Abstract
Exposure of reproductive-age women to toxic trace elements warrants attention because of their negative effects. This study aimed to investigate the levels of arsenic (As), cadmium (Cd), mercury (Hg), chromium (Cr), and lead (Pb) in the blood of 837 Taiwanese childbearing-age women and establish the correlation between their dietary pattern and heavy metal concentration. The concentrations of Cd and Pb were significantly higher in nonpregnant women than in pregnant women (Cd: 2.41 µg/L vs. 2.12 µg/L; Pb: 0.83 µg/dL vs. 0.73 µg/dL), whereas the concentration of Cr was significantly lower in nonpregnant women than in pregnant women (Cr: 0.98 µg/L vs. 1.05 µg/L). Otherwise, no significant differences in As (9.02 µg/L vs. 9.51 µg/L) and Hg (3.71 µg/L vs. 3.79 µg/L) were found between the nonpregnant and pregnant women. Overall, the blood metal concentrations of Cd and Hg showed a decreasing trend in the different pregnancy stages. The levels of As and Hg were highly correlated with seafood intake. Finally, lifestyle habits, such as burning incense usage and Chinese herb intake may contribute to metal accumulation in maternal or reproductive-age women. Preventive risk communication and educational strategies should be applied to these subgroups in view of food safety and public health concern.
Graphical Abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Heavy metals widely exist in our environment because they are difficult to degrade. Heavy metals may cause adverse health effects when converted into the food chain and accumulate in our body (Brender et al. 2006; Kolachi et al. 2011; Lin et al. 2014). Recent studies have revealed that lead (Pb) and mercury (Hg) exposure may contribute to an increased prevalence of dyslipidemia (Liu et al. 2022) whereas cadmium (Cd) exposure is related to the prevalence of coronary heart disease and stroke burden (Kim et al. 2022) in adults. For glucose balance, metals are involved in glucose metabolism, and arsenic (As) and Pb may alter glycemic regulation and are related to diabetes in women (Wang et al. 2020a). In addition, early pregnancy with exposure to barium (Ba) and Hg is associated with altered post-load glucose concentrations in later pregnancy (Zheng et al. 2021), and Cd has been related to diabetes (Filippini et al. 2022) and gestational diabetes (Zhou et al. 2022). A review article has highlighted that Pb, Cd, As, and Hg may produce reactive oxygen species; induce oxidative stress in the trophoblastic placental tissue; and lead to preterm birth (Singh et al. 2020). In addition, a case–control study showed that pregnant women exposed to chromium (Cr) and As exhibit an excessive association with preeclampsia (Wang et al. 2020c). Reducing the exposure of women to metals, especially Cd and Pb, during pregnancy is important because of its possible health impact (Osorio-Yanez et al. 2021). Pb exposure is harmful to the reproductive system in male and female; it can cause female infertility, abortion, premature birth, gestational hypertension, and inhibit embryonic growth and development (ATSDR 2019). In addition, As exposure of pregnant women might cause abortion and influence embryonic development (Vahter 2008). Meanwhile, a cohort study reported that maternal exposure to Hg, Cd, and cesium (Cs) in the first trimester is related to thyroid-stimulating hormone levels in mothers and newborns (Wang et al. 2020b). Moreover, the levels of As, Cd, Pb, Hg, aluminum (Al), tin (Sn), manganese (Mn), and antimony (Sb) are significantly higher in offspring/newborns with neural tube defects than in those without these conditions (Brender et al. 2006; Ovayolu et al. 2020; Ozel et al. 2019). Therefore, measuring the blood metal concentrations in reproductive-age women is important to promote the health of pregnant women and their newborns.
Human biomonitoring (HBM) is a method of measuring the chemicals, metabolites, or degraded compounds in body fluids, including blood, urine, saliva, sweat, excrement, hair, and nail; as it represents the total burden of the body after exposure and reflects the overall metabolism of the body dependent on personal exposure, metabolization, and excretion (Ladeira and Viegas 2016; WHO 2015). The National Health and Nutrition Examination Survey (NHANES) is the largest national survey for HBM in the United States, and their data showed an over 70% decrease in blood Pb levels in the general population in the past 20 years (CDC 2017). The Canadian Health Measures Survey also observed decreasing trends for Pb, Cd, Hg, and methylmercury in blood and As in urine from 2007 to 2019 (Health Canada 2013; 2015; 2019; 2021). In Europe, a study called “Consortium to Perform Human Biomonitoring on a European Scale” in 17 countries performed HBM in the urine of 1844 children (5–11 years of age) and their mothers in 2011–2012. Results showed that their biomarker concentrations are below the health-based guidance values and that the Hg and Cd levels are highly correlated between the mothers and their children (Joas et al. 2012; Den Hond et al. 2015). In Japan, a follow-up study measured Pb, Hg, Cd, Mn, and selenium (Se) concentrations in the blood of 20,000 pregnant women, and results showed that the blood Pb levels decreased 5–10 folds in the past 25 years (Nakayama et al. 2019). In China, the levels of Hg, Cd, and As in the plasma of women showed a 30%–40% decrease from 2005 to 2012 in a historically contaminated area in China (Jin et al. 2020). Overall, a decreasing trend of blood element levels was found in most countries worldwide.
The factors influencing high Hg body burden are agriculture and high-frequency water product consumption; high consumption of fresh fruits and eggs is also associated with high levels of Cd in the plasma (Jin et al. 2020). High seafood consumption contributes to elevated levels of As and Hg in the ovarian follicular fluid (FF) of women undergoing in vitro fertilization; in addition, diet source might contribute to As, Hg, Cd, and Pb in FF (Butts et al. 2020). A previous study showed that fish consumption is a predictor of blood Hg level (Lee et al. 2012; Nakayama et al. 2019). In addition, maternal age and alcohol drinks are predictors of blood Pb, whereas maternal age and smoking are predictors of Cd concentration (Nakayama et al. 2019). Overall, the dietary pattern of pregnant women is responsible for metal accumulation in maternal exposure and affects the performance of newborns.
The primary public health concern is related to maternal exposure to metals and how the high exposure to metals can be linked to offspring development, especially when these metals are ingested and bio-accumulated through different types of food, such as fish and other marine foods, which are popular and frequently consumed in Taiwan. Therefore, this study aims to evaluate the potential factors associated with metal accumulation in mothers or reproductive-age women. Preventive risk communication and educational strategies can be applied to these subgroups in view of food safety and public health concerns.
Materials and Methods
Participant Enrollment
This study recruited 849 women of reproductive-age between November 2019 and October 2020 from four medical centers and two universities around Taiwan. Inclusion criteria included living in the current area over one year and aged 20–49 years on the day of the questionnaire interview. Exclusion criteria included those with occupational exposure or living near industrial areas with metal exposure. Setting of this exclusion criteria according to the occupational and environmental exposures have been well-known to be linked to bioaccumulation in human body (Jin et al. 2020), therefore, the aim of this study was to try to find the major influencing factors in metals accumulation through food consumption. Finally, 837 women completed the questionnaire interviews and blood samplings in this study.
A stratified random sampling design was applied to recruit participants. The stratification was based on geographic regions in Taiwan, which were divided into northern, central, southern, and eastern Taiwan. The sample size of each region was inferred from the proportion of women aged 20–49 years in stratified regions to the total female population in Taiwan, which was recorded in the Ministry of Health and Welfare, Taiwan. Oversampling was implemented in the eastern region because of the relatively low density of women.
The participants enrolled in this study signed the informed consent form, which was approved by the Ethics Committee of the National Cheng Kung University Hospital (Tainan, Taiwan, encoded: A-ER-108-433), the Chung Shan Medical University Hospital (Taichung, Taiwan, encoded: CS2-20022), the Taipei Veterans General Hospital (Taipei, Taiwan, encoded: 2020-03-008B), and Hualien Tzu Chi Hospital, Buddhist Tzu Chi Medical Foundation (Hualien, Taiwan, encoded: IRB109-077-B). The questionnaire interviews and blood samplings were conducted for each participant.
Interviewer-Administered Questionnaire
Demographic information was obtained using face-to-face or online questionnaires. The questionnaires consisted of five parts: personal characteristics (age, occupation, education, pregnant status, etc.), pollution sources, personal living habits (cigarette use, secondhand smoke exposure, burning incense, cosmetics, amalgam, etc.), dietary pattern within a month, and family and personal disease histories. A food frequency questionnaire was used to collect consumption information based on intake frequency, such as how many times daily, weekly, or monthly. The fish item was grouped as large-sized fish (tuna, salmon, swordfish, codfish, etc.), small-sized fish, freshwater fish (mackerel, pomfret, etc.), cephalopods (squid, cuttlefish, etc.), shrimp, shellfish, oysters, and echinoderms (sea cucumber, etc.). Quantitative data for intake of milk and other drinks were calculated based on a cup, spoon for oil and sauces, and bowls for other food items.
Whole Blood Sampling and Analysis of Blood Heavy Metal Concentrations
Blood samples (8 mL) of the participants were collected by licensed nurses in vacuum tubes with K2EDTA and then stored at − 20 °C until analysis. The metal levels in the blood samples, including As, Cd, Cr, Hg, and Pb, were measured through inductively coupled plasma mass spectrometry (ICP-MS, Nexion 2000, PerkinElmer). A final volume of 4 mL was obtained by mixing 200 μL blood samples with 0.1% Triton X-100 and 0.1% HNO3 and then centrifuging at 3,000 rpm for 10 min. The ICP-MS system was used to analyze the metal concentrations of the suspension. Matrix-matched calibration curves ranging from 0.01 to 20 μg/L were built for blood samples in the ICP-MS analysis to reduce the matrix effects in the blood samples. The standard reference material 995d (SRM 995d), produced by the US National Institute of Science and Technology, was applied to evaluate the accuracy of determining essential elements of human blood. In this study, the recovery rates for the five metals were approximately 70%–120% in the applied SRM.
Statistical Analysis
The demographic characteristics were summarized using mean (standard deviation, SD) and number (%) for continuous and categorical variables, respectively. The distribution of blood metals levels was presented as median and range. Independent sample t and Chi-square tests were applied to compare the demographic characteristics between pregnant and nonpregnant women. The Mann–Whitney U test and Kruskal–Wallis test were used to assess the differences in blood metal distributions between different groups. Spearman’s rank correlation coefficients were used to evaluate correlations among different metal levels and continuous variables, including ages and dietary intake. Data were analyzed using IBM SPSS for Windows version 20.0 (IBM, Armonk, NY) and R 4.1.1.
Results
The demographics of 837 women are shown in Table 1. Four hundred and fifty-one (53.9%) participants were pregnant women. Of these women, 78.4% were aged between 25 and 39 years (average age = 33.2 years), and 44%, 24%, and 26% were living in northern, central, and southern Taiwan, respectively. In addition, 55% responded that they lived in the vicinity of polluted sources (vehicle exhaust, industrial emissions, night markets, etc.) within 2.5-km radius. Meanwhile, 34% of the participants responded that they used incense burning, 64% used cosmetic products, and 35% received dental amalgam. Significant differences in age, living areas, occupations, educational attainment, and incense burning between pregnant and nonpregnant were found in this study.
The concentrations of Cd and Pb were significantly higher in nonpregnant women than in pregnant women (Cd: 2.41 µg/L vs.2.12 µg/L; Pb: 0.83 µg/dL vs. 0.73 µg/dL), whereas the concentration of Cr was significantly lower in nonpregnant women than in pregnant women (Cr: 0.98 µg/L vs. 1.05 µg/L). Otherwise, no significant differences in As (9.02 µg/L vs. 9.51 µg/L) and Hg (3.71 µg/L vs. 3.79 µg/L) were found between the nonpregnant and pregnant women (Table 2).
Table S2 shows the distribution of blood metal levels for childbearing-age women eating-specific food or not. The As and Hg levels were both significantly higher in the childbearing-age women eating brown rice, grain rice, offal, fish/seafood, leaf vegetables, fruits, and marine plants than in those who did not eat these types of food. Higher Pb levels were found in the childbearing-age women eating small fish, seafood, offal, leaf vegetables, and marine plants than those who did not eat these types of food. Additionally, the current study found significant differences in blood Pb levels between those consuming Chinese medicine and those not consuming these medicines. Meanwhile, no significant differences were found in the Cd and Cr blood metal levels of childbearing-age women with different dietary patterns. Furthermore, the blood metal levels of childbearing-age women from low to high consumption in specific food are shown in Fig. 1. For As accumulation, the significant differences of As levels among their consumption quantity from less than Q1 to over Q3 groups were only found for brown rice, large fish, and marginal for freshwater fish; for Hg, the significant differences were found for large fish, small fish, freshwater fish, and cephalopods; for Pb, the marginal significant difference was only found for leaf crops in reproductive-age women.
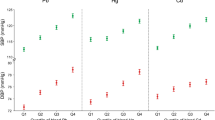
The distribution of blood metal levels in alternative trimester pregnant stages were further evaluated (Fig. 2). A significantly decreasing trend was found for Cd and Hg, and significant differences in Cd, Hg, and Pb were found among the alternative pregnancy stages. No significant differences in As and Cr were observed. Furthermore, As and Hg highly correlated with seafood intake (Fig. 3).
Discussion
Element Concentrations in Pregnant and Nonpregnant Women
As, Hg, Pb, Cd, and Cr are not only harmful to adults and children but also affect the fetus and infants. Exposure to these metals might be associated with birth outcomes, such as low birth weight, low birth length, spontaneous abortion, stillbirth, preterm birth, and head circumference (Cempaka et al. 2019; Gokoel et al. 2020; Milton et al. 2017; Murcia et al. 2016; Nyanza et al. 2020; Shih et al. 2021, 2020; Wang et al. 2021; Xia et al. 2016). For instance, in a previous study, the placenta of participants that underwent spontaneous abortion were found to contain significantly higher levels of As, Cd, Hg, Pb, Sb, Sn, Se, Mn, and cobalt (Co) compared with that of the control group (Baser et al. 2020). However, with exception of Cd, all levels were below the risk levels defined by the health authority (35 µg/L for As, 10 µg/L for Cr, 10 µg/L for Hg, 2 µg/L for Cd, and 10 µg/dL for Pb in blood of adults) (Rios et al. 2020). The effects of metals on pregnant women with high burden must be considered because those metals may be delivered to the embryo through umbilical cord blood and placenta during pregnancy (Li et al. 2019; Rudge et al. 2009) and cause neural tube and birth size defects in offspring (Araujo et al. 2020; Brender et al. 2006; Kippler et al. 2012). For Taiwanese infants, breastfeeding has been promoted by the Health Promotion Administration, Ministry of Health and Welfare since 2001 (Chen et al. 2020). About 40% and 29% of Taiwanese mothers reported exclusive breastfeeding at one month and two months postpartum, respectively (Chang et al. 2019). Although the advantages of breastfeeding are well-known, heavy metal toxicity has received increased research attention (Chao et al. 2014; Dix-Cooper and Kosatsky 2018; Jeong et al. 2017b; Vahidinia et al. 2019; Lin et al. 2022).
In this study, we found significant differences in blood Cd, Cr, and Pb levels and no significant differences in blood As and Hg levels between pregnant and nonpregnant women. The current results were partially consistent with those of Gong et al. (2021), who also reported significant differences in Cr and Pb (but not Cd) and no significant differences in As and Hg in the whole blood between 192 healthy pregnant women and 210 healthy nonpregnant women in China. Another study found that pregnant women in the US NHANES who have been exposed to several trace metals showed lower levels of blood Cd and Pb than nonpregnant women (Watson et al. 2020), which were similar to the current results. By contrast, the present results showed that the pregnant women had higher levels of Hg, along with As and Cr, than the nonpregnant women as opposed to the findings of Watson et al. (2020). However, the trend of increasing Hg levels by increasing their dental amalgams cannot be evidenced in the childbearing-age women (Table S1).
Blood Metal Levels Compared with Other Countries
We compared the blood essential element/metals in pregnant and nonpregnant women with measurements obtained from other countries (Table S3). The average level of As in Taiwan was slightly lower than that in the early report of Lei et al. (2015) in Taiwan, Brazil (Araujo et al. 2020), China (Wang et al. 2020c), and Iran (Vigeh et al. 2015), whereas the measured levels in this study were higher than those from other countries (Bank-Nielsen et al. 2019; Ettinger et al. 2017). The measurement data on Cd levels in the current study were lower than those in Egypt (Motawei and Gouda 2016) but higher than those in other countries (Lee et al. 2020; Ma et al. 2021; Pesce et al. 2021). The levels of Cr in the current study were lower than those obtained from China (Kim et al. 2020; Wang et al. 2020a) and Greenland (Bank-Nielsen et al. 2019). Our data on Hg were comparable with those obtained from Greenland (Bank-Nielsen et al. 2019), South Korea (Jeong et al. 2017a; Kim et al. 2013; Lee et al. 2021a,b, 2020; Song et al. 2016), and Japan (Kobayashi et al. 2019; Ma et al. 2021; Nakayama et al. 2019) but higher than those in other countries. For Pb, the current measured data were not higher than those found in other countries and slightly lower than the 2015 data in Taiwan (Lei et al. 2015). The blood metal concentrations varied in childbearing-age women worldwide. Overall, the levels of As, Hg, and Cd were higher in women from Taiwan than in those from other countries.
Association of Dietary Intake and Blood Metal Levels
Metals are emitted to the environment artificially because of industrialization and urbanization, and those metals might accumulate in humans through the soil, groundwater, atmosphere, and the food chain (Al-Saleh and Abduljabbar 2017). A cross-sectional study reported that age and body mass index are significantly negatively associated with plasma As and Cd (Ugwuja et al. 2020). Therefore, the different industrial developments in residential areas and the different demographic data of participants should be considered simultaneously.
As and Hg showed a similar accumulation trend in fish and seafood, and these metals accumulated in the pregnant women in this study. Given the high accumulation of inorganic As in crops and organic As in fish and seafood, crop intake was regarded as the main contributor and fish/seafood intake as the second contributor to As exposure (Ma et al. 2008; Nriagu and Lin 1995; Park and Lee 2013; Williams et al. 2007). On the other hand, previous studies have also shown that fish and crop are the main contributors to Hg exposure (Jiang et al. 2012; Li et al. 2010; Mahaffey et al. 2009), and the long sea line between Japan and Taiwan might be the major cause for the higher blood Hg levels of the residents eating large amounts of fish and seafood than those living in western countries (Bocca et al. 2020; de Figueiredo et al. 2020; Donohue et al. 2018; Park and Lee 2013). Furthermore, we found that Taiwanese breast milk donors who reported using incense had higher average breast milk levels of Hg and Pb compared with those that do not use incense in our recently published findings (Lin et al. 2022). This can be supported with the result of another study showing that incense burning affects the indoor Hg level (Shen et al. 2017). Thus, we suspect that incense usage might be another influencing factor for Hg exposure in Taiwanese reproductive-age women. Aside from the above different ways, human activity, such as coal-fired power plants, industrial processes, and coal combustions, are another major cause of Hg release and exposure. Hg released can enter aquatic and terrestrial systems, and the emitted inorganic Hg can be transformed to methyl Hg by bacteria, which can be absorbed by organisms, such as fish and crustaceans (Erickson and Lin 2015). Therefore, consumption of these marine organisms may result in the exposure and accumulation of methyl Hg in human bodies. Hg exposure causes neurological and immunological dysfunctions in adults and in development stage (Bjørklund et al. 2017; WHO 2017); hence, prenatal Hg exposure might cause adverse effects on pregnancy outcome (Vigeh et al. 2018). Overall, pregnant women and women of reproductive-age should avoid Hg exposure.
Pb is harmful in the central nervous system (Finkelstein et al. 1998; Zhou et al. 2020), renal function (Finkelstein et al. 1998), cardiovascular disease (Zheng et al. 2019), reproductive system (Sudjarwo and Giftania Wardani Sudjarwo 2017), and blood system (Alvarez-Ortega et al. 2019). The well-known sources of Pb exposure are mining, battery manufacturing, drinking water tubes, paint, makeup, and other consumer products (Cardoso et al. 2014; Nriagu et al. 1996; Piomelli 2002). Therefore, heavy metals can be found in the soil, water, air, and the food chain after disposal of those products. The exposure of Pb from daily life and occupation may enter the human body through inhalation of Pb particles discharged from industrial activities or ingestion of contaminated drinking water and food, which can be harmful to human health, especially for susceptible populations, such as child, infants, and pregnant women (Al Osman et al. 2019; Levallois et al. 2018). Furthermore, metal contaminants including As, Cd, Hg, and Pb are also regularly present in Chinese herbs according to previous findings (Cooper et al. 2007; Ernst and Coon 2001; Wang et al. 2019). It is noteworthy that a significantly higher Pb level in childbearing-age women consuming Chinese medicine was found in this study although the blood Pb levels of these women were not that high. Therefore, preventing the amount of Chinese medicine intake should still be given attention, especially for pregnant women.
Meanwhile, for Cd exposure, previous reports have mentioned that the main exposure route is tobacco exposure and food consumption in nonoccupational exposure (Järup 2003; UNEP 2010). However, Garner and Levallois (2016) reported that age and cigarette usage but not dietary intake are the major factors influencing Cd accumulation in Canadian population.
Association of Dietary Intake and Blood Metal Levels in Different Pregnancy Stages
A previous study collected urine samples during the first and third trimesters of pregnancy from 489 mothers living in a highly industrialized town in Spain. The concentrations of Mo, Se, Cd, Cs, and Sb were higher in the first than the third trimester, whereas the opposite trend was found for Co, Cu, and Zn (Fort et al. 2014). The decreasing Cd trend by the pregnancy stage was similar to the measurement of the current study. In contrast, the measured Cd level in 2738 pregnant Korean women was lower (1.41 µg/L) in the early pregnancy stage than in the later pregnancy stage (1.51 µg/L) (Jeong et al. 2017a). Furthermore, maternal blood Hg levels of Canadian and Japanese women also decreased significantly from first to third trimester of pregnancy (Morrissette et al. 2004; Vigeh et al. 2018), which is in accordance to the decreasing trend found in the current study. Meanwhile, for As, a previous study in Iran showed that the blood As levels increased from the first pregnancy stage to the third pregnancy stage (14.4 to 74.7 µg/L) (Vigeh et al. 2015), which is in opposition to our current findings that no significant differences in As levels were found among pregnancy stages. For Cr, our results agree with the findings of Gong et al. (2021), where no significant differences were found in the whole blood Cr concentrations among the different trimesters in Chinese pregnant women.
Furthermore, we assessed the correlations of metal levels of whole blood and their corresponding dietary intake of alternative foodstuffs. Results showed that the levels of As and Hg were highly correlated with seafood intake (Fig. 2), supporting the evidence that fish and seafood are the major contributors to As and Hg exposure (Jiang et al. 2012; Mahaffey et al. 2009; Park and Lee 2013; Williams et al. 2007). However, reasons responsible for the decreasing trends in Cd and Hg levels in the pregnancy stages of Taiwanese pregnant women are still unknown.
Strength and Limitations
The strength of this study was the recruitment of reproductive-age women, who may breastfeed their infants within a few hours after childbirth. Through this study, the participants can have knowledge of whether they have high metal accumulations and accordingly change their lifestyle or food consumption pattern to protect their infants from metal exposure through breastfeeding. However, one limitation of this study was that spot sample was obtained only once and the measured concentrations in the blood samples may not be enough to represent long-term exposure to all metals. Another limitation of this study was the potential issues of generalizability. In specific, we mainly focused on metal exposure through food consumption and excluded those with occupational or environmental exposures. However, the diet and lifestyle of those with occupational exposure versus those without may be different. To address these limitations, it is recommended for future studies that other human biological samples, such as urine or hair, can be additionally collected to assess metal exposure. In addition, metal exposure in reproductive-age women through other sources such as occupational and environmental exposures can be further clarified in future studies.
Conclusions
The concentrations of Cd and Pb were significantly higher in nonpregnant women than in pregnant women, whereas the concentration of Cr was significantly lower in nonpregnant women than in pregnant women. Although all levels in these childbearing-age women were lower than the risk levels defined by the health authority (except for Cd), the levels of As, Hg, and Cd were higher in women from Taiwan than in women from other countries. Moreover, the concentrations of As and Hg were highly correlated with seafood intake. Dietary diversification in addition to prevention of exposure is advocated (Ugwuja et al. 2020), and healthy diets and lifestyle, such as reducing burning incense usage and Chinese herb intake, should be advised for pregnant women. Prioritizing adequate nutrition and healthy dietary patterns in the risks and benefits of metals during pregnancy may play a key role for ensuring optimal health in a child. This topic warrants a follow-up in future studies.
Data Availability
The data set analysed and used during the current study is available with the corresponding author upon reasonable request.
Change history
08 June 2023
A Correction to this paper has been published: https://doi.org/10.1007/s12403-023-00571-y
02 October 2023
A Correction to this paper has been published: https://doi.org/10.1007/s12403-023-00606-4
References
Agency for Toxic Substances and Disease Registry (ATSDR), (2019) What is the biological fate of lead in the body? https://www.atsdr.cdc.gov/csem/leadtoxicity/biologic_fate.html#:~:text=Most%20inhaled%20lead%20in%20the,ultimately%2C%20in%20the%20feces. Accessed 11 March 2022
Al Osman M, Yang F, Massey IY (2019) Exposure routes and health effects of heavy metals on children. Biometals 32:563–573. https://doi.org/10.1016/j.ccr.2017.12.011
Al-Saleh I, Abduljabbar M (2017) Heavy metals (lead, cadmium, methylmercury, arsenic) in commonly imported rice grains (Oryza sativa) sold in Saudi Arabia and their potential health risk. Int J Hyg Environ Health 220:1168–1178. https://doi.org/10.1016/j.ijheh.2017.07.007
Alvarez-Ortega N, Caballero-Gallardo K, Olivero-Verbel J (2019) Toxicological effects in children exposed to lead: a cross-sectional study at the Colombian Caribbean coast. Environ Int 130:104809. https://doi.org/10.1016/j.envint.2019.05.003
Araujo MSD, Figueiredo ND, Camara VM, Asmus CIRF (2020) Maternal-child exposure to metals during pregnancy in Rio de Janeiro city, Brazil: the Rio birth cohort study of environmental exposure and childhood development (PIPA project). Environ Res 183:109155. https://doi.org/10.1016/j.envres.2020.109155
Bank-Nielsen PI, Long M, Bonefeld-Jørgensen EC (2019) Pregnant Inuit women’s exposure to metals and association with fetal growth outcomes: accept 2010–2015. Int J Environ Res Public Health 16(7):1171. https://doi.org/10.3390/ijerph16071171
Baser E, Kirmizi DA, Turksoy VA, Onat T, Caltekin MD, Kara M, Yalvac ES (2020) Environmental exposures in the etiology of abortion: placental toxic and trace element levels. Z Geburtshilfe Neonatol 224(06):339–347. https://doi.org/10.1055/a-1263-1698
Bjørklund G, Mutter J, Aaseth J (2017) Metal chelators and neurotoxicity: lead, mercury, and arsenic. Arch Toxicol 91(12):3787–3797. https://doi.org/10.1007/s00204-017-2100-0
Bocca B, Ruggieri F, Pino A, Rovira J, Calamandrei G, Mirabella F, Martinez MA, Domingo JL, Alimonti A, Schuhmacher M (2020) Human biomonitoring to evaluate exposure to toxic and essential trace elements during pregnancy. Part B: predictors of exposure. Environ Res 182:109108. https://doi.org/10.1016/j.envres.2019.109108
Brender JD, Suarez L, Felkner M, Gilani Z, Stinchcomb D, Moody K, Henry J, Hendricks K (2006) Maternal exposure to arsenic, cadmium, lead, and mercury and neural tube defects in offspring. Environ Res 101(1):132–139. https://doi.org/10.1016/j.envres.2005.08.003
Butts CD, Bloom MS, McGough A, Lenhart N, Wong R, Mok-Lin E, Parsons PJ, Galusha AL, Yucel RM, Feingold BJ, Browne RW, Fujimoto VY (2020) Seafood consumption is associated with higher follicular fluid arsenic (As) and mercury (Hg) concentrations in women undergoing in vitro fertilization (IVF). Environ Res 188:109753. https://doi.org/10.1016/j.envres.2020.109753
Cardoso OO, Julião FC, Alves RI, Baena AR, Díez IG, Suzuki MN, Celere BS, Nadal M, Domingo JL, Segura-Muñoz SI (2014) Concentration profiles of metals in breast milk, drinking water, and soil: relationship between matrices. Biol Trace Elem Res 160(1):116–122. https://doi.org/10.1007/s12011-014-0030-8
Cempaka AR, Tseng SH, Yuan KC, Bai CH, Tinkov AA, Skalny AV, Chang JS (2019) Dysregulated iron metabolism-associated dietary pattern predicts an altered body composition and metabolic syndrome. Nutrients 11(11):2733. https://doi.org/10.3390/nu11112733
Centers for Disease Control and Prevention (2017) About the National Health and Nutrition Examination Survey. https://www.cdc.gov/nchs/nhanes/about_nhanes.htm. Accessed 19 March 2022
Chang PC, Li SF, Yang HY, Wang LC, Weng CY, Chen KF, Chen W, Fan SY (2019) Factors associated with cessation of exclusive breastfeeding at 1 and 2 months postpartum in Taiwan. Int Breastfeed J 14:18. https://doi.org/10.1186/s13006-019-0213-1
Chao HH, Guo CH, Huang CB, Chen PC, Li HC, Hsiung DY, Chou YK (2014) Arsenic, cadmium, lead, and aluminium concentrations in human milk at early stages of lactation. Pediatr Neonatol 55(2):127–134. https://doi.org/10.1016/j.pedneo.2013.08.005
Chen CH, Chen RC, Chen MS, Chen LC, Wei HY (2020) Challenges and outcomes of using the ten steps to successful breastfeeding in the Mother-Baby Friendly Institute Program in Taiwan. J Hum Lact 36(1):187–191. https://doi.org/10.1177/0890334419839366
Cooper K, Noller B, Connell D, Yu J, Sadler R, Olszowy H, Golding G, Tinggi U, Moore MR, Myers S (2007) Public health risks from heavy metals and metalloids present in traditional Chinese medicines. J Toxicol Environ Health 70:1694–1699. https://doi.org/10.1080/15287390701434885
de Figueiredo ND, Araujo MS, Luiz RR, Camara VD, Jacob SD, dos Santos LMG, Vicentini SA, Asmus C (2020) Metal mixtures in pregnant women and umbilical cord blood at urban populations–Rio de Janeiro, Brazil. Environ Sci Pollut Res 27:40210–40218. https://doi.org/10.1007/s11356-020-10021-w
Den Hond E, Govarts E, Willems H, Smolders R, Casteleyn L, Kolossa-Gehring M, Schwedler G, Seiwert M, Fiddicke U, Castano A, Esteban M, Angerer J, Koch HM, Schindler BK, Sepai O, Exley K, Bloemen L, Horvat M, Knudsen LE, Joas A, Joas R, Biot P, Aerts D, Koppen G, Katsonouri A, Hadjipanayis A, Krskova A, Maly M, Morck TA, Rudnai P, Kozepesy S, Mulcahy M, Mannion R, Gutleb AC, Fischer ME, Ligocka D, Jakubowski M, Reis MF, Namorado S, Gurzau AE, Lupsa IR, Halzlova K, Jajcaj M, Mazej D, Tratnik JS, Lopez A, Lopez E, Berglund M, Larsson K, Lehmann A, Crettaz P, Schoeters G (2015) First steps toward harmonized human biomonitoring in Europe: demonstration project to perform human biomonitoring on a European scale. Environ Health Perspect 123(3):255–263. https://doi.org/10.1289/ehp.1408616
Dix-Cooper L, Kosatsky T (2018) Blood mercury, lead and cadmium levels and determinants of exposure among newcomer South and East Asian women of reproductive age living in Vancouver, Canada. Sci Total Environ 619–620:1409–1419. https://doi.org/10.1016/j.scitotenv.2017.11.126
Donohue A, Wagner CL, Burch JB, Rothenberg SE (2018) Blood total mercury and methylmercury among pregnant mothers in Charleston, South Carolina, USA. J Expo Sci Environ Epidemiol 28:494–504. https://doi.org/10.1038/s41370-018-0033-1
Erickson PR, Lin VS (2015) Research highlights: elucidation of biogeochemical factors influencing methylmercury production. Environ Sci Process Impacts 17:1708–1711. https://doi.org/10.1039/C5EM90037A
Ernst E, Coon JT (2001) Heavy metals in traditional Chinese medicines: a systematic review. Clin Pharm Therap 70(6):497–504. https://doi.org/10.1067/mcp.2001.120249
Ettinger AS, Arbuckle TE, Fisher M, Liang CL, Davis K, Cirtiu CM, Bélanger P, LeBlanc A, Fraser WD (2017) Arsenic levels among pregnant women and newborns in Canada: results from the Maternal-Infant Research on Environmental Chemicals (MIREC) cohort. Environ Res 153:8–16. https://doi.org/10.1016/j.envres.2016.11.008
Filippini T, Wise LA, Vinceti M (2022) Cadmium exposure and risk of diabetes and prediabetes: a systematic review and dose-response meta-analysis. Environ Int 158:106920. https://doi.org/10.1016/j.envint.2021.106920
Finkelstein Y, Markowitz ME, Rosen JF (1998) Low-level lead-induced neurotoxicity in children: an update on central nervous system effects. Brain Res Rev 27(2):168–176. https://doi.org/10.1016/S0165-0173(98)00011-3
Fort M, Cosin-Tomas M, Grimalt JO, Querol X, Casas M, Sunyer J (2014) Assessment of exposure to trace metals in a cohort of pregnant women from an urban center by urine analysis in the first and third trimesters of pregnancy. Environ Sci Pollut Res 21:9234–9241. https://doi.org/10.1007/s11356-014-2827-6
Garner R, Levallois P (2016) Cadmium levels and sources of exposure among Canadian adults. Health Rep 27(2):10–18
Gokoel AR, Zijlmans W, Covert HH, Abdoel Wahid F, Shankar A, MacDonald-Ottevanger MS, Hindori-Mohangoo AD, Wickliffe JK, Lichtveld MY, Harville EW (2020) Influence of prenatal exposure to mercury, perceived stress, and depression on birth outcomes in Suriname: results from the MeKiTamara study. Int J Environ Res Public Health 17(12):4444. https://doi.org/10.3390/ijerph17124444
Gong L, Yang Q, Liu CWB, Wang X, Zeng HL (2021) Assessment of 12 essential and toxic elements in whole blood of pregnant and non-pregnant women living in Wuhan of China. Biol Trace Elem Res 199:2121–2130. https://doi.org/10.1007/s12011-020-02337-y
Health Canada (2013) Second report on human biomonitoring of environmental chemicals in Canada: results of the Canadian Health Measures Survey Cycle 2 (2009–2011). https://www.canada.ca/en/health-canada/services/environmental-workplace-health/reports-publications/environmentalcontaminants/second-report-human-biomonitoring-environmental-chemicals-canada-health-canada-2013.html. Accessed 28 March 2022
Health Canada (2015) Third report on human biomonitoring of environmental chemicals in Canada. https://www.canada.ca/en/healthcanada/services/environmental-workplace-health/reports-publications/environmental-contaminants/third-report-human-biomonitoringenvironmental-chemicals-canada.html Accessed 28 March 2022
Health Canada (2019) Fifth report on human biomonitoring of environmental chemicals in Canada: results of the Canadian Health Measures Survey Cycle 5 (2016–2017). https://www.canada.ca/en/health-canada/services/environmental-workplace-health/reports-publications/environmentalcontaminants/fifth-report-human-biomonitoring.html. Accessed 28 March 2022
Health Canada (2021) Biomonitoring content summary for the Canadian Health Measures Survey: Cycles 1–6 (2007–2019). https://www.canada.ca/en/health-canada/services/environmental-workplace-health/reports-publications/environmental-contaminants/biomonitoringcontent-summary-canadian-health-measures-survey.html. Accessed 28 March 2022
Järup L (2003) Hazards of heavy metal contamination. Br Med Bull 68(1):167–182. https://doi.org/10.1093/bmb/ldg032
Jeong KS, Ha E, Shin JY, Park H, Hong YC, Ha M, Kim S, Lee SJ, Lee KY, Kim JH, Kim Y (2017a) Blood heavy metal concentrations in pregnant Korean women and their children up to age 5 years: Mothers’ and Children’s Environmental Health (MOCEH) birth cohort study. Sci Total Environ 605–606:784–791. https://doi.org/10.1016/j.scitotenv.2017.06.007
Jeong KS, Park H, Ha E, Shin J, Hong YC, Ha M, Park H, Kim BN, Lee B, Lee SJ, Lee KY, Kim JH, Kim Y (2017b) High maternal blood mercury level is associated with low verbal IQ in children. J Korean Med Sci 32(7):1097–1104. https://doi.org/10.3346/jkms.2017.32.7.1097
Jiang S, Shi C, Wu J (2011) (2012) Genotypic differences in arsenic mercury lead and cadmium in milled rice (Oryza sativa L.). Int J Food Sci Nutr. https://doi.org/10.3109/09637486636343
Jin L, YuZhang JRL, Ren AG (2020) Comparison of plasma concentrations of mercury, cadmium, and arsenic among women in 2005 and 2012 in a historically contaminated area in China. Biol Trace Elem Res 198:380–389. https://doi.org/10.1007/s12011-020-02075-1
Joas R, Casteleyn L, Biot P, Kolossa-Gehring M, Castano A, Angerer J, Schoeters G, Sepai O, Knudsen LE, Joas A, Horvat M, Bloemen L (2012) Harmonised human biomonitoring in Europe: activities towards an EU HBM framework. Int J Hyg Environ Health 215(2):172–175. https://doi.org/10.1016/j.ijheh.2011.08.010
Kim H, Kim KN, Hwang JY, Ha EH, Park H, Ha M, Kim Y, Hong YC, Chang N (2013) Relation between serum folate status and blood mercury concentrations in pregnant women. Nutrition 29(3):514–518. https://doi.org/10.1016/j.nut.2012.08.012
Kim SS, Xu X, Zhang Y, Zheng X, Liu R, Dietrich KN, Reponen T, Xie C, Sucharew H, Huo X, Chen A (2020) Birth outcomes associated with maternal exposure to metals from informal electronic waste recycling in Guiyu China. Environ in 137:105580. https://doi.org/10.1016/j.envint.2020.105580
Kim DW, Ock J, Moon KW, Park CH (2022) Association between Heavy Metal exposure and dyslipidemia among Korean adults: from the Korean National Environmental Health Survey, 2015–2017. Int J Environ Res Public Health. https://doi.org/10.3390/ijerph19063181
Kippler M, Tofail F, Gardner R, Rahman A, Hamadani JD, Bottai M, Vahter M (2012) Maternal cadmium exposure during pregnancy and size at birth: a prospective cohort study. Environ Health Persp 120(2):284–289. https://doi.org/10.1289/ehp.1103711
Kobayashi S, Kishi R, Saijo Y, Ito Y, Oba K, Araki A, Miyashita C, Itoh S, Minatoya M, Yamazaki K, Ait Bamai Y, Sato T, Yamazaki S, Nakayama SF, Isobe T, Nitta H (2019) Association of blood mercury levels during pregnancy with infant birth size by blood selenium levels in the Japan Environment and Children’s Study: a prospective birth cohort. Environ Int 125:418429. https://doi.org/10.1016/j.envint.2019.01.051
Kolachi NF, Kazi TG, Afridi HI, Kazi N, Khan S, Kandhro GA, Shah AQ, Baig JA, Wadhwa SK, Shah F, Jamali MK, Arain MB (2011) Status of toxic metals in biological samples of diabetic mothers and their neonates. Biol Trace Elem Res 143:196–212. https://doi.org/10.1007/s12011-010-8879-7
Ladeira C, Viegas S (2016) Human biomonitoring—an overview on biomarkers and their application in occupational and environmental health. Biomonitoring 3:15–24. https://doi.org/10.1515/bimo-2016-0003
Lee CC, Chang JW, Huang HY, Chen HL (2012) Factors influencing blood mercury levels of inhabitants living near fishing areas. Sci Total Environ 424:316–321. https://doi.org/10.1016/j.scitotenv.2012.01.049
Lee S, Hong YC, Park H, Kim Y, Ha M, Ha E (2020) Combined effects of multiple prenatal exposure to pollutants on birth weight: the Mothers and Children’s Environmental Health (MOCEH) study. Environ Res 181:108832. https://doi.org/10.1016/j.envres.2019.108832
Lee KS, Kim KN, Ahn YD, Choi YJ, Cho J, Jang Y, Lim YH, Kim JI, Shin CH, Lee Y (2021a) Prenatal and postnatal exposures to four metals mixture and IQ in 6-year-old children: a prospective cohort study in South Korea. Environ Int 157:106798. https://doi.org/10.1016/j.envint.2021.106798
Lee S, Park SK, Park H, Lee W, Kwon JH, Hong YC, Ha M, Kim Y, Lee B, Ha E (2021b) Prenatal heavy metal exposures and atopic dermatitis with gender difference in 6-month-old infants using multipollutant analysis. Environ Res 195:110865. https://doi.org/10.1016/j.envres.2021.110865
Lei HL, Wei HJ, Ho HY, Liao KW, Chien LC (2015) Relationship between risk factors for infertility in women and lead, cadmium, and arsenic blood levels: a cross-sectional study from Taiwan. BMC Public Health 15:1220. https://doi.org/10.1186/s12889-015-2564-x
Levallois P, Barn P, Valcke M, Gauvin D, Kosatsky T (2018) Public health consequences of lead in drinking water. Curr Environ Health Rep 5:255–262. https://doi.org/10.1007/s40572-018-0193-0
Li P, Feng X, Qiu G (2010) Methylmercury exposure and health effects from rice and fish consumption: a review. Int J Environ Res Public Health 7(6):2666–2691. https://doi.org/10.3390/ijerph7062666
Li AJ, Zhuang TF, Shi JB, Liang Y, Song MY (2019) Heavy metals in maternal and cord blood in Beijing and their efficiency of placental transfer. J Environ Sci 80:99–106. https://doi.org/10.1016/j.jes.2018.11.004
Lin YS, Ho WC, Caffrey JL, Sonawane B (2014) Low serum zinc is associated with elevated risk of cadmium nephrotoxicity. Environ Res 134:33–38. https://doi.org/10.1016/j.envres.2014.06.013
Lin YC, Chang WH, Li TC, Iwata O, Chen HL (2022) Health risk of infants exposed to lead and mercury through breastfeeding. Expos Health. https://doi.org/10.1007/s12403-022-00485-1
Liu J, Li Y, Li D, Wang Y, Wei S (2022) The burden of coronary heart disease and stroke attributable to dietary cadmium exposure in Chinese adults, 2017. Sci Total Environ 825:153997. https://doi.org/10.1016/j.scitotenv.2022.153997
Ma JF, Yamaji N, Mitani N, Xu XY, Su YH, McGrath SP, Zhao FJ (2008) Transporters of arsenite in rice and their role in arsenic accumulation in rice grain. Proc Natl Acad Sci USA 105(29):9931–9935. https://doi.org/10.1073/pnas.0802361105
Ma C, Iwai-Shimada M, Nakayama SF, Isobe T, Kobayashi Y, Tatsuta N, Taniguchi Y, Sekiyama M, Michikawa T, Yamazaki S, Kamijima M (2021) Association of prenatal exposure to cadmium with neurodevelopment in children at 2 years of age: the Japan Environment and Children’s Study. Environ Int 156:106762. https://doi.org/10.1016/j.envint.2021.106762
Mahaffey KR, Clickner RP, Jeffries RA (2009) Adult women’s blood mercury concentrations vary regionally in the United States: association with patterns of fish consumption (NHANES 1999–2004). Environ Health Persp 117(1):47–53. https://doi.org/10.1289/ehp.11674
Milton AH, Hussain S, Akter S, Rahman M, Mouly TA, Mitchell K (2017) A review of the effects of chronic arsenic exposure on adverse pregnancy outcomes. Int J Environ Res Public Health 14(6):556. https://doi.org/10.3390/ijerph14060556
Morrissette J, Takser L, St.Amour G, Smargiassi A, Lafond J, Mergler D, (2004) Temporal variation of blood and hair mercury levels in pregnancy in relation to fish consumption history in a population living along the St Lawrence River. Environ Res 95(3):363–374. https://doi.org/10.1016/j.envres.2003.12.007
Motawei SM, Gouda HE (2016) Screening of blood levels of mercury, cadmium, and copper in pregnant women in Dakahlia, Egypt: new attention to an old problem. Biol Trace Elem Res 171:308–314. https://doi.org/10.1007/s12011-015-0525-y
Murcia M, Ballester F, Enning AM, Iñiguez C, Valvi D, Basterrechea M, Rebagliato M, Vioque J, Maruri M, Tardon A, Riaño-Galán I, Vrijheid M, Llop S (2016) Prenatal mercury exposure and birth outcomes. Environ Res 151:11–20. https://doi.org/10.1016/j.envres.2016.07.003
Nakayama SF, Iwai-Shimada M, Oguri T, Isobe T, Takeuchi A, Kobayashi Y, Michikawa T, Yamazaki S, Nitta H, Kawamoto T (2019) Blood mercury, lead, cadmium, manganese and selenium levels in pregnant women and their determinants: the Japan Environment and Children’s Study (JECS). J Expo Sci Environ Epidemiol 29:633–647. https://doi.org/10.1038/s41370-019-0139-0
Nriagu JO, Lin TS (1995) Trace metals in wild rice sold in the United States. Sci Total Environ 172(2–3):223–228. https://doi.org/10.1016/0048-9697(95)04809-X
Nriagu JO, Blankson ML, Ocran K (1996) Childhood lead poisoning in Africa: a growing public health problem. Sci Total Environ 181(2):93–100. https://doi.org/10.1016/0048-9697(95)04954-1
Nyanza EC, Dewey D, Manyama M, Martin JW, Hatfield J, Bernier FP (2020) Maternal exposure to arsenic and mercury and associated risk of adverse birth outcomes in small-scale gold mining communities in Northern Tanzania. Environ Int 137:105450. https://doi.org/10.1016/j.envint.2019.105450
Osorio-Yanez C, Sanchez-Guerra M, Solano M, Baccarelli A, Wright R, Sanders AP, Tellez-Rojo MM, Tamayo-Ortiz M (2021) Metal exposure and bone remodeling during pregnancy: results from the PROGRESS cohort study. Environ Pollut 282:116962. https://doi.org/10.1016/j.envpol.2021.116962
Ovayolu A, Ovayolu G, Karaman E, Yuce T, Ozek MA, Turksoy VA (2020) Amniotic fluid levels of selected trace elements and heavy metals in pregnancies complicated with neural tube defects. Congenit Anom 60:136–141. https://doi.org/10.1111/cga.12363
Ozel S, Ozyer S, Aykut O, Cinar M, Yilmaz OH, Caglar A, Engin-Ustun Y (2019) Maternal second trimester blood levels of selected heavy metals in pregnancies complicated with neural tube defects. J Matern Fetal Neonatal Med 32(15):2547–2553. https://doi.org/10.1080/14767058.2018.1441280
Park S, Lee BK (2013) Strong positive associations between seafood, vegetables, and alcohol with blood mercury and urinary arsenic levels in the Korean adult population. Arch Environ Contam Toxicol 64:160–170. https://doi.org/10.1007/s00244-012-9808-x
Pesce G, Sesé L, Calciano L, Travert B, Dessimond B, Maesano CN, Ferrante G, Huel G, Prud’homme J, Guinot M, Soomro MH, Baloch RM, Lhote R, Annesi-Maesano I (2021) Foetal exposure to heavy metals and risk of atopic diseases in early childhood. Pediatr Allergy Immunol 32:242–250. https://doi.org/10.1111/pai.13397
Piomelli S (2002) Childhood lead poisoning. Pediatr Clin North Am 49(6):1285–1304. https://doi.org/10.1016/S0031-3955(02)00097-4
Rios JC, Villarroel L, Torres M, Astaburuaga JP, Leiva C, Cook P, Medel P, Cortes S (2020) Urinary heavy metals and blood lead levels among residents of Antofagasta, Chile. Rev Med Chil 148:746–754. https://doi.org/10.4067/s0034-98872020000600746
Rudge CV, Rollin HB, Nogueira CM, Thomassen Y, Rudge MC, Odland JO (2009) The placenta as a barrier for toxic and essential elements in paired maternal and cord blood samples of South African delivering women. J Environ Monit 11:1322–1330. https://doi.org/10.1039/B903805A
Shen HZ, Tsai CM, Yuan CS, Jen YH, Ie IR (2017) How incense and joss paper burning during the worship activities influences ambient mercury concentrations in indoor and outdoor environments of an Asian temple? Chemosphere 167:530–540. https://doi.org/10.1016/j.chemosphere.2016.09.159
Shih YH, Scannell Bryan M, Argos M (2020) Association between prenatal arsenic exposure, birth outcomes, and pregnancy complications: an observational study within the National Children’s Study cohort. Environ Res 183:109182. https://doi.org/10.1016/j.envres.2020.109182
Shih YH, Chen HY, Christensen K, Handler A, Turyk ME, Argos M (2021) Prenatal exposure to multiple metals and birth outcomes: an observational study within the National Children’s Study cohort. Environ Int 147:106373. https://doi.org/10.1016/j.envint.2020.106373
Singh L, Anand M, Singh S, Taneja A (2020) Environmental toxic metals in placenta and their effects on preterm delivery-current opinion. Drug Chem Toxicol 43(5):531–538. https://doi.org/10.1080/01480545.2018.1515216
Song Y, Lee CK, Kim KH, Lee JT, Suh C, Kim SY, Kim JH, Son BC, Kim DH, Lee S (2016) Factors associated with total mercury concentrations in maternal blood, cord blood, and breast milk among pregnant women in Busan Korea. Asia Pac J Clin Nutr 25(2):340–349
Sudjarwo SA, Giftania Wardani Sudjarwo K (2017) Protective effect of curcumin on lead acetate-induced testicular toxicity in Wistar rats. Res Pharm Sci 12(5):381–390. https://doi.org/10.4103/1735-5362.213983
Ugwuja EI, Uro-Chukwu HC, Ikaraoha IC, Aniagolu MO (2020) Association of life style and sociodemographic parameters with plasma heavy metals in apparently healthy adult in Ebonyi State, South East Nigeria. J Clin Diagn Res 14(7):BC01–BC06. https://doi.org/10.7860/JCDR/2020/43784.13832
United Nations Environment Programme (UNEP), (2010) Final review of scientific information on cadmium. https://www.unep.org/resources/report/final-review-scientific-information-cadmium
Vahidinia A, Samiee F, Faradmal J, Rahmani A, Taravati Javad M, Leili M (2019) Mercury, lead, cadmium, and barium levels in human breast milk and factors affecting their concentrations in Hamadan. Iran Biol Trace Elem Res 187:32–40. https://doi.org/10.1007/s12011-018-1355-5
Vahter MJB (2008) Health effects of early life exposure to arsenic. Basic Clin Pharmacol Toxicol 102(2):204–211. https://doi.org/10.1111/j.1742-7843.2007.00168.x
Vigeh M, Yokoyama K, Matsukawa T, Shinohara A, Ohtani K (2015) The relation of maternal blood arsenic to anemia during pregnancy. Women Health 55(1):42–57. https://doi.org/10.1080/03630242.2014.972016
Vigeh M, Nishioka E, Ohtani K, Omori Y, Matsukawa T, Koda S, Yokoyama K (2018) Prenatal mercury exposure and birth weight. Reprod Toxicol 76:78–83. https://doi.org/10.1016/j.reprotox.2018.01.002
Wang Z, Wang H, Wang H, Li Q, Li Y (2019) Heavy metal pollution and potential health risks of commercially available Chinese herbal medicines. Sci Total Environ 25(653):748–757. https://doi.org/10.1016/j.scitotenv.2018.10.388
Wang X, Karvonen-Gutierrez CA, Herman WH, Mukherjee B, Harlow SD, Park SK (2020a) Urinary metals and incident diabetes in midlife women: Study of Women’s Health Across the Nation (SWAN). BMJ Open Diabetes Res Care 8:e001233. https://doi.org/10.1136/bmjdrc-2020-001233
Wang X, Sun X, Zhang YQ, Chen MJ, Villanger GD, Aase H, Xia YK (2020b) Identifying a critical window of maternal metal exposure for maternal and neonatal thyroid function in China: a cohort study. Environ Int 139:105696. https://doi.org/10.1016/j.envint.2020.105696
Wang Y, Wang KK, Han TB, Zhang P, Chen X, Wu WW, Feng YL, Yang HL, Li M, Xie BJ, Guo PG, Warren JL, Shi XM, Wang SP, Zhang YW (2020c) Exposure to multiple metals and prevalence for preeclampsia in Taiyuan China. Environ Int 145:106098. https://doi.org/10.1016/j.envint.2020.106098
Wang X, Karvonen-Gutierrez CA, Mukherjee B, Herman WH, Park SK (2021) Urinary metals and adipokines in midlife women: the Study of Women’s Health Across the Nation (SWAN). Environ Res 196:110426. https://doi.org/10.1016/j.envres.2020.110426
Watson CV, Lewin M, Ragin-Wilson A, Jones R, Jarrett JM, Wallon K, Ward C, Hilliard N, Irvin-Barnwell E (2020) Characterization of trace elements exposure in pregnant women in the United States, NHANES 1999–2016. Environ Res 183:109208. https://doi.org/10.1016/j.envres.2020.109208
Williams P, Raab A, Feldmann J, Meharg A (2007) Market basket survey shows elevated levels of As in South Central US processed rice compared to California: consequences for human dietary exposure. Environ Sci Technol 41(7):2178–2183. https://doi.org/10.1021/es061489k
World Health Organization (WHO) (2015) Human biomonitoring: facts and figures. https://www.euro.who.int/en/media-centre/events/events/2015/04/ehp-mid-term-review/publications/human-biomonitoring-facts-and-figures. Accessed 28 March 2022
World Health Organization (WHO) (2017) Mercury and health. https://www.who.int/news-room/fact-sheets/detail/mercury-and-health. Accessed 25 July 2022
Xia W, Hu J, Zhang B, Li Y, Wise JP Sr, Bassig BA, Zhou A, Savitz DA, Xiong C, Zhao J, Du X, Zhou Y, Pan X, Yang J, Wu C, Jiang M, Peng Y, Qian Z, Zheng T, Xu S (2016) A case–control study of maternal exposure to chromium and infant low birth weight in China. Chemosphere 144:1484–1489. https://doi.org/10.1016/j.chemosphere.2015.10.006
Zheng X, Huo X, Zhang Y, Wang Q, Zhang Y, Xu X (2019) Cardiovascular endothelial inflammation by chronic coexposure to lead (Pb) and polycyclic aromatic hydrocarbons from preschool children in an e-waste recycling area. Environ Pollut 246:587–596. https://doi.org/10.1016/j.envpol.2018.12.055
Zheng Y, Lin PD, Williams PL, Weisskopf MG, Cardenas A, Rifas-Shiman SL, Wright RO, Amarasiriwardena C, Henn BC, Hivert MF, Oken E, James-Todd T (2021) Early pregnancy essential and non-essential metal mixtures and gestational glucose concentrations in the 2nd trimester: results from project Viva. Environ Int 155:106690. https://doi.org/10.1016/j.envint.2021.106690
Zhou F, Du G, Xie J, Gu J, Jia Q, Fan Y, Yu H, Zha Z, Wang K, Ouyang L (2020) RyRs mediate lead-induced neurodegenerative disorders through calcium signaling pathways. Sci Total Environ 701:134901. https://doi.org/10.1016/j.scitotenv.2019.134901
Zhou M, Peng L, Wang J, Cao R, Ou Z, Fang Y (2022) Cadmium exposure and the risk of GDM: evidence emerging from the systematic review and meta-analysis. Environ Sci Pollut Res Int 29(51):77253–77274. https://doi.org/10.1007/s11356-022-21171-4
Acknowledgements
We are in great debt to our colleagues at the Research Center of Environmental Trace Toxic Substances, College of Medicine, National Cheng Kung University for subject recruitment and analytical assistance.
Funding
This work was supported by the Health Promotion Administration, Ministry of Health and Welfare (Grant No. B1080723).
Author information
Authors and Affiliations
Contributions
T-HY: writing—original draft, investigation (subject recruitment, questionnaire interview). C-JH, C-JH, P-JW, C-CY, P-KH, and M-HW: investigation (subject recruitment, questionnaire interview). W-HC: methodology (questionnaire design), resources (analytical assistance). HH: formal analysis, visualization. J-WC: investigation (subject recruitment), resources (analytical assistance). C-TW: writing—original draft, investigation, formal analysis, visualization. RDA: writing—review & editing. TM: methodology (questionnaire design), writing—original draft, writing—review & editing. C–CL: writing—review & editing. H-LC: writing—review & editing, supervision, project administration, funding acquisition. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Ethical Approval
The participants enrolled in this study signed the informed consent form approved by the Ethics Committee of the National Cheng Kung University Hospital, the Chung Shan Medical University Hospital, the Taipei Veterans General Hospital, and Hualien Tzu Chi Hospital, Buddhist Tzu Chi Medical Foundation.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ying, TH., Huang, CJ., Hsieh, CJ. et al. Potential Factors Associated with the Blood Metal Concentrations of Reproductive-Age Women in Taiwan. Expo Health 16, 71–86 (2024). https://doi.org/10.1007/s12403-023-00541-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12403-023-00541-4