz/20190829030819
Zhao B-R, Wang X-X, Wang X-W (2022) Shoc2 recognizes bacterial flagellin and mediates antibacterial Erk/Stat signaling in an invertebrate. PLOS Pathog 18:e1010253. https://doi.org/10.1371/journal.ppat.1010253
Zhong Y, Zhao W, Tang Z, Huang L, Zhu X, Liang X, Yan A, Lu Z, Yu Y, Tang D, Wang D, Lu Z (2021) Comparative transcriptomic analysis of the different developmental stages of ovary in red swamp crayfish Procambarus clarkii. BMC Genom 22:199. https://doi.org/10.1186/s12864-021-07537-x
Zhou J, Zhao H, Huang Z, Ye X, Zhang L, Li Q, Zhao Z, Su X, Liu G, Du J (2020) Differential transcriptomic analysis of crayfish (Procambarus clarkii) from a rice coculture system challenged by Vibrio parahaemolyticus. Comp Biochem Physiol Genom Proteom 36:100741. https://doi.org/10.1016/j.cbd.2020.100741
Zimin AV, Salzberg SL (2020) The genome polishing tool POLCA makes fast and accurate corrections in genome assemblies. PLOS Comput Biol 16:e1007981. https://doi.org/10.1371/journal.pcbi.1007981
Zwart MP, Dieu BTM, Hemerik L, Vlak JM (2010) Evolutionary trajectory of white spot syndrome virus (WSSV) genome shrinkage during spread in Asia. PLoS ONE 5:e13400. https://doi.org/10.1371/journal.pone.0013400
Acknowledgements
This research was supported by Grants-in-Aid for Scientific Research from the Japan Society for Promotion of Science (JSPS) (JSPS KAKENHI Grant Numbers JP15H02462, JP19H00949, and 19J21518) and by the Science and Technology Research Partnership for Sustainable Development from the Japan Science and Technology Agency (SATREPS Grant Number JPMJSA1806).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kawato, S., Omine, R., Teruya, S. et al. Evolutionary genomics of white spot syndrome virus. Fish Sci 89, 769–783 (2023). https://doi.org/10.1007/s12562-023-01715-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12562-023-01715-4