Abstract
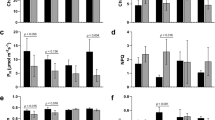
Miscanthus floridulus (Labiu.) Warb is the most cold-tolerant wild resource among the members of the Saccharum complex and has prospective applications in cold-tolerant sugarcane breeding. However, the physiological and transcriptomic basis of the cold tolerance of Miscanthus floridulus is still unclear. In this study, as part of a pot experiment, Miscanthus floridulus was subjected to low-temperature (3 °C) stress (normal temperature was used as a control), the main physiological characteristics were determined, and transcriptomic changes were analysed. Low-temperature stress significantly decreased the leaf relative water content, chlorophyll content, and cell membrane permeability of M. floridulus while increasing the proline, soluble sugar, soluble protein, and abscisic acid contents and peroxidase and superoxide dismutase activities. Analysis of differentially expressed genes (DEGs) showed that 13,298 genes were upregulated and that 7544 genes were downregulated upon low-temperature stress. These DEGs were enriched in many different metabolic pathways, in particular those involving enzymes related to plant hormone signal transduction and starch and sucrose metabolism. The activities of protein phosphatase, β-amylase and cell wall invertase increased under low-temperature stress. Our results revealed that M. floridulus might undergo extensive transcriptional reprogramming of genes involved in hormonal changes and sugar accumulation, which contributes synergistically or independently to the enhanced cold tolerance. The results of this study could provide a scientific basis for further application of M. floridulus in sugarcane cold tolerance breeding.





Similar content being viewed by others
Data Availability
The data supporting the conclusions of this article are provided within the paper and its additional files. All sequencing reads are deposited in the National Center for Biotechnology Information under the BioProject number PRJNA803453 with the Sequence Read Archive (SRA) study accession SRP358303.
References
Amalraj, V.A., and N. Balasundaram. 2006. On the taxonomy of the members of ‘Saccharum complex’. Genetic Resources & Crop Evolution 1 (53): 35–41. https://doi.org/10.1007/s10722-004-0581-1.
Anuradha, S., and S.R. Rao. 2001. Effect of brassinosteroids on salinity stress induced inhibition of seed germination and seedling growth of rice (Oryza sativa L.). Plant Growth Regulation 33: 151–153. https://doi.org/10.1023/A:1017590108484.
Belintani, N.G., J.T.S. Guerzoni, R.M.P. Moreira, and L.G.E. Vieira. 2012. Improving low-temperature tolerance in sugarcane by expressing the ipt gene under a cold inducible promoter. Biologia Plantarum 56 (1): 71–77. https://doi.org/10.1007/s10535-012-0018-1.
Cheng, S.F., C.Y. Huang, K.L. Chen, S.C. Lin, and Y.C. Lin. 2015. Phytoattenuation of lead-contaminated agricultural land using Miscanthus floridulus-an in situ case study. Desalination and Water Treatment 57 (2016): 7773–7779. https://doi.org/10.1080/19443994.2015.1033477.
Cristina, R.G.S., R.V. Rafael, A.G.S. Joaquim, C.M. Eduardo, O.M. Marcio, and M.M.A.L. Ana. 2013. Superoxide dismutase and ascorbate peroxidase improve the recovery of photosynthesis in sugarcane plants subjected to water deficit and low substrate temperature. Plant Physiology and Biochemistry 73: 326–336. https://doi.org/10.1016/j.plaphy.2013.10.012.
Costa, V., C. Angelini, I.D. Feis, and A. Ciccodicola. 2010. Uncovering the complexity of transcriptomes with RNA-Seq. Journal of Biomedicine and Biotechnology 2010: 1–19. https://doi.org/10.1155/2010/853916.
Dharshini, S., M. Chakravarthi, J. Ashwin Narayan, V.M. Manoj, M. Naveenarani, Ravinder Kumar, Minturam Meena, Bakshi Ram, and C. Appunu. 2016. De novo sequencing and transcriptome analysis of a low temperature tolerant Saccharum spontaneum clone IND 00–1037. Journal of Biotechnology 231: 280–294. https://doi.org/10.1016/j.jbiotec.2016.05.036.
Ditt, R.F., A. Gentile, R.G. Tavares, S.R. Camargo, J.H. Fernandez, M.J.D. Silva, and M. Menossi. 2011. Analysis of the stress-inducible transcription factor SsNAC23 in sugarcane plants. Scientia Agricola 68 (4): 454–461. https://doi.org/10.1590/S0103-90162011000400010.
Evans, D.L., and S.V. Joshi. 2016. Complete chloroplast genomes of Saccharum spontaneum, Saccharum officinarum and Miscanthus floridulus (Panicoideae: Andropogoneae) reveal the plastid view on sugarcane origins. Systematics and Biodiversity 14 (6): 1–24. https://doi.org/10.1080/14772000.2016.1197336.
Eremina, M., S.J. Unterholzner, A.I. Rathnayake, M. Castellanos, M. Khan, K.G. Kugler, S.T. May, K.F.X. Mayer, W. Rozhon, and B. Poppenberger. 2016. Brassinosteroids participate in the control of basal and acquired freezing tolerance of plants. Proceedings of the National Academy of Sciences 40 (113): 5982–5991. https://doi.org/10.1073/pnas.1611477113.
Elbein, A.D., Y.T. Pan, I. Pastuszak, and D. Carroll. 2003. New insights on trehalose: A multifunctional molecule. Glycobiology 13 (4): 17–27. https://doi.org/10.1093/glycob/cwg047.
Grivet, L., C. Daniels, J.C. Glaszmann, and A. D’Hont. 2004. A review of recent molecular genetics evidence for sugarcane evolution and domestication. Ethnobotany Research and Applications 2: 9–17. https://doi.org/10.17348/era.2.0.9-17.
Grabherr, M.G., B.J. Haa, M. Yassour, J.Z. Levin, D.A. Thompson, I. Amit, X. Adiconis, L. Fan, R. Raychowdhury, Q.D. Zeng, Z.H. Chen, E. Mauceli, N. Hacohen, A. Gnirke, N. Rhind, F.D. Palma, B.W. Birren, C. Nusbaum, K. Lindblad-Toh, N. Friedman, and A. Regev. 2013. Trinity: reconstructing a full-length transcriptome without a genome from RNA-Seq data. Nat Biotechnol 29 (7): 644–652. https://doi.org/10.1038/nbt.1883.
Gosti, F., N. Beaudoin, C. Serizet, A.A.R. Webb, N. Vartanian, and J. Giraudat. 1999. ABI1 protein phosphatase 2C is a negative regulator of abscisic acid signaling. The Plant Cell 11: 1897–1909. https://doi.org/10.2307/3871085.
Gusta, L.V., R. Trischuk, and C.J. Weiser. 2005. Plant cold acclimation: The role of abscisic acid. Journal of Plant Growth Regulation 24 (4): 308–318. https://doi.org/10.1007/s00344-005-0079-x.
Huang, Cl., C.W. Ho, Y.C. Chiang, Y. Shigemoto, T.W. Hsu, C.C. Hwang, X.J. Ge, C. Chen, T.H. Wu, C.H. Chou, H.J. Huang, T. Gojobori, N. Osada, and T.Y. Chiang. 2014. Adaptive divergence with gene flow in incipient speciation of Miscanthus floridulus/sinensis complex (Poaceae). The Plant Journal 80: 834–847. https://doi.org/10.1111/tpj.12676.
Huang, X., M.H. Chen, L.T. Yang, Y.R. Li, and J.M. Wu. 2015. Effects of exogenous abscisic acid on cell membrane and endogenous hormone contents in leaves of sugarcane seedlings under cold stress. Sugar Tech 17 (1): 59–64. https://doi.org/10.1007/s12355-014-0343-0.
Hetherington, B.A.M., and R.S. Quatrano. 1991. Mechanisms of action of abscisic acid at the cellular level. New Phytologist 119: 9–32. https://doi.org/10.1111/j.1469-8137.1991.tb01004.x.
Hao, X.Y., C. Yue, H. Tang, W.J. Qian, Y.C. Wang, L. Wang, X.C. Wang, and Y.J. Yang. 2017. Cloning of β- amylase gene (CsBAM3) and its expressio model response to cold stress in tea plant. Acta Agronomica Sinica 43 (10): 1417–1425. https://doi.org/10.3724/SP.J.1006.2017.01417.
Jain, M., A. Nijhawan, A.K. Tyagi, and J.P. Khurana. 2006. Validation of housekeeping genes as internal control for studying gene expression in rice by quantitative real-time PCR. Biochemical and Biophysical Research Communications 345: 646–651. https://doi.org/10.1016/j.bbrc.2006.04.140.
Kao, W.Y., T.T. Tsai, and W.H. Chen. 1998. A comparative study of Miscanthus floridulus (Labill) Warb and M. transmorrisonensis Hayata: photosynthetic gas exchange, leaf characteristics and growth in controlled environments. Annals of Botany 81: 295–299. https://doi.org/10.1006/anbo.1997.0553.
Kinoshita, T., A. Caño-Delgado, H. Seto, S. Hiranuma, S. Fujioka, S. Yoshida, and J. Chory. 2005. Binding of brassinosteroids to the extracellular domain of plant receptor kinase BRI1. Nature 433 (7022): 167–171. https://doi.org/10.1038/nature03227.
Li, P., H. Yang, L. Wang, H.J. Liu, H.Q. Huo, C.J. Zhang, A. Liu, A. Zhu, J.Y. Hu, Y.J. Lin, and L. Liu. 2019. Physiological and transcriptome analyses reveal short-term responses and formation of memory under drought stress in rice. Frontiers in Genetics 10: 55. https://doi.org/10.3389/fgene.2019.00055.
Li, S.J., X.H. Wang, and Q.H. Yang. 2017. Evaluation of cold tolerance in wild sugarcane germplasm resources under field conditions. Agricultural Biotechnology 6 (2): 22–25.
Li, B., and C.N. Dewey. 2011. RSEM: Accurate transcript quantification from RNA-Seq data with or without a reference genome. BMC Bioinformatics 12: 323. https://doi.org/10.1186/1471-2105-12-323.
Livak, K.J., and T.D. Schmittgen. 2001. Analysis of relative gene expression data using realtime quantitative PCR and the 2-△△Ct method. Methods 25 (4): 402–408. https://doi.org/10.1006/meth.2001.
Lyons, J.M. 1973. Chilling injury in plants. Annual Review of Plant Physiology 24: 445–460.
Luan, S. 2003. Protein phosphatases in plants. Annual Review of Plant Biology 54: 63–92. https://doi.org/10.1146/annurev.arplant.54.031902.134743.
Lunn, J.E., I. Delorge, C.M. Figueroa, P.V. Dijck, and M. Stitt. 2014. Trehalose metabolism in plants. The Plant Journal 79: 544–567. https://doi.org/10.1111/tpj.12509.
Manjunatha, T., K. Chandran, P. Mahesh, and K. Mohanraj. 2018. Characterization of Saccharum spontaneum accessions from North-Eastern India. Electronic Journal of Plant Breeding 9 (1): 38–50. https://doi.org/10.5958/0975-928X.2018.00005.4.
Nogueira, F.T.S. 2003. RNA expression profiles and data mining of sugarcane response to low temperature. Plant Physiology 132 (4): 1811–1824. https://doi.org/10.1104/pp.102.017483.
Nogueira, F.T.S., P.S. Schlo¨gl, S.R. Camargo, J.H. Fernandez, V.E.D.P. Pompermayer, and P. Arruda. 2005. SsNAC23, a member of the NAC domain protein family, is associated with cold, herbivory and water stress in sugarcane. Plant Science 169 (1): 93–106. https://doi.org/10.1016/j.plantsci.2005.03.008.
Oliver, S.N., J.T.V. Dongen, S.C. Alfred, E.A. Mamun, X.C. Zhao, H.S. Saini, S.F. Fernandes, C.L. Blanchard, B.G. Sutton, P. Geigenberger, E.S. Dennis, and R. Dolferus. 2005. Cold-induced repression of the rice anther-specific cell wall invertase gene OSINV4 is correlated with sucrose accumulation and pollen sterility. Plant, Cell and Environment 28: 1534–1551. https://doi.org/10.1111/j.1365-3040.2005.01390.x.
Park, J.W., T.R. Benatti, T. Marconi, Q.Y. Yu, N.S. Gracia, V. Mora, and A.D. Silva. 2015. Cold responsive gene expression profiling of sugarcane and Saccharum spontaneum with functional analysis of a cold inducible saccharum homolog of NOD26-like intrinsic protein to salt and water stress. PLoS One 10 (5): e0125810. https://doi.org/10.1371/journal.pone.0125810.
Popov, V.N., O.V. Antipina, and E.A. Burakhanova. 2013. Involvement of cell-wall invertase in low-temperature hardening of tobacco plants. Russian Journal of Plant Physiology 60 (2): 221–226. https://doi.org/10.1134/S1021443713020167.
Proels, R.K., and R. Hückelhoven. 2014. Cell-wall invertases, key enzymes in the modulation of plant metabolism during defence responses. Molecular Plant Pathology 15 (8): 858–864. https://doi.org/10.1111/mpp.12139.
Silva, J.A.D. 2017. The importance of the wild cane Saccharum Spontaneum for bioenergy genetic breeding. Sugar Tech 19 (3): 229–240. https://doi.org/10.1007/s12355-017-0510-1.
Selvarajan, D., C. Mohan, V. Dhandapani, G. Nerkar, A.N. Jayanarayanan, M.V. Mohanan, N. Murugan, L. Kaur, M. Chennappa, R.K.M. Meena, B. Ram, and A. Chinnaswamy. 2018. Differential gene expression profiling through transcriptome approach of Saccharum spontaneum L under low temperature stress reveals genes potentially involved in cold acclimation. 3 Biotech 8 (4): 195. https://doi.org/10.1007/s13205-018-1194-2.
Shingote, P.R., P.G. Kawar, M.C. Pagariya, P.R. Rathod, and S.B. Kharte. 2017. Ectopic expression of SsMYB18, a novel MYB transcription factor from Saccharum spontaneum augments salt and cold tolerance in tobacco. Sugar Tech 19 (3): 270–282. https://doi.org/10.1007/s12355-016-0466-6.
Schweighofer, A., H. Hirt, and I. Meskiene. 2004. Plant PP2C phosphatases: Emerging functions in stress signaling. TRENDS in Plant Science 9 (5): 236–243. https://doi.org/10.1016/j.tplants.2004.03.007.
Sreeramulu, S., Y. Mostizky, S. Sunitha, E. Shani, H. Nahum, D. Salomon, L.B. Hayun, C. Gruetter, D. Rauh, N. Ori, and G. Sessa. 2013. BSKs are partially redundant positive regulators of brassinosteroid signaling in Arabidopsis. The Plant Journal 74: 905–919. https://doi.org/10.1111/tpj.12175.
Strauss, G., and H. Hauser. 1985. Stabilization of lipid bilayer vesicles by sucrose during freezing. Proceedings of the National Academy of Sciences 83: 2422–2426. https://doi.org/10.1073/pnas.83.8.2422.
Thiebaut, F., C.A. Rojas, K.L. Almeida, C. Grativol, G.C. Domiciano, C.R.C. Lamb, J.D.A. Engler, A.S. Hemerly, and P.C.G. Ferreira. 2012. Regulation of miR319 during cold stress in sugarcane. Plant Cell & Environment 35 (3): 502–512. https://doi.org/10.1111/j.1365-3040.2011.02430.x.
Wei, T.L., Y. Wang, Z.Z. Xie, D. Guo, C.W. Chen, Q.J. Fan, X.D. Deng, and J.H. Liu. 2019. Enhanced ROS scavenging and sugar accumulation contribute to drought tolerance of naturally occurring autotetraploids in Poncirus trifoliata. Plant Biotechnology Journal 17 (7): 1–14. https://doi.org/10.1111/pbi.13064.
Wang, T.J., X.H. Wang, and Q.H. Yang. 2020. Comparative analysis of drought-responsive transcriptome in different genotype Saccharum spontaneum L. Sugar Tech 22 (21): 1–17. https://doi.org/10.1007/s12355-019-00774-1.
Xin, Z., and J. Browse. 2000. Cold comfort farm: The acclimation of plants to freezing temperatures. Plant, Cell & Environment 23 (9): 893–902. https://doi.org/10.1046/j.1365-3040.2000.00611.x.
Yang, Y.T., X. Zhang, Y.C. Su, J.K. Zou, Z.T. Wang, L.P. Xu, and Y.X. Que. 2017. miRNA alteration is an important mechanism in sugarcane response to low-temperature environment. BMC Genomics 18 (1): 833. https://doi.org/10.1186/s12864-017-4231-3.
Yang, H.L., T.J. Wang, X.H. Yu, Y. Yang, C.F. Wang, Q.H. Yang, and X.H. Wang. 2020a. Enhanced sugar accumulation and regulated plant hormone signalling genes contribute to cold tolerance in hypoploid Saccharum spontaneum. BMC Genomics 21 (1): 507. https://doi.org/10.1186/s12864-020-06917-z.
Yang, Y.T., X. Zhang, Y. Chen, J.L. Guo, H. Ling, S.W. Gao, Y.C. Su, Y.X. Que, and L.P. Xu. 2016. Selection of reference genes for normalization of microRNA expression by RT-qPCR in sugarcane buds under cold stress. Frontiers in Plant Science 7: 86. https://doi.org/10.3389/fpls.2016.00086.
Yu, X.H., X.H. Wang, and Q.H. Yang. 2019. Genetic Diversity and phylogenetic relationship of Saccharum Spontaneum L. with different ploidy levels based on SRAP Markers. Sugar Tech 21 (5): 1–13. https://doi.org/10.1007/s12355-019-00700-5.
Yang, H.I., X.H. Yu, C.F. Wang, Y. Yang, X.H. Wang, and Q.H. Yang. 2020. Evaluation of the cold tolerance of Saccharum spontaneum L. clones with different ploidy levels on the basis of morphological and physiological indices. Plant Biology 22 (2020b): 623–633. https://doi.org/10.1111/plb.13110.
Zhang, J.S., X.T. Zhang, H.B. Tang, Q. Zhang, X.T. Hua, X.K. Ma, F. Zhu, T. Jones, X.G. Zhu, J. Bowers, et al. 2018. Allele-defined genome of the autopolyploid sugarcane Saccharum spontaneum L. Nature Genetics 50 (11): 1565–1573. https://doi.org/10.1038/s41588-018-0237-2.
Zhang, B.Q., L.T. Yang, and Y.R. Li. 2015. Physiological and biochemical characteristics related to cold resistance in sugarcane. Sugar Tech 17 (1): 49–58. https://doi.org/10.1007/s12355-014-0340-3.
Zhang, L., Q. Lin, Y.Z. Feng, X.M. Fan, D.Y. Yuan, X.C. Zeng, and H.P. Cao. 2016. Transcriptomic identification and expression of starch and sucrose metabolism genes in the seeds of chinese chestnut (Castanea mollissima). Journal of Agricultural and Food Chemistry 63 (3): 929–942. https://doi.org/10.1021/jf505247d.
Acknowledgements
We thank AJE for its linguistic assistance during the preparation of this manuscript. This study was financially supported by the National Natural Science Foundation of China (No. 32160492, No. 31460372 and No. 31760417).
Funding
National Natural Science Foundation of China, Grant/Award Numbers: 32160492, 31460372 and 31760417.
Author information
Authors and Affiliations
Contributions
Xian-Hong Wang, Hong-Li Yang and Qing-Hui Yang designed the experiments; Hong-Li Yang performed the research, analysed the data and wrote the manuscript; Zhi-Jun Guo, Mo Chen and Qing Liu treated the experimental materials; and Lu Jiang, Jun-Jun He, Xing-Hua Yu and Xian-Hong Wang revised the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
12355_2022_1193_MOESM2_ESM.docx
Additional file 2: Table S2. Primers used in quantitative real-time RT-qPCR validation of gene expression data as revealed by RNAseq analysis (DOCX 72 kb)
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yang, HL., Guo, ZJ., Jiang, L. et al. How the Wild Sugarcane Resource Miscanthus floridulus Responds to Low-Temperature Stress: A Physiological and Transcriptomic Analysis. Sugar Tech 25, 398–409 (2023). https://doi.org/10.1007/s12355-022-01193-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12355-022-01193-5