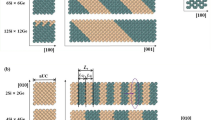
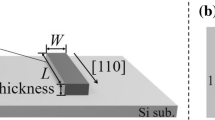
Abstract
Various silicon crystal structures with different atomic arrangements from that of diamond have been observed in chemically synthesized nanowires. The structures are typified by mixed stacking mismatches of closely packed Si dimers. Instead of viewing them as defects, we define the concept of hexagonality and describe these structures as Si polymorphs. The small transverse dimensions of a nanowire make this approach meaningful. Unique among the polymorphs are cubic symmetry diamond and hexagonal symmetry wurtzite structures. Electron diffraction studies conducted with Au as an internal reference unambiguously confirm the existence of the hexagonal symmetry Si nanowires.
Cohesive energy calculations suggest that the wurtzite polymorph is the least stable and the diamond polymorph is the most stable. Cohesive energies of intermediate polymorphs follow a linear trend with respect to their structural hexagonality. We identify the driving force in the polymorph formations as the growth kinetics. Fast longitudinal elongation during the growth freezes stacking mismatches and thus leads to a variety of Si polymorphs. The results are expected to shed new light on the importance of growth kinetics in nanomaterial syntheses and may open up ways to produce structures that are uncommon in bulk materials.

Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Park, C. H.; Cheong, B. H.; Lee, K. H.; Chang, K. J. Structural and electronic properties of cubic, 2H, 4H, and 6H SiC. Phys. Rev. B: Condens. Matter 1994, 49, 4485–4493.
Yeh, C.-Y.; Lu, Z. W.; Froyen, S.; Zunger, A. Zinc-blende-wurtzite polytypism in semiconductors. Phys. Rev. B: Condens. Matter 1992, 46, 10086–10097.
Raffy, C.; Furthmüller, J.; Bechstedt, F. Properties of hexagonal polytypes of group-IV elements from first-principles calculations. Phys. Rev. B: Condens. Matter 2002, 66, 075201.
Wentorf, R. H.; Kasper, J. S. Two new forms of silicon. Science 1963, 139, 338–339.
Jennings, H. M.; Richman, M. H. A hexagonal (wurtzite) form of silicon. Science 1976, 193, 1242–1243.
Kasper, J. S.; Wentorf, R. H. Hexagonal (wurtzite) silicon. Science 1977, 197, 599.
Sánchez-Dehesa, J.; Vergés, J. A.; Tejedor, C. Diamond structure versus wurtzite structure for silicon. Solid State Commun. 1981, 38, 871–873.
Zhang, D. B.; Hua, M.; Dumitrică, T. Stability of polycrystalline and wurtzite Si nanowires via symmetry-adapted tight-binding objective molecular dynamics. J. Chem. Phys. 2008, 128, 084104.
Morral, A. F. I.; Arbiol, J.; Prades, J. D.; Cirera, A.; Morante, J. R. Synthesis of silicon nanowires with wurtzite crystalline structure by using standard chemical vapor deposition. Adv. Mater. 2007, 19, 1347–1351.
Arbiol, J.; Morral, A. F. I.; Estradé, S.; Peiró, F.; Kalache, B.; Cabarrocas, P. R. I.; Morante, J. R. Influence of the (111) twinning on the formation of diamond cubic/diamond hexagonal heterostructures in Cu-catalyzed Si nanowires. J. Appl. Phys. 2008, 104, 064312.
Arbiol, J.; Kalache, B.; Cabarrocas, P. R. I.; Morante, J. R.; Morral, A. F. I. Influence of Cu as a catalyst on the properties of silicon nanowires synthesized by the vapor-solid-solid mechanism. Nanotechnology 2007, 18, 305606.
Zhao, H. Z.; Zhou, S.; Hasanali, Z. H.; Wang, D. W. Influence of pressure on silicon nanowire growth kinetics. J. Phys. Chem. C 2008, 112, 5695–5698.
Cui, Y.; Duan, X. F.; Hu, J. T.; Lieber, C. M. Doping and electrical transport in silicon nanowires. J. Phys. Chem. B 2000, 104, 5213–5216.
Stadelmann, P. A. EMS—A software package for electron diffraction analysis and HREM image simulation in materials science. Ultramicroscopy 1987, 21, 131–145.
Parthé, E. Crystal Chemistry of Tetrahedral Structures; Taylor & Francis, US, 1964.
Cayron, C.; Den Hertog, M.; Latu-Romain, L.; Mouchet, C.; Secouard, C.; Rouviere, J.-L.; Rouviere, E.; Simonato, J.-P. Odd electron diffraction patterns in silicon nanowires and silicon thin films explained by microtwins and nanotwins. J. Appl. Crystallogr. 2009, 42, 242–252.
Wang, Z. W.; Li, Z. Y. Structures and energetics of indium-catalyzed silicon nanowires. Nano Lett. 2009, 94, 1467–1471.
Glas, F. A simple calculation of energy changes upon stacking fault formation or local crystalline phase transition in semiconductors. J. Appl. Phys. 2008, 104, 093520.
Wei, D. P.; Chen, Q. Metal-catalyzed CVD method to synthesize silicon nanobelts. J. Phys. Chem. C 2008, 112, 15129–15133.
Reyes-Gasga, J.; Gómez-Rodríguez, A.; Gao, X. X.; José-Yacamán, M. On the interpretation of the forbidden spots observed in the electron diffraction patterns of flat Au triangular nanoparticles. Ultramicroscopy 2008, 108, 929–936.
Wang, Z. W.; Daemen, L. L.; Zhao, Y. S.; Zha, C. S.; Downs, R. T.; Wang, X. D.; Wang, Z. L.; Hemley, R. J. Morphology-tuned wurtzite-type ZnS nanobelts. Nat. Mater. 2005, 4, 922–927.
Johansson, J.; Karlsson, L. S.; Dick, K. A.; Bolinsson, J.; Wacaser, B. A.; Deppert, K.; Samuelson, L. Effects of growth conditions on the crystal structure of gold-seeded GaP nanowires. J. Cryst. Growth 2008, 310, 5102–5105.
Hao, Y. F.; Meng, G. W.; Wang, Z. L.; Ye, C. H.; Zhang, L. D. Periodically twinned nanowires and polytypic nanobelts of ZnS: The role of mass diffusion in vapor-liquid-solid growth. Nano Lett. 2006, 6, 1650–1655.
Morales, A. M.; Lieber, C. M. A laser ablation method for the synthesis of crystalline semiconductor nanowires. Science 1998, 279, 208–211.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at Springerlink.com
Electronic supplementary material
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License ( https://creativecommons.org/licenses/by-nc/2.0 ), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Liu, X., Wang, D. Kinetically-induced hexagonality in chemically grown silicon nanowires. Nano Res. 2, 575–582 (2009). https://doi.org/10.1007/s12274-009-9058-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12274-009-9058-z