Abstract
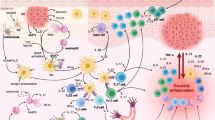
This is a summary of the original article “An Anti-OX40 Antibody to Treat Moderate-to-Severe Atopic Dermatitis: a Multicentre, Double-blind, Placebo-Controlled Phase 2b Study”. Atopic dermatitis (AD) is an inflammatory skin disease caused by a complex interplay of genetic factors, alterations to the skin microenvironment, and immune dysregulation, including T cells that have become uncontrolled. Rocatinlimab is an investigational agent that blocks OX40, a receptor on activated T cells that has an important role in inflammatory conditions such as AD. This summary of research provides an overview of a previously published article on the results of a phase 2b study of patients with moderate-to-severe AD who were treated with different doses of rocatinlimab or placebo and followed for up to 56 weeks. Rocatinlimab significantly improved the symptoms of AD and was well tolerated. The most common adverse events were fever, nasopharyngitis, and chills. This study supports rocatinlimab as a potentially safe and effective treatment for moderate-to-severe AD.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Data Availability
Individual participant data that underlie the results reported in this article, after deidentification (text, tables, figures, and appendices), and the study protocol, reporting and analysis plan, dataset specifications, and clinical study report will be available to investigators whose proposed use of the data has been approved by an independent review committee identified for this purpose. Data will be available online beginning 3 months and ending 20 years after article publication, marketing approval received in North America, Europe, and Japan, and completion of worldwide development programs.
Reference
Guttman-Yassky E, Simpson EL, Reich K, Kabashima K, Igawa K, Suzuki T, Mano H, Matsui T, Esfandiari E, Furue M. An anti-OX40 antibody to treat moderate-to-severe atopic dermatitis: a multicentre, double-blind, placebo-controlled phase 2b study. Lancet. 2023;401(10372):204–14. https://doi.org/10.1016/S0140-6736(22)02037-2.
Acknowledgements
The authors acknowledge the contributions of Michael Croft and thank the patients and study investigators, without whom this work could not have been completed.
Medical Writing and Editorial Assistance
Medical writing and editorial assistance were provided by Rebecca Lane and Kristin Carlin of Peloton Advantage, an OPEN Health company, and by Shannon Rao of Amgen Inc. Medical writing support was funded by Amgen Inc. and Kyowa Kirin Co., Ltd.
Funding
This study was funded by Kyowa Kirin, Co., Ltd. Publication fees were funded by Amgen, Inc. and Kyowa Kirin, Co., Ltd.
Author information
Authors and Affiliations
Contributions
Emma Guttman-Yassky, Eric L. Simpson, Kristian Reich, Kenji Kabashima, Ken Igawa, Tetsuya Suzuki, Hirotaka Mano, Takeshi Matsui, Ehsanollah Esfandiari, and Masutaka Furue reviewed and approved the final document.
Corresponding author
Ethics declarations
Conflict of Interest
Please see original article for full author disclosures.
Ethical Approval
The study protocol was approved by the Institutional Review Board or Independent Ethics Committee and regulatory health authorities in accordance with local regulations before study commencement. The principal investigator and sub-investigators were responsible for conducting the study in full accordance with the Declaration of Helsinki and the Good Clinical Practice Guidelines. All patients provided written informed consent before participating.
Additional information
Prior Publication: This is a summary of a peer-reviewed article previously published in the journal Lancet. 2023; 401(10372):204–214. doi: https://doi.org/10.1016/S0140-6736(22)02037-2.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Guttman-Yassky, E., Simpson, E.L., Reich, K. et al. Summary of Research: An Anti-OX40 Antibody to Treat Moderate-to-Severe Atopic Dermatitis: A Multicentre, Double-Blind, Placebo-Controlled Phase 2b Study. Adv Ther 41, 928–931 (2024). https://doi.org/10.1007/s12325-023-02755-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-023-02755-z