Abstract
Introduction
Chorea is characterized by sudden, involuntary movements that interfere with quality of life (QOL). Utility values measure preferences for different health states and reflect societal perceived disease severity. To date, no studies have reported utility values specifically for Huntington’s disease (HD) chorea. We estimated impact on QOL of HD chorea severity using utility values from the general population.
Methods
Participants were enrolled using computer-assisted telephone interviews. Participants read vignettes describing four health states for varying levels of chorea severity, with the same underlying HD severity. Time trade-off (TTO) methods were used to estimate utility values, which range from −1 (worse than death) to +1 (perfect health) and represent the number of years in an imperfect health state an individual is willing to give up to live in full health. TTO utilities were augmented with visual analog scale (VAS) participant responses. The primary outcome was HD chorea utility estimated by TTO.
Results
Mean ± SD TTO-derived utility values were 0.07 ± 0.52, 0.26 ± 0.50, 0.48 ± 0.47, and 0.64 ± 0.41 for severe, moderate/severe, moderate/mild, and mild chorea severity, respectively. Differences between each health state and its adjacent less severe health state were statistically significant (all P < 0.0001). Respondents were willing to give up 3.6, 5.2, 7.4, and 9.3 years during a 10-year life span to avoid living with mild, mild/moderate, moderate/severe, and severe chorea, respectively. VAS and TTO results were consistent.
Conclusions
Significant decreases in utility values were seen as HD chorea severity increased. These data can be leveraged for cost-effectiveness modeling to better understand the value of treatments for chorea.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Why carry out this study? |
Patients with Huntington’s disease (HD) often experience abnormal involuntary movements, or chorea, which can interfere with daily functioning and impair quality of life. |
This study examined the impact of HD chorea severity on quality of life using utility values estimated from the general population. |
What was learned from this study? |
Survey respondents were able to recognize that chorea negatively affects patients with HD, with greater impact seen in patients with more severe chorea. |
The utility values that were determined in this study may be beneficial for decision makers to quantify the impact of symptoms and potential benefits of treatments specifically for chorea associated with HD. |
Introduction
Huntington’s disease (HD) is a hereditary neurodegenerative disorder that typically manifests in adulthood and is characterized by motor, cognitive, and psychiatric symptoms [1, 2]. The prevalence of HD varies across different regions, with higher rates in Western countries (10.6–13.7 per 100,000 population) and lower rates in Asian countries (0.4 per 100,000 population) [1, 3,4,5]. Over the course of the disease, approximately 90% of patients will present with chorea, a prominent motor symptom of HD [3]. Chorea associated with HD is characterized by sudden, involuntary movements that can affect any muscle and can interfere with daily functioning, as well as increase the risk of injury [3]. Thus, HD can significantly affect quality of life (QOL), with fear of falling, loss of independence, and impact on family life being the key concerns among patients with HD and chorea associated with HD [6, 7].
Currently, there is no cure for HD and disease management focuses on controlling the triad of progressive symptoms. Two drug therapies, tetrabenazine and deutetrabenazine, are approved by the US Food and Drug Administration for the treatment of chorea associated with HD [8, 9]. These medications reduce the severity of chorea symptoms and improve patients’ QOL, but do not cure the cause of the involuntary movements [8, 9].
Utility values provide insight on the impact of disease states on QOL by measuring the preferences for different health states. There are several methods for estimating utility values, including time trade-off (TTO) and standard gamble (SG). The TTO method has been widely used to measure utilities for health states and has been shown to have less total bias than methods such as SG [10,11,12,13]. Moreover, SG is associated with significant cognitive burden and has been shown to overestimate utilities [10,11,12].
Several existing studies examine the utility of different stages of HD [14,15,16]; however, no studies to date have reported utility values specifically for HD chorea or measured the decreased QOL experienced by patients with HD due to chorea. This study aims to address the gap in literature by estimating the utility of several health states related to chorea associated with HD using the TTO method, while considering important factors mediating QOL.
Methods
Study Design and Population
This study used a one-time computer-assisted telephone interview (CATI) to estimate utilities for health states of chorea associated with HD from the societal perspective in the USA. Health states described varying levels of chorea severity with the same underlying HD severity (Table 1). Participants were shown written vignettes depicting the four health states of chorea (i.e., mild, mild/moderate, moderate/severe, severe). Each vignette described a different health state of chorea, with the same underlying HD severity, based on movement symptoms and the QOL associated with the health state (Table S1 in the Supplementary Material). To ensure that vignettes provided accurate and comprehensive descriptions of health states, feedback was obtained from four external physicians via one-on-one telephone interviews. Eligible external physicians were licensed neurologists in the USA who have treated patients with HD for at least 5 years and have treated at least five patients with HD over the past 12 months. After reading the vignette for a particular health state, participants answered a series of TTO questions to assess their perceived utility of the health state (see Supplementary Appendix in the Supplementary Material for the TTO data collection procedure). Each participant completed a training task and practice task prior to evaluating the four health states to ensure that they understood the concept of TTO.
The source population consisted of the general US public. Participants were recruited by SurveyEngine via an online panel. Individuals willing to participate responded to an initial online survey that verified eligibility according to the inclusion criteria. Eligible participants were aged at least 18 years without a diagnosis of HD and able to speak and read English. If participants were deemed eligible, informed consent was collected prior to proceeding onto the main survey questions and scheduling a CATI. The survey terminated if informed consent was not provided.
Outcomes and Analyses
Participant demographic characteristics were collected during the initial online survey. The primary outcome was utilities estimated from the TTO. TTO questions were illustrated using visual aids (Fig. S1 in the Supplementary Material), and participants were randomly assigned to see the health states in one of two orders, either from most severe chorea to least severe or from least severe to most severe chorea. Utility values for TTO range from −1 (worse than death) to +1 (perfect health), with the smallest difference being 0.05. TTO utilities were validated using utilities obtained from the visual analog scale (VAS), which ranges from 0 (worst imaginable health) to 100 (best imaginable health).
Participant characteristics, including age, sex, race, region, education, employment, income, comorbidities, familiarity with HD, and general health status, were summarized descriptively.
VAS ratings were divided by 100 to obtain utility values (rescaled 0–1). Utility values for TTO were calculated on the basis of responses and summarized across participants separately for each health state. VAS and TTO ratings were assessed by testing results for logical errors. Logical errors were defined as a participant rating a more severe health state (e.g., HD with severe chorea) as better than a less severe health state (e.g., HD with moderate/severe chorea). To avoid response bias, all ratings by any participant with logical errors were censored from the primary analyses. Sensitivity analyses were conducted by only removing answers with logical errors, while retaining participants’ logical answers, and by keeping all answers from all participants to assess the impact of removing logical errors on utility values.
For the primary analysis, statistical comparisons were conducted for adjacent health states (e.g., comparing the utility of HD with severe chorea to the utility of HD with moderate/severe chorea) using paired t tests with P values. To examine the potential ordering effect on the ratings, utility values from participants randomly assigned to see health states in each order (i.e., from most severe to least severe, and from least severe to most severe) were analyzed separately. The utilities for each health state were compared between the two orderings using two-sample t tests, and P values were calculated.
Compliance with Ethics Guidelines
The interview guide and survey instrument were approved by the Western Institutional Review Board (1-1308175-1). This study was performed in accordance with the Declaration of Helsinki of 1964 and its later amendments. Eligible respondents provided informed consent to participate. Each individual’s consent to participate in the study was collected at the end of the informed consent and the responses were recorded in the survey database. The survey terminated if individuals indicated that they were not willing to participate. Individuals who provided consent moved to the main survey questions and were scheduled for a CATI. Only individuals who completed the full survey and the CATI received compensation for participation.
Results
Participant Characteristics
Overall, 205 participants took the study survey, with 50 respondents excluded from this analysis because of logical errors in their responses. The remaining 155 participants provided valid responses to all TTO and VAS questions. The mean (standard deviation [SD]) age was 47 (18) years and 51.6% were female (Table 2). Overall, 36.8% of participants reported no comorbidities. Among participants with a comorbidity, the most common conditions were depression (24.5%) and hypertension (23.2%). According to participant responses, mean (SD) current health status was 0.79 (0.16), for which a score of 1 represents best imaginable health.
Utility Values
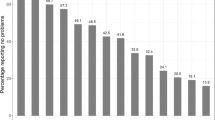
Utility increments from a more severe health state to its adjacent less severe health state ranged from 0.16 to 0.22 and were all statistically significant (Table 3; P < 0.0001 for each comparison). The distribution of TTO responses for each health state are shown in Fig. S2 in the Supplementary Material. According to utility values, participants were willing to give up 9.3, 7.4, 5.2, and 3.6 years during a 10-year life span to avoid living with severe, moderate/severe, moderate/mild, and mild chorea, respectively (Fig. 1).
VAS utility increments from a more severe to less severe chorea health state ranged from 0.12 to 0.15, depending on the severity of chorea. Differences between each health state and its adjacent health states were found to be significant (P < 0.0001 for each comparison).
Sensitivity analyses showed that excluding illogical participant responses did not significantly alter TTO or VAS responses (Table S2 in the Supplementary Material). For TTO utilities, no ordering effect was found in any of the health states (P > 0.3 for all health states); however, participants who saw health states from most to least severe had significantly higher VAS scores for all chorea health states compared to participants who saw health states from least to most severe (P < 0.05 for all health states; Table S3 in the Supplementary Material).
Discussion
Although several studies have examined the utility of different stages of HD [14,15,16], none have focused specifically on chorea or measured the decreases in QOL due to chorea among patients with HD. By incorporating non-chorea HD symptoms into the vignettes and keeping these symptoms constant across the four health states, we were able to isolate and analyze utility specifically associated with chorea. This study found significant decreases in utility as the severity of chorea worsened, with the utility increment from a more severe health state to its adjacent less severe health state ranging from 0.16 to 0.22 based on responses to TTO questions. These findings suggest that participants were willing to give up 1.6 to 2.2 years during a 10-year life span to avoid living with more severe chorea. Although HD chorea is known to affect patient QOL, it is difficult to compare our findings with previous literature because most studies were focused on HD overall. However, in a previous study that elicited utility values for HD overall using the 36-item Short Form Health Survey (SF-36) [16], higher utility values were reported compared with the utilities estimated in this study. The previous study reported values of 0.39 (after conversion to a 0–1 scale) for the two most severe stages of HD combined compared to 0.07 for the most severe chorea health state in the current study [16]. Potential reasons for this difference include self-reporting limitations and varying severities of each health state across the two studies. Results from this study were consistent with those observed in utility studies for other severe movement disorders, such as Parkinson’s disease [17,18,19]. For example, in one study, the utility for the most severe health state in Parkinson’s disease ranged from −0.13 to 0.13 [17]. This utility estimate is consistent with the utility associated with the most severe chorea health states in the current study (i.e., mean TTO utility = 0.07).
Estimated utility values from the current study reflect the impact of chorea on both motor function and QOL, leading to more accurate estimates of the values of chorea associated with HD. Additionally, this study used the TTO method rather than the SG method to elicit utility values. Although SG is grounded in expected utility theory, it is associated with significant cognitive burden because respondents may have difficulty interpreting and assessing probabilities [20]. TTO has become the more commonly used method in recent years because it has been shown to be associated with less total bias than SG [11].
This study has some limitations to consider. First, previous studies have evaluated HD overall, limiting the comparability of results from this study, which focused on chorea associated with HD. One important limitation of TTO analyses is that respondents frequently find TTO surveys cognitively burdensome, and thus may have difficulty providing logical and accurate responses to survey questions [21]. Here, we have taken this effect into account by performing a sensitivity analysis, comparing results with illogical answers included to those with illogical answers excluded; no significant differences were observed (Table S2 in the Supplementary Material). Additionally, there may have been discrepancies between participants’ understanding of the health states and patients’ real-life experiences despite physicians providing feedback on the vignettes, which informed participants from the general population about health states. Lastly, the current study only included four health states and was not able to assess utility values for all potential HD chorea symptoms and levels of severity. Importantly, HD chorea symptoms may vary greatly among individuals; thus, it is difficult to capture the full range of experiences of those with HD chorea.
Conclusions
In this survey-based study with participants representative of the US general population, significant decreases in utility values were seen as severity of chorea associated with HD worsened. These findings suggest that participants recognize the negative impact of HD chorea on daily functioning and QOL. The utility values obtained from this study may assist in estimating the impact of chorea and the benefit of treatment in patients with HD and can be leveraged by decision makers for future cost-effectiveness analyses.
References
Bates GP, Dorsey R, Gusella JF, et al. Huntington disease. Nat Rev Dis Primers. 2015;1:15005. https://doi.org/10.1038/nrdp.2015.5.
Ross CA, Aylward EH, Wild EJ, et al. Huntington disease: natural history, biomarkers and prospects for therapeutics. Nat Rev Neurol. 2014;10:204–16. https://doi.org/10.1038/nrneurol.2014.24.
Haddad MS, Cummings JL. Huntington’s disease. Psychiatr Clin N Am. 1997;20:791–807. https://doi.org/10.1016/s0193-953x(05)70345-2.
Pringsheim T, Wiltshire K, Day L, Dykeman J, Steeves T, Jette N. The incidence and prevalence of Huntington’s disease: a systematic review and meta-analysis. Mov Disord. 2012;27:1083–91. https://doi.org/10.1002/mds.25075.
McColgan P, Tabrizi SJ. Huntington’s disease: a clinical review. Eur J Neurol. 2018;25:24–34. https://doi.org/10.1111/ene.13413.
Thorley EM, Iyer RG, Wicks P, et al. Understanding how chorea affects health-related quality of llfe in Huntington disease: an online survey of patients and caregivers in the United States. Patient. 2018;11:547–59. https://doi.org/10.1007/s40271-018-0312-x.
Ho AK, Hocaoglu MB, European Huntington’s Disease Network Quality of Life Working Group. Impact of Huntington’s across the entire disease spectrum: the phases and stages of disease from the patient perspective. Clin Genet. 2011;80:235–9. https://doi.org/10.1111/j.1399-0004.2011.01748.x.
XENAZINE® (tetrabenazine) [package insert] Fontaine-les-Dijon, France: Recipharm Fontaine SAS; Sept 2017.
Austedo® (deutetrabenazine) tablets [prescribing information]. Parsippany, NJ: Teva Pharmaceuticals USA, Inc.; 2021.
Bleichrodt H, Johannesson M. Standard gamble, time trade-off and rating scale: experimental results on the ranking properties of QALYs. J Health Econ. 1997;16:155–75. https://doi.org/10.1016/s0167-6296(96)00509-7.
Bleichrodt H. A new explanation for the difference between time trade-off utilities and standard gamble utilities. Health Econ. 2002;11:447–56. https://doi.org/10.1002/hec.688.
van Osch SM, Wakker PP, van den Hout WB, Stiggelbout AM. Correcting biases in standard gamble and time tradeoff utilities. Med Decis Mak. 2004;24:511–7. https://doi.org/10.1177/0272989X04268955.
Wright DR, Wittenberg E, Swan JS, Miksad RA, Prosser LA. Methods for measuring temporary health states for cost-utility analyses. Pharmacoeconomics. 2009;27:713–23. https://doi.org/10.2165/11317060-000000000-00000.
Dorey J, Clay E, Khemiri A, Belhadj A, Cubillo PT, Toumi M. The quality of life of Spanish patients with Huntington’s disease measured with H-QoL-I and EQ-5D. J Mark Access Health Policy. 2016. https://doi.org/10.3402/jmahp.v4.27356.
van Walsem MR, Howe EI, Ruud GA, Frich JC, Andelic N. Health-related quality of life and unmet healthcare needs in Huntington’s disease. Health Qual Life Outcomes. 2017;15:6. https://doi.org/10.1186/s12955-016-0575-7.
Hawton A, Green C, Goodwin E, Harrower T. Health state utility values (QALY weights) for Huntington’s disease: an analysis of data from the European Huntington’s Disease Network (EHDN). Eur J Health Econ. 2019;20:1335–47. https://doi.org/10.1007/s10198-019-01092-9.
Lowin J, Bergman A, Chaudhuri KR, et al. A cost-effectiveness analysis of levodopa/carbidopa intestinal gel compared to standard care in late stage Parkinson’s disease in the UK. J Med Econ. 2011;14:584–93. https://doi.org/10.3111/13696998.2011.598201.
Palmer CS, Schmier JK, Snyder E, Scott B. Patient preferences and utilities for ‘off-time’ outcomes in the treatment of Parkinson’s disease. Qual Life Res. 2000;9:819. https://doi.org/10.1023/a:1008903126315.
Pietzsch JB, Garner AM, Marks WJ Jr. Cost-effectiveness of deep brain stimulation for advanced Parkinson’s disease in the United States. Neuromodulation. 2016;19:689–97. https://doi.org/10.1111/ner.12474.
Brazier J, Ratcliffe J, Saloman J, Tsuchiya A. Measuring and valuing health benefits for economic evaluation. Oxford: Oxford University Press; 2016.
Lugner AK, Krabbe PFM. An overview of the time trade-off method: concept, foundation, and the evaluation of distorting factors in putting a value on health. Expert Rev Pharmacoecon Outcomes Res. 2020;20(4):331–42. https://doi.org/10.1080/14737167.2020.1779062.
Acknowledgements
Funding
This study and the journal's Rapid Service and Open Access fees were funded by Teva Pharmaceutical Industries Ltd., Tel Aviv, Israel. Teva Pharmaceutical Industries Ltd. developed deutetrabenazine and markets it for the treatment of chorea associated with Huntington’s disease.
Medical Writing and Editorial Assistance
We thank Caryn Gordon, PharmD (Cello Health Communications/MedErgy, with funding from Teva Pharmaceuticals), for medical writing and editorial assistance in the preparation of this manuscript.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Author Contributions
Daniel O. Claassen contributed to the interpretation of the data and the drafting of the manuscript. Debbie Goldschmidt, Mo Zhou, and Rajeev Ayyagari contributed to the conception and design of the analysis, analysis and interpretation of the data, and the drafting of the manuscript. Sam Leo and Rinat Ribalov contributed to the conception and design of the analysis, interpretation of the data, and the drafting of the manuscript.
Prior Presentation
This study was previously presented, in part, at the Academy of Managed Care Pharmacy 2021 Virtual National Meeting (April 12–16, 2021) and the American Academy of Neurology 2021 Virtual Annual Meeting (April 17–22, 2021).
Disclosures
Daniel O. Claassen has received research support from Griffin Foundation, Huntington’s Disease Society of America, Michael J. Fox Foundation, National Institute of Neurological Disorders and Stroke, and National Institute on Aging; pharmaceutical grant support from AbbVie, Acadia, Biogen, Bristol Myers Squibb, Cerecor, Eli Lilly, Jazz Pharmaceuticals, Lundbeck, Teva Neuroscience, Vaccinex, and Wave Life Sciences; and is a consultant or advisory board member of Acadia, Adamas, Alterity, Lundbeck, Neurocrine, Teva Neuroscience, and Wave Life Sciences. Rajeev Ayyagari is an employee of Analysis Group, Inc. Debbie Goldschmidt is an employee of Analysis Group, Inc. Mo Zhou is an employee of Analysis Group, Inc. Sam Leo is an employee of Teva Pharmaceuticals. Rinat Ribalov is an employee of Teva Pharmaceutical Industries Ltd.
Compliance with Ethics Guidelines
The interview guide and survey instrument were approved by the Western Institutional Review Board (1-1308175-1). This study was performed in accordance with the Declaration of Helsinki of 1964 and its later amendments. Eligible respondents provided informed consent to participate. Each individual’s consent to participate in the study was collected at the end of the informed consent and the responses were recorded in the survey database. The survey terminated if individuals indicated that they were not willing to participate. Individuals who provided consent moved to the main survey questions and were scheduled for a CATI. Only individuals who completed the full survey and the CATI received compensation for participation.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Author information
Authors and Affiliations
Corresponding author
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Claassen, D.O., Ayyagari, R., Goldschmidt, D. et al. Defining Utility Values for Chorea Health States in Patients with Huntington’s Disease. Adv Ther 39, 1784–1793 (2022). https://doi.org/10.1007/s12325-022-02046-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-022-02046-z