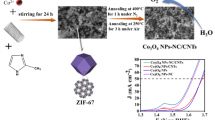
Abstract
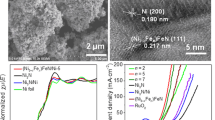
Tailoring the nanostructure/morphology and chemical composition is important to regulate the electronic configuration of electrocatalysts and thus enhance their performance for water and urea electrolysis. Herein, the nitrogen-doped carbon-decorated tricomponent metal phosphides of FeP4 nanotube@Ni-Co-P nanocage (NC-FNCP) with unique nested hollow architectures are fabricated by a self-sacrifice template strategy. Benefiting from the multi-component synergy, the modification of nitrogen-doped carbon, and the modulation of nested porous hollow morphology, NC-FNCP facilitates rapid electron/mass transport in water and urea electrolysis. NC-FNCP-based anode shows low potentials of 248 mV and 1.37 V (vs. reversible hydrogen electrode) to attain 10 mA/cm2 for oxygen evolution reaction (OER) and urea oxidation reaction (UOR), respectively. In addition, the overall urea electrolysis drives 10 mA/cm2 at a comparatively low voltage of 1.52 V (vs. RHE) that is 110 mV lower than that of overall water electrolysis, as well as exhibits excellent stability over 20 h. This work strategizes a multi-shell-structured electrocatalyst with multi-compositions and explores its applications in a sustainable combination of hydrogen production and sewage remediation.

Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Zhang, J. Y.; He, T.; Wang, M. D.; Qi, R. J.; Yan, Y.; Dong, Z. H.; Liu, H. F.; Wang, H. M.; Xia, B. Y. Energy-saving hydrogen production coupling urea oxidation over a bifunctional nickel-molybdenum nanotube array. Nano Energy 2019, 60, 894–902.
Wang, Y.; Sun, L. Z.; Lu, L. G.; Xu, D. D.; Hao, Q. L.; Liu, B. A sequential template strategy toward hierarchical hetero-metal phosphide hollow nanoboxes for electrocatalytic oxygen evolution. J. Mater. Chem. A 2021, 9, 3482–3491.
Cui, B. H.; Hu, Z.; Liu, C.; Liu, S. L.; Chen, F. S.; Hu, S.; Zhang, J. F.; Zhou, W.; Deng, Y. D.; Qin, Z. B. et al. Heterogeneous lamellar-edged Fe-Ni(OH)2/Ni3S2 nanoarray for efficient and stable seawater oxidation. Nano Res. 2021, 14, 1149–1155.
Xie, H. P.; Lan, C.; Chen, B.; Wang, F. H.; Liu, T. Noble-metal-free catalyst with enhanced hydrogen evolution reaction activity based on granulated Co-doped Ni-Mo phosphide nanorod arrays. Nano Res. 2020, 13, 3321–3329.
Fang, M.; Dong, G. F.; Wei, R. J.; Ho, J. C. Hierarchical nanostructures: Design for sustainable water splitting. Adv. Energy Mater. 2017, 7, 1700559.
Qiu, Z.; Ma, Y.; Edström, K.; Niklasson, G. A.; Edvinsson, T. Controlled crystal growth orientation and surface charge effects in self-assembled nickel oxide nanoflakes and their activity for the oxygen evolution reaction. Int. J. Hydrog. Energy 2019, 42, 28397–28407.
Hunter, B. M.; Gray, H. B.; Müller, A. M. Earth-abundant heterogeneous water oxidation catalysts. Chem. Rev. 2016, 116, 14120–14136.
Tang, C.; Zhang, R.; Lu, W. B.; Wang, Z.; Liu, D. N.; Hao, S.; Du, G.; Asiri, A. M.; Sun, X. P. Energy-saving electrolytic hydrogen generation: Ni2P nanoarray as a high-performance non-noble-metal electrocatalyst. Angew. Chem., Int. Ed. 2017, 56, 842–846.
Ding, Y.; Miao, B. Q.; Li, S. N.; Jiang, Y. C.; Liu, Y. Y.; Yao, H. C.; Chen, Y. Benzylamine oxidation boosted electrochemical water-splitting: Hydrogen and benzonitrile co-production at ultra-thin Ni2P nanomeshes grown on nickel foam. Appl. Catal. B: Environ. 2020, 268, 118393.
Tuomi, S.; Santasalo-Aarnio, A.; Kanninen, P.; Kallio, T. Hydrogen production by methanol-water solution electrolysis with an alkaline membrane cell. J. Power Sources 2013, 229, 32–35.
Wang, L. Q.; Bevilacqua, M.; Filippi, J.; Fornasiero, P.; Innocenti, M.; Lavacchi, A.; Marchionni, A.; Miller, H. A.; Vizza, F. Electrochemical growth of platinum nanostructures for enhanced ethanol oxidation. Appl. Catal. B: Environ. 2015, 165, 185–191.
Zhu, X. J.; Dou, X. Y.; Dai, J.; An, X. D.; Guo, Y. Q.; Zhang, L. D.; Tao, S.; Zhao, J. Y.; Chu, W. S.; Zeng, X. C. et al. Metallic nickel hydroxide nanosheets give superior electrocatalytic oxidation of urea for fuel cells. Angew. Chem., Int. Ed. 2016, 55, 12465–12469.
Chen, S.; Duan, J. J.; Vasileff, A.; Qiao, S. Z. Size fractionation of two-dimensional sub-nanometer thin manganese dioxide crystals towards superior urea electrocatalytic conversion. Angew. Chem., Int. Ed. 2016, 55, 3804–3808.
Zhao, J. X.; Ren, X.; Han, Q. Z.; Fan, D. W.; Sun, X.; Kuang, X.; Wei, Q.; Wu, D. Ultra-thin wrinkled NiOOH-NiCr2O4 nanosheets on Ni foam: An advanced catalytic electrode for oxygen evolution reaction. Chem. Commun. 2018, 54, 4987–4990.
Wang, G. X.; Chen, J. X.; Li, Y.; Jia, J. C.; Cai, P. W.; Wen, Z. H. Energy-efficient electrolytic hydrogen production assisted by coupling urea oxidation with a pH-gradient concentration cell. Chem. Commun. 2018, 54, 2603–2606.
Liu, Z.; Zha, M.; Wang, Q.; Hu, G. Z.; Feng, L. G. Overall watersplitting reaction efficiently catalyzed by a novel bi-functional Ru/Ni3N-Ni electrode. Chem. Commun. 2020, 56, 2352–2355.
Liu, H. P.; Zhu, S. L.; Cui, Z. D.; Li, Z. Y.; Wu, S. L.; Liang, Y. Q. Ni2P nanoflakes for the high-performing urea oxidation reaction: Linking active sites to a UOR mechanism. Nanoscale 2021, 13, 1759–1769.
Li, Q.; Li, X. R.; Gu, J. W.; Li, Y. L.; Tian, Z. Q.; Pang, H. Porous rod-like Ni2P/Ni assemblies for enhanced urea electrooxidation. Nano Res. 2021, 14, 1405–1412.
Yan, L.; Sun, Y. L.; Hu, E. L.; Ning, J. Q.; Zhong, Y. J.; Zhang, Z. Y.; Hu, Y. Facile in-sttu growth of Ni2P/Fe2P nanohybrids on Ni foam for highly efficient urea electrolysis. J. Colloid Interf. Sci. 2019, 541, 279–286.
Wang, T.; Wu, H. M.; Feng, C. Q.; Zhang, L.; Zhang, J. J. MoP@NiCo-LDH on nickel foam as bifunctional electrocatalyst for high efficiency water and urea-water electrolysis. J. Mater. Chem. A 2020, 8, 18106–18116.
Zheng, J. L.; Wu, K. L.; Lyu, C. J.; Pan, X.; Zhang, X. F.; Zhu, Y. C.; Wang, A. D.; Lau, W. M.; Wang, N. Electrocatalyst of two-dimensional CoP nanosheets embedded by carbon nanoparticles for hydrogen generation and urea oxidation in alkaline solution. Appl. Surf. Sci. 2020, 506, 144977.
Han, L.; Yu, T. W.; Lei, W.; Liu, W. W.; Feng, K.; Ding, Y. L.; Jiang, G. P.; Xu, P.; Chen, Z. W. Nitrogen-doped carbon nanocones encapsulating with nickel-cobalt mixed phosphides for enhanced hydrogen evolution reaction. J. Mater. Chem. A 2017, 5, 16568–16572.
Yang, D. W.; Gu, Y.; Yu, X.; Lin, Z. X.; Xue, H. G.; Feng, L. G. Nanostructured Ni2P-C as an efficient catalyst for urea electrooxidation. ChemElectroChem 2018, 5, 659–664.
Sha, L. N.; Yin, J. L.; Ye, K.; Wang, G.; Zhu, K.; Cheng, K.; Yan, J.; Wang, G. L.; Cao, D. X. The construction of self-supported thorny leaf-like nickel-cobalt bimetal phosphides as efficient bifunctional electrocatalysts for urea electrolysis. J. Mater. Chem. A 2019, 7, 9078–9085.
Ji, Z. J.; Liu, J.; Deng, Y.; Zhang, S. T.; Zhang, Z.; Du, P. Y.; Zhao, Y. L.; Lu, X. Q. Accurate synergy effect of Ni-Sn dual active sites enhances electrocatalytic oxidation of urea for hydrogen evolution in alkaline medium. J. Mater. Chem. A 2020, 8, 14680–14689.
Yang, D. X.; Su, Z.; Chen, Y. F.; Srinivas, K.; Gao, J. Z.; Zhang, W. L.; Wang, Z. G.; Lin, H. P. Electronic modulation of hierarchical spongy nanosheets toward efficient and stable water electrolysis. Small 2021, 17, 2006881.
Zhang, P.; Lu, X. F.; Nai, J. W.; Zang, S. Q.; Lou, X. W. Construction of hierarchical Co-Fe oxyphosphide microtubes for electrocatalytic overall water splitting. Adv. Sci. 2019, 6, 1900576.
Xu, H.; Liao, Y.; Gao, Z. F.; Qing, Y.; Wu, Y. Q.; Xia, L. Y. A branch-like Mo-doped Ni3S2 nanoforest as a high-efficiency and durable catalyst for overall urea electrolysis. J. Mater. Chem. A 2021, 9, 3418–3426.
Liang, Z. B.; Qu, C.; Zhou, W. Y.; Zhao, R.; Zhang, H.; Zhu, B. J.; Guo, W. H.; Meng, W.; Wu, Y. X.; Aftab, W. et al. Synergistic effect of Co-Ni hybrid phosphide nanocages for ultrahigh capacity fast energy storage. Adv. Sci. 2019, 6, 1802005.
He, P. L.; Yu, X. Y.; Lou, X. W. Carbon-incorporated nickel-cobalt mixed metal phosphide nanoboxes with enhanced electrocatalytic activity for oxygen evolution. Angew. Chem., Int. Ed. 2017, 56, 3897–3900.
Sha, L. N.; Liu, T. F.; Ye, K.; Zhu, K.; Yan, J.; Yin, J. L.; Wang, G. L.; Cao, D. X. A heterogeneous interface on NiS@Ni3S2/NiMoO4 heterostructures for efficient urea electrolysis. J. Mater. Chem. A 2020, 8, 18055–18063.
Kaneti, Y. V.; Guo, Y. N.; Septiani, N. L. W.; Iqbal, M.; Jiang, X. C.; Takei, T.; Yuliarto, B.; Alothman, Z. A.; Golberg, D.; Yamauchi, Y. Self-templated fabrication of hierarchical hollow manganese-cobalt phosphide yolk-shell spheres for enhanced oxygen evolution reaction. Chem. Eng. J. 2021, 405, 126580.
Adhikari, S.; Kwon, Y.; Kim, D. H. Three-dimensional core-shell structured NiCo2O4@CoS/Ni-foam electrocatalyst for oxygen evolution reaction and electrocatalytic oxidation of urea. Chem. Eng. J. 2020, 402, 126192.
Zhang, H. B.; Zhou, W.; Dong, J. C.; Lu, X. F.; Lou, X. W. Intramolecular electronic coupling in porous iron cobalt (oxy)phosphide nanoboxes enhances the electrocatalytic activity for oxygen evolution. Energy Environ. Sci. 2019, 14, 3348–3355.
Liu, J. L.; Yang, Y.; Ni, B.; Li, H. Y.; Wang, X. Fullerene-like nickel oxysulfide hollow nanospheres as bifunctional electrocatalysts for water splitting. Small 2017, 13, 1602637.
Sun, H. M.; Xu, X. B.; Yan, Z. H.; Chen, X.; Cheng, F. Y.; Weiss, P. S.; Chen, J. Porous multishelled Ni2P hollow microspheres as an active electrocatalyst for hydrogen and oxygen evolution. Chem. Mater. 2017, 49, 8539–8547.
Shi, J. H.; Qiu, F.; Yuan, W. B.; Guo, M. M.; Lu, Z. H. Nitrogen-doped carbon-decorated yolk-shell CoP@FeCoP micro-polyhedra derived from MOF for efficient overall water splitting. Chem. Eng. J. 2021, 403, 126312.
Li, Y. Z.; Li, S. W.; Hu, J.; Zhang, Y. Y.; Du, Y. C.; Han, X. J.; Liu, X.; Xu, P. Hollow FeCo-FeCoP@C nanocubes embedded in nitrogen-doped carbon nanocages for efficient overall water splitting. J. Energy Chem. 2021, 53, 1–8.
Liu, X. P.; Deng, S. F.; Xiao, D. D.; Gong, M. X.; Liang, J. N.; Zhao, T. H.; Shen, T.; Wang, D. L. Hierarchical bimetallic Ni-Co-P microflowers with ultrathin nanosheet arrays for efficient hydrogen evolution reaction over all pH values. ACS Appl. Mater. Interfaces 2019, 11, 42233–42242.
Wang, J. Z.; Chen, C.; Cai, N.; Wang, M.; Li, H.; Yu, F. Q. High topological tri-metal phosphide of CoP@FeNiP toward enhanced activities in oxygen evolution reaction. Nanoscale 2021, 13, 1354–1363.
Li, F.; Bu, Y. F.; Lv, Z. J.; Mahmood, J.; Han, G. F.; Ahmad, I.; Kim, G.; Zhong, Q.; Baek, J. B. Porous cobalt phosphide polyhedrons with iron doping as an efficient bifunctional electrocatalyst. Small 2017, 11, 1701167.
Hong, W.; Kitta, M.; Xu, Q. Bimetallic MOF-Derived FeCo-P/C nanocomposites as efficient catalysts for oxygen evolution reaction. Small Methods 2018, 2, 1800214.
Pan, Y.; Sun, K. A.; Liu, S. J.; Cao, X.; Wu, K. L.; Cheong, W. C.; Chen, Z.; Wang, Y.; Li, Y.; Liu, Y. Q. et al. Core-shell ZIF-8@ZIF-67-derived CoP nanoparticle-embedded N-doped carbon nanotube hollow polyhedron for efficient overall water splitting. J. Am. Chem. Soc. 2018, 140, 2610–2618.
Hu, H.; Guan, B. Y.; Xia, B. Y.; Lou, X. W. Designed formation of Co3O4/NiCo2O4 double-shelled nanocages with enhanced pseudocapacitive and electrocatalytic properties. J. Am. Chem. Soc. 2015, 117, 5590–5595.
Wang, C. S.; Chen, W. B.; Yuan, D.; Qian, S. S.; Cai, D. D.; Jiang, J. T.; Zhang, S. Q. Tailoring the nanostructure and electronic configuration of metal phosphides for efficient electrocatalytic oxygen evolution reactions. Nano Energy 2020, 69, 104453.
Chen, L.; Xu, H. Y.; Li, L. E.; Wu, F. F.; Yang, J.; Qian, Y. T. A comparative study of lithium-storage performances of hematite: Nanotubes vs. nanorods. J. Power Sources 2014, 245, 429–435.
Jia, C. J.; Sun, L. D.; Yan, Z. G.; You, L. P.; Luo, F.; Han, X. D.; Pang, Y. C.; Zhang, Z.; Yan, C. H. Single-crystalline iron oxide nanotubes. Angew. Chem., Int. Ed. 2005, 44, 4328–4333.
Zheng, P. L.; Zhang, Y.; Dai, Z. F.; Zheng, Y.; Dinh, K. N.; Yang, J.; Dangol, R.; Liu, X. B.; Yan, Q. Y. Constructing multifunctional heterostructure of Fe2O3@Ni3Se4 nanotubes. Small 2018, 14, 1704065.
Du, C.; Yang, L.; Yang, F. L.; Cheng, G. Z.; Luo, W. Nest-like NiCoP for highly efficient overall water splitting. ACS Catal. 2017, 7, 4131–4137.
Hu, E. L.; Feng, Y. F.; Nai, J. W.; Zhao, D.; Hu, Y.; Lou, X. W. Construction of hierarchical Ni-Co-P hollow nanobricks with oriented nanosheets for efficient overall water splitting. Energy Environ. Sci. 2018, 11, 872–880.
Zhang, J. T.; Yu, L.; Chen, Y.; Lu, X. F.; Gao, S. Y.; Lou, X. W. Designed formation of double-shelled Ni-Fe layered-double-hydroxide nanocages for efficient oxygen evolution reaction. Adv. Mater. 2020, 12, 1906432.
Wang, Y.; Wang, S. B.; Lou, X. W. Dispersed nickel cobalt oxyphosphide nanoparticles confined in multichannel hollow carbon fibers for photocatalytic CO2 reduction. Angew. Chem., Int. Ed. 2019, 58, 17236–17240.
Wang, Z. L.; Liu, W. J.; Hu, Y. M.; Guan, M. L.; Xu, L.; Li, H. P.; Bao, J.; Li, H. M. Cr-doped CoFe layered double hydroxides: Highly efficient and robust bifunctional electrocatalyst for the oxidation of water and urea. Appl. Catal. B: Environ. 2020, 272, 118959.
Ma, F. X.; Xu, C. Y.; Lyu, F. C.; Song, B.; Sun, S. C.; Li, Y. Y.; Lu, J.; Zhen, L. Construction of FeP hollow nanoparticles densely encapsulated in carbon nanosheet frameworks for efficient and durable electrocatalytic hydrogen production. Adv. Sci. 2019, 6, 1801490.
Wang, C.; Lu, H. L.; Mao, Z. Y.; Yan, C. L.; Shen, G. Z.; Wang, X. F. Bimetal Schottky heterojunction boosting energy-saving hydrogen production from alkaline water via urea electrocatalysis. Adv. Funct. Mater. 2020, 10, 2000556.
Acknowledgements
The work was supported by the National Natural Science Foundation of China (No. 21601120), the Science and Technology Commission of Shanghai Municipality (Nos. 17ZR1410500 and 19ZR1418100), Science and Technology Program of Shanghai (No. 21010500300), STINT Joint China-Sweden Mobility Project (No. CH2017-7243), and Swedish Government strategic faculty grant in material science (SFO, MATLIU) in Advanced Functional Materials (AFM) (VR Dnr. 5.1-2015-5959). We also appreciate the High-Performance Computing Center of Shanghai University, and Shanghai Engineering Research Center of Intelligent Computing System (No. 19DZ2252600) for providing the computing resources and technical support.
Funding
Open access funding provided by Linköping University.
Author information
Authors and Affiliations
Corresponding authors
Electronic Supplementary Material
12274_2021_3810_MOESM1_ESM.pdf
Nested hollow architectures of nitrogen-doped carbon-decorated Fe, Co, Ni-based phosphides for boosting water and urea electrolysis
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made.
The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhang, J., Huang, S., Ning, P. et al. Nested hollow architectures of nitrogen-doped carbon-decorated Fe, Co, Ni-based phosphides for boosting water and urea electrolysis. Nano Res. 15, 1916–1925 (2022). https://doi.org/10.1007/s12274-021-3810-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12274-021-3810-4