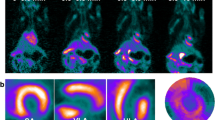
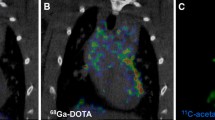
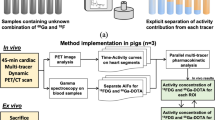
Abstract
Objectives
We aimed to develop a dynamic imaging technique for a novel PET superoxide tracer, [18F]DHMT, to allow for absolute quantification of myocardial reactive oxygen species (ROS) production in a large animal model.
Methods
Six beagle dogs underwent a single baseline dynamic [18F]DHMT PET study, whereas one animal underwent three serial dynamic studies over the course of chronic doxorubicin administration (1 mg·kg−1·week−1 for 15 weeks). During the scans, sequential arterial blood samples were obtained for plasma metabolite correction. The optimal compartment model and graphical analysis method were identified for kinetic modeling. Values for the left ventricular (LV) net influx rate, Ki, were reported for all the studies and compared with the LV standard uptake values (SUVs) and the LV-to-blood pool SUV ratios from the 60 to 90 minute static images. Parametric images were also generated.
Results
[18F]DHMT followed irreversible kinetics once oxidized within the myocardium in the presence of superoxide, as evidenced by the fitting generated by the irreversible two-tissue (2Ti) compartment model and the linearity of Patlak analysis. Myocardial Ki values showed a weak correlation with LV SUV (R2 = 0.27), but a strong correlation with LV-to-blood pool SUV ratio (R2 = 0.92). Generation of high-quality parametric images showed superior myocardial to blood contrast compared to static images.
Conclusions
A dynamic PET imaging technique for [18F]DHMT was developed with full and simplified kinetic modeling for absolute quantification of myocardial superoxide production in a large animal model.








Similar content being viewed by others
Abbreviations
- ROS:
-
Reactive oxygen species
- [18F]DHMT:
-
[18F]-6-(4-((1-(2-fluoroethyl)-1H-1,2,3-triazol-4-yl)methoxy)phenyl)-5-methyl-5, 6-dihydrophenanthridine-3, 8-diamine
- VOI:
-
Volume of interest
- TAC:
-
Time activity curve
- SUV:
-
Standard uptake value
- 2Ti:
-
Two-tissue irreversible
- DOX:
-
Doxorubicin
- LVEF:
-
Left ventricular ejection fraction
- K i :
-
Net influx rate
- AIC:
-
Akaike information criterion
References
Sugamura K, Keaney JF Jr. Reactive oxygen species in cardiovascular disease. Free Radic Biol Med 2011;51:978-92.
Frijhoff J, Winyard PG, Zarkovic N, Davies SS, Stocker R, Cheng D, et al. Clinical relevance of biomarkers of oxidative stress. Antioxid Redox Signal 2015;23:1144-70.
Zhang W, Cai Z, Li L, Ropchan J, Lim K, Boutagy NE, et al. Optimized and automated radiosynthesis of [(18)F]DHMT for translational imaging of reactive oxygen species with positron emission tomography. Molecules 2016;21:1759.
Boutagy NE, Wu J, Cai Z, Zhang W, Booth CJ, Kyriakides TC, et al. In vivo reactive oxygen species detection with a novel positron emission tomography tracer, (18)F-DHMT, allows for early detection of anthracycline-induced cardiotoxicity in rodents. JACC Basic Transl Sci 2018;3:378-90.
Mohy-Ud-Din H, Boutagy NE, Stendahl JC, Zhuang ZW, Sinusas AJ, Liu C. Quantification of intramyocardial blood volume with (99m)Tc-RBC SPECT-CT imaging: A preclinical study. J Nucl Cardiol 2018;25:2096-111.
Gunn RN, Gunn SR, Cunningham VJ. Positron emission tomography compartmental models. J Cereb Blood Flow Metab 2001;21:635-52.
Marquardt DW. An algorithm for least-squares estimation of nonlinear parameters. J Soc Indust Appl Math 1963;11:431-41.
Strother SC, Casey ME, Hoffman EJ. Measuring PET scanner sensitivity: Relating countrates to image signal-to-noise ratios using noise equivalents counts. IEEE Trans Nucl Sci 1990;37:783-8.
Wu J, Lin SF, Gallezot JD, Chan C, Prasad R, Thorn SL, et al. Quantitative analysis of dynamic 123I-mIBG SPECT imaging data in healthy humans with a population-based metabolite correction method. J Nucl Med 2016;57:1226-32.
Wu J, Gallezot JD, Lu Y, Ye Q, Liu H, Esserman DA, et al. Simplified quantification and acquisition protocol for (123)I-MIBG dynamic SPECT. J Nucl Med 2018;59:1574-80.
Patlak CS, Blasberg RG, Fenstermacher JD. Graphical evaluation of blood-to-brain transfer constants from multiple-time uptake data. J Cereb Blood Flow Metab 1983;3:1-7.
Houstis N, Rosen ED, Lander ES. Reactive oxygen species have a causal role in multiple forms of insulin resistance. Nature 2006;440:944-8.
Carroll V, Michel BW, Blecha J, VanBrocklin H, Keshari K, Wilson D, et al. A boronate-caged [(1)(8)F]FLT probe for hydrogen peroxide detection using positron emission tomography. J Am Chem Soc 2014;136:14742-5.
Huang HJ, Isakow W, Byers DE, Engle JT, Griffin EA, Kemp D, et al. Imaging pulmonary inducible nitric oxide synthase expression with PET. J Nucl Med 2015;56:76-81.
Klein LJ, Visser FC, Knaapen P, Peters JH, Teule GJ, Visser CA, et al. Carbon-11 acetate as a tracer of myocardial oxygen consumption. Eur J Nucl Med 2001;28:651-68.
Acknowledgments
We would like to thank Dr. Eva Romito, Tsa Shelton and Christi Hawley for their participation and support of the animal studies.
Disclosure
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The authors of this article have provided a PowerPoint file, available for download at SpringerLink, which summarizes the contents of the paper and is free for re-use at meetings and presentations. Search for the article DOI on SpringerLink.com.
Funding
This work is supported by an internal funding from the Department of Radiology and Biomedical Imaging at Yale University, and the NIH grants R01HL123949 and T32HL098069.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wu, J., Boutagy, N.E., Cai, Z. et al. Feasibility study of PET dynamic imaging of [18F]DHMT for quantification of reactive oxygen species in the myocardium of large animals. J. Nucl. Cardiol. 29, 216–225 (2022). https://doi.org/10.1007/s12350-020-02184-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12350-020-02184-3