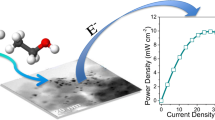
Abstract
Carbon-supported Pt/C, Pt/Re/C, Pt/SnO2/C and Pt/Re/SnO2/C, with 20 wt.% overall metal loading were prepared and their electrochemical activity towards ethanol oxidation reaction (EOR) was investigated. Transmission electron microscopy (TEM) combined with energy dispersive X-ray spectroscopy (EDS) revealed, that indeed binary and ternary combinations of the designed nanoparticles (NPs) were formed and successfully uniformly deposited on a carbon support. Fourier transform infrared spectroscopy (FTIR) allowed to assess the chemical composition of the nanocatalysts and X-ray diffraction (XRD) allowed to determine the catalyst structure. Potentiodynamic and chronoamperometric measurements were used to establish its catalytic activity and stability. The influence of Re addition on the electrochemical activity towards ethanol oxidation reaction (EOR) was verified. Indeed, the addition of Re to the binary Pt/SnO2/C catalyst leads to the formation of ternary Pt/Re/SnO2/C with physical contact between the individual NPs, enhancing the EOR. Furthermore, the onset potential of the synthesized ternary catalyst is shifted to more negative potentials and the current densities and specific activity are nearly 11 and 5 times higher, respectively, than for commercial Pt catalyst. Additionally ternary Pt/Re/SnO2/C catalyst retained 96% of its electrochemical surface area.

Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Jiang, K. Z.; Bu, L. Z.; Wang, P. T.; Guo, S. J.; Huang, X. Q. Trimetallic PtSnRh wavy nanowires as efficient nanoelectrocatalysts for alcohol electrooxidation. ACS Appl. Mater. Interfaces2015, 7, 15061–15067.
Erini, N.; Rudi, S.; Beermann, V.; Krause, P.; Yang, R. Z.; Huang, Y. H.; Strasser, P. Exceptional activity of a Pt-Rh-Ni ternary nano-structured catalyst for the electrochemical oxidation of ethanol. ChemElectroChem2015, 2, 903–908.
Yang, G. X.; Frenkel, A. I.; Su, D.; Teng, X. W. Enhanced electrokinetics of C-C bond splitting during ethanol oxidation by using a Pt/Rh/Sn catalyst with a partially oxidized Pt and Rh core and a SnO2 shell. ChemCatChem2016, 8, 2876–2880.
Comignani, V.; Sieben, J. M.; Sanchez, M. D.; Duarte, M. M. E. Influence of carbon support properties on the electrocatalytic activity of PtRuCu nanoparticles for methanol and ethanol oxidation. Int. J. Hydrogen Energy2017, 42, 24785–24796.
Spinacé, E. V.; Linardi, M.; Neto, A. O. Co-catalytic effect of nickel in the electro-oxidation of ethanol on binary Pt-Sn electrocatalysts. Electrochem. Commun.2005, 7, 365–369.
Spinacé, E. V.; Dias, R. R.; Brandalise, M.; Linardi, M.; Neto, A. O. Electro-oxidation of ethanol using PtSnRh/C electrocatalysts prepared by an alcohol-reduction process. Ionics2010, 16, 91–95.
Yang, G. X.; Namin, L. M.; Aaron Deskins, N.; Teng, X. W. Influence of *OH adsorbates on the potentiodynamics of the CO2 generation during the electro-oxidation of ethanol. J. Catal.2017, 353, 335–348.
Silva, J. C. M.; Parreira, L. S.; De Souza, R. F. B.; Calegaro, M. L.; Spinacé, E. V.; Neto, A. O.; Santos, M. C. PtSn/C alloyed and non-alloyed materials: Differences in the ethanol electro-oxidation reaction pathways. Appl. Catal. B: Environ.2011, 110, 141–147.
Antolini, E.; Colmati, F.; Gonzalez, E. R. Ethanol oxidation on carbon supported (PtSn)alloy/SnO2 and (PtSnPd)alloy/SnO2 catalysts with a fixed Pt/SnO2 atomic ratio: Effect of the alloy phase characteristics. J. Power Sources2009, 193, 555–561.
Perez, J.; Paganin, V. A.; Antolini, E. Particle size effect for ethanol electro-oxidation on Pt/C catalysts in half-cell and in a single direct ethanol fuel cell. J. Electroanal. Chem.2011, 654, 108–115.
Zhou, W. P.; An, W.; Su, D.; Palomino, R.; Liu, P.; White, M. G.; Adzic, R. R. Electrooxidation of methanol at SnOx-Pt interface: A tunable activity of tin oxide nanoparticles. J. Phys. Chem. Lett.2012, 3, 3286–3290.
Colmati, F.; Antolini, E.; Gonzalez, E. R. Preparation, structural characterization and activity for ethanol oxidation of carbon supported ternary Pt-Sn-Rh catalysts. J. Alloys Compd.2008, 456, 264–270.
Antolini, E.; Colmati, F.; Gonzalez, E. R. Effect of Ru addition on the structural characteristics and the electrochemical activity for ethanol oxidation of carbon supported Pt-Sn alloy catalysts. Electrochem. Commun.2007, 9, 398–404.
Jiang, L. H.; Sun, G. Q.; Sun, S. G.; Liu, J. G.; Tang, S. H.; Li, H. Q.; Zhou, B.; Xin, Q. Structure and chemical composition of supported Pt-Sn electrocatalysts for ethanol oxidation. Electrochim. Acta2005, 50, 5384–5389.
Antolini, E. Catalysts for direct ethanol fuel cells. J. Power Sources2007, 170, 1–12.
Antolini, E.; Gonzalez, E. R. Effect of synthesis method and structural characteristics of Pt-Sn fuel cell catalysts on the electro-oxidation of CH3OH and CH3CH2OH in acid medium. Catal. Today2011, 160, 28–38.
Li, M.; Kowal, A.; Sasaki, K.; Marinkovic, N.; Su, D.; Korach, E.; Liu, P.; Adzic, R. R. Ethanol oxidation on the ternary Pt-Rh-SnO2/C electrocatalysts with varied Pt:Rh:Sn ratios. Electrochim. Acta2010, 55, 4331–4338.
Du, W. X.; Yang, G. X.; Wong, E.; Deskins, N. A.; Frenkel, A. I.; Su, D.; Teng, X. W. Platinum-tin oxide core-shell catalysts for efficient electro-oxidation of ethanol. J. Am. Chem. Soc.2014, 136, 10862–10865.
Wang, C.; Jusys, Z.; Behm, R. J. Ethanol electro-oxidation on carbon-supported Pt, PtRu and Pt3Sn catalysts: A quantitative DEMS study. J. Power Sources2006, 154, 351–359.
Li, M.; Cullen, D. A.; Sasaki, K.; Marinkovic, N. S.; More, K.; Adzic, R. R. Ternary electrocatalysts for oxidizing ethanol to carbon dioxide: Making Ir capable of splitting C-C bond. J. Am. Chem. Soc.2013, 135, 132–141.
Li, M.; Zhou, W. P.; Marinkovic, N. S.; Sasaki, K.; Adzic, R. R. The role of rhodium and tin oxide in the platinum-based electrocatalysts for ethanol oxidation to CO2. Electrochim. Acta2013, 104, 454–461.
Kowal, A.; Li, M.; Shao, M.; Sasaki, K.; Vukmirovic, M. B.; Zhang, J.; Marinkovic, N. S.; Liu, P.; Frenkel, A. I.; Adzic, R. R. Ternary Pt/Rh/SnO2 electrocatalysts for oxidizing ethanol to CO2. Nat. Mater.2009, 8, 325–330.
Higuchi, E.; Takase, T.; Chiku, M.; Inoue, H. Preparation of ternary Pt/Rh/SnO2 anode catalysts for use in direct ethanol fuel cells and their electrocatalytic activity for ethanol oxidation reaction. J. Power Sources2014, 263, 280–287.
Tayal, J.; Rawat, B.; Basu, S. Effect of addition of rhenium to Pt-based anode catalysts in electro-oxidation of ethanol in direct ethanol PEM fuel cell. Int. J. Hydrogen Energy2012, 37, 4597–4605.
Simonetti, D. A.; Kunkes, E. L.; Dumesic, J. A. Gas-phase conversion of glycerol to synthesis gas over carbon-supported platinum and platinum-rhenium catalysts. J. Catal.2007, 247, 298–306.
Ciftci, A.; Ligthart, D. A. J. M.; Sen, A. O.; Van Hoof, A. J. F.; Friedrich, H.; Hensen, E. J. M. Pt-Re synergy in aqueous-phase reforming of glycerol and the water-gas shift reaction. J. Catal.2014, 311, 88–101.
Ramstad, A.; Strisland, F.; Raaen, S.; Borg, A.; Berg, C. CO and O2 adsorption on the Re/Pt(111) surface studied by photoemission and thermal desorption. Surf. Sci.1999, 440, 290–300.
Duke, A. S.; Xie, K. M.; Monnier, J. R.; Chen, D. A. Superior long-term activity for a Pt-Re alloy compared to Pt in methanol oxidation reactions. Surf. Sci.2017, 657, 35–43.
Infoplease: Math & Science, Chemistry, Interactive Periodic Table [Online]. https://www.infoplease.com/transitionmetals/rhenium&https://www.infoplease.com/transition-metals/platinum (accessed Feb 20, 2020).
Erini, N; Loukrakpam, R.; Petkov, V.; Baranova, E. A.; Yang, R. Z.; Teschner, D.; Huang, Y. H; Brankovic, S. R.; Strasser, P. Ethanol electro-oxidation on ternary platinum-rhodium-tin nanocatalysts: Insights in the atomic 3D structure of the active catalytic phase. ACS Catal.2014, 4, 1859–1867.
Alnot, M.; Gorodetskii, V.; Cassuto, A.; Ehrhardt, J. J. Auger electron spectroscopy, X-ray photoelectron spectroscopy, work function measurements and photoemission of adsorbed xenon on thin films of Pt-Re(111) alloys. Thin Solid Films1987, 151, 251–262.
Duke, A. S.; Galhenage, R. P.; Tenney, S. A.; Sutter, P.; Chen, D. A. In situ studies of carbon monoxide oxidation on platinum and platinum-rhenium alloy surfaces. J. Phys. Chem. C2015, 119, 381–391.
Drzymala, E.; Gruzel, G; Pajor-Swierzy, A; Depciuch, J.; Socha, R; Kowal, A.; Warszyński, P.; Parlinska-Wojtan, M. Design and assembly of ternary Pt/Re/SnO2 NPs by controlling the zeta potential of individual Pt, Re, and SnO2 NPs. J. Nanopart. Res.2018, 20, 144.
Roth, C; Papworth, A. J.; Hussain, I.; Nichols, R. J.; Schiffrin, D. J. A Pt/Ru nanoparticulate system to study the bifunctional mechanism of electrocatalysis. J. Electroanal. Chem.2005, 581, 79–85.
Parlinska-Wojtan, M.; Drzymala, E.; Gruzel, G; Depciuch, J.; Donten, M.; Kowal, A. Ternary Pt/Re/SnO2 nanoparticles for ethanol oxidation reaction: Understanding the correlation between the synthesis route and the obtained material. Appl. Catal. A: Gen.2019, 570, 319–328.
De Souza, E. A; Giz, M. J.; Camara, G A; Antolini, E.; Passos, R. R. Ethanol electro-oxidation on partially alloyed Pt-Sn-Rh/C catalysts. Electrochim. Acta2014, 147, 483–489.
Sivaiah, K.; B. Hemalatha Rudramadevi, B.; Buddhudu, S.; Bhaskar Kumar, G; Varadarajulu, A Structural, thermal and optical properties of Cu2+ and Co2+: PVP polymer films. Indian J. Pure Appl. Phys.2010, 48, 658–662.
Krishnan, K; Krishnan, R. S. Raman and infrared spectra of ethylene glycol. Proc. Indian Acad. Sci. - Sect. A1966, 64, 111.
Plyler, E. K. Infrared spectra of methanol, ethanol, and n-propanol. J. Res. Natl. Bur. Stand.1952, 48, 281–286.
Mariammal, R. N; Rajamanickam, N; Ramachandran, K. Synthesis and characterization of undoped and Co-Doped SnO2 nanoparticles. J. Nano- Electron. Phys.2011, 3, 92–100.
Chen, D. L.; Gao, L. A. Novel synthesis of well-dispersed crystalline SnO2 nanoparticles by water-in-oil microemulsion-assisted hydrothermal process. J. Colloid Interface Sci.2004, 279, 137–142.
Krishnakumar, T.; Pinna, N; Kumari, K. P.; Perumal, K; Jayaprakash, R. Microwave-assisted synthesis and characterization of tin oxide nanoparticles. Mater. Lett.2008, 62, 3437–3440.
Solymosi, F.; Block, J. H. Catalytic decomposition of HClO4 vapor over CuO by field ion mass spectrometry. J. Catal.1976, 42, 173–176.
Karelin, A. I.; Grigorovich, Z. I. Vibrational spectra of perchloric acid—II. Modifications and phase transitions of solid HClO4 and DClO4. Spectrochim. Acta Part A: Mol. Spectrosc.1976, 32, 851–857.
Dou, M. L.; Hou, M.; Liang, D.; Lu, W. T.; Shao, Z. G; Yi, B. L. SnO2 nanocluster supported Pt catalyst with high stability for proton exchange membrane fuel cells. Electrochim. Acta2013, 92, 468–473.
Lim, D. H.; Choi, D. H.; Lee, W. D.; Lee, H. I. A new synthesis of a highly dispersed and CO tolerant PtSn/C electrocatalyst for low-temperature fuel cell; its electrocatalytic activity and long-term durability. Appl. Catal. B: Environ.2009, 89, 484–493.
Binninger, T.; Fabbri, E.; Kötz, R.; Schmidt, T. J. Determination of the electrochemically active surface area of metal-oxide supported platinum catalyst. J. Electrochem. Soc.2014, 161, H121–H128.
Li, H. Q.; Sun, G. Q.; Cao, L.; Jiang, L. H.; Xin, Q. Comparison of different promotion effect of PtRu/C and PtSn/C electrocatalysts for ethanol electro-oxidation. Electrochim. Acta2007, 52, 6622–6629.
De Lima, R. B.; Paganin, V.; Iwasita, T.; Vielstich, W. On the electrocatalysis of ethylene glycol oxidation. Electrochim. Acta2003, 49, 85–91.
Kowal, A.; Gojkovic, S. L.; Lee, K. S.; Olszewski, P.; Sung, Y. E. Synthesis, characterization and electrocatalytic activity for ethanol oxidation of carbon supported Pt, Pt-Rh, Pt-SnO2 and Pt-Rh-SnO2 nanoclusters. Electrochem. Commun.2009, 11, 724–727.
Luo, J. H.; Alexander, B.; Wagner, T. R.; Maggard, P. A. Synthesis and characterization of ReO4-containing microporous and open framework structures. Inorg. Chem.2004, 43, 5537–5542.
Gong, Y.; Zhou, M. F. Infrared spectra of transition-metal dioxide anions: MO2- (M = Rh, Ir, Pt, Au) in solid argon. J. Phys. Chem. A2009, 113, 4990–4995.
Lima, F. H. B.; Gonzalez, E. R. Ethanol electro-oxidation on carbon-supported Pt-Ru, Pt-Rh and Pt-Ru-Rh nanoparticles. Electrochim. Acta2008, 53, 2963–2971.
Jiang, L.; Colmenares, L.; Jusys, Z.; Sun, G. Q.; Behm, R. J. Ethanol electrooxidation on novel carbon supported Pt/SnOx/C catalysts with varied Pt:Sn ratio. Electrochim. Acta2007, 53, 377–389.
St. John, S.; Boolchand, P.; Angelopoulos, A. P. Improved electrocatalytic ethanol oxidation activity in acidic and alkaline electrolytes using size-controlled Pt-Sn nanoparticles. Langmuir2013, 29, 16150–16159.
Vigier, F.; Coutanceau, C.; Hahn, F.; Belgsir, E. M.; Lamy, C. On the mechanism of ethanol electro-oxidation on Pt and PtSn catalysts: Electrochemical and in situ IR reflectance spectroscopy studies. J. Electroanal. Chem.2004, 563, 81–89.
Mai, P. T.; Haze, A.; Chiku, M.; Higuchi, E.; Inoue, H. Ethanol oxidation reaction on tandem Pt/Rh/SnOx catalyst. Catalysts2017, 7, 246.
Davenport, W. H.; Kollonitsch, V.; Klein, C. H. Advances in rhenium catalysts. Ind. Eng. Chem.1968, 60, 10–19.
Kirilin, A. V.; Tokarev, A. V.; Manyar, H.; Hardacre, C.; Salmi, T.; Mikkola, J. P.; Murzin, D. Y. Aqueous phase reforming of xylitol over Pt-Re bimetallic catalyst: Effect of the Re addition. Catal. Today2014, 223, 97–107.
Raciti, D.; Kubal, J.; Ma, C.; Barclay, M.; Gonzalez, M.; Chi, M. F.; Greeley, J.; More, K. L.; Wang, C. Pt3Re alloy nanoparticles as electrocatalysts for the oxygen reduction reaction. Nano Energy2016, 20, 202–211.
Goel, J.; Basu, S. Pt-Re-Sn as metal catalysts for electro-oxidation of ethanol in direct ethanol fuel cell. Energy Procedia2012, 28, 66–77.
Zhang, L.; Karim, A. M.; Engelhard, M. H.; Wei, Z. H.; King, D. L.; Wang, Y. Correlation of Pt-Re surface properties with reaction pathways for the aqueous-phase reforming of glycerol. J. Catal.2012, 287, 37–43.
Gharibi, H.; Sadeghi, S.; Golmohammadi, F. Electrooxidation of Ethanol on highly active and stable carbon supported PtSnO2 and its application in passive direct ethanol fuel cell: Effect of tin oxide synthesis method. Electrochim. Acta2016, 190, 1100–1112.
Colmati, F.; Antolini, E.; Gonzalez, E. R. Effect of temperature on the mechanism of ethanol oxidation on carbon supported Pt, PtRu and Pt3Sn electrocatalysts. J. Power Sources2006, 157, 98–103.
Colmati, F.; Antolini, E.; Gonzalez, E. R. Pt-Sn/C electrocatalysts for methanol oxidation synthesized by reduction with formic acid. Electrochim. Acta2005, 50, 5496–5503.
Crabb, E. M.; Marshall, R.; Thompsett, D. Carbon monoxide electro-oxidation properties of carbon-supported PtSn catalysts prepared using surface organometallic chemistry. J. Electrochem. Soc.2000, 147, 4440–4447.
An, K.; Somorjai, G. A. Nanocatalysis I: Synthesis of metal and bimetallic nanoparticles and porous oxides and their catalytic reaction studies. Catal. Lett.2015, 145, 233–248.
Calvillo, L.; Celorrio, V.; Moliner, R.; Lázaro, M. J. Influence of the support on the physicochemical properties of Pt electrocatalysts: Comparison of catalysts supported on different carbon materials. Mater. Chem. Phys.2011, 127, 335–341.
Rizo, R.; Sebastián, D.; Lázaro, M. J.; Pastor, E. On the design of Pt-Sn efficient catalyst for carbon monoxide and ethanol oxidation in acid and alkaline media. Appl. Catal. B: Environ.2017, 200, 246–254.
Alegre, C.; Gálvez, M. E.; Baquedano, E.; Pastor, E.; Moliner, R.; Lázaro, M. J. Influence of support’s oxygen functionalization on the activity of Pt/carbon xerogels catalysts for methanol electro-oxidation. Int. J. Hydrogen Energy2012, 37, 7180–7191.
Sebastián, D.; Suelves, I.; Moliner, R.; Lázaro, M. J. The effect of the functionalization of carbon nanofibers on their electronic conductivity. Carbon2010, 48, 4421–4431.
Acknowledgments
The authors thank the Institute of Engineering Materials and Biomaterials of the Silesian University of Technology, for the use of the Titan FEI TEM instrument. Many thanks also to the Department of Chemistry, University of Warsaw for the access to the Talos F200 FEI TEM instrument. Dr. Ewa Juszynska-Galazka from Department of Soft Matter Research Institute of Nuclear Physics Polish Academy of Sciences is also acknowledged for her help in FTIR measurements. Many thanks to Dr. Alexey Maximenko for running the XRD measurements. Financial support from the Polish National Science Centre (NCN), grant UMO-2014/13/B/ST5/04497 is acknowledged.
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made.
The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Drzymała, E., Gruzeł, G., Depciuch, J. et al. Ternary Pt/Re/SnO2/C catalyst for EOR: Electrocatalytic activity and durability enhancement. Nano Res. 13, 832–842 (2020). https://doi.org/10.1007/s12274-020-2704-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12274-020-2704-1