Abstract
Epidemiological studies focusing on metal(oid) exposure and excess body weight among children show inconsistent results and the influence of metal(loid) mixtures has been little studied. We aimed to explore the effect of 10 metal(loid)s, individually and in combinations, on overweight and obesity among children aged 6–12. Urinary levels of these metal(oid)s from 92 controls and 51 cases with overweight and obesity were analyzed. Metal(loid) levels were log-transformed and categorized into tertiles according to the control group cutoff points. Two logistic regression models and weighted quantile sum regressions (WQS) were run: model 1 adjusting for age, sex, creatinine, energy intake and physical activity and model 2 additionally adjusting for maternal education, rice and fish consumption. In the single metal(oid) exposure model, there was a trend of significant negative association for urinary cobalt (Co), where children in the third tertile had lower odds of present overweight and obesity than those in the first (Odds Ratio, OR = 0.43; 95% Confidence Interval, CI = 0.20–0.93). Urinary chromium (Cr) levels were borderline-significant negatively associated with overweight and obesity (ORa = 1.70; 95% CI = 0.97–2.98). Molybdemun, Cr and Co had a major contribution to the inverse association between metal(loid) mixture and overweight and obesity as well as lead, cadmium and total arsenic in the positive relationship. Our findings in this explorative study suggested an inverse association of high urinary Co levels with overweight and obesity. Moreover, metal(loid) mixture exposure may have influence on overweight and obesity with an important contribution of Co in the potential negative effect.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Excess body weight (overweight or obesity) among children is currently a major worldwide public health concern due to its prevalence and associated health problems, such as cardiovascular, musculoskeletal, endocrine diseases, autoimmune disorders and cancer (De Bont et al. 2022). Worldwide the prevalence of overweight and obesity, in 2016 was over 340 million children and adolescents aged 5–19 (WHO 2021). Concretely, in Spain, the prevalence of childhood excess body weight has continued to rise in the last decade (Bravo-Saquicela et al. 2022). In fact, in 2018 Spain ranked second within the WHO European Region in childhood overweight and obesity prevalence (WHO 2018). This high prevalence cannot be solely attributed to traditional risk factors such as changes in diet, physical activity and underlying genetic susceptibility (Tang-Péronard et al. 2011). Over recent years, it has been investigated the role of obesogens (chemicals that lead to increase white adipose tissue mass after their exposure (Heindel et al. 2022)) on the development of obesity especially during the critical development window (embryo, fetus or young child). In this period, some of the environmental substances such as bisphenol A and analogs, phthalates, tributyltin, flame retardants, polybrominated diphenyl ether (PBDEs), pesticides, dioxin and PCBs (polychlorinated biphenyl), per and poly-fluoroalkyl substances (PFAS), and metal(loid)s may play an important role (Iavicoli et al. 2009; Heindel and Blumberg 2019; Heindel et al. 2022). Concretely, metal(loid)s are ubiquitous elements of natural and anthropogenic origins, with widespread environmental distribution. They can be introduced into the body in multiple ways including via food consumption, drinking water and air inhalation (Rosin 2009). Recent studies have found an association between metals exposure and obesity prevalence (Tinkov et al. 2021). Thus, metal(loid)s such as arsenic (As), cadmium (Cd), lead (Pb), mercury (Hg) and nickel (Ni), considered toxic elements, could contribute to obesity displacing essential metals such as zinc, chromium (Cr), copper (Cu), iron (Fe) and magnesium (Mg) that might affect to the energy production and carbohydrate tolerance (Katzen-Luchenta 2007) generally leading to the disturbance of metabolic process (Padilla et al. 2010). Moreover, deficiencies of certain essential metals such as Cr, Cu, Fe and Mg can increase adiposity (Wildman and Mao 2001; Guerrero-Romero and Rodríguez-Morán 2006; Yanoff et al. 2007; Komolova et al. 2008; Padmavathi et al. 2010).
Biomarkers are commonly used in epidemiological studies to assess metal(loid) exposure and its internal dose as they consider all possible sources in the population (Boogaard et al. 2012; Martinez-Morata et al. 2023). Concretely, urine is the non-invasive biological matrix most widely used for biomonitoring metal exposure (Esteban and Castaño 2009). Due to the discharge of metals in urine is high and there are no ethical considerations or lack of access to it, it is considered the best and most accessible biological matrix (Nasab et al. 2022). In fact, may provide useful information for biomonitoring studies carried out in certain population groups such as children where blood samples are more difficult to obtain (Molina-Villalba et al. 2015). Several metals such as As, Mn, Ni, Cr have a short life-biological in urine while others such as urinary Cd represent chronic and cumulative exposure (Vogel et al. 2021). Nonetheless, under steady conditions, urinary metals seem to be appropriate long-term exposure biomarkers (Castiello et al. 2020). Previous research has studied the potential relationship of metal(loid)s exposure with overweight and obesity in sensitive subpopulation groups like infants or children and has displayed inconsistent results because some did not find an association with Hg levels (Rothenberg et al. 2015; Fan et al. 2017; Fábelová et al. 2018) whereas others reported a positive association (Geier et al. 2016; Shin et al. 2018; Cho 2021). In the same manner, some studies have found a negative association between As concentrations and overweight and obesity (Su et al. 2012; Lin et al. 2014), others positive (Nasab et al. 2022; Hashemi et al. 2023) and one did not find relationship (Lewis et al. 2018). Instead, for other metals such as Ni (Tascilar et al. 2011; Błażewicz et al. 2013), Mn (Fan et al. 2017; Ge et al. 2020), Cu (Fan et al. 2017; Ge et al. 2020) and Cd (Padilla et al. 2010; Shao et al. 2017; Shan 2022) few epidemiological studies have assessed their potential role in overweight and obesity. Besides, these studies have typically focused on metal(loid)s individual analyses. However, in the real-world scenario humans are simultaneously exposed to multiple metals and the effects of one element can depend on their interactions with the rest (Notario-Barandiaran et al. 2023). Given the hypothesis of our study is based on the urinary metal(loid) concentrations might contribute to overweight and obesity among Spanish children, this study aimed to assess the potential individual and mixture effects of a selection of metal(loid)s, measured in urine samples, on overweight and obesity of Spanish children aged 6–12.
Materials and Methods
Study Design and Setting
The present research is a case–control study designed to assess the influence of environmental factors affecting overweight and obesity among Spanish children. Study participants were recruited from January 2020 to January 2022. Both cases and controls were recruited from 15 primary care centers and 3 schools located in areas with different socioeconomic level and randomly selected from the province of Granada that covers an area of1 2.531 m2 in Southern Spain. Concretely, 28% of cases were recruited from schools and 72% from primary care centers. No relevant differences were observed between participants recruited in primary care centers and those recruited from schools, except for physical activity (data no shown).
Personal identifiers in the dataset were removed to guarantee confidentiality.
Study Participants
Eligible cases met the following inclusion criteria: (1) overweight or obesity diagnosis; (2) prepuberal children aged between 6 and 12 years-old; (3) having resided continuously in the study areas for at least 6 months. The controls met the same inclusion criteria, with the exception of overweight and obesity diagnosis. Exclusion criteria included obesity as a symptom of other pathologies or as a side effect of pharmacological treatment.
Data Collection and Variables
Covariates were collected from a structured questionnaire by face-to-face trained interviewers to the participants’ parents or legal guardians including maternal education (primary, secondary and university) and smoker/s in the household (yes/no) using the household members smokers ≥ 1 as a proxy for secondhand smoke. The following data were collected from children: gender (boys/girls), age (years), physical activity out-of-school (yes/no) and fish and rice intake (g/day). Dietary information for the 12 months prior to the interview was obtained using a semi-quantitative food frequency questionnaire (FFQ) that was addressed to the parents or legal guardians of participants and completed by nutrition professionals. This questionnaire included the following food groups: dairy products, eggs, meat and meat derivatives, fish and fish derivatives, vegetables, tubers, fruits and nuts, legumes, cereals, precooked or ultra-processed food, bakery products, pastries and sweets, fats and oils, non-alcoholic beverages and miscellaneous. The energy intake was calculated using three 24 h dietary recalls.
In addition, child anthropometry assessment was performed at the recruitment moment and was carried out by trained field staff including height (cm) and weight (kg). Weight was measured with the participants wearing light clothing and without shoes, using a portable Tanita scale (model MC 780-S MA). Height was measured in the standing position with a stadiometer (model SECA 214 (20–207 cm). During height measurements, care was taken that the participants’ backs, buttocks and heels were in contact with the wall. Body mass index (BMI) was calculated as weight in kilograms divided by height squared, in meters. The participants were classified as underweight, normal weight, overweight and obese according the standards proposed by the International Obesity Task Force, as described by Cole et al. (2007, 2000). Thus, we divided the participants into two groups: cases (overweight and obese children) and controls (normal-weight children). The cut-off point for overweight and obesity was established on the equivalent value of 25 kg/m2 for adults.
Sampling and Metal(loid) Analysis
A single and first urine of the day from study participants in a polypropylene bottle was provided by their parents or legal guardians. The urine sample was stored in the participants’ homes under frozen conditions until collection within a 1 to 4 days-period after the interview. For this study, nine metals (Cd, Co, Cr, Cu, Hg, Mn, Mo, Ni and Pb) and one metal(loid) (As) were analyzed. A calibration curve was prepared in ultrapure water (Milli-Q, Merck, Darmstadt, Germany) with 2% HNO3 (Merck, Darmstadt, Germany) and 1% HCl (Merck) using appropriate metal standard solutions (Agilent Technologies, Santa Clara, CA, USA). Urine samples were diluted 1:10 in ultrapure water (Milli-Q) with 2% HNO3 (Merck) and 1% HCl (Merck). Appropriate blanks were analyzed to correct the results.
The multi-element analyses were performed on an Agilent 8900 triple quadrupole Inductively coupled plasma mass spectrometry (ICP-MS) (Agilent Technologies). The instrument was tuned and performance parameters were checked prior to analysis. To ensure the quality of the results, a multi-element 400 µg/L internal standard solution with Sc, Ge, Ir and Rh was added online to the samples. Furthermore, a suitable certified reference material [Seronorm (Sero, Billingstad, Norway) Trace Elements Urine L2 (reference 210,705)] was reanalyzed along with a blank and an intermediate calibration standard every 12 samples. National Institute of Standards and Technology NIST (Gaithersburg, MD, USA) Trace Elements in Natural Water Standard Reference Material SRM 1640a was also used as certified reference material and analyzed at the beginning and the end of each sequence. Additionally, one in every 12 samples was reanalyzed at the end of each session. Limits of detection (LOD) (μg/L) were obtained for As (0.07), Cd (0.04), Co (0.01), Cr (0.2); Cu (0.5), Hg (0.08), Mn (0.08), Mo (0.03), Ni (0.1), Pb (0.1). Metal(loid) concentrations below the LOD were replaced by the LOD divided by the square root of 2 (CDC 2015). Co, Ni, As and Mo were detected in 100% of the samples while the detection rate for Mn and Cd was the lowest in both cases and controls (27.2% and 56.5%, respectively). For the rest of the metal(loid)s, the detection rate ranged from 66.3% to 96.7% The classical Jaffé method (Weber and Van Zanten 1991; Peake and Whiting 2006) based on the photometric measurement of kinetics of creatinine reaction with picric acid at 37 °C was used for determination of creatinine concentration in urine. For it, a kit of reagents was provided by Biosystems (Barcelona, Spain).
Statistical Analysis
For descriptive purposes, the arithmetic mean, median, standard deviation (SD) and interquartile range (25–75th percentiles) were used for continuous variables, whereas absolute and the percentages for categorical variables. To assess the significance level of the differences observed between cases and controls, the Chi-squared test was used in categorical variables and, for continuous variables, the Mann–Whitney U or Student’s t-test was calculated.
Metal(loid) concentrations were natural log-transformed and evaluated as continuous and as categorical variables to relax the linearity assumption. The metal(loid) levels were categorized in tertiles (T) built from the cutoff points according to the control group’s distribution (report the expected frequencies), with the lowest concentration (T1) used as the reference category. Cd and Mn were dichotomized into < LOD and ≥ LOD because their detection frequency was 20%–61%. Next, the assessment of the associations of individual metal(loid)s exposure (independent variable) with overweight and obesity (dependent variable) was performed through multivariable logistic regression models. Odds ratios (OR) and their corresponding 95% confidence intervals (95% CI) were estimated.
The p-trend values across categories were calculated by introducing metal(loid)s levels in tertiles as an ordinal level indicator in the models. To control urinary dilution, instead of using creatinine-adjusted urinary metal(loid) levels (creatinine standardization) we used urinary creatinine levels as an independent variable in regression models, as this approach is less likely to produce a biased effect estimate (HBM (Human Biomonitoring) Commission 2005; Barr et al. 2005). The selection of covariates to be included as confounders in the models was based on the existing bibliography (Fan et al. 2017; Ashley-Martin et al. 2019; Ge et al. 2020; García-Villarino et al. 2022; Nasab et al. 2022) and those whose inclusion in any model produced changes > 10% in the estimations. Thus, model 1 was adjusted for creatinine (g/L), age (years), gender (boys/girls), energy intake (Kcal/day) and physical activity (yes/no) and model 2 was additionally adjusted for maternal education (categorized as primary, secondary and university), rice and fish consumption (g/day). Sensitivity analyses were also performed, fitting the same model 2, but adding smoker/s in the household (yes/no). In addition, we studied the potential modifying effect of gender by including in each model the product term of this variable*metal(loid)s concentrations as well as by means of stratified analyses. The rational for this is based on previous investigations supporting that gender could modify the effect of metal(loid) levels on overweight and obesity (Shin et al. 2018; Ashley-Martin et al. 2019; Tetsuka et al. 2022; Nasab et al. 2022).
Finally, we assessed the potential mixture effect of the different metal(loid)s on overweight and obesity using Weighted Quantile Sum Regression (WQS) approach (Czarnota et al. 2015b; Carrico et al. 2015). This statistical approach calculates a weighted index based on the individual associations of several exposures with the outcome, and estimates the individual weight of each metal(loid) in the mixture, expressing it as a percentage. Thus, associations between the WQS index and the outcome were studied using multivariable logistic regression adjusted for the same covariates included in the two individual models (model 1 and 2). We performed WQS index calculation by gender, and therefore the individual contribution of each element shows its relative effect on girls and boys. WQS analyses were performed using tertile scored metal(loid)s concentrations using a training set defined by a 40% random sample of the dataset being the remaining 60% used for model validation. A total of 200 bootstrap samples were used for calculating the final weight. Since WQS regression require prior selection of the expected direction of individual associations within the mixture, we calculated two models (positive and negative).
Statistical analyses were performed using Stata v.15 statistical package software (Stata Corp., 2017, College Station, TX, USA), whereas WQS analyses were performed using R statistical computing environment v 4.1.1 (R Core Team 2021) with gWQS package v3.0.1 (Renzetti et al. 2021). The two-sided significance level was set at p < 0.05 and p < 0.10 for borderline significance.
Results
A total of 143 children (92 controls and 51 cases) had metal(loid) concentrations available in their urine samples. Table 1 shows the comparison of the sociodemographic and lifestyle characteristics for cases and controls. Both groups were similar for all variables studied, both in the overall population and according to gender (Table S1), except for maternal education and age. There were also no statistically significant differences in general characteristics between the included cases and controls and those without metal(loid) level data (Table S2).
As shown in Table 2, the controls had higher urinary Hg levels than the cases both in the overall population (0.6 vs 0.3 µg/g, p-value < 0.001) and according to gender (boys: 0.6 vs 0.4 µg/g, p-value = 0.001; girls: 0.7 vs 0.3 µg/g, p-value = 0.013) (Table S3). Moreover, controls showed slightly higher urinary Co levels than cases with a borderline significance level (p-value = 0.086). As levels also seemed to be higher in the control group than in the case group, although without reaching statistical significance (p-value = 0.332). For the rest of metal(loid)s, we did not find statistically significant differences between the study groups.
Individual chemical models of the influence of urinary metal(loid)s on overweight and obesity in overall participants are shown in Table 3. There was a general trend of non-significant negative associations with the exception of Co (p-trend = 0.039), for which the likelihood of having overweight and obesity was 84% lower for those individuals in the third tertile compared to the first (p = 0.040). This inverse association was supported by the analysis of urinary Co levels as a continuous variable (ORa = 0.43; 95% CI = 0.20–0.93). Children in the second tertile of As levels showed lower odds of present overweight and obesity compared to those in the first tertile (ORa T2 vs T1 = 0.25; 95% CI = 0.07–1.00). Urinary Hg and Cr levels were negatively and positively associated with overweight and obesity, respectively, with the latter metal showing borderline significance. The association of Co and Hg levels with overweight and obesity lost statistical significance in the model adjusted for maternal education, fish and rice consumption (model 2). However, the magnitude and direction of the association remained the same.
These results did not differ substantially from those obtained in the sensitivity analyses, adjusting also for smoker/s in the household (Table S4).
Although the low number of participants by strata is small, we found differences in the potential relationship between metal(loid)s and overweight andobesity according to gender. Thus, the above-mentioned negative associations of urinary Co and Hg levels with overweight and obesity seems to be stronger among boys (Co: ORa = 0.14; 95% CI = 0.02–0.76, p-interaction = 0.420); Hg: ORa = 0.35; 95% CI = 0.13–0.96, p-interaction = 0.407). Additionally, we observed a suggestive negative association of urinary Ni levels and detectable Mn concentrations with overweight and obesity, respectively (Ni: ORa = 0.17; CI95% = 0.03–0.94, p-interaction = 0.252; Mn: ORa = 0.10; CI95% = 0.00–0.83, p-interaction = 0.100) (Table S5). Despite the lack of statistical significance at the 95% level, probably due to the limited sample size, we found a suggestive borderline positive association of detectable urinary Cd levels with overweight and obesity in girls (ORa = 4.22; 95% CI = 0.77–23.04, p-interaction = 0.125) (Table S6).
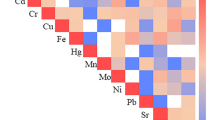
With regards to the multi-pollutant model, the “positive” WQS index showed a significant association with overweight and obesity in the overall population (OR = 2.68; 95% CI = 1.24–5.09) being Pb, Cd and As the metal(loid)s that contributed the most in both model 1 and 2. In addition, the “negative” WQS was significantly associated to overweight and obesity in overall participants (OR = 0.39; 95% IC 0.11–0.75) being mainly represented by Mo, Cr and Co in both models (Fig. 1). After the stratification by gender, multi-pollutant associations seem to be evident both in girls and boys and were mainly accounted by the same metal(loid)s than overall population (Fig. S1). We performed a sensitivity analysis using continuous metal(loid) concentrations and excluding Mn and Cd because they had a low detection rate. Despite it was observed some differences in estimations and metal(loid) contributions in the mixtures, the trend and significance of the associations were similar (Fig. S2 and Fig. S3).
Estimation of the mixture effect of metal(loid)s on excess weight among the overall population (a) Model 1: Adjusted for creatinine (g/L), age (years), gender (boys/girls), energy intake (Kcal/day) and physical activity (yes/no), (b) Model 2: Adjusted for creatinine (g/L), age (years), gender (boys/girls), energy intake (Kcal/day), physical activity (yes/no), maternal education (primary, secondary and university), rice and fish consumption (g/day). Weighted Quantile Sum Regression (WQS) models
Discussion
To the best of our knowledge, this study carried out on Spanish children is among the first to assess the associations between urinary metal(loid)s and overweight and obesity, combining both individual and mixture effects. Our findings suggested that urinary Co levels were related to a lower likelihood of presenting overweight and obesity. On the other hand, WQS analysis revealed a potential positive mixture effect of metal(loid)s on overweight and obesity mainly driven by Pb, Cd and As as well as a potential negative mixture effect mainly represented by Mo, Cr and Co in the overall population.
Mo showed the highest median concentration in both cases (58.1 µg/g) and controls (58.4 µg/g) followed by arsenic with 25.6 µg/g in cases and 12.9 µg/g in controls. Compared to other studies performed in Spain, urinary metal(loid) levels in our study population were in line with earlier results obtained from the INMA—INfancia y Medio Ambiente [Environment and Childhood]—Granada cohort from the same geographic area (Castiello et al. 2020). Urinary Co, As, Mo and Cd concentrations were also similar to those found in other studies performed in Asturias and Valencia in children aged 4 and 6–11 years, respectively (Roca et al. 2016; Junqué et al. 2022). However, we found higher urinary As levels and lower Cd and Cu concentrations than in other earlier studies performed on children living near our study area and aged 5–17 (Aguilera et al. 2010; Molina-Villalba et al. 2015). Even though the aforementioned studies also included Spanish children from the region of Andalusia, a direct comparison of metal(loid) concentrations between studies is difficult due to differences in study design and variations in the analytical laboratory procedures, including both techniques and LODs.
In comparison with studies carried out in other countries such as Italy, Mexico, Uruguay, Iran, China and the United States, our study sample showed similar urinary Cd, Co and Mn levels (Protano et al. 2016; Kafaei et al. 2017; Lewis et al. 2018; CDC 2021; Shan 2022), lower Pb levels (Protano et al. 2016; Lewis et al. 2018; Kordas et al. 2018), but higher As and Hg concentrations (Zhang et al. 2017; Kafaei et al. 2017; Lewis et al. 2018; Kordas et al. 2018; CDC 2021). Given that we measured the total As levels (including both inorganic and organic forms) being the last highly associated with fish intake, we expect to find higher concentrations in a relatively high fish-consuming population. There were no significant differences in metal(loid) concentrations between both study groups except for Hg, which were found at higher levels in controls than in cases. This may be due to differences in social class as healthy diet and weight control practices are more frequent in individuals with higher social class. In fact, it has been reported that children from the lowest social class present more risk of obesity compared to those from a higher social status (Lamerz et al. 2005). In this sense, fish intake seems to be different also according to social class. Thus, it has been reported that those with low education and income, and low education and high income consumed fish less regularly than weekly, whereas those with high education and low income and high education and high income were weekly. Since fish is a source of mercury exposure, it could explain the differences found (Skuland 2015). An in vivo study has shown that HgCl2 administration in mice decreased adipose tissue content and adipocyte size. Moreover, it’s been shown that repeated exposure to HgCl2 decreased proliferator-activated receptor (PPAR)α and PPARγ expression in adipocytes which play an important role in the regulation of lipid metabolism (Kawakami et al. 2012).
Our analysis revealed that Co was one of the elements with a major contribution to the negative association of the metal(loid)s mixture on overweight and obesity. This is in line with the results of the individual models where Co follows a U-shape relationship with overweight and obesity showing a higher protective effect at low and high exposure levels. Nevertheless, only high levels of Co (third tertile) had a significant effect. This shows that among many factors related to overweight and obesity, moderate levels (second tertile) of Co were not substantially influential so children seem to be more susceptible to high dose as previously reported (Shan 2022). Moreover, our results are consistent with previous epidemiological studies carried out in children and adolescents (Padilla et al. 2010; Shao et al. 2017; Vrijheid et al. 2020; Tetsuka et al. 2022; Shan 2022). In vivo studies have shown that Co increases serum leptin and adiponectin levels in mice, two hormones produced by adipose tissue and related to lipid metabolism. In this sense, leptin produces a potent inhibition of appetite so its deficiency promotes adiposity (Kawakami et al. 2012). Moreover, Co can regulate glycogen deposits through suppression of glucagon signaling, and thus influence body weight (Tascilar et al. 2011).
Similarly, we found a significant negative association between urinary Hg levels and overweight and obesity. Our findings were not consistent with those of other previously conducted studies showing no association between blood Hg levels and obesity in children aged 2–19 years (Rothenberg et al. 2015; Fan et al. 2017) or a positive association with BMI (Geier et al. 2016; Shin et al. 2018; Cho 2021). The different biological matrices used might explain the differences found in our findings from the aforementioned studies as methylmercury is the predominant species in the blood whereas inorganic Hg is mainly found in the urine (Berglund et al. 2005). In this sense, adverse health effects of Hg could be related to its chemical form (Clarkson and Magos 2006). Few studies have analyzed this association in children and the mechanism underlying the effect of Hg on obesity is poorly understood so further research is needed to understand this relationship.
Regarding the role of other metal(loid)s addressed in this study, the second vs first tertile of As levels was associated with a lower odds of having overweight and obesity. In line of our results, an inverse association between inorganic As and BMI was reported in Taiwanese adolescents (Su et al. 2012; Lin et al. 2014). Nevertheless, our results were contradictory with previous studies such as two cross-sectional studies performed on children and adolescents aged 6–18 years residing in Iran that revealed a positive association between urinary As levels and BMI (Nasab et al. 2022; Hashemi et al. 2023). Likewise, in another study urinary As levels were not related to BMI among children in Mexico city (Lewis et al. 2018). The fact that in Spanish are large fish consumers, makes As to be assessed as non-toxic forms, such as arsenobetain (major As species), arsenocholine and monomethylarsonic acid (Moreda–Piñeiro et al. 2012). This could explain the differences found between our results and from other studies.
Few studies have investigated the influence of Mn levels on obesity or overweight. The National Health and Nutrition Examination Survey (NAHNES) 2011–2014 and 2011–2016 data from a sample of 5404 children and 2000 adolescents aged 6–19, reported that high blood Mn levels were positively associated with overweight and obesity (Fan et al. 2017; Ge et al. 2020). On the contrary, our study found that detectable urinary Mn concentrations were inversely associated with overweight and obesity in boys. In spite of mechanisms underlying the relationship between Mn levels and obesity remain unclear, it’s known that Mn is one of the antioxidant nutrients that play an important role in the metabolism of carbohydrates, protein and lipids (Choi and Bae 2013).
The discrepancy between our findings and previous studies could be explained by metal(loid) exposure differences, the control of covariates in regression models, the different biological matrices used for biomonitoring as well as genetic variations modulating metabolism and excretion metal(loid)s.
In our analysis, we found that metal(loid) joint exposure was significantly associated with overweight and obesity where Pb, Cd and As were weighted highly in the positive association whereas Mo, Cr and Co were in the negative relationship. Nevertheless, those results are very difficult to interpret so Cd was only detected in 56.5% and 60.8% of the samples from controls and cases, respectively, urine is not the best biological matrix to measure Pb exposure (Téllez-Rojo et al. 2019) and the results were contradictory to those found in the individual models. Moreover, should be taken into account that the mixed effects of elements with different associations cannot be analyzed simultaneously. Thus, if an element negatively associated to excess body weight is misclassified in the WQS model, would be assigned a negligible weight in the WQS index positively associated to overweight and obesity (Czarnota et al., 2015a; b). Another limitation of WQS regression is that it assesses the effect of expositions taking into account that associations are linear and additive and potential synergic effects cannot be addressed (Zhang et al. 2019). Nevertheless, in spite of multivariable logistic regression models are frequently used to assess the human health effects of chemicals (Buser et al. 2014; Liu et al. 2017) because results are easy to understand and interpreted if the joint effects of chemicals are not taken into account it could contribute to false positive or false negative results (Czarnota et al. 2015a). Thus, the relationship of As, Pb, Mo and Cr with overweight and obesity in individual models can be associated to the confounding effect of the other chemical element. However, including all metal(loid)s of our interest in a single regression model could be not correct due high correlation between them which could lead to result distortion (Marill 2004). Moreover, interactions between metal(loid)s cannot be covered in an individual model (Zhang et al. 2019).
Our study also had some limitations. Our findings were based on a modest sample size thus, the statistical power was reduced and could have precluded the detection of significant associations, especially in stratified analyses. However, we were able to detect trends that could be reproducible in larger samples of participants. A further potential limitation could be that the use of spot urine per individual may have contributed to exposure misclassification. Some detected metals could represent current exposure or a recently absorbed amount, due to their short biological half-lives of urinary metals such as urinary Pb, Hg, As, Ni, Mn, Cr. Nevertheless, in steady-state conditions, urinary metal(loid) concentrations can be suitable biomarkers of long-term exposure for many metals (García-Villarino et al. 2022). Moreover, in spite of urinary metal(loid)s are used as valid biomarkers of these elements exposure (Esteban and Castaño 2009; Padilla et al. 2010; Shao et al. 2017; Lewis et al. 2018; Castiello et al. 2020), maybe urine is not the most recommended matrix for some elements such as Pb, Hg and Mn (Lucchini et al. 2015; Téllez-Rojo et al. 2019) so those results must be interpreted with caution. Nonetheless, urine is a non-invasive matrix frequently used in environmental studies such as NHANES and the German Environmental Survey for Children (Esteban and Castaño 2009; Padilla et al. 2010; Shao et al. 2017) and may provide useful information in the children population where blood collection is more difficult to carry out (Molina-Villalba et al. 2015). Additional limitations of our study are the non-availability the metal(loid) speciation data, particularly for As, which would have allowed us to distinguish the predominant chemical form and its toxicity as well as its relationship to the focus of the study. Finally, co-exposure to other pollutants or unidentified factors can’t be ruled out. Lastly, Cd is one of the metals that drive the positive association between metal mixtures and overweight and obesity, however we need to bear in mind that Cd is only detected in 56.5% and 60.8% of the samples from controls and cases.
The study’s strengths include the pioneering aspect, to the best of our knowledge, of assessing metal(loid) mixtures on overweight and obesity in children. The multi-pollutant model complements the results obtained for single chemical models, and is therefore an important tool for exploring the derived effects of simultaneous exposure to metal(loid)s. Of particular note is the case–control design of the study, establishing a temporal association between exposure and outcome. In this regard, the majority of published evidence on the associations between metal(loid)s and obesity are cross-sectional studies (Padilla et al. 2010; Scinicariello et al. 2013; Fan et al. 2017; Shao et al. 2017; Ge et al. 2020; Nasab et al. 2022; Shan 2022). Another of the study’s advantages is that it focuses on children, an understudied, sensitive and expanding group.
Conclusion
The findings of the present study evidenced that the mixture of metal(loid)s might be associated with overweight and obesity. Pb, Cd and As were the metal(oid)s from the mixture contributing the most to an increase in the probability of present overweight/obesity and Mo, Cr and Co in the decrease. Considering the results of individual models, it was found an inverse association between high Co levels and overweight and obesity. This study combined traditional and new statistical techniques to assess the potential role of metal(loid) exposure (individually and as a mixture) on overweight and obesity in a specially vulnerable population such as children due their rapid growth and still developing detoxification mechanisms (Rodríguez-Barranco et al. 2013). However, further research with larger samples is still needed to confirm our findings due to the explorative nature of this study.
Data Availability
The data that are used in this study are available from the corresponding authors upon reasonable request.
References
Aguilera I, Daponte A, Gil F et al (2010) Urinary levels of arsenic and heavy metals in children and adolescents living in the industrialised area of Ria of Huelva (SW Spain). Environ Int 36:563–569. https://doi.org/10.1016/j.envint.2010.04.012
Ashley-Martin J, Dodds L, Arbuckle TE et al (2019) Blood metal levels and early childhood anthropometric measures in a cohort of Canadian children. Environ Res 179:108736. https://doi.org/10.1016/j.envres.2019.108736
Barr DB, Wilder LC, Caudill SP et al (2005) Urinary creatinine concentrations in the U.S. population: implications for urinary biologic monitoring measurements. Environ Health Perspect 113:192–200. https://doi.org/10.1289/ehp.7337
Berglund M, Lind B, Björnberg KA et al (2005) Inter-individual variations of human mercury exposure biomarkers: a cross-sectional assessment. Environ Health 4:20. https://doi.org/10.1186/1476-069X-4-20
Błażewicz A, Klatka M, Astel A et al (2013) Differences in trace metal concentrations (Co, Cu, Fe, Mn, Zn, Cd, and Ni) in whole blood, plasma, and urine of obese and nonobese children. Biol Trace Elem Res 155:190–200. https://doi.org/10.1007/s12011-013-9783-8
Boogaard PJ, Aylward LL, Hays SM (2012) Application of human biomonitoring (HBM) of chemical exposure in the characterisation of health risks under REACH. Int J Hyg Environ Health 215:238–241. https://doi.org/10.1016/j.ijheh.2011.09.009
Bravo-Saquicela DM, Sabag A, Rezende LFM, Rey-Lopez JP (2022) Has the prevalence of childhood obesity in Spain plateaued? A systematic review and meta-analysis. Int J Environ Res Public Health 19:5240. https://doi.org/10.3390/ijerph19095240
Buser MC, Murray HE, Scinicariello F (2014) Age and sex differences in childhood and adulthood obesity association with phthalates: analyses of NHANES 2007–2010. Int J Hyg Environ Health 217:687–694. https://doi.org/10.1016/j.ijheh.2014.02.005
Carrico C, Gennings C, Wheeler DC, Factor-Litvak P (2015) Characterization of weighted quantile sum regression for highly correlated data in a risk analysis setting. JABES 20:100–120. https://doi.org/10.1007/s13253-014-0180-3
Castiello F, Olmedo P, Gil F et al (2020) Association of urinary metal concentrations with blood pressure and serum hormones in Spanish male adolescents. Environ Res 182:108958. https://doi.org/10.1016/j.envres.2019.108958
CDC (2015) Fourth national report on human exposure to environmental chemicals updated tables
CDC (2021) Fourth national report on human exposure to environmental chemicals. Updated tables, volume three: analysis of pooled serum samples for select chemicals, NHANES 2005–2016
Cho KY (2021) Association of blood mercury levels with the risks of overweight and high waist-to-height ratio in children and adolescents: data from the korean national health and nutrition examination survey. Children 8:1087. https://doi.org/10.3390/children8121087
Choi M-K, Bae Y-J (2013) Relationship between dietary magnesium, manganese, and copper and metabolic syndrome risk in Korean adults: The Korea national health and nutrition examination survey (2007–2008). Biol Trace Elem Res 156:56–66. https://doi.org/10.1007/s12011-013-9852-z
Clarkson TW, Magos L (2006) The toxicology of mercury and its chemical compounds. Crit Rev Toxicol 36:609–662. https://doi.org/10.1080/10408440600845619
Cole TJ, Bellizzi MC, Flegal KM, Dietz WH (2000) Establishing a standard definition for child overweight and obesity worldwide: international survey. BMJ 320:1240. https://doi.org/10.1136/bmj.320.7244.1240
Cole TJ, Flegal KM, Nicholls D, Jackson AA (2007) Body mass index cut offs to define thinness in children and adolescents: international survey. BMJ 335:194. https://doi.org/10.1136/bmj.39238.399444.55
Czarnota J, Gennings C, Colt JS et al (2015a) Analysis of environmental chemical mixtures and non-hodgkin lymphoma risk in the NCI-SEER NHL Study. Environ Health Perspect 123:965–970. https://doi.org/10.1289/ehp.1408630
Czarnota J, Gennings C, Wheeler DC (2015b) Assessment of weighted quantile sum regression for modeling chemical mixtures and cancer risk. Cancer Inform. https://doi.org/10.4137/CIN.S17295
De Bont J, Bennett M, León-Muñoz LM, Duarte-Salles T (2022) The prevalence and incidence rate of overweight and obesity among 2.5 million children and adolescents in Spain. Revista Española De Cardiología (english Edition) 75:300–307. https://doi.org/10.1016/j.rec.2021.07.002
Esteban M, Castaño A (2009) Non-invasive matrices in human biomonitoring: a review. Environ Int 35:438–449. https://doi.org/10.1016/j.envint.2008.09.003
Fábelová L, Vandentorren S, Vuillermoz C et al (2018) Hair concentration of trace elements and growth in homeless children aged <6years: Results from the ENFAMS study. Environ Int 114:318–325. https://doi.org/10.1016/j.envint.2017.10.012
Fan Y, Zhang C, Bu J (2017) Relationship between selected serum metallic elements and obesity in children and adolescent in the U.S. Nutrients 9:104. https://doi.org/10.3390/nu9020104
García-Villarino M, Signes-Pastor AJ, Karagas MR et al (2022) Exposure to metal mixture and growth indicators at 4–5 years A study in the INMA-Asturias cohort. Environ Res 204:112375. https://doi.org/10.1016/j.envres.2021.112375
Ge W, Liu W, Liu G (2020) The relationships between serum copper levels and overweight/total obesity and central obesity in children and adolescents aged 6–18 years. J Trace Elem Med Biol 61:126557. https://doi.org/10.1016/j.jtemb.2020.126557
Geier DA, Kern JK, Homme KG et al (2016) Thimerosal-containing hepatitis B vaccine exposure is highly associated with childhood obesity: a case-control study using the vaccine safety datalink. N Am J Med Sci 8:297. https://doi.org/10.4103/1947-2714.187148
Guerrero-Romero F, Rodríguez-Morán M (2006) Hypomagnesemia, oxidative stress, inflammation, and metabolic syndrome. Diabetes Metab Res Rev 22:471–476. https://doi.org/10.1002/dmrr.644
Hashemi M, Rajabi S, Eghbalian M et al (2023) Demographic and anthropometric characteristics and their effect on the concentration of heavy metals (arsenic, lead, chromium, zinc) in children and adolescents. Heliyon 9:e13621. https://doi.org/10.1016/j.heliyon.2023.e13621
HBM (Human Biomonitoring) Commission (2005) Standardization of substance contents in urine–creatinine. Statement of the Commission Human Biomonitoring of the Environmental Agency. Bundesgesundheitsbl Gesundheitsforsch Gesundheitsschutz 48:616–618
Heindel JJ, Blumberg B (2019) Environmental obesogens: mechanisms and controversies. Annu Rev Pharmacol Toxicol 59:89–106. https://doi.org/10.1146/annurev-pharmtox-010818-021304
Heindel JJ, Howard S, Agay-Shay K et al (2022) Obesity II: Establishing causal links between chemical exposures and obesity. Biochem Pharmacol 199:115015. https://doi.org/10.1016/j.bcp.2022.115015
Iavicoli I, Fontana L, Bergamaschi A (2009) The effects of metals as endocrine disruptors. J Toxicol Environ Health B Crit Rev 12:206–223. https://doi.org/10.1080/10937400902902062
Junqué E, Tardón A, Fernandez-Somoano A, Grimalt JO (2022) Environmental and dietary determinants of metal exposure in four-year-old children from a cohort located in an industrial area (Asturias, Northern Spain). Environ Res 214:113862. https://doi.org/10.1016/j.envres.2022.113862
Kafaei R, Tahmasbi R, Ravanipour M et al (2017) Urinary arsenic, cadmium, manganese, nickel, and vanadium levels of schoolchildren in the vicinity of the industrialised area of Asaluyeh. Iran Environ Sci Pollut Res Int 24:23498–23507. https://doi.org/10.1007/s11356-017-9981-6
Katzen-Luchenta J (2007) The declaration of nutrition, health, and intelligence for the child-to-be: adapted from the declaration of Olympia on nutrition and fitness, 28–29 May 1996 in Ancient Olympia, Greece, an Article by Artemis P. Simopoulos, MD, The Center for Genetics, Nutrition and Health, Washington, DC, USA. Nutr Health 19:85–102. https://doi.org/10.1177/026010600701900212
Kawakami T, Hanao N, Nishiyama K et al (2012) Differential effects of cobalt and mercury on lipid metabolism in the white adipose tissue of high-fat diet-induced obesity mice. Toxicol Appl Pharmacol 258:32–42. https://doi.org/10.1016/j.taap.2011.10.004
Komolova M, Bourque SL, Nakatsu K, Adams MA (2008) Sedentariness and increased visceral adiposity in adult perinatally iron-deficient rats. Int J Obes 32:1441–1444. https://doi.org/10.1038/ijo.2008.97
Kordas K, Roy A, Vahter M et al (2018) Multiple-metal exposure, diet, and oxidative stress in Uruguayan school children. Environ Res 166:507–515. https://doi.org/10.1016/j.envres.2018.06.028
Lamerz A, Kuepper-Nybelen J, Wehle C et al (2005) Social class, parental education, and obesity prevalence in a study of six-year-old children in Germany. Int J Obes 29:373–380. https://doi.org/10.1038/sj.ijo.0802914
Lewis RC, Meeker JD, Basu N et al (2018) Urinary metal concentrations among mothers and children in a Mexico City birth cohort study. Int J Hyg Environ Health 221:609–615. https://doi.org/10.1016/j.ijheh.2018.04.005
Lin H-C, Huang Y-K, Shiue H-S et al (2014) Arsenic methylation capacity and obesity are associated with insulin resistance in obese children and adolescents. Food Chem Toxicol 74:60–67. https://doi.org/10.1016/j.fct.2014.08.018
Liu B, Lehmler H-J, Sun Y et al (2017) Bisphenol A substitutes and obesity in US adults: analysis of a population-based, cross-sectional study. Lancet Planet Health 1:e114–e122. https://doi.org/10.1016/S2542-5196(17)30049-9
Lucchini RG, Aschner M, Yangho K, Šarić M (2015) Chapter 45—Manganese. In: Nordberg GF, Fowler BA, Nordberg M (eds) Handbook on the toxicology of metals, 4th edn. Academic Press, San Diego, pp 975–1011
Marill KA (2004) Advanced statistics: linear regression, Part II: multiple linear regression. Acad Emerg Med 11:94–102. https://doi.org/10.1197/j.aem.2003.09.006
Martinez-Morata I, Sobel M, Tellez-Plaza M et al (2023) A state-of-the-science review on metal biomarkers. Curr Environ Health Rep. https://doi.org/10.1007/s40572-023-00402-x
Molina-Villalba I, Lacasaña M, Rodríguez-Barranco M et al (2015) Biomonitoring of arsenic, cadmium, lead, manganese and mercury in urine and hair of children living near mining and industrial areas. Chemosphere 124:83–91. https://doi.org/10.1016/j.chemosphere.2014.11.016
Moreda-Piñeiro J, Alonso-Rodríguez E, Romarís-Hortas V et al (2012) Assessment of the bioavailability of toxic and non-toxic arsenic species in seafood samples. Food Chem 130:552–560. https://doi.org/10.1016/j.foodchem.2011.07.071
Nasab H, Rajabi S, Eghbalian M et al (2022) Association of As, Pb, Cr, and Zn urinary heavy metals levels with predictive indicators of cardiovascular disease and obesity in children and adolescents. Chemosphere 294:133664. https://doi.org/10.1016/j.chemosphere.2022.133664
Notario-Barandiaran L, Irizar A, Begoña-Zubero M et al (2023) Association between mediterranean diet and metal(loid) exposure in 4–5-year-old children living in Spain. Environ Res 233:116508. https://doi.org/10.1016/j.envres.2023.116508
Padilla MA, Elobeid M, Ruden DM, Allison DB (2010) An examination of the association of selected toxic metals with total and central obesity indices: NHANES 99–02. Int J Environ Res Public Health 7:3332–3347. https://doi.org/10.3390/ijerph7093332
Padmavathi IJN, Rao KR, Venu L et al (2010) Chronic maternal dietary chromium restriction modulates visceral adiposity: probable underlying mechanisms. Diabetes 59:98–104. https://doi.org/10.2337/db09-0779
Peake M, Whiting M (2006) Measurement of serum creatinine–current status and future goals. Clin Biochem Rev 27:173–184
Protano C, Astolfi ML, Canepari S, Vitali M (2016) Urinary levels of trace elements among primary school-aged children from Italy: the contribution of smoking habits of family members. Sci Total Environ 557–558:378–385. https://doi.org/10.1016/j.scitotenv.2016.03.073
R Core Team (2021) A language and environment for statistical computing. r foundation for statistical computing, Vienna, Austria. https://www.R-project.org/
Renzetti S, Curtin P, Just A, et al (2021) gWQS: generalized wrighted quantile sum regression. https://CRAN.R-project.org/package=gWQS
Roca M, Sánchez A, Pérez R et al (2016) Biomonitoring of 20 elements in urine of children. Levels Predic Expos Chemosph 144:1698–1705. https://doi.org/10.1016/j.chemosphere.2015.10.008
Rodríguez-Barranco M, Lacasaña M, Aguilar-Garduño C et al (2013) Association of arsenic, cadmium and manganese exposure with neurodevelopment and behavioural disorders in children: a systematic review and meta-analysis. Sci Total Environ 454–455:562–577. https://doi.org/10.1016/j.scitotenv.2013.03.047
Rosin A (2009) The long-term consequences of exposure to lead. Israel Med Assoc J 11:689–694
Rothenberg SE, Korrick SA, Fayad R (2015) The influence of obesity on blood mercury levels for U.S. non-pregnant adults and children: NHANES 2007–2010. Environ Res 138:173–180. https://doi.org/10.1016/j.envres.2015.01.018
Scinicariello F, Buser MC, Mevissen M, Portier CJ (2013) Blood lead level association with lower body weight in NHANES 1999–2006. Toxicol Appl Pharmacol 273:516–523. https://doi.org/10.1016/j.taap.2013.09.022
Shan Q (2022) Trend analysis of the association of urinary metals and obesity in children and adolescents. Chemosphere 307:135617. https://doi.org/10.1016/j.chemosphere.2022.135617
Shao W, Liu Q, He X et al (2017) Association between level of urinary trace heavy metals and obesity among children aged 6–19 years: NHANES 1999–2011. Environ Sci Pollut Res 24:11573–11581. https://doi.org/10.1007/s11356-017-8803-1
Shin Y-Y, Ryu I-K, Park M-J, Kim S-H (2018) The association of total blood mercury levels and overweight among Korean adolescents: analysis of the Korean National Health and Nutrition Examination Survey (KNHANES) 2010–2013. Korean J Pediatr 61:121–128. https://doi.org/10.3345/kjp.2018.61.4.121
Skuland SE (2015) Healthy eating and barriers related to social class. the case of vegetable and fish consumption in Norway. Appetite 92:217–226. https://doi.org/10.1016/j.appet.2015.05.008
Su C-T, Lin H-C, Choy C-S et al (2012) The relationship between obesity, insulin and arsenic methylation capability in Taiwan adolescents. Sci Total Environ 414:152–158. https://doi.org/10.1016/j.scitotenv.2011.10.023
Tang-Péronard JL, Andersen HR, Jensen TK, Heitmann BL (2011) Endocrine-disrupting chemicals and obesity development in humans: a review. Obes Rev 12:622–636. https://doi.org/10.1111/j.1467-789X.2011.00871.x
Tascilar ME, Ozgen IT, Abaci A et al (2011) Trace elements in obese Turkish children. Biol Trace Elem Res 143:188–195. https://doi.org/10.1007/s12011-010-8878-8
Téllez-Rojo MM, Bautista-Arredondo LF, Trejo-Valdivia B et al (2019) Reporte nacional de niveles de plomo en sangre y uso de barro vidriado en población infantil vulnerable. Salud Pública de México 61:787–797. https://doi.org/10.21149/10555
Tetsuka J, Matsukawa T, Yokoyama K et al (2022) Effects of trace elements on anthropometric characteristics of children: cobalt and childhood body mass index. Juntendo Med J 68:251–260. https://doi.org/10.14789/jmj.JMJ21-0043-OA
Tinkov AA, Aschner M, Ke T et al (2021) Adipotropic effects of heavy metals and their potential role in obesity. Fac Rev 10:32. https://doi.org/10.12703/r/10-32
Vogel N, Murawski A, Schmied-Tobies MIH et al (2021) Lead, cadmium, mercury, and chromium in urine and blood of children and adolescents in Germany—Human biomonitoring results of the German Environmental Survey 2014–2017 (GerES V). Int J Hyg Environ Health 237:113822. https://doi.org/10.1016/j.ijheh.2021.113822
Vrijheid M, Fossati S, Maitre L et al (2020) Early-life environmental exposures and childhood obesity: an exposome-wide approach. Environ Health Perspect 128:067009. https://doi.org/10.1289/EHP5975
Weber JA, Van Zanten AP (1991) Interferences in current methods for measurements of creatinine. Clin Chem 37:695–700
WHO (2018) WHO European Childhood Obesity Surveillance Initiative (COSI). Factsheet. Highlights 2015–2017.
WHO (2021) Obesity and overweight. https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight. Accessed 13 Aug 2022
Wildman RE, Mao S (2001) Tissue-specific alterations in lipoprotein lipase activity in copper-deficient rats. Biol Trace Elem Res 80:221–229. https://doi.org/10.1385/BTER:80:3:221
Yanoff LB, Menzie CM, Denkinger B et al (2007) Inflammation and iron deficiency in the hypoferremia of obesity. Int J Obes 31:1412–1419. https://doi.org/10.1038/sj.ijo.0803625
Zhang X, Cui X, Lin C et al (2017) Reference levels and relationships of nine elements in first-spot morning urine and 24-h urine from 210 Chinese children. Int J Hyg Environ Health 220:227–234. https://doi.org/10.1016/j.ijheh.2016.10.013
Zhang Y, Dong T, Hu W et al (2019) Association between exposure to a mixture of phenols, pesticides, and phthalates and obesity: comparison of three statistical models. Environ Int 123:325–336. https://doi.org/10.1016/j.envint.2018.11.076
Acknowledgements
This study would have not been possible without the participation of the children and their parents.
Funding
Funding for open access publishing: Universidad de Granada/CBUA. This research was carried out within the framework of the Project PI20/01278, funded by Instituto de Salud Carlos III (ISCIII) and co-funded by the European Regional Development Fund (ERDF/FEDER), FEDER-Consejería de Salud y Familias, Junta de Andalucía PE-0250–2019, FEDER/Junta de Andalucía-Consejería de Transformación Económica, Industria, Conocimiento y Universidades/Proyecto P18-RT-4247 and INTRAIBS-2021–02 funded by ibs.GRANADA.
Author information
Authors and Affiliations
Contributions
Inmaculada Salcedo-Bellido: Data curation, Methodology, Formal analysis, Writing -original draft, Writing—review & editing, Helga Castillo Bueno: Investigation, Writing—review & editing, Pablo Olmedo: Methodology, Writing—review & editing, Fernando Gil: Methodology, Writing—review & editing, Francisco M. Ocaña-Peinado: Formal analysis, Writing—review & editing, Lourdes Rodrigo: Investigation, Methodology, Supervision, Writing—review & editing, Ana Rivas: Conceptualization, Project administration, Funding acquisition, Supervision, Writing—review & editing.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no relevant financial or non-financial interests.
Ethics Approval
The Ethics Committee of all participating centers approved the study protocol, in conformity to the principles of the Declaration of Helsinki. The study protocol was approved by the Ethics Committee of the University of Granada.
Consent to Participate
All parents or legal guardians of the study participants were fully informed about the study objectives and signed an informed consent.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Salcedo-Bellido, I., Castillo Bueno, H., Olmedo, P. et al. Metal (loid) Exposure and Overweight and Obesity in 6–12-Year-Old Spanish Children. Expo Health (2024). https://doi.org/10.1007/s12403-024-00638-4
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12403-024-00638-4