Abstract
Introduction
Several viscosupplement treatments are available for patients suffering from painful osteoarthritis (OA) of the knee, but few comparative clinical trials have been conducted. The primary objective of the trial was to demonstrate the non-inferiority of Synolis VA (80 mg hyaluronic acid and 160 mg sorbitol) (Group HA1) to Synvisc-One (48 mg hylan GF-20) (Group HA2) at Day 168 in terms of pain relief efficacy in patients with knee OA (Kellgren and Lawrence radiological stage II or III) in whom oral treatment with analgesics, NSAIDs or weak opioids provided insufficient clinical responses or were poorly tolerated.
Methods
This was a prospective, multicentre, comparative, randomized, double-blinded trial comparing the two previously indicated viscosupplements, HA1 and HA2. The average VAS pain score (1–100) was 62.5 at baseline (Day 0). The patients were randomized into two parallel groups at Day 0 and followed until Day 168. They received one injection of either HA1 or HA2. The primary end point was the evolution of the Western Ontario and McMaster University (WOMAC) pain index at D168 in the groups. One of the secondary end points was the daily assessment of this index by the patient for 7 days following the injection and thereafter at Day 14. The other secondary end points were the WOMAC pain, stiffness, function and total scores assessed at Day 28, Day 84 and Day 168. At Day 168, efficacy and satisfaction were assessed by the evaluator and by the patient using a Likert scale (7 points). Moreover, the number of strict responders in each group was evaluated according to the The Osteoarthritis Research Society International (OARSI) Standing Committee for Clinical Trials Response Criteria Initiative and the Outcome Measures in Rheumatology (OMERACT) criteria (OMERACT-OARSI). The per protocol (PP) population was used for the primary analysis.
Results
A total of 202 patients were randomized. The patients were predominantly female (66%). The median age of the whole population was 65 years, and the median body mass index was 27.4 kg/m2. No statistically significant differences between the two treatment groups were observed for any of the demographic criteria. At Day 168, 197 had had no protocol violations (94 in the HA1 group and 103 in the HA2 group). The WOMAC pain score decreased in the two groups: − 29.2 ± 24.1 (SD) in the HA1 group and − 31.6 ± 25.5 (SD) in the HA2 group, confirming the non-inferiority of Synolis VA (P = 0.57 for the difference between groups). Regarding the secondary end points, no significant difference was observed at Day 14, Day 28, Day 84 or Day 168 for all the outcomes except stiffness at Day 28 (P = in favour of treatment received in HA2). The rate of responders was comparable between the two groups: 79% for HA1 and 77% for HA2. Both products were well tolerated. Serious adverse events were reported by four patients in the HA1 group and 3 in the HA2 group.
Conclusion
In this trial, we confirmed the non-inferiority of Synolis VA compared to Synvisc-One at Day 168 according to the WOMAC pain score. Safety was satisfying and comparable in the two groups.
Trial Registration
2017-A00034-49.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Why carry out this study? |
Osteoarthritis (OA) is a degenerative disease mainly affecting the knee joints. It highly restricts the ability of patients to perform their activities of daily living, walking disability being the main factor. |
Both OARSI and ESCEO recommendations show that intra-articular hyaluronic acid has established its place in the arsenal of non-surgical treatment of OA. However, with many viscosupplements (VS) being available in the market there are remarkably few clinical trials directly comparing different VS. |
This study was conducted to compare two different VS. for knee OA treatment, Synolis VA (80mg HA/160 mg sorbitol) and SynviscOne (hylan G-F 20). |
What was learned from the study? |
The study has demonstrated the non-inferiority of a single intra-articular injection of non-crosslinked HA plus Sorbitol vs. a single intra-articular injection of crosslinked hylan G-F 20, which had been previously been shown to be superior to intra-articular placebo. |
The study showed significant clinical benefit of both tested VS, in both the short and long term (months) in patient with symptomatic knee OA. |
This suggests focusing future research on determining the long-term effects of repeated IAHA injections on patient-reported outcomes and total knee replacement-sparing effects, on the assessment of disease-modifying effects and on the potential for combination therapy with other pharmacological and non-pharmacological therapies to optimize the management of knee OA. |
Digital Features
This article is published with digital features, including a summary slide, to facilitate understanding of the article. To view digital features for this article go to https://doi.org/10.6084/m9.figshare.13690432.
Introduction
Osteoarthritis (OA) is a degenerative disease characterized by progressive articular destruction that causes pain, joint stiffness and functional impairment [1]. Numerous pharmacological and non-pharmacological treatment modalities are available to manage OA. Recently, two international societies, namely the European Society for Clinical and Economic Aspects of Osteoporosis and Osteoarthritis (ESCEO) and Osteoarthritis Research Society International (OARSI), have provided guidelines for the management of knee OA [2, 3]. Even though there are some differences between these two guidelines, they emphasize that intra-articular injection of hyaluronic acid (HA) could be one of the treatment modalities. Although several reviews reported a clinically significant effect of the saline solution injection on pain relief [4], the most recent systematic reviews and meta-analyses on this topic concluded that HA provides an improvement in pain and function compared to a placebo [5,6,7]. Interestingly, some of these studies also suggest that HA is most likely comparable to or more effective than intra-articular corticosteroids for long-term pain relief, although these two modalities are not used for the same patients [8, 9]. More recently there has been an increased usage of platelet-rich plasma (PRP) injections, whether leucocyte-poor or not, and several studies demonstrated their efficacy in knee OA [10]. There is however an ongoing debate about the optimal volume of the PRP injected into a knee with reported variations going from 2 ml to as much as 8 ml per injection.
HA is a polysaccharide macromolecule belonging to the family of glycosaminoglycans and has a two-helix structure in the solid state or under certain physiological conditions in solution [11]. HA, produced by synoviocytes and chondrocytes, is the primary component of synovial joint fluid. A healthy knee contains ~ 2 ml of synovial fluid with an HA concentration of 2.5–4.0 mg/ml [12]. In fact, it acts as a lubricant and shock absorber, protecting the articular cartilage and joint structures from compressive and shear forces, thereby reducing articular cartilage wear. The rheological properties (i.e., viscosity and elasticity) are essential for good joint functionality. Furthermore, it supplies oxygen and nutrients to the surrounding tissues and removes carbon dioxide and metabolic wastes. The synovial fluid’s rheological properties, i.e., viscosity and elasticity, decrease over time because the concentration of HA and the average molecular weight (MW) decrease over time. Osteoarthritis leads to the progressive deterioration of articular cartilage [12, 13]. In addition to the viscoelastic properties of HA, the molecule has demonstrated anti-inflammatory properties, such as the inhibition of phagocytosis and reduction of inflammatory mediators [13].
The primary aim of viscosupplementation is first to improve the mechanical function of the joint by restoring the rheological properties. However, it is unclear how exogenous HA provides long-term effects knowing that after 48 h the solution is no longer detected in the affected joint [12]. However, recent studies indicate that the protective physiochemical functions of exogenous HA may have disease-modifying, long-term effects on articular cartilage associated with long-lasting symptom relief, particularly in the initial phases of OA. The current mechanism of action of exogenous HA is not fully understood, but HA may provide biological actions, including anti-inflammatory, antihyperalgesic, anti-nociceptive and anabolic effects by suppressing matrix metalloprotease (MMP) and interleukin 1 (IL-1) activity [14]. The American College of Rheumatology (ACR), European Society for Clinical and Economic Aspects of Osteoporosis and Osteoarthritis (ESCEO) task force, The European League Against Rheumatism (EULAR) and OARSI consider that there is currently no clinical evidence supporting an advantage in the efficacy and safety of one product over another. Furthermore, if certain intrinsic properties, such as the molecular weight of particular intraarticular HA (IAHA) preparations, provide beneficial results compared with other HA products, characteristics related to the individual patient are likely to prevail [13]. Surprisingly, few comparative clinical trials on different HA compounds have been conducted. In this trial, we compared hylan G-F20 (Synvisc-One®), a G-F20 preparation that comprises two crosslinked derivatives of hyaluronan with a molecular weight of 6 MDa12, and HA1 (Synolis VA®), a chemically non-modified sodium hyaluronate, with a molecular weight of 2 MDa, combined with sorbitol (4%).
Methods
This is a prospective, multicentre, phase IIIb, comparative, randomized, double-blinded trial comparing two viscosupplements, one containing a solution of 80 mg HA-160 mg sorbitol (HA1) and the other containing 48 mg hylan GF-20 (HA2), in two parallel groups over a period of 24 weeks. HA1 is a viscoelastic, sterile, apyrogenic, isotonic, buffered, 2% solution of sodium hyaluronate. Sodium hyaluronate used in HA1 is obtained from bacterial fermentation and has a high mean molecular weight (MW) of 2 MDa. HA2 is a sterile, nonpyrogenic, elastoviscous fluid containing hylan G-F 20 composed of Hylan A and Hylan B (8.0 mg ± 2.0 mg per ml) in buffered physiological sodium chloride solution (pH 7.2 ± 0.3).
Here, we opted for a non-inferiority trial design, which appeared to us to be the most appropriate design. Non-inferiority trials test whether a new treatment is not unacceptably less efficient than an active control treatment already in use. With continuous improvements in health technologies, the incremental benefits of newly developed treatments may be only marginal over existing treatments.
The primary end point was the evolution of the The Western Ontario and McMaster Universities Arthritis Index (WOMAC) pain index at Day 168 post-injection in both groups. This is a 24-item, condition-specific questionnaire available in a 5-point Likert and 100 mm visual analog scale (VAS) as well as an 11-box numerical rating scale. It has three subscales: pain (5 questions), stiffness (2 questions) and physical function (17 questions). Here, we used a 100 mm VAS rating scale with a pre-defined margin of non-inferiority as < 8 mm.
One of the secondary end points was the daily assessment of this index by the patient for 7 days following the injection and thereafter at Day 14. The other secondary end points were the WOMAC pain, stiffness, function and total scores assessed at Day 28, Day 84 and Day 168. At D168, efficacy and satisfaction were assessed by the evaluator and by the patient using a 7-point Likert scale.
The trial was conducted in accordance with the ethics principals of the Declaration of Helsinki. It was approved and registered under no. 2017-A00034-49 to the ANSM, and ethical approval was obtained from CPP Ile-de-France VI.
The patients were randomized into two parallel groups at Day 0 and followed until Day 168. Patients were randomized to one of the two treatment groups (HA1 or HA2) in a 1:1 ratio. Randomization was performed per centre and by block of four under the responsibility of Sylia Stat, Bourg la reine, France.
The investigator evaluator assessed the patient’s eligibility according to the inclusion and exclusion criteria. The same day (or within 24 h after inclusion), the injector physician injected the investigational product into the patient. Sealed patient unit boxes were undistinguishable. The patient was not directly in contact with the product unit. As such, both the patient and the evaluating investigator were unaware of the nature of the investigational product in the boxes. As a consequence, at the time of the primary efficacy criteria assessment at Day 168, neither the patient nor the investigator was aware of the allocated treatment.
The trial was conducted in three phases:
Information period: Day 1, the investigator proposed that the patient participate in this clinical trial and provided all relevant information related to this trial. An information notice and informed consent form were handed over to the patient.
Recruitment period: The patient gave his/her consent (signed informed consent), and the evaluator assessed the patient’s eligibility according to the inclusion and exclusion criteria. On the same day, Day 0, or within 24 h, viscosupplementation was administered by the physician injector.
Follow-up phase: At Day 1, Day 7, Day 28, Day 84 and Day 168, OA symptoms were evaluated (pain, functionality, stiffness), the safety of each product was monitored, and AEs were recorded.
The following patients were enrolled in the trial:
-
Men and women aged between 45 and 80 years with knee OA based on the American College of Rheumatology classification (i.e., knee pain, osteophyte and morning stiffness and/or crackling while walking) with:
-
Radiographically defined osteoarthritis: joint space narrowing and osteophyte on x-rays taken < 1 year previously with modified Kellgren-Lawrence grade Il–III where grade II showed definite osteophytes and definite narrowing of the joint space and grade III moderate multiple osteophytes and definite narrowing of joint spaces with some sclerosis and deformity of the bone contour.
-
Patients having had symptoms for at least 6 months prior to inclusion.
-
Subjects who were intolerant to or for whom the treatment with analgesics and/or NSAIDs and/or weak opioids was insufficient.
-
Subjects with WOMAC pain ≥ 40 mm on a visual analogue scale (VAS) ranging from 0–100 mm in the knee to treat and, if OA ws bilateral, < 10 mm (VAS) on the contralateral knee.
It was recommended that the use of concomitant medications should be kept to a minimum during the trial period. However, if it was considered necessary for patient well-being and would not interfere with the investigational product, the patients could be prescribed at the discretion of the investigator.
Patients were excluded if they had inflammatory joint disorder, had received previous viscosupplementation treatment within 6 months or corticosteroids within 3 months, infection in or around the study knee, relevant skin disease in the area of the injection site, injury or trauma of the study knee, or a history of allergy or intolerance to study preparations.
Statistical Analysis
The following hypotheses to determine the non-inferiority of HA1 2*2 ml versus HA2 6 ml were as follows: (1) power (1 − β) = 90%, (2) one-sided alpha significance level = 2.5%, (3) non-inferiority upper limit of the 95% confidence interval < 8, (4) standard deviation (SD): 16 on the WOMAC pain score and (5) minimum number of evaluable patients per group n = 85. The primary efficacy analysis was conducted in the per protocol (PP) population. Non-inferiority (HA1 vs. HA2) will be demonstrated if the upper limit of the 95% CI of the difference in the WOMAC pain score [Day 168-Day 0] HA1 - [Day 168-Day 0] HA2 is < 8 (mm). This analysis was also performed on the ITT patient population using the WOMAC pain score. The analysis of all secondary criteria was performed on the ITT and PP patient populations.
For quantitative variables, Student’s t-test was applied, or if the parametric test conditions were not fulfilled, a Mann-Whitney nonparametric test was used (Shapiro-Wilk test < 1%). For qualitative variables, a chi² test or Fisher’s correction was used as needed. The 95% confidence interval (CI) of the difference between the two treatment groups—[HA1: Day 168-Day 0] – [HA2: Day 168-Day 0], [HA1: Day 84-Day 0] – [HA2: Day 84-Day 0], [HA1: Day 28-Day 0] – [HA2: Day 28-Day 0]—was calculated. Primary statistical analysis was performed at a probability threshold of 2.5% and 5% for secondary statistical analysis for significance using two-sided tests or two-sided confidence intervals. All summaries and statistical analyses were generated using SAS (version 9.4, Cary, NC, USA).
Results
Subject Disposition, Demographics and Disease Characteristics at Baseline
The observation period of the trial covered 168 days of ambulatory care. For several reasons (protocol violations and premature discontinuation), data were analysed in different data sets: full population, intention to treat, safety ITT, per-protocol population and PP efficacy.
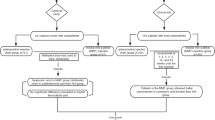
A total of 202 subjects were enrolled and randomized to HA1 (n = 96) and HA2 (n = 106) (Fig. 1). The ITT populations consisted of 201 (HA1 [n = 96], HA2 [n = 105]); all subjects received the trial injection in accordance with the randomization scheme. The PP population consisted of 177 (87.6%) subjects [HA1 (n = 85), HA2 (n = 89)]; 4 subjects were excluded from the PP analysis (2 in each group) presenting a deviation from the inclusion criteria. From the D84 visit, four patients (two in each group) were excluded from the PP population because of corticosteroid injection. From Day 168 visits, 17 patients (8 in the HA1 group and 9 in the HA2 group) were excluded: 11 patients because of visit times outside predefined windows and six patients because of corticosteroid injection.
Baseline demographic data for the PP population at the time of randomization are presented by treatment group in Table 1. Patients were predominantly female (66%). The median age of the whole population was 65 years, and the median body mass index was 27.4 kg/m2. More than half (70%) had a Kellgren-Lawrence (K-L) OA grade of III. No statistically significant difference between the two treatment groups was observed for any demographic criteria.
In the PP population, the means of the pain assessment scoring (VAS) were 62.1 ± 13.7 for the HA1 group (N = 94) and 63.3 ± 14.1 for the HA2® group (N = 103) (NS: Mann-Whitney test). The WOMAC criteria (pain, stiffness and functionality) show homogeneity of symptoms among patients between the groups at inclusion. This homogeneity was similar in both the PP and ITT populations, neither of which showed any statistically significant difference.
Primary Efficacy Outcome
The primary efficacy variable was the comparison between HA1 and HA2 regarding the change between Day 168 and Day 0 of the overall WOMAC pain score, which was used to assess the level of pain reported by the patient for the knee injected at Day 0. In the PP dataset (n = 177), both treatment groups showed a significant decrease in pain at Day 168, with an overall WOMAC pain score of − 29.9 ± 23.3 (SD) in the HA1 group and − 31.2 ± 25.2 (SD) in the HA2 group, with no significant difference between the treatment groups (P = 0.5).
The intergroup difference for the primary efficacy end point was − 1.6 mm (95% CI: -7.2, 4.0; P = 0.5) in the PP dataset (Fig. 2). As the upper limit of the 95% CI is < 8, it can be concluded that HA1 is not inferior to HA2 for this primary efficacy end point.
The analysis performed on the ITT population (n = 197) confirms the results observed in the PP population, demonstrating the non-inferiority of HA1 vs. HA2 on the primary end point.
Secondary Efficacy Outcomes
One of the secondary outcomes in this trial was the evolution of pain at Day 7 post-injection in the ITT population. Remarkably, after 1 week, a significant decrease in pain was observed in both groups, with no significant differences between the HA1 and HA2 groups. The decrease continued throughout the trial. A maximum pain decrease was observed at Day 14 in both treatment groups, and the benefit of pain reduction was maintained over time until the end of the follow-up at Day 168 (Fig. 3). No significant difference was observed at each time point in either the PP or ITT population. Nevertheless, the curve profile of both groups shows a slight difference; the pain reduction with HA1 seems gradual and constant until Day 168.
Regarding the components of the WOMAC, both groups showed a significant score improvement in stiffness and function, as well as in the total score, with no difference between treatment groups from baseline to Day 168 in the PP and ITT datasets. The WOMAC total score difference compares Day 0 -27.24 and -27.53 for HA1 and HA2, respectively. For both treatment groups, the maximum intensity of stiffness and physical function decrease was observed at Day 28 in the PP population, with no significant difference.
The OMERACT-OARSI score also showed clear clinical meaningfulness. At Day 168, the responder rates of HA1 and HA2 were 78.8% and 76.9%, respectively, with no significant difference between the two groups (P = 0.762).
Finally, the patient-assessed global pain satisfaction was similar for both treatments at Day 168. A total of 89.2% of HA1-treated patients and 88% of HA2-treated patients were satisfied with their treatment (P = 0.9). Investigator assessment of treatment efficacy at Day 168 using the Likert scale showed similar results for the two treatments [92.9% for HA1 and 93.4% for HA2 (P = 0.8)], with no statistically significant difference between the groups.
Safety Outcomes
Adverse event (AE) reporting and collection of other safety data were performed for the ITT safety analysis population of 202 patients (96 patients in the HA1 group and 106 in the HA2 group).
Eighty-six patients reported AEs during the study, 46 AEs in the HA1 group and 40 AEs in the HA2 group. Forty-six of those were related to the treatment (HA1 n = 26/HA2 n = 20), with 37 related to underlying musculoskeletal disorders and 9 related to injection site abnormalities. There was no statistically significant difference between the groups. The frequencies of such AEs are in line with those described in other clinical studies. Eleven AEs occurred post-injection, and 75 occurred during the follow-up. The rate of study discontinuation due to adverse events was low. The AEs that led to early discontinuation for three patients (2 in the HA1 group and 1 in the HA2 group) were not treatment related: one patient in the HA1 group had a myocardial infarction, and another suffered an ocular trauma, while one patient in the HA2 group had acute chest pain due to coronary heart disease. There was no statistically significant difference between the groups. No death occurred during the trial.
Discussion
The heterogeneity of the effects of HA treatments highlighted by various meta-analyses calls into question the relevance of the classical meta-analytic approach and suggests the need for product-specific clinical evaluations. In the present trial, we showed a significant decrease in observed pain with the use of two viscosupplements: one containing a solution of HA (HA1 80 mg HA and 160 mg sorbitol) and the other containing a solution of hylan (HA2 48 mg hylan GF-20). The effect was observed quickly within days following the injection, with a maximum time of between 2 and 3 weeks. Interestingly, the effect was maintained over time up to the end of follow-up. The curves remained parallel and steady throughout the observation. The statistical test applied with an upper limit of the 95% CI of 7.227 (< 8) allowed us to conclude that HA1 is non-inferior to HA2 on this parameter in the PP population. The same test applied to the ITT population equally demonstrated non-inferiority, with an upper limit of the 95% CI of 7.8859. For the secondary end points, such as stiffness, functionality and WOMAC total score, the amplitude and duration of the improvement were very similar, and significant differences between treatments were not significant except for WOMAC stiffness at Day 28 in the PP and ITT populations. Satisfaction assessed on a Likert scale by the patients and efficacy assessed by the investigators at Day 168 were high and comparable. Taking into account the responders according to the OMERACT OARSI definition, no statistically significant difference was observed at any time between treatment groups. In terms of safety, both products were well tolerated. No case of allergy or infection was reported.
According to the ESCEO task force, there are more than 80 marketed HA preparations worldwide [11]. They have different origins (animal versus biofermentation), concentrations, dosing regimens, molecular weights (MW) and volumes of injection. Moreover, some of the preparations include different concentrations of additives, such as mannitol, sorbitol or chondroitin sulphate. While each of these parameters may theoretically have an impact on the effect of IAHA treatment, the clinical results are quite heterogeneous. The mode of action is probably not sufficient to explain the possible different clinical outcomes of different HAs [11]. In fact, it has been suggested that several mechanisms overlap and interact to relieve OA pain.
However, regarding the molecular weight (MW), there are suggestions that within the spectrum of available HA derivatives, the efficacy of HA products with a high molecular weight (HMW) is superior to that of derivatives with a low molecular weight [14]. HA preparation could also differ according to molecular structure (linear, crosslinked and a mix of both) and method of crosslinking. It has been shown that crosslinking increases the time the substance stays in the joint synovial fluid, which could, in turn, increase the efficacy and duration of the treatment effect [14]. Some studies have suggested that the increase in the MW, stability and viscosity of HA by crosslinking results in an extended duration of action with fewer intra-articular injections [15,16,17]. However, in this study, which directly compared a non-crosslinked HA (HA1) with a crosslinked HA (HA2), we observed no significant difference between the two.
In most formulations, HA is derived from rooster comb tissue, and dosing schedules are typically 3, 4 or 5 intra-articular injections. In contrast, more advanced HA preparations have evolved to provide durable activity and require fewer injections. Single HA injections have been suggested to be associated with patient convenience (e.g., patients not returning to complete the injection series), compliance and even safety. Moreover, it mechanically reduces the probability of an infectious complication (septic arthritis). The question therefore arises whether a single injection of HA has the same level of efficacy as multi-injection regimens, particularly compared to an injected placebo. In a recent meta-analysis based on 28 clinical trials, it was suggested that one injection produces results similar to those of multiple injections of HA in terms of pain relief in the treatment of knee OA [17]. For a single dose regimen, chemically crosslinked HA and avian-derived HA injections could be prepared. In a recent trial, it was shown that crosslinked HA was superior to avian-derived HA in improving physical function and pain [13]. Recently, another trial demonstrated that a single crosslinked injection was noninferior to three weekly injections of linear high molecular weight HA in terms of pain reduction [18].
For this trial, the chosen comparator was Synvisc-One, which is currently indicated for use as a single injection and is also a leader in the main European markets. Indeed, hylan GF-20, as one of the most evaluated products and available in Europe, was considered the most appropriate reference product for the present trial. In the present trial, we decided to use HA for 6 months, even though there have been some studies that have assessed the effect of HA at 3 months only [19, 20].
In their recent guidelines, ESCEO and OARSI support the use of HA as one of the treatment modalities [2, 3]. In addition, the ESCEO task force encourages the use of repeated cycles of HA in patients who respond to the first injection, starting a new treatment cycle as soon as the first symptoms appear.
We acknowledge some limitations in our trial. First, the absence of a placebo arm does not allow us to assess the real effectiveness of the two HA products. Second, the period of 6 months could have increased the drop-out rate, which might have increased the bias in obtaining optimal data in this kind of clinical trial. Our retention rate was nevertheless excellent, with 202 patients evaluated after the 6-month follow-up. Third, many independent centres participated in this trial, which could increase the representativity of the population but could also increase the risk of protocol violations. However, 24 violations were observed in this trial. In fact, 20 patients were excluded for major deviations, with 9 patients for HA1 and 11 patients for HA2, 3 for exit from the study after Day 84, with 1 patient for HA1 and 2 patients for HA2, and 1 for major deviation and exit from study after Day 84 for HA1.
For future research, the ESCEO task force calls for additional well-conducted clinical trials (randomized controlled studies, cohort studies) to define the predictive factors (i.e., patient phenotypes, treatment characteristics) associated with an optimal response to treatment [11]. Currently, limited available evidence suggests that IAHA injections tend to be more effective if the patient has moderate, radiologically advanced OA, is not too old (< 60 years of age), has a high level of symptoms and has no effusion. The same group of experts suggests focusing future research on determining the long-term effects of repeated IAHA injections on patient-reported outcomes and total knee replacement-sparing effects, on the assessment of disease-modifying effects and on the potential for combination therapy with other pharmacological and non-pharmacological therapies to optimize the management of knee OA.
Conclusions
In this randomized double-blind trial comparing Synolis VA with Synvisc-One, both products provided quick and clinically significant improvements in symptoms related to knee OA in patients who experienced failure or intolerance of previous analgesic and anti-inflammatory treatments. The observed safety profile of both products was satisfactory. The non-inferiority of Synolis VA to Synvisc-One on the single primary efficacy end point was demonstrated in the present trial, and other secondary end point analyses confirmed these results. The demonstrated efficacy of both products supports their use as a therapeutic option in knee OA, as mentioned in the recent recommendations of the scientific community.
References
Clynes MA, Jameson KA, Edwards MH, Cooper C, Dennison EM. Impact of osteoarthritis on activities of daily living: does joint site matter? Aging Clin Exp Res. 2019;31(8):1049–56. https://doi.org/10.1007/s40520-019-01163-0.
Bruyere O, et al. An updated algorithm recommendation for the management of knee osteoarthritis from the European Society for Clinical and Economic Aspects of Osteoporosis, Osteoarthritis and Musculoskeletal Diseases (ESCEO). Semin Arthritis Rheum. 2019;49(3):337–50.
Bannuru RR, et al. OARSI guidelines for the non-surgical management of knee, hip, and polyarticular osteoarthritis. Osteoarthri Cartil. 2019;27(11):1578–89.
Saltzman BM. The therapeutic effect of intra-articular normal saline injections for knee osteoarthritis: a meta-analysis of evidence level 1 studies. Am J Sports Med. 2017;45(11):2647–53.
Maheu E, et al. Why we should definitely include intra-articular hyaluronic acid as a therapeutic option in the management of knee osteoarthritis: Results of an extensive critical literature review. Semin Arthritis Rheum. 2019;48(4):563–72.
Campbell KA, et al. Is local viscosupplementation injection clinically superior to other therapies in the treatment of osteoarthritis of the knee: a systematic review of overlapping meta-analyses. Arthroscopy. 2015;31(10):2036-45.e14. https://doi.org/10.1016/j.arthro.2015.03.030.
Bowman S, et al. Recent advances in hyaluronic acid based therapy for osteoarthritis. Clin Transl Med. 2018;7:6. https://doi.org/10.1186/s40169-017-0180-3.
He WW, et al. Efficacy and safety of intraarticular hyaluronic acid and corticosteroid for knee osteoarthritis: a meta-analysis. Int J Surg. 2017;39:95–103.
Ran J, et al. Comparison of intra-articular hyaluronic acid and methylprednisolone for pain management in knee osteoarthritis: a meta-analysis of randomized controlled trials. Int J Surg. 2018;53:103–10.
Cole BJ, et al. Hyaluronic acid versus platelet-rich plasma: a prospective, double-blind randomized controlled trial comparing clinical outcomes and effects on intra-articular biology for the treatment of knee osteoarthritis. Am J Sports Med. 2017;45(2):339–46.
Altman RD, et al. Product differences in intra-articular hyaluronic acids for osteoarthritis of the knee. Am J Sports Med. 2016;44(8):2158–65.
Guo Y, Yang P, Liu L. Origin and efficacy of hyaluronan injections in knee osteoarthritis: randomized, double-blind. Trial Med Sci Monit. 2018;24:4728–37.
Cooper C, et al. Use of intraarticular hyaluronic acid in the management of knee osteoarthritis in clinical practice. Arthritis Care Res (Hoboken). 2017;69(9):1287–96.
Rutjes AW, et al. Viscosupplementation for osteoarthritis of the knee: a systematic review and meta-analysis. Ann Intern Med. 2012;157(3):180–91.
Chou CW, et al. Hylan G-F 20 has better pain relief and cost-effectiveness than sodium hyaluronate in treating early osteoarthritic knees in Taiwan. J Formos Med Assoc. 2009;108(8):663–72.
Strand V, et al. A multicenter, randomized controlled trial comparing a single intra-articular injection of Gel-200, a new cross-linked formulation of hyaluronic acid, to phosphate buffered saline for treatment of osteoarthritis of the knee. Osteoarthr Cartil. 2012;20(5):350–6.
Vincent P. Intra-articular hyaluronic acid in the symptomatic treatment of knee osteoarthritis: a meta-analysis of single-injection products. Curr Ther Res Clin Exp. 2019;90:39–51.
Ha CW, et al. Efficacy and safety of single injection of cross-linked sodium hyaluronate vs. three injections of high molecular weight sodium hyaluronate for osteoarthritis of the knee: a double-blind, randomized, multi-center, non-inferiority trial. BMC Musculoskelet Disord. 2017;18(1):223.
Petrella RJ, Petrella M. A prospective, randomized, double-blind, placebo controlled trial to evaluate the efficacy of intraarticular hyaluronic acid for osteoarthritis of the knee. J Rheumatol. 2006;33(5):951–6.
Galluccio F, Barskova T, Cerinic MM. Short-term effect of the combination of hyaluronic acid, chondroitin sulfate, and keratin matrix on early symptomatic knee osteoarthritis. Eur J Rheumatol. 2015;2(3):106–8.
Acknowledgments
We thank the physicians who performed the intraarticular injections and follow-up visits. We also thank the participants of the study.
Funding
The study costs and fees, the Rapid Service Fee and the Open Access Fee were supported by Aptissen SA.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Disclosures
This trial was supported by Aptissen. Olivier Bruyere: Grant research (in the last 5 years): Biophytis, IBSA, MEDA, Servier, SMB. Consulting or lecture fees (in the last 5 years): Amgen, Aptissen, Biophytis, IBSA, MEDA, Novartis, Sanofi, Servier, SMB, TRB Chemedica, UCB. The author declares no conflict of interest. Bernard Cortet: No financial interests. Consulting or lecture fees (in the last 5 years): Alexion, Amgen, Ferring, Kyowa-Kirin, Lilly, MSD, Medtronic, Mylan, Novartis, Roche diagnostic, Theramex, Sublimed, UCB. The author declares no conflict of interest. Eduard Vidovic: Consulting or lecture fees (in the last 5 years): Aptissen, Inflamalps, Lymphatica, Pharmotech. The author declares no conflict of interest. Sandrine Lombion: The author declares no conflict of interest. Bernina Naissant: The author declares no conflict of interest.
Compliance with Ethics Guidelines
The trial was conducted in accordance with the ethics principals of the Declaration of Helsinki. It was approved and registered under no. 2017-A00034-49 to the ANSM, and ethical approval was obtained from CPP Ile-de-France VI. Ref: CPP/6-17 -ID RCB: 2017-A00034-49.
Data Availability
The datasets generated and analyzed during the current study are stored at the Aptissen site, Plan-les-Ouates, Switzerland, and can be made available upon reasonable request to the corresponding author.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Cortet, B., Lombion, S., Naissant, B. et al. Non-Inferiority of a Single Injection of Sodium Hyaluronate Plus Sorbitol to Hylan G-F20: A 6-Month Randomized Controlled Trial. Adv Ther 38, 2271–2283 (2021). https://doi.org/10.1007/s12325-021-01648-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-021-01648-3