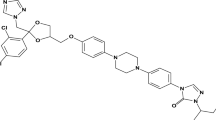
Abstract
Solution thermodynamics and solubility of glibenclamide (GBN) in binary co-solvent mixtures of Transcutol + water at temperature range of 298.15–333.15 K were investigated in present study. The modified Apelblat model was used to predict the solubility of GBN in co-solvent mixtures at various temperatures. The highest and lowest solubility of GBN were observed in pure Transcutol and pure water, respectively. Moreover, all co-solvent mixtures had highest solubility at 333.15 K. The experimental solubility data of GBN was correlated well with the modified Apelblat model at each temperature studied with relative absolute deviation in the range of 0.008–5.903 %. The correlation coefficients in co-solvent mixtures were observed in the range of 0.995–0.999 which indicated good fitting of experimental data with calculated one. The enthalpies and entropies for GBN dissolution were observed in the range of 2.012–38.215 kJ mol−1 and 6.748–114.709 J mol−1 K−1, respectively indicating its dissolution is endothermic and an entropy-driven process. These results indicated that Transcutol can be used as a co-solvent in preformulation studies and formulation development of GBN.


Similar content being viewed by others
References
Abdelbary, A.A., H.F. Salem, and M.E. Maher. 2011. In vitro and in vivo evaluation of glibenclamide using surface solid dispersion (SSD) approach. British Journal of Pharmacology and Toxicology 2: 51–62.
Apelblat, A., and E. Manzurola. 1999. Solubilities of o-acetylsalicylic, 4-aminosalicylic, 3,5-dinitrosalicylic and p-toluic acid and magnesium-dl-aspartate in water from T = (278–48) K. The Journal of Chemical Thermodynamics 31: 85–91.
Bachhav, Y.G., and V.B. Patravale. 2009. SMEDDS of glyburide: formulation, in vitro evaluation and stability studies. AAPS Pharmaceutical Science & Technology 10: 482–487.
Cantillo, E.A., D.R. Delgado, and F. Martinez. 2013. Solution thermodynamics of indomethacin in ethanol + propylene glycol mixtures. Journal of Molecular Liquids 181: 62–67.
Gowrisankar, D., J.M. Rajendra-Kumar, and P.V.M. Latha. 2005. Spectrophotometric estimation of glibenclamide. Asian Journal of Chemistry 17: 1334–1336.
Sardari, F., and A. Jouyban. 2013. Solubility of nifedipine in ethanol + water and propylene glycol + water mixtures at 293.2–313.2 K. Industrial and Engineering Chemistry Research. doi:10.1021/ie402588t.
Seedher, N., and M. Kanojia. 2009. Co-solvent solubilization of some poorly-soluble antidiabetic drugs. Pharmaceutical Development and Technology 14: 185–192.
Shafiq, S., F. Shakeel, S. Talegaonkar, F.J. Ahmad, R.K. Khar, and M. Ali. 2007. Development and bioavailability assessment of ramipril nanoemulsion formulation. European Journal of Pharmaceutics and Biopharmaceutics 66: 227–243.
Shakeel, F., and M.S. Faisal. 2010. Nanoemulsion: a promising tool for solubility and dissolution enhancement of celecoxib. Pharmaceutical Development and Technology 15: 53–56.
Shakeel, F. 2010. Criterion for excipients screening in the development of nanoemulsion formulation of three anti-inflammatory drugs. Pharmaceutical Development and Technology 15: 131–138.
Shakeel, F., S. Shafiq, N. Haq, F.K. Alanazi, and I.A. Alsarra. 2012. Nanoemulsions as potential vehicles for transdermal and dermal delivery of hydrophobic compounds: an overview. Expert Opinion on Drug Delivery 9: 953–974.
Shakeel, F., F.K. Alanazi, I.A. Alsarra, and N. Haq. 2013a. Thermodynamics-based mathematical model for solubility prediction of glibenclamide in ethanol–water mixtures. Pharmaceutical Development and Technology. doi:10.3109/10837450.2013.823992.
Shakeel, F., N. Haq, M. Elbadry, F.K. Alanazi, and I.A. Alsarra. 2013b. Ultra fine super self-nanoemulsifying drug delivery system (SNEDDS) enhanced solubility and dissolution of indomethacin. Journal of Molecular Liquids 180: 89–94.
Shakeel, F., N. Haq, F.K. Alanazi, and I.A. Alsarra. 2013c. Self-nanoemulsifying performance of two grades of Lauroglycol (Lauroglycol-90 and Lauroglycol-FCC) in the presence of mixed nonionic surfactants. Pharmaceutical Development and Technology. doi:10.3109/10837450.2013.829099.
Sohrabi, M.R., N. Kamali, and M. Khakpour. 2011. Simultaneous spectrophotometric determination of metformin hydrochloride and glibenclamide in binary mixtures using combined discrete and continuous wavelet transforms. Analytical Sciences 27: 1037–1041.
Soleymani, J., D. Djozan, F. Martinez, and A. Jouyban. 2013. Solubility of ranitidine hydrochloride in solvent mixtures of PEG 200, PEG 400, ethanol and propylene glycol at 25 °C. Journal of Molecular Liquids 182: 91–94.
Strickley, R.G. 2004. Solubilizing excipients in oral and injectable formulations. Pharmaceutical Research 21: 201–230.
Sunsandee, N., M. Hronec, M. Stolcova, N. Leepipatpiboon, and U. Pancharoen. 2013. Thermodynamics of the solubility of 4-acetyl benzoic acid in different solvents from 303.15 to 373.15 K. Journal of Molecular Liquids 180: 252–259.
Swanson, B.N. 1985. Medical use of dimethyl sulfoxide (DMSO). Review Clinical Basic Pharmacology 5: 1–33.
Wei, H., and R.L. Lobenberg. 2006. Biorelevant dissolution media as a predictive tool for glyburide a class II drug. European Journal of Pharmaceutical Sciences 19: 45–62.
Zhang, C.L., F. Zhao, and Y. Wang. 2011. Thermodynamics of the solubility of sulfamethazine in methanol, ethanol, 1-propanol, acetone and chloroform from 293.15 to 333.15 K. Journal of Molecular Liquids 159: 170–172.
Acknowledgments
The authors would like to extend their sincere appreciation to the Deanship of Scientific Research at King Saud University for its funding the work through the research group project No. RGP-VPP-139.
Conflict of interest
The authors report no declaration of interest. The authors alone are responsible for the content and writing of the paper.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shazly, G.A., Haq, N. & Shakeel, F. Solution thermodynamics and solubility prediction of glibenclamide in Transcutol + water co-solvent mixtures at 298.15–333.15 K. Arch. Pharm. Res. 37, 746–751 (2014). https://doi.org/10.1007/s12272-013-0314-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-013-0314-0