Abstract
Background
Data on coronary plaque observations on multi-slice computed tomography (MSCT) in patients with type 2 diabetes are scarce.
Methods and Results
In total, 60 patients (19 with diabetes) underwent 64-slice MSCT, followed by conventional coronary angiography with intravascular ultrasound (IVUS). Non-invasively, the extent of coronary atherosclerosis and 3 plaque types (non-calcified, calcified, mixed) were visually evaluated on MSCT. Invasively, plaque burden was assessed on gray-scale IVUS. Plaque composition was evaluated on virtual histology intravascular ultrasound (VH IVUS). Concerning geometrical plaque data, diabetic patients showed more plaques on MSCT (7.1 ± 3.2 vs 4.9 ± 3.2 in non-diabetic patients, P = .01). On gray-scale IVUS, diabetes was associated with a larger plaque burden (48.7 ± 10.7% vs 40.0 ± 12.1%, P = .003). Concerning plaque composition, diabetes was associated with more calcified plaques on MSCT (52% vs 24%). Relatively more fibrocalcific plaques in diabetic patients (29% versus 9%) were observed on VH IVUS. Moreover, these plaques contained more necrotic core (10.8 ± 5.9% vs 8.6 ± 5.2%, P = .01).
Conclusion
A higher plaque extent and more calcified lesions were observed in diabetic patients on MSCT. The findings were confirmed on gray-scale and VH IVUS. Thus, MSCT may potentially be used to explore patterns of coronary atherosclerosis in diabetic patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Type 2 diabetes is strongly associated with the development of coronary artery disease (CAD).1 Thus far, functional testing has been used to detect CAD in diabetic patients, including myocardial perfusion scintigraphy and stress echocardiography. Although abnormal test results have been clearly associated with the development of cardiovascular events on follow-up, elevated event rates have also been observed in patients without detectable ischemia.2,3 These findings suggest that to some extent the increased cardiovascular risk in patients with diabetes could be attributed to the presence of diffuse CAD rather than focal high-grade lesions.
Accordingly, direct visualization of coronary atherosclerosis could be superior to detection of ischemia for risk stratification of diabetic patients. Both non-invasive and invasive modalities are currently available and may be useful for this purpose. The available studies thus far have mainly addressed the quantification of plaque burden. Indeed, diabetes was demonstrated to be a strong predictor of increased atherosclerotic volume on gray-scale intravascular ultrasound (IVUS).4 Non-invasively, increased calcific plaque burden was observed in diabetic patients on electron beam computed tomography.5 Nonetheless, although the above techniques allow estimation of coronary plaque extent, they are suboptimal for the assessment of plaque composition.
Recently, multi-slice computed tomography (MSCT) coronary angiography was introduced, which in addition to visualization of obstructive and non-obstructive plaques allows assessment of plaque composition.6,7 Invasively, plaque composition may be evaluated by virtual histology IVUS (VH IVUS).8 However, only limited data are available on plaque characterization by both techniques in diabetic patients. Accordingly, the purpose of the study was to evaluate coronary plaque extent and composition on MSCT and to compare these findings with patterns of coronary atherosclerosis on invasive angiography, gray-scale IVUS, and VH IVUS. Patients with and without diabetes were compared.
Methods
Patients and Study Protocol
A total of 60 patients, 19 (32%) patients with type 2 diabetes and 41 (68%) patients without diabetes, were included in the study. All patients presented with chest pain suggestive of CAD. Diabetes was diagnosed according to the criteria as set by the American Diabetes Association:9 (1) a fasting plasma glucose level of ≥7.0 mmol/L or (2) symptoms of diabetes and a casual plasma glucose level of ≥11.1 mmol/L. The patients with a long history of diabetes and requiring insulin and oral hypoglycemic agents were also diagnosed as having diabetes. Patients underwent 64-slice MSCT coronary angiography, followed within a month by conventional coronary angiography in combination with IVUS of one to three vessels. The clinical history of the patients was evaluated prior to conventional coronary angiography to ensure that neither acute coronary events nor worsening of angina occurred between the examinations.
Patients were excluded from the study if contraindications for MSCT were present.10 IVUS examination was not performed if severe vessel tortuousness, severe luminal narrowing precluding the insertion of the IVUS catheter or vessel occlusion were present. Informed consent was obtained from all patients.
MSCT
Image acquisition
MSCT coronary angiography was performed using a 64-detector row Toshiba Aquilion (Toshiba Medical Systems, Tokyo, Japan) scanner. A helical scan protocol with electrocardiographic gating was applied as described previously.11 Additional oral β-blockers (metoprolol, 50 or 100 mg, single dose, 1 hour prior to the examination) were provided if the heart rate was ≥60 beats/minute and no contraindications for the use of β-blockers were present. The image dataset was initially reconstructed at the diastolic phase of the R-R interval (75% of the R-R interval). In case of the presence of motion artifacts additional phases were explored. The middle segment of the right coronary artery was verified in the end-systolic image reconstruction dataset in all patients with good image quality at end-diastolic phase. Image reconstructions obtained with either smooth or medium filters were used for the evaluation of non-calcified plaques in all patients. In the presence of extensive calcifications, reconstructions obtained with a sharp filter were additionally analyzed to verify calcium containing plaques.
Image analysis
Images were evaluated on a remote workstation with dedicated software (Vitrea 2, Vital Images, Minnetonka, Minnesota, USA and Advantage, GE Healthcare, Boston, Massachusetts, USA). Two experienced observers evaluated the examinations side-by-side in consensus. The presence, extent and type of coronary plaques were assessed using a qualitative visual approach as previously described.12 Briefly, the plaques were assessed using axial images and curved multiplanar reconstructions. Plaques were defined as structures >1 mm2 within and/or adjacent to the coronary artery lumen, which could be clearly distinguished from the vessel lumen and the surrounding tissue. Plaques were visually classified as obstructive or not, using a threshold of 50% luminal narrowing. Three types of plaques were distinguished: non-calcified (composed only of low-density component), mixed (composed of both low- and high-density components), and calcified (composed only of high-density component).
Conventional Coronary Angiography and Quantitative Coronary Angiography Analysis
Conventional invasive coronary angiography was performed according to standard protocols. An experienced observer performed off-line quantitative coronary angiography (QCA) analyses, using dedicated software (QCA CMS 6.0, Medis Medical imaging systems, Leiden, The Netherlands). For each vessel examined both with MSCT and IVUS, percent luminal narrowing was reported at the site of the minimal lumen area (MLA).
Gray-scale and VH IVUS
Image acquisition
IVUS examinations were performed with a 20 MHz, 2.9 F phased-array IVUS catheter (Eagle Eye, Volcano Corporation, Rancho Cordova, California, USA). After intracoronary administration of nitrates, the catheter was introduced to the distal coronary artery and withdrawn at a continuous speed of 0.5 mm/second to the coronary ostium using automated pullback device (Track Back, Volcano Corporation, Rancho Cordova, California, USA). To define the starting position of the IVUS catheter, cine runs before and during contrast injection were performed. The images were stored for off-line analysis.
Image analysis
Gray-scale IVUS. To evaluate geometrical plaque parameters, gray-scale IVUS datasets were evaluated on a vessel level by an experienced observer, using dedicated software (QCU CMS 4.0 Medis Medical imaging systems, Leiden, The Netherlands). Cross-sectional images spaced 0.5 mm apart in the pullback were analyzed within the full length of the examined vessel. First, the contours of the external elastic membrane (EEM) were identified and the mean EEM area was calculated in the examined vessel. Subsequently, lumen-intima interface was identified and mean lumen area was calculated. Plaque area was enclosed by the contours of the EEM and the lumen-intima interface and mean plaque area was identified per vessel. Plaque burden was calculated as the percentage of the EEM occupied by plaque [Plaque burden (%) = Σ(EEMarea − lumenarea)/ΣEEMarea × 100]. Percent of abnormal images (having a maximal plaque thickness >0.5 mm) was calculated in each examined vessel.13 Finally, remodeling index was calculated by dividing the EEM area at MLA site of the vessel by the EEM area at proximal reference site. The latter was defined as the frame with largest lumen area located within 10 mm from the MLA frame with no major intervening side branches.14
Of note, the accuracy of the IVUS pullback device was assessed by imaging single stents of known length (8-18 mm stents) in a subpopulation of 15 patients (5 stents in the right coronary artery, 5 stents in the left anterior descending coronary artery, 5 stents in the left circumflex coronary artery). In all included patients, the pullback was available at least 10 mm beyond the distal end of the stent and at least 10 mm proximal to the stent. The stents were measured between the distal and proximal IVUS frames with complete circumferential appearances of the stent struts.
VH IVUS. An experienced observer performed quantitative VH IVUS image analysis on a plaque level using dedicated software (pcVH 2.1, Volcano Corporation, Rancho Cordova, California, USA). Qualitative VH IVUS analysis was performed in consensus by two experienced observers. First, four tissues were differentiated and labeled with different colors (fibrotic, fibro-fatty tissues, necrotic core, and dense calcium), as described and validated previously.8,15 The mean percentage of each plaque component was obtained in the full length of each plaque observed on MSCT. In addition, plaques were visually assessed in three consecutive frames within 10 mm from the MLA site of plaque and were classified into four different types based on geometrical and compositional parameters as well as the location of specific components in the plaque: (1) pathological intimal thickening, (2) fibroatheroma, (3) thin cap fibroatheroma, and (4) fibrocalcific plaque.16,17
Plaques were matched between MSCT and VH IVUS as previously described.18 First, plaques were visually identified on MSCT. The distances from the established landmarks (coronary ostia, side-branches) to the starting point and the termination point of the plaques were measured on multiplanar reconstructions of the vessels on MSCT. These distances were subsequently measured on the longitudinal mode of IVUS datasets. The transversal IVUS sections were further inspected and the starting and the termination frames of the lesions were depicted.
Statistical Analysis
Categorical variables are expressed as numbers (percentages) and compared between groups with 2-tailed Chi-square test. When normally distributed, continuous variables are expressed as means (standard deviation) and compared with the 2-tailed t-test for independent samples. When not normally distributed, continuous variables are expressed as medians (interquartile range) and compared with the 2-tailed non-parametric Mann-Whitney test. To evaluate the reproducibility of coronary plaque evaluation on MSCT and VH IVUS Cohen’s kappa coefficient was calculated. The agreement of QCA analysis was expressed as the difference of percent diameter stenosis between the measurements.
P-values <.05 were considered as statistically significant. Statistical analyses were performed using SPSS software (version 14.0, SPSS Inc, Chicago, Ill, USA).
Results
Baseline clinical characteristics of patients with diabetes and without diabetes are provided in Table 1.
Geometrical Data of Coronary Atherosclerosis (Plaque Extent)
MSCT
MSCT coronary angiograms were of diagnostic quality in all patients. A good intra- and inter-observer agreement was observed for the detection of obstructive plaques (Cohen’s kappa coefficient 0.81 for intra-observer variability and 0.81 for inter-observer variability). The results of the geometrical analysis on MSCT are presented in Table 2. The average number of coronary plaques was higher in diabetic patients as compared with patients without diabetes. In particular, a trend for a higher number of non-obstructive plaques was observed in diabetic patients (5.2 ± 2.7 in patients with diabetes versus 3.8 ± 2.6 in patients without diabetes, P = .06).
QCA and gray-scale IVUS
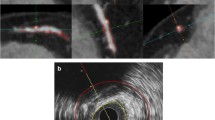
A good intra- and inter-observer agreement was observed for the evaluation of stenosis severity on QCA (mean difference 7.4 ± 7.2% and 8.1 ± 7.4%, respectively). Similarly, a good accuracy of the IVUS pullback device was observed. The difference in known and measured stent length was 1.7 ± 0.8 mm (P = .4), whereas the deviation from known stent length was 10.9 ± 3.5%. Geometrical coronary plaque data obtained by QCA and gray-scale IVUS are presented in Table 2. Briefly, on gray-scale IVUS, more diffuse atherosclerosis was observed in diabetic patients as reflected by a larger plaque burden in the presence of diabetes. In addition, the number of abnormal IVUS frames was also higher in patients with diabetes. An example of diffuse atherosclerosis in a patient with type 2 diabetes on MSCT and gray-scale IVUS is provided in Figure 1.
An example of coronary atherosclerosis in a patient with type 2 diabetes as demonstrated by MSCT (A), gray-scale IVUS (B), and VH IVUS (C). A, MSCT multiplanar reconstruction of the right coronary artery, demonstrating diffuse atherosclerosis along the course of the artery. A mixed plaque is observed in the proximal part of the vessel both on longitudinal and the transversal images. B, The presence of diffuse atherosclerosis was confirmed on the longitudinally reconstructed gray-scale IVUS image. C, The corresponding image on VH IVUS at the site of MLA of the plaque, depicting a lesion with the features of fibrocalcific plaque. IVUS, Intravascular ultrasound; MSCT, multi-slice computed tomography; MLA, minimal lumen area; VH IVUS, virtual histology intravascular ultrasound
Compositional Data of Coronary Atherosclerosis
MSCT
A good agreement was observed for the classification of plaque type (Cohen’s kappa coefficient 0.85 for intra-observer variability and 0.74 for inter-observer variability). As shown in Table 3, relatively more calcified plaques were observed in diabetic patients as compared with patients without diabetes. The proportion of mixed plaques was similar between the two patient populations, whereas non-calcified plaques were relatively more prevalent in patients without diabetes.
VH IVUS
The results of coronary plaque composition on VH IVUS are presented in Table 3. Briefly, the amount of dense calcium was larger in the plaques of patients with diabetes as compared with patients without diabetes. Moreover, the plaques of diabetic subjects contained more necrotic core.
A good agreement was observed for the classification of plaque type (Cohen’s kappa coefficient 0.75 for intra-observer variability and 0.87 for inter-observer variability). Qualitative assessment of lesions revealed a higher prevalence of fibrocalcific plaques in diabetic patients. In contrast, in patients without diabetes relatively more lesions were classified as pathological intimal thickening. No differences, however, were observed in the prevalence of fibroatheroma and thin cap fibroatheroma. An example of a mixed plaque on MSCT and the corresponding plaque demonstrating features of fibrocalcific plaque on VH IVUS is provided in Figure 1.
Discussion
The findings of coronary plaque characterization using MSCT angiography, gray-scale, and VH IVUS may be summarized as follows. First, the extent of coronary plaques was higher in diabetic patients on MSCT. This finding corresponded with more diffuse atherosclerosis and a larger plaque burden on gray-scale IVUS examination. Second, the proportion of completely calcified plaques on MSCT was larger in patients with diabetes as compared with patients without diabetes. On VH IVUS, this corresponded with a larger amount of dense calcium and a higher prevalence of fibrocalcific plaques. Finally, a larger amount of necrotic core was observed in the plaques of diabetic patients, whereas the prevalence of TCFA was identical between the two patient populations.
Plaque Extent
In the present study, a higher extent of coronary atherosclerosis was observed in diabetic patients both on MSCT and on gray-scale IVUS. A trend toward a higher number of non-obstructive plaques was observed although the finding was not statistically significant. This finding is in line with a previous study exploring non-invasive coronary plaque characterization on MSCT angiography.12 Indeed, in this study including 215 patients (86 patients with type 2 diabetes) diabetes was associated with a higher prevalence of non-obstructive coronary plaques.12 Moreover, the observation on MSCT in the present study was paralleled by a more diffuse atherosclerosis and a larger plaque burden on gray-scale IVUS. Similar observations were reported by Nicholls et al4 who reported on the measures of coronary plaques on gray-scale IVUS in 654 patients, of which 128 had diabetes. The authors reported a significantly larger plaque burden in diabetic patients. Increased plaque extent on MSCT as well as on gray-scale IVUS (namely due to non-obstructive and thus predominantly non-flow limiting plaques) might explain a higher event rate in diabetic population. Indeed, it has been observed that plaques leading to myocardial infarction are most often non-obstructive on invasive angiography months prior to the occurrence of the event.19 Moreover, pathological studies of the victims of sudden cardiac death have shown that the cross sectional area luminal narrowing was below 75% (luminal narrowing of less than 50%) in more than 80% of plaques referred to as vulnerable to rupture.20 Nevertheless, it remains to be determined in follow-up studies whether a higher plaque extent as observed on MSCT may be able to predict the increased cardiovascular event rate in diabetic patients.
A trend for a larger EEM area was observed in the diabetic patients of the present study population. Moreover, in accordance to the observations by Nicholls et al,4 the degree of stenosis on QCA and the remodeling index of the lesions of the present study did not differ between the two patient populations, although more plaque was observed in diabetic patients. The discrepancy could be explained by the presence of diffuse atherosclerosis also in the reference segment of plaques of diabetic patients, as previously reported.21 As a consequence, the QCA measurements as well as the remodeling index may underestimate the actual plaque burden. Moreover, previous studies showed that coronary arteries of diabetic patients tend to undergo negative remodeling.21,22 Nevertheless, no correlation between vessel shrinkage and the presence of diabetes was observed in the present study. Indeed, contrary to previous studies the majority of coronary arteries that were examined invasively in the present study contained non-obstructive plaques. In addition, only a small proportion of patients in the current study were insulin dependent, whereas negative remodeling may be more strongly related to insulin treatment than to non-insulin dependent diabetes.22
Plaque Composition
In the present study, the proportion of completely calcified plaques on MSCT was larger in patients with diabetes as compared with patients without diabetes. Similar observations on coronary calcifications were reported in previous studies using electron beam computed tomography and MSCT.5,12 Indeed, in a recent MSCT study 49% of plaques were classified as completely calcified in diabetic patients.12 In contrast to the above study, however, diabetic patients in the present study contained a larger proportion of mixed plaques and a smaller proportion of non-calcified plaques. This finding could be explained by a higher risk profile of patients as compared to the population of the previous study. Accordingly, atherosclerosis could be more advanced (with more extensive calcifications) in these patients. As a consequence, the number of mixed plaques was identical in diabetic patients and patients without diabetes.
In accordance with the findings on MSCT, a larger amount of dense calcium was observed in the plaques of diabetic patients on VH IVUS. This observation is in line with a pathological study by Burke et al23 who demonstrated that type 2 diabetes was associated with a larger area of calcified matrix. Similar findings were recently reported by Nasu et al24 who investigated coronary plaque composition on VH IVUS in the target vessels of 36 type 2 diabetic patients and compared the findings with patients without diabetes; the authors reported a larger amount of dense calcium in the lesions of diabetic patients. On visual evaluation, more plaques of diabetic patients in the present study contained features of fibrocalcific plaque. Indeed, based on pathological investigations fibrocalcific plaque may resemble the healing process following previous plaque ruptures or erosions and accordingly represent advanced atherosclerotic lesions.17
Finally and importantly, together with the presence of extensive calcifications a larger amount of necrotic core was observed in the plaques of diabetic patients on VH IVUS. Indeed, increased amount of necrotic core was shown to be a feature of increased risk to plaque rupture.20 Nevertheless, the proportion of TCFA (the precursor lesion of plaque rupture) was identical in diabetic patients and in patients without diabetes. The explanation of these findings may be twofold. First, the patients of the study were extensively treated with cardiovascular medication, which possibly could have resulted in a more stabilized coronary plaque profile and thus decreased development of TCFA. Second, the coexistence of more extensive calcifications and the absence of direct evidence of vulnerable plaque morphology on VH IVUS may imply different pathophysiological mechanisms of plaque instability in diabetes.23 Further studies are necessary to better understand the mechanisms that lead to plaque instability and cardiovascular events in diabetic patients.
Limitations
The findings of the study are based on a relatively small patient population. Concerning MSCT, the technique is still associated with an elevated x-ray dose, while also the administration of contrast material is required. Also, no validated algorithms that allow quantification of plaque stenosis, volume, and composition are available for MSCT at present. Thus, in the present study MSCT studies were analyzed using a qualitative visual approach without the use of dedicated software algorithms or attenuation value measurements. The limitation of VH IVUS is the fact that the technique is relatively new, expensive and not widely available. As a result, the current observations need confirmation in future studies. In addition, both gray-scale IVUS and VH IVUS are invasive techniques and accordingly associated with risk. As a result their use remains restricted to symptomatic patients. Importantly, these investigations in larger patient cohorts should also include follow-up, as data on the potential prognostic value of both MSCT and VH IVUS are limited. Further studies should address whether certain plaque characteristics in diabetic patients may indeed be related to adverse outcome.
Conclusions
In conclusion, differences in coronary plaque patterns were observed on MSCT, gray-scale, and VH IVUS between patients with versus without diabetes. Diabetes was associated with a higher plaque extent as determined both on MSCT and gray-scale IVUS. Concerning plaque composition, more calcified plaques were observed on MSCT in patients with diabetes. On VH IVUS, plaques of diabetic patients contained a larger amount of dense calcium and were of a more advanced stage based on visual qualification. Thus, MSCT may potentially be used to explore patterns of coronary atherosclerosis in diabetic patients.
References
Haffner SM, Lehto S, Rönnemaa T, Pyörälä K, Laakso M. Mortality from coronary heart disease in subjects with type 2 diabetes and in nondiabetic subjects with and without prior myocardial infarction. N Engl J Med 1998;339:229-34.
Giri S, Shaw LJ, Murthy DR, Travin MI, Miller DD, Hachamovitch R, et al. Impact of diabetes on the risk stratification using stress single-photon emission computed tomography myocardial perfusion imaging in patients with symptoms suggestive of coronary artery disease. Circulation 2002;105:32-40.
Elhendy A, Arruda AM, Mahoney DW, Pellikka PA. Prognostic stratification of diabetic patients by exercise echocardiography. J Am Coll Cardiol 2001;37:1551-7.
Nicholls SJ, Tuzcu EM, Crowe T, Sipahi I, Schoenhagen P, Kapadia S, et al. Relationship between cardiovascular risk factors and atherosclerotic disease burden measured by intravascular ultrasound. J Am Coll Cardiol 2006;47:1967-75.
Schurgin S, Rich S, Mazzone T. Increased prevalence of significant coronary artery calcification in patients with diabetes. Diabetes Care 2001;24:335-8.
Schroeder S, Kopp AF, Baumbach A, Meisner C, Kuettner A, Georg C, et al. Noninvasive detection and evaluation of atherosclerotic coronary plaques with multislice computed tomography. J Am Coll Cardiol 2001;37:1430-5.
Leber AW, Becker A, Knez A, von Ziegler F, Sirol M, Nikolaou K, et al. Accuracy of 64-slice computed tomography to classify and quantify plaque volumes in the proximal coronary system: A comparative study using intravascular ultrasound. J Am Coll Cardiol 2006;47:672-7.
Nasu K, Tsuchikane E, Katoh O, Vince DG, Virmani R, Surmely JF, et al. Accuracy of in vivo coronary plaque morphology assessment: A validation study of in vivo virtual histology compared with in vitro histopathology. J Am Coll Cardiol 2006;47:2405-12.
American Diabetes Association. Diagnosis and classification of diabetes mellitus. Diabetes Care 2008;31:S55-60.
Schuijf JD, Bax JJ, Salm LP, Jukema JW, Lamb HJ, van der Wall EE, et al. Noninvasive coronary imaging and assessment of left ventricular function using 16-slice computed tomography. Am J Cardiol 2005;95:571-4.
Schuijf JD, Pundziute G, Jukema JW, Lamb HJ, van der Hoeven BL, de Roos A, et al. Diagnostic accuracy of 64-slice multislice computed tomography in the noninvasive evaluation of significant coronary artery disease. Am J Cardiol 2006;98:145-8.
Pundziute G, Schuijf JD, Jukema JW, Boersma E, Scholte AJ, Kroft LJ, et al. Noninvasive assessment of plaque characteristics with multislice computed tomography coronary angiography in symptomatic diabetic patients. Diabetes Care 2007;30:1113-9.
Tuzcu EM, Kapadia SR, Tutar E, Ziada KM, Hobbs RE, McCarthy PM, et al. High prevalence of coronary atherosclerosis in asymptomatic teenagers and young adults: Evidence from intravascular ultrasound. Circulation 2001;103:2705-10.
Mintz GS, Nissen SE, Anderson WD, Bailey SR, Erbel R, Fitzgerald PJ, et al. American College of Cardiology Clinical expert consensus document on standards for acquisition, measurement and reporting of intravascular ultrasound studies (IVUS). A report of the American College of Cardiology task force on clinical expert consensus documents. J Am Coll Cardiol 2001;37:1478-92.
Nair A, Kuban BD, Tuzcu EM, Schoenhagen P, Nissen SE, Vince DG. Coronary plaque classification with intravascular ultrasound radiofrequency data analysis. Circulation 2002;106:2200-6.
Carlier SG, Mintz GS, Stone GW. Imaging of atherosclerotic plaque using radiofrequency ultrasound signal processing. J Nucl Cardiol 2006;13:831-40.
Virmani R, Kolodgie FD, Burke AP, Farb A, Schwartz SM. Lessons from sudden coronary death: A comprehensive morphological classification scheme for atherosclerotic lesions. Arterioscler Thromb Vasc Biol 2000;20:1262-75.
Pundziute G, Schuijf JD, Jukema JW, Decramer I, Sarno G, Vanhoenacker PK, et al. Head-to-head comparison of coronary plaque evaluation between multislice computed tomography and intravascular ultrasound radiofrequency data analysis. J Am Coll Cardiol Intv 2008;1:176-82.
Falk E, Shah PK, Fuster V. Coronary plaque disruption. Circulation 1995;92:657-71.
Kolodgie FD, Virmani R, Burke AP, Farb A, Weber DK, Kutys R, et al. Pathologic assessment of the vulnerable human coronary plaque. Heart 2004;90:1385-91.
Jensen LO, Thayssen P, Mintz GS, Maeng M, Junker A, Galloe A, et al. Intravascular ultrasound assessment of remodelling and reference segment plaque burden in type-2 diabetic patients. Eur Heart J 2007;28:1759-64.
Kornowski R, Mintz GS, Lansky AJ, Hong MK, Kent KM, Pichard AD, et al. Paradoxic decreases in atherosclerotic plaque mass in insulin-treated diabetic patients. Am J Cardiol 1998;81:1298-304.
Burke AP, Kolodgie FD, Zieske A, Fowler DR, Weber DK, Varghese PJ, et al. Morphologic findings of coronary atherosclerotic plaques in diabetics: A postmortem study. Arterioscler Thromb Vasc Biol 2004;24:1266-71.
Nasu K, Tsuchikane E, Katoh O, Fujita H, Surmely JF, Ehara M, et al. Plaque characterization by virtual histology intravascular ultrasound analysis in type II diabetic patients. Heart 2007;94:429-33.
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Author information
Authors and Affiliations
Corresponding author
Additional information
Gabija Pundziute received training fellowship grant from the European Society of Cardiology.
Jacob M. van Werkhoven received a research grant from the Netherlands Society of Cardiology.
Gaetano Nucifora received fellowship grant from the European Association of Percutaneous Cardiovascular Interventions.
Jeroen J. Bax received grants from GE Healthcare, BMS medical imaging, St Jude, Medtronic, Boston Scientific, Biotronik, and Edwards Lifesciences
See related editorial, doi: 10.1007/s12350-009-9069-x
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Pundziute, G., Schuijf, J.D., Jukema, J.W. et al. Type 2 diabetes is associated with more advanced coronary atherosclerosis on multislice computed tomography and virtual histology intravascular ultrasound. J. Nucl. Cardiol. 16, 376–383 (2009). https://doi.org/10.1007/s12350-008-9046-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12350-008-9046-9