Abstract
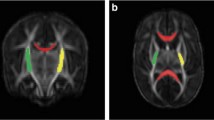
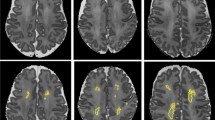
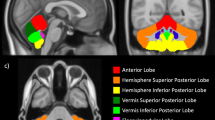
This study aims to evaluate in extremely premature infants the severity of brain structural injury causing total absence or near-total absence of cerebellar hemispheres by using MRI visual and volumetric scoring systems. It also aims to assess the role of the score systems in predicting motor outcome. We developed qualitative and quantitative MRI scoring systems to grade the overall brain damage severity in 16 infants with total absence or near-total absence of cerebellar hemispheres. The qualitative scoring system assessed the severity of macrostructural abnormalities of cerebellum, brainstem, supratentorial gray and white matters, ventricles while the quantitative scoring system weighted the loss of brain tissue volumes, and gross motor function classification system (GMFCS) was used to assess motor function at 1- and 5-year follow-ups.
Positive correlations between both MRI scores and GMFCS scales were detected at follow-ups (p > 0.05), but only the volumetric score could identify those infants developing higher levels of motor impairment.
Brain volumetric MRI offers an unbiassed assessment of prenatal brain damage. The quantitative scoring system, performed at term equivalent age, can be a helpful tool for predicting the long-term motor outcome in extremely preterm infants with a near-total absence of cerebellum.


Similar content being viewed by others
Data Availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Miall LS, Cornette LG, Tanner SF, Arthur RJ, Levene MI. Posterior fossa abnormalities seen on magnetic resonance brain imaging in a cohort of newborn infants. J Perinatol Off J Calif Perinat Assoc. 2003;23:396–403.
Poretti A, Prayer D, Boltshauser E. Morphological spectrum of prenatal cerebellar disruptions. Eur J Paediatr Neurol EJPN Off J Eur Paediatr Neurol Soc. 2009;13:397–407.
Benbir G, Kara S, Yalcinkaya BC, Karhkaya G, Tuysuz B, Kocer N, et al. Unilateral cerebellar hypoplasia with different clinical features. Cerebellum Lond Engl. 2011;10:49–60.
Grunnet ML, Shields WD. Cerebellar hemorrhage in the premature infant. J Pediatr. 1976;88:605–8.
Mercuri E, He J, Curati WL, Dubowitz LM, Cowan FM, Bydder GM. Cerebellar infarction and atrophy in infants and children with a history of premature birth. Pediatr Radiol. 1997;27:139–43.
Rollins NK, Wen TS, Dominguez R. Crossed cerebellar atrophy in children: a neurologic sequela of extreme prematurity. Pediatr Radiol. 1995;25(Suppl 1):S20–5.
Tam EWY, Ferriero DM, Xu D, Berman JI, Vigneron DB, Barkovich AJ, et al. Cerebellar development in the preterm neonate: effect of supratentorial brain injury. Pediatr Res. 2009;66:102–6.
Gano D, Barkovich AJ. Cerebellar hypoplasia of prematurity: causes and consequences. Handb Clin Neurol. 2019;162:201–16.
Lee C, Kim DW, Jeon GS, Roh EJ, Seo JH, Wang KC, et al. Cerebellar alterations induced by chronic hypoxia: an immunohistochemical study using a chick embryonic model. Brain Res. 2001;901:271–6.
Robins JB, Mason GC, Watters J, Martinez D. Case report: cerebellar hemi-hypoplasia. Prenat Diagn. 1998;18:173–7.
Hiller L, McGahan JP, Bijan B, Melendres G, Towner D. Sonographic detection of in utero isolated cerebellar hemorrhage. J Ultrasound Med Off J Am Inst Ultrasound Med. 2003;22:649–52.
Boltshauser E. Cerebellum-small brain but large confusion: a review of selected cerebellar malformations and disruptions. Am J Med Genet A. 2004;126A:376–85.
Poretti A, Limperopoulos C, Roulet-Perez E, Wolf NI, Rauscher C, Prayer D, et al. Outcome of severe unilateral cerebellar hypoplasia. Dev Med Child Neurol. 2010;52:718–24.
Poretti A, Boltshauser E, Doherty D. Cerebellar hypoplasia: differential diagnosis and diagnostic approach. Am J Med Genet C: Semin Med Genet. 2014;166C:211–26.
Poretti A, Boltshauser E. Terminology in morphological anomalies of the cerebellum does matter. Cerebellum Ataxias. 2015;2:8.
Yu F, Jiang Q, Sun X, Zhang R. A new case of complete primary cerebellar agenesis: clinical and imaging findings in a living patient. Brain J Neurol. 2015;138:e353.
Kidokoro H, Neil JJ, Inder TE. New MR imaging assessment tool to define brain abnormalities in very preterm infants at term. AJNR Am J Neuroradiol. 2013;34:2208–14.
Kidokoro H, Anderson PJ, Doyle LW, Woodward LJ, Neil JJ, Inder TE. Brain injury and altered brain growth in preterm infants: predictors and prognosis. Pediatrics. 2014;134:e444–53.
Miller SP, Ferriero DM, Leonard C, Piecuch R, Glidden DV, Partridge JC, et al. Early brain injury in premature newborns detected with magnetic resonance imaging is associated with adverse early neurodevelopmental outcome. J Pediatr. 2005;147:609–16.
Inder TE, Wells SJ, Mogridge NB, Spencer C, Volpe JJ. Defining the nature of the cerebral abnormalities in the premature infant: a qualitative magnetic resonance imaging study. J Pediatr. 2003;143:171–9.
Sie LTL, Hart AAM, van Hof J, de Groot L, Lems W, Lafeber HN, et al. Predictive value of neonatal MRI with respect to late MRI findings and clinical outcome. A study in infants with periventricular densities on neonatal ultrasound. Neuropediatrics. 2005;36:78–89.
George JM, Fiori S, Fripp J, Pannek K, Bursle J, Moldrich RX, et al. Validation of an MRI brain injury and growth scoring system in very preterm infants scanned at 29- to 35-week postmenstrual age. AJNR Am J Neuroradiol. 2017;38:1435–42.
Limperopoulos C, Chilingaryan G, Sullivan N, Guizard N, Robertson RL, du Plessis AJ. Injury to the premature cerebellum: outcome is related to remote cortical development. Cereb Cortex N Y N. 1991;2014(24):728–36.
Tam EWY, Rosenbluth G, Rogers EE, Ferriero DM, Glidden D, Goldstein RB, et al. Cerebellar hemorrhage on magnetic resonance imaging in preterm newborns associated with abnormal neurologic outcome. J Pediatr. 2011;158:245–50.
Nguyen The Tich S, Anderson PJ, Shimony JS, Hunt RW, Doyle LW, Inder TE. A novel quantitative simple brain metric using MR imaging for preterm infants. AJNR Am J Neuroradiol. 2009;30:125–31.
Sener RN, Jinkins JR. Subtotal agenesis of the cerebellum in an adult: MRI demonstration. Neuroradiology. 1993;35:286–7.
Poretti A, Boltshauser E, Huisman TAGM. Prenatal cerebellar disruptions: neuroimaging spectrum of findings in correlation with likely mechanisms and etiologies of injury. Neuroimaging Clin N Am. 2016;26:359–72.
Calandrelli R, D’Apolito G, Marco P, Zampino G, Tartaglione T, Colosimo C. Costello syndrome: analysis of the posterior cranial fossa in children with posterior fossa crowding. Neuroradiol J. 2015;28:254–8.
Yushkevich PA, Piven J, Hazlett HC, Smith RG, Ho S, Gee JC, et al. User-guided 3D active contour segmentation of anatomical structures: significantly improved efficiency and reliability. Neuroimage. 2006;31:1116–28.
Calandrelli R, Panfili M, D’Apolito G, Zampino G, Pedicelli A, Pilato F, et al. Quantitative approach to the posterior cranial fossa and craniocervical junction in asymptomatic children with achondroplasia. Neuroradiology. 2017;59:1031–41.
Calandrelli R, D’Apolito G, Panfili M, Massimi L, Caldarelli M, Colosimo C. Role of “major” and “minor” lambdoid arch sutures in posterior cranial fossa changes: mechanism of cerebellar tonsillar herniation in infants with multisutural craniosynostosis. Childs Nerv Syst ChNS Off J Int Soc Pediatr Neurosurg. 2016;32:451–9.
Calandrelli R, Pilato F, Massimi L, Panfili M, Di Rocco C, Colosimo C. Quantitative analysis of cranial-orbital changes in infants with anterior synostotic plagiocephaly. Childs Nerv Syst ChNS Off J Int Soc Pediatr Neurosurg. 2018;34:1725–33.
Calandrelli R, Pilato F, Massimi L, Panfili M, Colosimo C. A systematic quantitative morpho-volumetric analysis in infants with sagittal craniosynostosis and relationship with the severity of scaphocephalic deformity. Radiol Med (Torino). 2020;125:585–94.
Balikci A, May-Benson TA, Aracikul Balikci AF, Tarakci E, Ikbal Dogan Z, Ilbay G. Evaluation of Ayres Sensory Integration® intervention on sensory processing and motor function in a child with rubinstein-taybi syndrome: a case report. Clin Med Insights Case Rep. 2023;16:11795476221148866.
Salavati M, Krijnen WP, Rameckers EAA, Looijestijn PL, CGB M, van der Schans CP, et al. Reliability of the modified gross motor function measure-88 (GMFM-88) for children with both spastic cerebral palsy and cerebral visual impairment: a preliminary study. Res Dev Disabil. 2015;45–46:32–48.
Papile LA, Burstein J, Burstein R, Koffler H. Incidence and evolution of subependymal and intraventricular hemorrhage: a study of infants with birth weights less than 1,500 gm. J Pediatr. 1978;92:529–34.
Limperopoulos C, Robertson RL, Sullivan NR, Bassan H, du Plessis AJ. Cerebellar injury in term infants: clinical characteristics, magnetic resonance imaging findings, and outcome. Pediatr Neurol. 2009;41:1–8.
Bodensteiner JB, Johnsen SD. Cerebellar injury in the extremely premature infant: newly recognized but relatively common outcome. J Child Neurol. 2005;20:139–42.
Messerschmidt A, Brugger PC, Boltshauser E, Zoder G, Sterniste W, Birnbacher R, et al. Disruption of cerebellar development: potential complication of extreme prematurity. AJNR Am J Neuroradiol. 2005;26:1659–67.
Kline JE, Sita Priyanka Illapani V, He L, Parikh NA. Automated brain morphometric biomarkers from MRI at term predict motor development in very preterm infants. NeuroImage Clin. 2020;28:102475.
Parikh NA, Lasky RE, Kennedy KA, McDavid G, Tyson JE. Perinatal factors and regional brain volume abnormalities at term in a cohort of extremely low birth weight infants. PLoS One. 2013;8:e62804.
Woodward LJ, Anderson PJ, Austin NC, Howard K, Inder TE. Neonatal MRI to predict neurodevelopmental outcomes in preterm infants. N Engl J Med. 2006;355:685–94.
Balakrishnan U, Amboiram P, Ninan B, Chandrasekar A, Rangasami R. Correlation among magnetic resonance imaging parameters of brain in preterm neonates at term equivalent age. Indian J Pediatr. 2017;84:13–9.
Volpe JJ. Brain injury in premature infants: a complex amalgam of destructive and developmental disturbances. Lancet Neurol. 2009;8:110–24.
Calandrelli R, Marco P, Tran HE, Colosimo C, Pilato F. A novel radiological score system to assess the clinical severity in patients with acute cerebellitis. Cerebellum Lond Engl. 2023;22:173–82.
Jiang X, Faber J, Giordano I, Machts J, Kindler C, Dudesek A, et al. Characterization of cerebellar atrophy and resting state functional connectivity patterns in sporadic adult-onset ataxia of unknown etiology (SAOA). Cerebellum Lond Engl. 2019;18:873–81.
Zhang H, Ji S, Ren S, Liu M, Ran W, Zhang X, et al. Cerebellar atrophy in multiple system atrophy (cerebellar type) and its implication for network connectivity. Cerebellum Lond Engl. 2020;19:636–44.
Peterson BS, Vohr B, Staib LH, Cannistraci CJ, Dolberg A, Schneider KC, et al. Regional brain volume abnormalities and long-term cognitive outcome in preterm infants. JAMA. 2000;284:1939–47.
Narberhaus A, Segarra D, Caldú X, Giménez M, Pueyo R, Botet F, et al. Corpus callosum and prefrontal functions in adolescents with history of very preterm birth. Neuropsychologia. 2008;46:111–6.
ten Donkelaar HJ, Lammens M, Wesseling P, Thijssen HOM, Renier WO. Development and developmental disorders of the human cerebellum. J Neurol. 2003;250:1025–36.
Molinari M, Filippini V, Leggio MG. Neuronal plasticity of interrelated cerebellar and cortical networks. Neuroscience. 2002;111:863–70.
Mehrkanoon S, Boonstra TW, Breakspear M, Hinder M, Summers JJ. Upregulation of cortico-cerebellar functional connectivity after motor learning. NeuroImage. 2016;128:252–63.
Yamamoto T, Hayashi T, Murata Y, Ose T, Higo N. Premotor cortical-cerebellar reorganization in a macaque model of primary motor cortical lesion and recovery. J Neurosci. 2019;39:8484–96.
Pagnozzi AM, van Eijk L, Pannek K, Boyd RN, Saha S, George J, et al. Early brain morphometrics from neonatal MRI predict motor and cognitive outcomes at 2-years corrected age in very preterm infants. NeuroImage. 2023;267:119815.
Author information
Authors and Affiliations
Contributions
Rosalinda Calandrelli: project development, data collection, and manuscript writing; Laura Tuzza: data collection; Domenico Marco Romeo: data collection; Chiara Arpaia: data collection; Cesare Colosimo: project development; Fabio Pilato: data collection, statistical analysis, and manuscript writing. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics Approval and Consent to Participate
We declare that all procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Competing Interests
Rosalinda Calandrelli, Laura Tuzza, Domenico Marco Romeo, Chiara Arpaia, and Fabio Pilato declare that they have no conflict of interests. Cesare Colosimo declares that he is a scientific consultant for Bracco Diagnostics Inc. and Bayer HealthCare.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Calandrelli, R., Tuzza, L., Romeo, D.M. et al. Extremely Preterm Infants with a Near-total Absence of Cerebellum: Usefulness of Quantitative Magnetic Resonance in Predicting the Motor Outcome. Cerebellum (2023). https://doi.org/10.1007/s12311-023-01593-7
Accepted:
Published:
DOI: https://doi.org/10.1007/s12311-023-01593-7