Abstract
The climate crisis has accelerated the frequency and intensity of droughts in the Mediterranean areas, impacting the hydrology and the ecology of their basins. Maule River is the largest watershed in the Chilean Mediterranean Zone. The growing anthropic and environmental pressure has led to changes in the structure and function of its terrestrial ecosystem. However, little is known about its influence on aquatic ecosystems. The effect of the recent extreme drought on the streamflow and its influence on water quality and the benthic macroinvertebrate community structure were evaluated in the Maule River’s lower section. Beyond temporal and spatial fluctuations, healthy and permanently well-oxygenated waters prevailed in the study area. However, during the megadrought period, evidenced by a significant decrease in both precipitation and river streamflow, a greater salt wedge influence was observed, at least, in the last 7 km of the river. Although with certain asynchrony, a change in the structure of the benthic macroinvertebrate community was observed in the upstream stations. This marinization reveals an increase in the domain of marine taxa associated with the Polychaeta group and a clear decrease in typical freshwater Oligochaeta individuals. This modification in the structure of the benthic community is expected to affect the function of this estuarine system. The results presented here highlight the importance of considering the variability associated with biological components for the management of water resources and related ecosystem services, considering that one of the main problems associated with changes in land use and the pressures caused by the climate crisis is the decrease in the quality and availability of water in this and other Mediterranean basins globally.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Mediterranean zones worldwide are characterized by a marked seasonal variability, with heavy rains and floods during the cold and wet winter months and hot and very dry conditions during summer (e.g., Belda et al. 2014). Nevertheless, the climate crisis has accelerated the frequency and intensity of droughts in these zones, impacting the hydrology and the ecology of their basins (Hallett et al. 2018; McKee et al. 1993). Furthermore, it has been widely demonstrated that ecological status of the Mediterranean freshwater ecosystems is being affected by various pressures resulting from agriculture, urban expansion, climate change, and the alteration of their hydrological cycle (Newton et al. 2018; Perennou et al. 2018; Martínez-Megías and Rico 2022). These threats have several socio-ecological and economic implications since freshwater systems provide essential ecosystem services (Herbert et al. 2015; Perennou et al. 2018; Novoa et al. 2019; Morant et al. 2020).
The Chilean Mediterranean Zone (ChMZ) extends between the basins of the Aconcagua and Biobío rivers (32°S–39°S; Di Castri 1981), though these geographical limits can contract or expand due to the occurrence of El Niño or La Niña events (Luebert and Pliscoff 2004, 2006). This area is the most densely populated in the country and is where the greatest economic growth occurs, sustained mainly by the exploitation of its natural resources (Figueroa et al. 2007; Fierro et al. 2019). Here, water is one of the main support matrices, since it plays a fundamental role in the development of agricultural, forestry, and other industrial activities, as well as a source for human consumption (Rubio-Álvarez and McPhee 2010). Therefore, the freshwater systems are essential for the Chilean development due to the variety of economic services they support (Novoa et al. 2019).
Drought is a natural phenomenon that can occur in all climatic zones and is defined as a sustained and regionally extensive occurrence of below-normal natural water availability (Tallaksen and van Lanen 2004). However, and as a result of the climate crisis, a change in the magnitude, frequency, intensity, and spatiotemporal distribution of rainfall is also expected (Maity and Maity 2022; Hallett et al. 2018). Most studies on the impacts of drought on water quality have been carried out in North America, Europe, and Australia (Mosley 2015). However, considering the intensification of droughts globally, it is necessary to study these impacts in other regions of the world such as Asia, Africa, and South America.
The impacts of climate change in the ChMZ are expressed as extensive periods of drought in summer and intense rains concentrated in short periods of time in winter (FAO 2010; Henríquez et al. 2016). In this sense, since 2010 to date (2010–2023), the ChMZ has been affected by a prolonged and intense drought that is unprecedented in the historical record of the last millennium (CR2 2015; Garreaud et al. 2017), with an annual precipitation reduction that could reach up to 30% during this period (Bozkurt et al. 2018).
Drier and warmer conditions have impacted the hydrological processes in the ChMZ area (Garreaud et al. 2017; Alvarez-Garreton et al. 2021), and this uninterrupted rainfall deficit has affected several aspects, including the structure and distribution of its natural vegetation (Arroyo et al. 2020; Garreaud et al. 2017), the fire regime (González et al. 2018), the water supply (Muñoz et al. 2020), and even the dynamics with their interface zones with the ocean (Pérez et al. 2015; Masotti et al. 2018; Vargas et al. 2020).
The second most extensive basin in the ChMZ corresponds to the Maule River (2,105,200 ha), which represents the third most important Chilean catchment (Dirección General de Aguas 2016). Due to the climatic conditions, as well as its geomorphological characteristics, various hydroelectric projects were developed in this basin (Pacheco et al. 2022), as well as several agricultural activities, that represent 26% of the land use, which are an important proportion considering that the native forest represents only 20% (CTHA 2014). During the last decade, the growing anthropic pressure has also led to other changes in the land use mosaic, especially to forestry (Pliscoff 2020). These changes in the landscape generate changes in the hydrological regime, which have been intensified due to the extensive drought; thus, a lower streamflow in the river is expected to decrease both the transport capacity and the dilution volume and therefore increases the concentration of the elements, including pollutants, downstream of the discharge points (Caruso 2002). A lower dilution effect could also increase the concentration of organic compounds, with an intensification in the biochemical oxygen demand and, therefore, a lower oxygen availability (Whitehead et al. 2009). Furthermore, the diminished streamflow could favor the continued presence and extension of marine water (i.e., marinization) with increases in salinity and variations in temperature; that together with the factors mentioned above constitutes a threat to the structure and hence to the functioning of estuarine ecosystems (Whitehead et al. 2009). All these factors cause cumulative or synergistic effects that will ultimately impact the vulnerability of this watershed (Arriagada et al. 2019).
Evaluation of aquatic ecosystems in this region has been developed mainly under a traditional approach, based on the comparison of the ranges of environmental parameters obtained in situ, mainly physicochemical, with local normatives (Kannel et al. 2007). However, this approach does not adequately reflect the health status of an aquatic environment (Debels et al. 2005). Suitable ecological management requires an evaluation not only of the physicochemical attributes of the waters but also of the composition and abundance of their biological communities (Karr and Chu 1998; Fierro et al. 2018), because they integrate both structural and functional characteristics and reflect the health of the studied system (He et al. 2020; Bonada et al. 2006). Benthic macroinvertebrates, mainly annelids, mollusks, insects, and crustaceans (> 0.5 mm in size), are an essential link between the nutrient cycles and the energy transferred to higher trophic levels (Welsh 2003) that have proven to be a proper bioindicator to assess spatial and/or temporal trends of water quality in rivers, lakes, and wetlands (Tampo et al. 2021; Mereta et al. 2013). At the spatial level, a longitudinal zonification of the benthic communities is observed as the characteristics of the environment vary, which can be explained by the concept of a “continuous river,” which proposes that the characteristic biological communities of a certain fluvial section are established in harmony with the dynamic physicochemical conditions of the river (Vannote et al. 1980). Among the factors that influence the composition, abundance, biomass, and distribution of these communities, the type of vegetation, basin land use, type of substrate, temperature, dissolved oxygen, and streamflow magnitude have been described, being the alterations in the magnitude of the streamflow the one that generates the most important ecological changes (Poff and Zimmerman 2010). Despite its importance, the diversity of the benthic macroinvertebrate community of many rivers in the ChMZ remains completely unexplored. Taking into account that several threats (e.g., changes in land use, introduction of exotic species, habitat loss and degradation, pollution, and climate change) have been identified as affecting the aquatic environments of the ChMZ (Fierro et al. 2019), the risk that many species go from being unknown to disappearing is high (Figueroa et al. 2013). Therefore, policies focused on the protection and conservation of the biodiversity of these aquatic environments are urgently required.
The main objectives of this research were, thus, to assess the megadrought effect on the hydrological regime, water quality, and structure of the benthic macroinvertebrate communities of Maule River’s lower reach, through a biannual sampling (i.e., summer–winter) from 2008 to 2020. Given the study encompasses data throughout the intensification of the megadrought period, it is possible to assess whether there has been any response from the benthic community to this threat. A substantial change in the streamflow of the river is expected as a direct effect of the prolonged drought period, while changes in the water quality and the structure of the benthic macroinvertebrate communities are also expected to have occurred from the hydrological variation.
Methods
Study Area
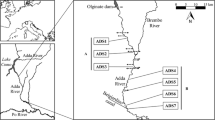
The Maule River is in the ChMZ between 35°10′ and 36°30′ of latitude south and between 70°25′ and 72°30′ of longitude west. Its basin covers 21,052 km2 and at its lower reach it registers an average streamflow of 532 m3/s (Urrutia et al. 2011). This river is born in the Laguna del Maule, and its main tributaries are the Puelche, Los Cipreses, Claro, and Melado rivers. In its intermediate part, the main tributary is the Loncomilla River, from which the Maule River runs confined by the Costal Range. In the last 10 km, the river widens into an estuary that is about 900 m wide at its mouth, where a natural-formed sandbar is modulated by the river streamflow (Fig. 1). The natural hydrological regime of the Maule River was described as of the mixed type in its upper part—pluvial floods in winter, spring, and early summer and pronounced low water in autumn, while in its middle and lower part a pluvial type regime prevailed (Niemeyer and Cereceda 1984). Furthermore, the estuarine zone has low tidal amplitude (1–2 m) with a mixed regime (Araya-Vergara 1982) and a water column height oscillating between 2 and 10 m depth. The main land uses in its basin are for agricultural and grazing (33.5%), scrubs (16.3%), native forests (20.0%), plantations (18.1%), and unused lands (17.2%), while a small proportion (1.0%) is represented by mixed forest (CTHA 2014).
Hydroclimatology
Watershed precipitation and streamflow river data was obtained from the CR2 platform (http://explorador.cr2.cl/), an online tool that allows viewing systematized data from various government information sources such as the Dirección General de Aguas (DGA) and the Dirección Meteorológica de Chile (DMC). The average daily streamflow data was obtained from the flow meter station in Forel (station code: DGA 07383001), which is the closest gauging station to the Maule River’s mouth and has information from 1985 to date. Air temperature and precipitation data from the Maule River station in Forel was complemented with data from other stations located within the basin, such as the Talca U.C. (station code: DGA 07383001), which has uninterrupted information since 1982.
Benthic Communities and Water Column Sampling
The information associated with the benthic macroinvertebrate communities and the physicochemical characterization of the lower reach of the river was obtained from the Programa de Monitoreo Marino Costero-Planta de Constitución, Arauco Company. This program collected environmental information since 2008 to date (2023), but the data from 2008 to 2020 are used for this study. Samples were collected on a six-monthly, seasonal basis (summer/winter), using a fixed sampling design consisting of three sampling stations: at the mouth (Maule-1) and 7 km (Maule-2) and 13 km (Maule-3) upstream from there (Fig. 1).
Samples for the study of the benthic macroinvertebrate communities were obtained in triplicate with a Van Veen grab sampler of 0.025 m2. Subsequently, samples were washed and sieved through a 0.5 mm mesh. Samples were fixed in 10% formalin and kept and transported in polyethylene bags. Samples were analyzed within a period not exceeding 1 week after sampling, in order to prevent the breakdown of calcium carbonate shells. In the laboratory, a Rose Bengal dye solution was added to facilitate visualization and separation of the individuals, which were identified to the lowest possible taxonomic level. Separation, identification, and counting of the different species or taxa were carried out with a stereomicroscope (Olympus SZ). Identification of the individuals was carried out using specialized literature (Cerda and Castilla 2001; Fauchald 1977; Forcelli 2000; Lancellotti and Vásquez 2000; Retamal 1981; Rozbaczylo 1980) and verified in the World Register of Marine Species (http://www.marinespecies/org).
The hydrographic characterization of the water column was carried out through vertical deployments of a CTD-O profiler (Sea-Bird 19 Plus probe), which registers the following parameters at a high frequency: conductivity-salinity, temperature (°C), pressure (dbar), and dissolved oxygen (mg/L) from the surface to the riverbed. Salinity was measured using the Practical Salinity Scale. Just one CTD profile (the downward) was used by station. In addition, surface water samples were collected in triplicate by station for water quality analysis, following the recommendations contained in the Chilean normative: Norm No. 411/2 Of 96 (“Water quality–Sampling–Part 2: Guide on sampling technique”), Norm No. 411/3 Of 96 (Water quality–Sampling–Part 3: Guide for the conservation and management of samples), and Norm No. 411/6 Of 96 (Water quality–Sampling–Part 6: Guide for sampling rivers and watercourses). Then, in the laboratory, analyses of pH, true color (Pt.Co), total solids (mg/L), total suspended solids (mg/L), oils and fats (mg/L), and total and fecal coliforms (MPN/100 mL) were carried out, following the standard methods established in APHA-AWWA-WPCF (1995).
Data Analyses
In order to validate the information obtained and detect the existence of possible anomalous data, a preliminary analysis of the hydroclimatic data was carried out following the recommendations of Zuur et al. (2007) and Borcard et al. (2011). The streamflow and precipitation series were analyzed using the Mann–Kendall non-parametric trend test, complemented with the Sen slope estimator. The non-parametric Mann–Kendall test is a convenient linear procedure for non-normally distributed data and for data containing extreme values and non-linear trends. The null hypothesis (Ho) is that the data (× 1, × 2, …, xn) is an independent and identically distributed sample of random variables. The objective of these tests is to detect the tendency of the data to increase or decrease. The complemented Sen test corresponds to a non-parametric method, which has certain advantages, such as that it allows missing values or that the sample does not have to fit a particular distribution (Kendall 1975; Mann 1945). These methodologies are widely used in the analysis of environmental time series, since they are reliable with a small sample size and are tolerable to variations (i.e., noise) in the time series (e.g., Andrades et al. 2019). Additionally, to identify the significant frequencies within the hydroclimatic series and their location in the time domain, a wavelet analysis was performed (Cazelles et al. 2008), and the Standardized Precipitation Index was calculated according to McKee et al. (1993).
The seasonal variability (summer–winter) and the temporal trends of the physicochemical parameters were evaluated through the non-parametric analysis of Wilcoxon rank sum tests and a Mann–Kendall and Sen’s slope test, respectively. In order to evaluate the possible existence of space–time variations in the environmental variables, an analysis of variance based on multiple permutations (PERMANOVA) (Anderson et al. 2016) was carried out using the software PRIMER v7, considering the factors Year (from 2008 to 2020), Season (summer and winter), and Location (Maule-1, Maule-2, and Maule-3), using the distance matrix of the environmental variables, obtained based on the Euclidean distances of the normalized data, considering 9999 permutations.
Principal component analysis (PCA) was used to analyze the physicochemical data of the water. This technique reduces the dimensionality of a data set by keeping the elements that contribute the most to its variance, a method that has been widely used in the interpretation associated with the study of water quality (Mishra 2010). This analysis identifies underlying relationships between the monitored parameters, as well as segregations and/or clusters among them. The Water Quality Index (WQI) was also estimated, following the recommendations in Cade-Idepe (2005) and Debels et al. (2005). This management tool simplifies the presentation of the quality results, summarizing a series of analyzed parameters in a value. This index was calculated for each sampling station and campaign using all the environmental parameters measured (see Table 1).
To analyze the benthic macroinvertebrate community data, the following univariate community descriptors were determined: abundance, taxa richness (S), Shannon–Wiener diversity (H′), Simpson dominance (λ), and Pielou uniformity (J′). Complementarily, with the abundance data of all taxa, a Bray–Curtis similarity matrix was constructed transforming the data to the fourth root. A PERMANOVA test was then carried out to evaluate the existence of possible temporal and spatial variations in the structure of these communities, considering the factors Year (2008 to 2020), Season (summer and winter), and Location (Maule-1, Maule-2, and Maule-3) and running 9999 permutations. The significant differences detected with the PERMANOVA were evaluated by a paired comparison analysis (pairwise test; Anderson et al. 2016). Additionally, to determine which taxa contributed more to the dissimilarity between the levels of the temporal or spatial factors that presented significant differences, a Similarity Percentage (SIMPER) analysis was applied using the Bray–Curtis similarity index (Clarke 1993). Finally, to determine the relationships between the environmental parameters and the temporal and/or spatial changes observed in the structure of the benthic macroinvertebrate communities’ abundances, a Distance-based Linear Model (DistLM) analysis was carried out, which considers linear models based on the matrices of distance (Anderson et al. 2016). For this analysis, 9999 iterations were considered, and the Bayesian information criterion (BIC) was used to select the best models. The level applied to all tests to determine significance was p < 0.05. For this procedure, the Primer 7 BEST routine was considered (Clarke & Gorley 2015). Furthermore, a Spearman analysis was developed to evaluate the correlation between the physicochemical parameters evaluated.
Results
Hydroclimatic Variability
The entire time series (1985–2020) of the accumulated monthly precipitation exhibits marked interannual fluctuations, with a significant downward trend (p value < 0.001; n = 449) for the 2006–2020 period (Fig. 2a). The highest records were found during the winter months (June–August), whereas comparatively the lowest levels characterized the summer months (December to February). Between the years 1985 and 2006, specifically during the years 1987 (July), 1992 (June), 1997 (June), and 2000 (June), a maximum rainfall in winter > 250 mm stands out; however, in the last 11 years of the series (2009–2020), these maximums did not exceed 200 mm per month.
Temporal variability of a accumulated monthly precipitation in the Maule River basin, b monthly riverine streamflow, and c Standardized Precipitation Index (considering a scale of 12 months) for the period 1985–2020. The gray area in a and b indicates the moment at which the decline in the rainfall and streamflow trends begins, respectively
A temporal pattern like that described for the precipitation was observed in the streamflow of the Maule River (Fig. 2b). The monthly streamflow between 1985 and the beginning of 2020 showed high interannual variability, where the maximums (> 2000 m3/s) were registered during the winter months (June–August) of the years 1987 (July), 1997 (June), 2001 (July), 2002 (August), and 2006 (July). However, after 2007, the streamflow exhibited a significant downward trend (p value < 0.001; n = 426). In fact, the lowest average streamflow, registered during summer (December to February), changed from 214.3 ± 142.7 m3/s for the period 1985 to 2006 to 175.5 ± 78.8 m3/s from 2007 onwards.
The Standardized Precipitation Index (SPI) showed the presence of moderately humid conditions (SPI > 1), interspersed with moderately dry periods (SPI < −1), between January 1985 and mid-2007 (Fig. 2c). During this period, the noticeable highlights include an extremely dry condition (SPI < −2) between the middle of 1998 and the middle of 1999. However, after 2007 and until the end of the temporal record (i.e., 2020), a recurrent dry condition (SPI < −1) prevailed, reaching an extremely dry condition at the beginning of 2020. It should be noted that during the latter period no SPI magnitudes associated with humid conditions were detected.
The wavelet analysis of the monthly accumulated precipitation and monthly discharge showed the persistence of a significant annual cycle (confidence level of 0.95), which was more consistent for precipitation (Fig. S1a and b). However, in some years, significant pulses associated with variations in the precipitation and streamflow due to seasonal changes stand out. Nevertheless, these signals disappear after 2006 and 2009 for the precipitation and streamflow, respectively.
Water Quality of the Maule River
Table 1 presents the average values and the standard deviation associated with the parameters evaluated in the waters of the Maule River by sampling location for the study period (2008–2020) and the results of the seasonal and temporal analysis. Significant seasonal variability was found in all sampling stations for total coliforms, pH, and temperature, while total solids only was significant in Maule-1 and Maule-2. Otherwise, seasonal differences in salinity were just observed in Maule-2 and Maule-3. The average concentration of oils and fats and the microbiological load (i.e., total and fecal coliforms) were comparatively higher during winter while higher values were registered during the summer period in total solids, pH, and temperature. It should be noted that salinity in the station closest to the river mouth (Maule-1) did not present notable seasonal variations; however, temporal fluctuations were detected at the Maule-2 and Maule-3 stations. In contrast, slight seasonal variations were detected in the parameters true color, total suspended solids, and dissolved oxygen at all stations. The temporal analysis (i.e., Mann–Kendall test and Sen slope) by variable for each sampling station indicated that significant trends (view p values and sample size in Table 1) were detected for oils and fats (Maule-1 and Maule-3) and total suspended solids (Maule-1, Maule-2, and Maule-3) that exhibited downward trends, while salinity presented a significant upward trend (Maule-2).
The homoscedasticity test (PERMDISP) for the set of environmental variables was significant (F12,65 = 3.36, p value = 0.019) only in the factor Year. However, the pairwise analysis of this factor was only significant in less than 34% of all comparisons; hence, there would be no multivariate global over-dispersion for the data. The PERMANOVA results indicate that the main factor that explains the variation of the environmental data was Season (60.8%MS) (Table 2a).
The PCA indicated that there are five main components with values > 1, which, together, explain 82.4% of the total variability of the information. Figure 3 represents PC1 versus PC2 in two dimensions, which together are the components that explain the highest percentage of the variability of the information (44.5%). Temperature and pH showed strong correlation (r > 0.69) with PC1, with higher records during the summer periods. Microbiological load (i.e., total and fecal coliforms) and oils and fats have moderate correlations (r > 0.60) with PC2. However, the dispersion of the samples did not show a clear seasonal behavior.
Two-dimensional representation of the first two main components (PC1: first principal component; PC2: second principal component) generated with the environmental information collected between the years 2008 and 2020. In brackets the percentage of variance explained by each component. CE: conductivity, DO: dissolved oxygen, Oil_fats: oil and fats, TSS: total suspended solid, Tcol: total coliform, Fecol: fecal coliform, TS: total solid
Temporal behavior of the physicochemical parameters that significantly explain the variability of the benthic macroinvertebrate community obtained from the DistLM (see details of this analysis in “Benthic Macroinvertebrate Communities”) is observed in Fig. 4. Oils and fats, dissolved oxygen, and total suspended solids showed a similar variability among stations, with a clear decrease in oil and fats and total suspended solids starting in the winter 2015 and summer 2013, respectively, which lasted until the end of the series. On the other hand, dissolved oxygen remains at high concentrations throughout the study period. Salinity, by contrast, showed clear differences among sampling stations. Regardless of the variability of the streamflow or the seasonality of the sampling, a large part of the water column in the station Maule-1 was dominated by brackish-type waters (0.5–17), with marine-type waters (> 17) in the bottom layer. Freshwaters (< 0.5) were occasionally recorded on the surface during winters of 2009 and 2011. Previous to winter 2012, values < 3 dominated the entire water column in the station Maule-2, varying between brackish and fresh waters. However, since the summer of 2013 and until the end of the series, the presence of brackish waters increased remarkably, dominating almost the entire water column, even with marine-type water (> 17) registered at the bottom during the summer campaigns. On the contrary, 13 km upstream from the mouth of the Maule River, in the locality Maule-3, regardless of the seasonality or the variability of the streamflow, the entire water column was dominated by fresh or inland waters (< 0.5) during all the studied period (2008–2020).
The WQI denotes a general trend with high-quality waters recorded in the summer periods relative to what was observed during winters. Furthermore, it was observed a longitudinal gradient throughout the study area during most of the study period, with waters varying between very good quality in Maule-2 and Maule-3 and good quality in Maule-1. Nevertheless, and superimposed to this seasonal and spatial trend, the water quality in each location appears to improve over time (Fig. 5).
Benthic Macroinvertebrate Communities
A total of 31,682 specimens representing 69 invertebrate taxa were recorded, with a clear decline in abundance from 2015, despite slight punctual increases (Fig. S2a and b). Considering only the major taxonomic groups, Polychaeta had the highest abundance followed by Oligochaeta (17,144 and 13,755 individuals, respectively). Crustacean and Mollusca were noticeably less represented with 601 and 120 individuals found, respectively. These groups together represented 99.8% of the total number of individuals. The total abundance of invertebrates varied throughout the study period, as well as between seasons, and among stations (Fig. S2a). The highest number of invertebrates occurred at Maule-3 in the summer of 2011 (11,369 individuals; out of scale in Fig. S2a), while invertebrates were absent at select sites on four sampling occasions (i.e., summers of 2017–2018 at Maule-2 and Maule-3 and winter of 2018 at Maule-1 and Maule-3).
The community indexes of diversity (H′), dominance (λ), and uniformity (J′) were highly variable spatiotemporally throughout the study period, with recurrent ascending and descending cycles in all localities (Fig. S3). Nevertheless, H′ in Maule-1 seems to show a decreasing trend between winter 2009 and winter 2016 and then exhibited irregular behavior until the end of the series. H′ in Maule-2, in general, was quite irregular; however, an upward trend stands out between summer 2019 and winter 2020. H′ in Maule-3 was also highly irregular over time, although in general it was higher, compared to Maule-2, from the winter campaign of 2012. Though the community structure parameters J′ and λ presented highly irregular spatiotemporal distributions, recurrently, between summer 2008 and winter 2013, the highest values of these indices were found in Maule-1, while between summer 2014 and winter 2020 the highest were registered in Maule-3.
The PERMDISP analysis for the abundance data was significant (p < 0.05) in the factors Year and Location. Nevertheless, after evaluating the paired analysis for the factor Year, significant values (F12,65 = 2.96, p value = 0.027) were only detected in less than 16.7% of all comparisons, while in the case of the factor Location for the three sampling sites, only one was significant (Maule-2). Therefore, there is no multivariate global over-dispersion in the biotic data. The PERMANOVA results indicate that the main factor that explains the variation of the benthic macroinvertebrate community’s data was Location (46.6%MS) (Table 2b). The SIMPER confirms community differences among stations. The greatest dissimilarity among localities occurred between Maule-1 and Maule-3, followed by Maule-2 and Maule-3, and finally between Maule-1 and Maule-2 (Table 3). The most representative taxa at Maule-1 were Prionospio, Phoxocephalidae, and Gammaridea. However, after winter 2016, these groups were occasionally registered, while Nereididae, Capitella, Cossura, Microphoxus, and Mulinia contributed more to the total abundance (Fig. 6). In Maule-2, Prionospio also dominated, but after 2016, Nereididae and occasionally Phoxocephalidae were also abundant. In Maule-3, by contrast, Oligochaeta was the most represented taxa until 2012, being only recorded occasionally after this year (i.e., winter of 2016 and 2019), when Capitella and Prionospio dominated the community (Fig. 6).
The DistLM assessing which environmental variables best described changes in the invertebrate community in isolation or combination are presented in Table 4. The parameters that significantly explained the variability in the benthic community in descending order of importance were oils and fats, dissolved oxygen, total suspended solids, and salinity (Fig. 4). In addition, the BIC indicated that the best adjusted model was obtained with the levels of oils and fats, followed by the model that also included dissolved oxygen, while the third best model also included salinity (Table 5). Nevertheless, the Spearman correlation analysis indicates that there is significant relationship between oils and fats and total suspended solids and between dissolved oxygen and salinity (Fig. S4).
Discussion
Droughts are defined as a condition where the availability of water is below normal in a sustained and spatially extensive manner, which can occur in different climatic zones (Tallaksen and van Lanen 2004). In the Mediterranean areas of the world, intense and short-term droughts (1 to 2 years) are a common phenomenon, although longer dry periods have been recorded in recent decades (Garreaud et al. 2020; Hallett et al. 2018; McKee et al. 1993). This increase in the temporal extension of droughts has also been described for the Chilean Mediterranean Zone (ChMZ), where an uninterrupted sequence of dry years has been recorded since 2010 (CR2 2015; Garreaud et al. 2017, 2020). This megadrought has been characterized by annual rainfall deficits that oscillate between 25 and 45%, a drop in Andean snow cover, and a decrease in the river’s streamflow, volumes of reservoirs, and groundwater levels (CR2 2015; Garreaud et al. 2017, 2020; Alvarez-Garreton et al. 2021). Although it was established, through streamflow reconstructions (Urrutia et al. 2011), that the Maule River basin has had at least 4 years with very low streamflow levels (i.e., 1698, 1908, 1924, and 1998), classified among the ten driest in the last 800 years (Muñoz et al. 2016), the current drought affecting the ChMZ is unprecedented in the last century due to its intensity and duration (Garreaud et al. 2017, 2020). However, based on our hydroclimatic variability results (1985–2020), the sign of megadrought in the study area started earlier than described for the ChMZ (e.g., CR2 2015; Garreaud et al. 2017, 2020; Alvarez-Garreton et al. 2021). A significant decline in both precipitation and streamflow, registered since 2006 in the basin and 2007 in the Maule River, respectively, resulted in a transitional period from moderately to extremely dry. These sustained decreases in these hydroclimatic parameters occur concomitantly with other natural disturbances and the multiple anthropogenic stressors that modulate the behavior of the freshwater ecosystems in the basin, causing cumulative or synergistic effects that deteriorate the health of these systems and increase the vulnerability of the watershed (Arriagada et al. 2019).
The present investigation, which considers a 12-year follow-up of the last 13 km of the Maule River, revealed that, regardless of the monitoring point, during the summer periods higher levels of total solids, pH, temperature, and conductivity-salinity were generally recorded. On the contrary, during winter periods, it was common to find a higher microbiological load (i.e., total and fecal coliforms), as well as higher levels of oils and fats, parameters that evidence the influence on the river of human activities that occur in the basin. These changes in the riverine water quality are mainly related to seasonal variations in water inputs to the basin. A lower streamflow during summer implies a diminishing in the transport capacity and increases the water residence times along with a lower dilution volume (Whitehead et al. 2009); hence, during this period, a higher concentration of solids is expected, especially in the lower parts of the river, as well as a greater spatial influence of marine waters due to a higher tide amplitude. Likewise, there is an increase in temperature because of the intensification in radiation and in pH as a result, among others, of a greater autotrophic activity and/or changes in the solubility of bicarbonate (Hall et al. 2023). On the other hand, the increase in freshwater flows as a result of punctual precipitation events that occur during winter periods increases the dragging of pollutants from the basin into the river. Nevertheless, and despite this seasonal variability, the study area recurrently exhibited waters that ranged from good to very good quality according to the WQI, although with temporary variations where these quality conditions are affected, mainly associated with periods with greater saline influence or more punctual records associated with increases in the total and fecal coliforms and high levels of oils and fats. Nevertheless, most of the evaluated parameters oscillated within the ranges that have been reported for the Maule River (Cade-Idepe 2004a), as well as in other ChMZ basins (Cade-Idepe 2004b, c, d; Debels et al. 2005; Torres-Ramírez et al. 2017; Fierro et al. 2021).
Superimposed to the seasonal trend, the river water quality throughout the study period improves, evidencing a lower impact of the activities that occur in the basin (mainly anthropogenic). This improvement in quality is the result of less washing and dragging due to the diminished in river streamflow, as a consequence of the sustained decrease in rainfall due to the drought. Furthermore, this decrease in streamflow has favored the influence of waters of marine origin upstream—“marinization” (Hallett et al. 2018), influence that has been increasing as the megadrought has developed. Reducing the contributions of pollutants from diffuse sources during periods of drought could improve the quality of the water of the receiving body (Caruso 2002), although because of the lower dilution, during these periods of low streamflow the pollutants could increase its concentration in point sources (Zwolsman and van Bokhoven 2007; Peña-Guerrero et al. 2020). In fact, there are several studies that demonstrate that the water quality of freshwater systems could decrease during periods of drought (e.g., Mosley 2015). However, the magnitude of the impacts on water quality depends on many factors beyond the human activities that occur in their basins, among them it is worth highlighting the type of water body, its shape, size, depth, water regime, and water residence times, among others (Van Vliet and Zwolsman 2008; Delpla et al. 2009; Li et al. 2017). Nevertheless, this impact can be exacerbated during drought periods if the water quality of the system is already deteriorated (Zwolsman and van Bokhoven 2007). Therefore, the impact of drought on the water quality of freshwater systems is complex and spatiotemporally variable (Mosley 2015).
A study carried out in the estuarine sector of the Maule River estimated that the tidal could reach approximately 16 km upstream from the mouth (Araya-Vergara 1981). A hydrodynamic modeling work determines that the advance of this salt wedge is affected by the sandbar that forms naturally at the mouth and that is modulated by the river streamflow, which also plays an important role in restricting the advance of this wedge upriver (Arce 2014). Nevertheless, prior to 2012, no marine-type waters (> 17) had been detected in the locality of Maule-2; therefore, the persistence of a dry environmental condition together with a lower streamflow would be favoring the influence of the marine waters upstream (at least 7 km), with the consequences that the entry of these salt wedges entails by affecting the ecosystem services provided (e.g., water used for agriculture, human consuming, or aquaculture). In fact, the main cause of salinity intrusion in estuarine systems is the freshwater scarcity (Herbert et al. 2015). The effects of saltwater intrusion on ecosystem services have received less attention, perhaps because shifts in water chemistry are invisible to the general public. Nonetheless, marine salt inputs to previously freshwater-dominated systems have profound impacts on ecosystem biogeochemistry, leading to coastal forest loss, species invasions, reductions in agricultural productivity, changes in coastal water quality, and marsh migration (Mosley 2015; Tully et al. 2019; Martínez-Megías and Rico 2022). Several adverse environmental effects are triggered by the intrusion of the salt wedge, because saline wedge intrudes not only longitudinally, but also laterally, could degrade nearby aquifers by salinizing coastal groundwater (Barlow 2003). In addition, the vertical exchange of dissolved oxygen with the bottom layer is interrupted due to the isolation of this lower, denser salt layer (Watanabe et al. 2014), leading to bottom anoxia and consequently deteriorating the water quality (D’Adamo and Lukatelich 1985; Tweedley et al. 2015). These anoxic conditions could also trigger the release of nutrients and trace metals due to the change in the redox condition of the sediments (McAuliffe et al. 1993; Kraal et al. 2013). Nevertheless, despite the marinization, the estuary of the Maule River remains oxygenated (> 4 mg/L) throughout the study period, even at the bottom (data not shown), due likely to the wind influence that could mix the entire water column avoiding the stratification. However, abrupt saltwater intrusions affect the structure, distribution, and function of the benthic community along the riverine systems (Sylaios et al. 2006).
Rivers of the ChMZ have been poorly studied and knowledge of their biodiversity is scarce and fragmented, especially in relation to benthic macroinvertebrates (Fierro et al. 2019), where the existing information is concentrated mainly in the groups of insects, mollusks, and crustaceans (Figueroa et al. 2013). Regarding the environmental factors that regulate the biodiversity of these aquatic communities, it has been widely demonstrated that the natural streamflow regime, which in turn partially modulates the water quality, has a profound influence not only on the structure but also on the behavior of these organisms; however, there is a series of interrelated mechanisms that exert control over these characteristics that operate on different temporal and spatial scales (Poff and Zimmerman 2010; Bunn and Arthington 2002). Among the factors that regulate these mechanisms, the quality of the waters and the structures and the complexity of the riverine environments are important modulators of the zoning patterns of the communities that live there (Habit et al. 2007). The results of this study indicate that the system presents waters which quality increased through time, but with substantial and persistent changes in the magnitude of the streamflow that allow a growing, both in magnitude and extension, influence of marine waters, which, consequently, modifies the benthic macroinvertebrate community structure upstream.
Several investigations that have evaluated the composition and structure of benthic macroinvertebrate communities in other basins within the ChMZ have been developed mainly in the intermediate depression or in the upper part of the basins (Figueroa et al. 2013; Soria et al. 2019; Fierro et al. 2019). These studies describe a high number of taxa (> 50), within which the Insecta group stands out in abundance and diversity, with the contributions of the groups Crustacea, Mollusca, and Oligochaeta being poorly represented (Habit 1998; Figueroa et al. 2006, 2007, 2010, 2013; Soria et al. 2019; Fierro et al. 2021). In the present study, the first developed in the lower part of a ChMZ river, a typical assemblage of estuarine benthic fauna was registered during the first part of the study (before 2010–2012). Insecta and Oligochaeta were identified in the sector furthest from the coast, in Maule-3, but after 2012 typical marine taxa dominated and Oligochaeta was found just occasionally during winter periods. On the contrary, Polychaeta was the most abundant group in the entire study area, especially in the localities Maule-1 and Maule-2, coinciding with what has been reported for some coastal freshwater systems (i.e., lakes, lagoons, and estuaries) in southern central Chile, where the Polychaeta stands out for its abundance and diversity (Bertrán and Parra 2001; Bertrán et al. 2016; Fierro et al. 2014; Torres-Ramírez et al. 2017).
The physicochemical analysis of the lower section of the Maule River indicates that, beyond the temporal (winter-summer) and spatial fluctuations, healthy and permanently well-oxygenated waters (> 4 mg/L) prevailed in the study area, with quality parameters oscillating, in general, between ranges allowed by the Chilean regulations on water for agricultural irrigation and for recreational use with direct contact (NCh. 1333). In relation to the environmental parameters that significantly explain the variability of the benthic macroinvertebrate community structure, it seems that the main underlying factor modulating this variability is related to the marinization of the system, i.e., a decrease in the streamflow as a result of the megadrought, with a concomitant downward trend in total suspended solids and in oil and fats (the latter as a proxy of anthropogenic influence) and an upward trend in salinity in permanently oxygenated waters due to the increasing spatiotemporal influence of the salt wedge. Changes in the benthic macroinvertebrate community structure upstream (Maule-2 and Maule-3) occurred during winter 2010 and summer 2013, respectively, revealing an increasing presence of taxa associated with the Polychaeta group, with dominant organisms that are typical of the marine environments (e.g., Prionospio, Capitella), along with a decline of the Oligochaeta group, characteristic of freshwaters. This apparent delay between changes in the waters’ physicochemical signal (at least with the timing when it is possible to register it) and the response of the benthic community’s structure would be explained mainly by the biological characteristics and lifestyles of these organisms. Classified as good indicators of the function of the river systems and sentinels of disturbances that are not detected through traditional evaluations (e.g., chemical characterization), benthic organisms respond to the cumulative effects of disturbances over time (Figueroa et al. 2013; Tweedley et al. 2015; Fierro et al. 2018, 2021; Elgueta 2020), capturing longer-term changes in the environment than are not recorded by sporadic water quality monitoring (Mosley 2015). Therefore, to the threats that would be affecting the Chilean Mediterranean aquatic ecosystems identified by Fierro et al. (2019), and already described (see “Introduction”), the diminishing of the riverine streamflow as a result of the current megadrought condition and, in consequence, the marinization of the rivers should also be considered. Thus, this saline influence modifies not just the physicochemical characteristics altering the habitat of the freshwater systems but the structure and likely the function of their biological communities due to the osmotic stress caused (Anton-Pardo and Armengol 2012; Hallett et al. 2018). Peña-Guerrero et al. (2020) indicate that during periods of drought it is necessary to evaluate the effect of anthropogenic stressors on water quality through chemical analysis of pertinent environmental variables; nonetheless, the results presented here highlight the importance of also considering the variability associated with biological components that are important for the functioning of aquatic ecosystems, such as benthic macroinvertebrate communities.
This research is a contribution to the fragmented knowledge of the estuarine environments of the Mediterranean areas, especially in relation to variations in both quality and availability of water, as well as changes in benthic macroinvertebrate communities in response to the incidence of saline waters, caused by the changes in the hydrodynamic regime due to the projected intensification and more recurrent drought events. Anthropic activity, product of economic development, has generated important changes in land use (e.g., agricultural, forestry, mining, and urban), which is one of the main threats to biodiversity worldwide and a detrimental factor not only on the quality but also on the quantity of the water present in the basins (Cuevas et al. 2014). This study area in particular has presented important variations in the conformation of the coastal landscape, registering a significant loss of deciduous forest, mainly due to the increase in forestry plantations (Miranda et al. 2017; Pliscoff 2020). These tensors exacerbated by the current megadrought and the increasing pressure on the scarce water resources require the implementation of permanent monitoring programs that provide information about the condition of different environmental matrices, in order to develop and implement policies that allow preserving the diverse ecosystem services of the basins present in the ChMZ.
Conclusions
Megadrought has produced profound changes in the hydrological regime—, i.e., a marked and sustained diminishing in precipitation and riverine streamflow, which have allowed not just a growing upstream influence of the marine salt wedge but an important alteration of the benthic macroinvertebrate communities structure in the Maule River’s lower reach. This modification in the composition of the benthic community, whose spatial influence seems to be controlled by the intensity and duration of the drought, is expected to affect the function of the ecosystems associated with the lower part of the river, mainly those associated with the estuarine zone, and consequently the services that these ecosystems offer.
Finally, and bearing in mind that one of the main problems associated with landscape changes and the pressures caused by the climate crisis is the decrease in the quality and availability of water—, a problem that is exacerbated in the current context of a megadrought, the results reported here provide useful information for the management of the water resources in this and other Chilean Mediterranean basins. For a comprehensive evaluation, it is recommended, in addition to a permanent chemical characterization of the waters, the incorporation of other complementary elements that provide environmentally relevant information, such as the analysis of the different land uses and the study of the aquatic communities (e.g., benthic macroinvertebrates, fish, and/or diatoms) present throughout the entire basin.
Data Availability
The data that supports the findings of this study are available upon request from the authors.
References
Alvarez-Garreton, C., J.P. Boisier, R. Garreaud, J. Seibert, and M. Vis. 2021. Progressive water deficits during multiyear droughts in basins with long hydrological memory in Chile. Hydrology and Earth System Science 25: 429–446. https://doi.org/10.5194/hess-25-429-2021.
Anderson, M.J., R.N. Gorley, and K.R. Clarke. 2016. PERMANOVA+ for PRIMER: guide to software and statistical methods. PRIMER-E, Plymouth, UK.
Andrades, J., H. Torres, J. López, and M. Rojas. 2019. Análisis espacio temporal de la homogeneidad de estaciones de precipitación en una zona árida y semiárida del centro occidente de Venezuela. Revista Ciencia e Ingeniería 40 (2): 185–194.
Anton-Pardo, M., and X. Armengol. 2012. Effects of salinity and water temporality on zooplankton community in coastal Mediterranean ponds. Estuarine Coastal and Shelf Science 114: 93–99.
APHA-AWWA-WPCF. 1995. Standard methods for the examination of water and wastewater, 18th ed. American Water Works Association, Water Environmental Federation: American Public Health Association.
Araya-Vergara, J.F. 1981. El concepto de “delta en ría” y su significado en la evolución litoral (ejemplo en Chile central). Informaciones Geográficas Chile 28: 71–102.
Araya-Vergara, J.F. 1982. Análisis de la localización de los procesos y formas predominantes de la línea litoral de Chile: Observación preliminar. Informaciones Geográficas Chile 29: 35–55.
Arce, D.A. 2014. Caracterización en terreno y por modelación numérica de la hidrodinámica del estuario del río Maule, Chile. M.S. Thesis, Universidad de Chile.
Arriagada, L., O. Rojas, J.L. Arumí, J. Munizaga, C. Rojas, L. Farias, and C. Vega. 2019. A new method to evaluate the vulnerability of watersheds facing several stressors: A case study in Mediterranean Chile. Science of the Total Environment 651: 1517–1533.
Arroyo, M.T.K., V. Robles, Í. Tamburrino, J. Martínez-Harms, R.D. Garreaud, P. Jara-Arancio, P. Pliscoff, A. Copier, J. Arenas, J. Keymer, and K. Castro. 2020. Extreme drought affects visitation and seed set in a plant species in the central Chilean Andes heavily dependent on hummingbird pollination. Plants 9: 1–28. https://doi.org/10.3390/plants9111553.
Barlow, P. 2003. Saltwater intrusion from the Delaware River during drought-implications for the effect of sea-level rise on coastal aquifers. In Ground water in freshwater–saltwater environments of the Atlantic coast. 46–48.US Geological Survey, Reston, Virginia.
Belda, M., E. Holtanová, T. Halenka, and J. Kalvová. 2014. Climate classification revisited: From Köppen to Trewartha. Climate Research 59: 1–13. https://doi.org/10.3354/cr01204.
Bertrán, J., and O. Parra. 2001. Macrofauna del curso inferior y estuario del río Biobío (Chile): Cambios asociados a variabilidad estacional del caudal hídrico. Revista Chilena De Historia Natural 74: 331–340.
Bertrán, C., P. Fierro, E. Encalada, F. Peña-Cortés, J. Tapia, E. Hauenstein, and L. Vargas-Chacoff. 2016. Macrobenthos of the coastal Budi Lagoon, southern Chile: Changes associated with seasonal environmental variation. Brazilian Journal of Oceanography 64 (3): 239–248.
Bonada, N., N. Prat, V.H. Resh, and B. Statzner. 2006. Developments in aquatic insect biomonitoring: A comparative analysis of recent approaches. Annual Review of Entomology 51: 495–523. https://doi.org/10.1146/annurev.ento.51.110104.151124.
Borcard, D., F. Gillet, and P. Legendre. 2011. Numerical ecology with R. Springer, New York. https://doi.org/10.1007/978-1-4419-7976-6.
Bozkurt, D., M. Rojas, J.P. Boisier, and J. Valdivieso. 2018. Projected hydroclimate changes over Andean basins in central Chile from downscaled CMIP5 models under the low and high emission scenarios. Climatic Change 150 (3): 131–147.
Bunn, S.E., and A.H. Arthington. 2002. Basic principles and ecological consequences of altered flow regimes for aquatic biodiversity. Environmental Management 30 (4): 492–507.
Cade-Idepe. Consultores en Ingeniería. 2004a. Diagnóstico y clasificación de los cursos de agua según objetivos de calidad. Cuenca del río Maule. Dirección General de Aguas. Ministerio de Obras Públicas. Gobierno de Chile.
Cade-Idepe. Consultores en Ingeniería. 2004b. Diagnóstico y clasificación de los cursos de agua según objetivos de calidad. Cuenca del río Mataquito. Dirección General de Aguas. Ministerio de Obras Públicas. Gobierno de Chile.
Cade-Idepe. Consultores en Ingeniería. 2004c. Diagnóstico y clasificación de los cursos de agua según objetivos de calidad. Cuenca del río Aconcagua. Dirección General de Aguas. Ministerio de Obras Públicas. Gobierno de Chile.
Cade-Idepe. Consultores en Ingeniería. 2004d. Diagnóstico y clasificación de los cursos de agua según objetivos de calidad. Cuenca del río Maipo. Dirección General de Aguas. Ministerio de Obras Públicas. Gobierno de Chile.
Cade-Idepe. Consultores en Ingeniería. 2005. Diagnóstico y clasificación de los cursos de agua y cuerpos de agua según objetivos de calidad. Dirección General de Aguas. Ministerio de Obras Públicas. Gobierno de Chile.
Caruso, B.S. 2002. Temporal and spatial patterns of extreme low flows and effects on stream ecosystems in Otago, New Zealand. Journal of Hydrology 257 (1–4): 115–133.
Cazelles, B., M. Chavez, D. Berteaux, F. Ménard, J.O. Vik, S. Jenouvrier, and N.C. Stenseth. 2008. Wavelet analysis of ecological time series. Oecologia 156 (2): 287–304.
Cerda, M., and J.C. Castilla. 2001. Diversidad y biomasa de macro-invertebrados intermareales del tunicado Pyura praeputialis (Heller, 1878) en la bahía de Antofagasta. Chile. Revista Chilena De Historia Natural 74: 841–853.
Clarke, K.R. 1993. Non-parametric multivariate analyses of changes in community structure. Australian Journal of Ecology 18: 117–143.
Clarke, K.R., and R.N. Gorley. 2015. PRIMER v7: user manual/tutorial. PRIMER-E. Plymouth, UK.
CR2. 2015. Informe a la Nación: La megasequía 2010–2015. Una lección para el futuro. Centro de Ciencia del Clima y Resiliencia (CR)2. Universidad de Chile, Santiago, Chile.
CTHA. 2014. Informe Final: Estudio de análisis temporal y espacial de la producción de agua superficial en subcuencas con aprovechamiento humano, en las regiones del Maule, Biobío y La Araucanía y su relación con el uso actual del suelo. Centro Tecnológico de Hidrología Ambiental. Universidad de Talca.
Cuevas, J.G., J. Huertas, C. Leiva, L. Paulino, J. Dörner, and J.L. Arumi. 2014. Nutrient retention in a microcatchment with low levels of anthropogenic pollution. Bosque 35: 76–88.
D’Adamo, N., and R. Lukatelich. 1985. Water quality of the Murray River estuary: Summary Report Waterways Commission, 6. Perth, Western Australia: Centre for Water Research, The University of Western Australia, Report.
Debels, P., R. Figueroa, R. Urrutia, R. Barra, and X. Niell. 2005. Evaluation of water quality in the Chillán River (central Chile) using physicochemical parameters and modified water quality index. Environmental Monitoring and Assessment 110: 301–322.
Delpla, I., A.V. Jung, E. Baures, M. Clement, and O. Thomas. 2009. Impacts of climate change on surface water quality in relation to drinking water production. Environment International 35 (8): 1225–1233. https://doi.org/10.1016/j.envint.2009.07.001.
Di Castri, F. 1981. Mediterranean type shrublands of the world. In Ecosystems of the world, 11, Mediterranean type shrublands, ed. F. di Castri, D.W. Goodball, and R.L. Specht, 1–43. Amsterdam: Elsevier.
Dirección General de Aguas. 2016. Atlas del Agua. Gobierno de Chile, Ministerio de Obras Públicas: Santiago, Chile.
Elgueta, A.V. 2020. Efecto de las alteraciones del régimen de caudal en la estructura de ensambles de macroinvertebrados y peces: un enfoque taxonómico y funcional. Doctoral thesis, Universidad de Concepción.
FAO. 2010. Gestión del riesgo de sequía y otros eventos climáticos extremos en Chile: estudio piloto sobre la vulnerabilidad y la gestión local del riesgo. Santiago, Chile: Oficina de Estudios y Políticas Agrarias (ODEPA)/Food and Agriculture Organization (FAO).
Fauchald, K. 1977. The polychaeta worms. Definitions and keys to the orders, families and genera. Science Series 28: 188.
Fierro, P., C. Bertrán, D. Martínez, C. Valdovinos, and L. Vargas-Chacoff. 2014. Ontogenetic and temporal changes in the diet of the Chilean silverside Odontesthes regia (Atherinidae) in southern Chile. Cahiers De Biologie Marine 55 (3): 323–332.
Fierro, P., I. Arismendi, R. Hughes, C. Valdovinos, and A. Jara-Flores. 2018. A benthic macroinvertebrate multimetric index for Chilean Mediterranean streams. Ecological Indicators 91: 18–23.
Fierro, P., C. Valdovinos, I. Arismendi, G. Díaz, M. Ruiz de Gamboa, and L. Arriagada. 2019. Assessment of anthropogenic threats to Chilean Mediterranean freshwater ecosystems: Literature review and expert opinions. Environmental Impact Assessment Review 77: 114–121.
Fierro, P., C. Valdovinos, C. Lara, and G.S. Saldías. 2021. Influence of intensive agriculture on benthic macroinvertebrate assemblages and water quality in the Aconcagua river basin (central Chile). Water 13 (4): 492.
Figueroa, R., V. Ruiz, X. Niell, E. Araya, and A. Palma. 2006. Invertebrate colonization patterns in a Mediterranean Chilean stream. Hydrobiologia 429 (1): 151–156.
Figueroa, R., A. Palma, V. Ruiz, and X. Niell. 2007. Comparative analysis of biotic indexes used to evaluate water quality in a Mediterranean river of Chile: Chillán River, VIII Region. Revista Chilena De Historia Natural 80 (2): 225–242.
Figueroa, R., V.H. Ruiz, P. Berríos, A. Palma, P. Villegas, and A. Andreu-Soler. 2010. Trophic ecology of native and introduced fish species from the Chillán River. South-Central Chile. Journal of Applied Ichthyology 26 (1): 78–83.
Figueroa, R., N. Bonada, M. Guevara, P. Pedreros, F. Correa-Araneda, M.E. Díaz, and V.H. Ruiz. 2013. Freshwater biodiversity and conservation in mediterranean climate streams of Chile. Hydrobiologia 719: 269–289.
Forcelli. D. 2000. Moluscos Magallánicos, Guía de los moluscos de la Patagonia y del sur de Chile, ed. Vázquez Mazzini. Buenos Aires.
Garreaud, R.D., C. Alvarez-Garreton, J. Barichivich, J.P. Boisier, D. Christie, M. Galleguillos, C. LeQuesne, J. McPhee, and M. Zambrano-Bigiarini. 2017. The 2010–2015 megadrought in central Chile: Impacts on regional hydroclimate and vegetation. Hydrology and Earth System Science 21: 6307–6327.
Garreaud, R.D., J.P. Boisier, R. Rondanelli, A. Montecinos, H.H. Sepúlveda, and D. Veloso-Aguila. 2020. The Central Chile Mega Drought (2010–2018): A climate dynamics perspective. International Journal of Climatology 40 (1): 421–439.
González, M.E., S. Gómez-González, A. Lara, R. Garreaud, and I. Díaz-Hormazábal. 2018. The 2010–2015 Megadrought and its influence on the fire regime in central and south-central Chile. Ecosphere 9: 1–17. https://doi.org/10.1002/ecs2.2300.
Habit, E. 1998. Análisis de la dieta de Percilia gillissi en ambientes de río y canales de riego (cuenca del río Itata, Chile). Theoria 7: 33–46.
Habit, E., M. Belk, P. Victoriano, and E. Jaque. 2007. Spatio-temporal distribution patterns and conservation of fish assemblages in a Chilean coastal river. Biodiversity Conservation 16 (11): 3179–3191.
Hall, N., J. Testa, M. Li, and H. Paerl. 2023. Assessing drivers of estuarine pH: A comparative analysis of the continental USA’s two largest estuaries. Limnology and Oceanography. https://doi.org/10.1002/lno.12418.
Hallett, C.S., A.J. Hobday, J.R. Tweedley, P.A. Thompson, K. McMahon, and F.J. Valesini. 2018. Observed and predicted impacts of climate change on the estuaries of south-western Australia, a Mediterranean climate region. Regional Environmental Change 18: 1357–1373.
He, S., J. Soininen, K. Chen, and B. Wang. 2020. Environmental factors override dispersal-related factors in shaping diatom and macroinvertebrate communities within stream networks in China. Frontiers in Ecology and Evolution 8: 141. https://doi.org/10.3389/fevo.2020.00141.
Henríquez, C., N. Aspee, and J. Quense. 2016. Zonas de catástrofe por eventos hidrometeorológicos en Chile y aportes para un índice de riesgo climático. Revista De Geografía Norte Grande 63: 27–44.
Herbert, E.R., P. Boon, A.J. Burgin, S.C. Neubauer, R.B. Franklin, M. Ardón, K.N. Hopfensperger, L.P.M. Lamers, and P. Gell. 2015. A global perspective on wetland salinization: Ecological consequences of a growing threat to freshwater wetlands. Ecosphere 6 (10): 1–43.
Kannel, P.R., S. Lee, Y.S. Lee, S.R. Kanel, and S.P. Khan. 2007. Application of water quality indices and dissolved oxygen as indicators for river water classification and urban impact assessment. Environmental Monitoring and Assessment 132: 93–110.
Karr, J.R., and E.W. Chu. 1998. Restoring life in running waters. Washington, DC: Island Press.
Kendall, M.G. 1975. Rank correlation methods, 4th ed. London: Charles Griffin.
Kraal, P., E.D. Burton, and R.T. Bush. 2013. Iron monosulfate accumulation and pyrite formation in eutrophic estuarine sediments. Geochemical Et Cosmochimica Acta 122: 75–88.
Lancellotti, D., and J. Vásquez. 2000. Zoogeografía de macroinvertebrados bentónicos de la costa de Chile: Contribución para la conservación marina. Revista Chilena De Historia Natural 73: 99–129.
Li, S., R.T. Bush, R. Mao, L. Xiong, and C. Ye. 2017. Extreme drought causes distinct water acidification and eutrophication in the Lower Lakes (Lakes Alexandrina and Albert), Australia. Journal of Hydrology 544: 133–146.
Luebert, F., and P. Pliscoff. 2004. Clasificación de pisos de vegetación y análisis de representatividad ecológica de áreas propuestas para la protección en la ecorregión Valdiviana. Valdivia: Serie de Publicaciones WWF Chile.
Luebert, F., and P. Pliscoff. 2006. Sinopsis bioclimática y vegetacional de Chile. Santiago: Editorial Universitaria.
Maity, S.S., and R. Maity. 2022. Changing pattern of intensity–duration–frequency relationship of precipitation due to climate change. Water Resources Management 36: 5371–5399. https://doi.org/10.1007/s11269-022-03313-y.
Mann, H.B. 1945. Non-parametric tests against trend. Econometrica 13: 163–171.
Martínez-Megías, C., and A. Rico. 2022. Biodiversity impacts by multiple anthropogenic stressors in Mediterranean coastal wetlands. Science of the Total Environment 818: 151712. https://doi.org/10.1016/j.scitotenv.2021.151712.
Masotti, I., P. Aparicio-Rizzo, M.A. Yevenes, R. Garreaud, L. Belmar, and L. Farías. 2018. The influence of river discharge on nutrient export and phytoplankton biomass off the central Chile coast (33–37°S): Seasonal cycle and interannual variability. Frontiers in Marine Science 5: 423.
McAuliffe, T.F., R.J. Lukatelich, and N.A. Hill. 1993. The role of sediments in phosphorus cycling in the Swan River Estuary. In Swan River Trust—The Future, Workshop Proceedings. 75–87.
McKee, T.B., N.J., Doesken, and J. Kleist. 1993. The relationship of drought frequency and duration to time scales. In: Proceeding of the 8th Conference on Applied Climatology 17: 179–184.
Mereta, T.S., P. Boets, L. De Meester, and P.L.M. Goethals. 2013. Development of a multimetric index based on benthic macroinvertebrates for the assessment of natural wetlands in Southwest Ethiopia. Ecological Indicators 29: 510–521. https://doi.org/10.1016/j.ecolind.2013.01.026.
Miranda, A., A. Altamirano, L. Cayuela, A. Lara, and M. González. 2017. Native forest loss in the Chilean biodiversity hotspot: Revealing the evidence. Regional Environmental Change 17 (1): 285–297.
Mishra, A. 2010. Assessment of water quality using principal component analysis: A case study of the river Ganges. Journal of Water Chemistry and Technology 32: 227–234.
Morant, D., A. Picazo, C. Rochera, A.C. Santamans, J. Miralles-Lorenzo, A. Camacho-Santamans, C. Ibañez, M. Martínez-Eixarch, and A. Camacho. 2020. Carbon metabolic rates and GHG emissions in different wetland types of the Ebro Delta. PLoS ONE 15 (4): e0231713.
Mosley, L.M. 2015. Drought impacts on the water quality of freshwater systems; review and integration. Earth-Science Reviews 140: 203–214.
Muñoz, A.A., A. González-Reyes, A. Lara, D. Sauchyn, D. Christie, P. Puchi, R. Urrutia-Jalabert, I. Toledo-Guerrero, I. Aguilera-Betti, I. Mundo, P.R. Sheppard, D. Stahle, R. Villalba, P. Szejner, C. LeQuesne, and J. Vanstone. 2016. Streamflow variability in the Chilean Temperate-Mediterranean climate transition (35°S–42°S) during the last 400 years inferred from tree-ring records. Climate Dynamics 47 (12): 4051–4066.
Muñoz, A.A., K. Klock-Barría, C. Alvarez-Garreton, I. Aguilera-Betti, Á. González-Reyes, J.A. Lastra, R.O. Chávez, P. Barría, D. Christie, M. Rojas-Badilla, and C. LeQuesne. 2020. Water crisis in Petorca Basin, Chile: The combined effects of a mega-drought and water management. Water 12: 1–17. https://doi.org/10.3390/w12030648.
Newton, A., A.C. Brito, J.D. Icely, V. Derolez, I. Clara, S. Angus, G. Schernewski, M. Ignácio, A.I. Lillebo, A.I. Sousa, B. Béjaoui, C. Solidoro, M. Tosci, M. Cañedp-Argüelles, M. Yamamuro, S. Reizopoulou, HCh. Tseng, D. Canu, L. Roselli, M. Maanan, S. Cristina, A.C. Ruiz-Fernández, R.F. de Lima, B. Kjerfve, N. Rubio-Cisneros, A. Pérez-Ruzafa, C. Marcos, R. Pastres, F. Pranovi, M. Snoussi, J. Turpie, Y. Tuchkovenko, B. Dyack, J. Brookes, R. Povilanskas, and V. Khokhlov. 2018. Assessing, quantifying and valuing the ecosystem services of coastal lagoons. Journal for Nature Conservation 44: 50–65.
Niemeyer, H., and P. Cereceda. 1984. Geografía de Chile. Instituto Geográfico Militar. Santiago, Chile.
Novoa, V., R. Ahumada-Rudolph, O. Rojas, K. Sáez, F. de la Barrera, and J.L. Arumí. 2019. Sustainability assessment of the agricultural water footprint in the Cachapoal River basin, Chile. Ecological Indicators 98: 19–28.
Pacheco, F., O. Rojas, E. Hernández, and D. Caamaño. 2022. Effects on fluvial geomorphology and vegetation cover following hydroelectric power plant operation: A case study in the Maule River (Chile). Water 14 (11): 1673. https://doi.org/10.3390/w14111673.
Peña-Guerrero, M.D., A. Nauditt, C. Muñoz-Robles, L. Ribbe, and F. Meza. 2020. Drought impacts on water quality and potential implications for agricultural production in the Maipo River Basin. Central Chile. Hydrological Sciences Journal 65 (6): 1005–1021.
Perennou, C., E. Gaget, T. Galewski, I. Geijzendorffer, and A. Guelmami. 2018. Evolution of wetlands in Mediterranean region. Water Resources in the Mediterranean Region, 297–320. Elsevier Inc.
Pérez, C.A., M.D. DeGrandpre, N.A. Lagos, G.S. Saldías, E.K. Cascales, and C.A. Vargas. 2015. Influence of climate and land use in carbon biogeochemistry in lower reaches of rivers in central southern Chile: Implications for the carbonate system in river-influenced rocky shore environments. Journal of Geophysical Research: Biogeosciences 120 (4): 673–692.
Pliscoff, P. 2020. Análisis del estado actual de los ecosistemas terrestres, asociados a dos cuencas en Chile central: Maipo y Maule. Escenarios Hídricos 2030, Chile.
Poff, N.L., and J.K.H. Zimmerman. 2010. Ecological responses to altered flow regimes: A literature review to inform the science and management of environmental flows. Freshwater Biology 55: 194–205. https://doi.org/10.1111/j.1365-2427.2009.02272.x.
Retamal, M. 1981. Catalogo Ilustrado de los Crustáceos Decápodos de Chile. Gayana Zoología 44: 1–110.
Rozbaczylo, N. 1980. Clave para el Reconocimiento de Familias de Anélidos Poliquetos del Mar Chileno. Studies on Neotropical Fauna and Environment 15: 167–196.
Rubio-Álvarez, E., and J. McPhee. 2010. Patterns of spatial and temporal variability in streamflow records in south central Chile in the period 1952–2003. Water Resources Research 46 (W05514): 2010. https://doi.org/10.1029/2009WR00782.
Soria, M., M.E. Díaz, M. Beltrán, and R. Figueroa. 2019. Importancia de la conservación de los ríos tributarios de cabecera como fuente de biodiversidad: Río Lía (Santa Juana, región del Biobío, Chile). Ecosistemas: Revista Cietifica y Tecnica De Ecologia y Medio Ambiente 28 (3): 208–212.
Sylaios, G., E. Koutrakis, and A. Kallianiotis. 2006. Hydrographic variability, nutrient distribution and water mass dynamics in Strymonikos Gulf (Northern Greece). Continental Shelf Research 26 (2): 217–235.
Tallaksen, L.M., and H.A. J. Van Lanen. 2004. Hydrological drought. Processes and estimation methods for streamflow and groundwater. Developments in Water Science, 48. Elsevier Science B.V., 579 p.
Tampo, L., I. Kaboré, E.H. Alhassan, and A, Ouéda, L.M. Bawa, L, and G. Djaneye-Boundjo. 2021. Benthic macroinvertebrates as ecological indicators: Their sensitivity to the water quality and human disturbances in a tropical river. Frontiers in Water 3: 662765.
Torres-Ramírez, P., J. Ortiz, S. Figueroa, R. Jerez, and D. Arcos. 2017. Characterization of the coastal wetland Mataquito, Curicó province, Maule Region. In The ecology and natural history of Chilean saltmarshes, ed. J. M. Fariña, and A. Camaño. Springer International Publishing AG 2017. doi: https://doi.org/10.1007/978-3-319-63877-5.
Tully, K., K. Gedan, R. Epanchin-Niell, A. Strong, E.S. Bernhardt, T. BenDor, M. Mitchell, J. Kominoski, T.E. Jordan, S.C. Neubauer, and N.B. Weston. 2019. The invisible flood: The chemistry, ecology, and social implications of coastal saltwater intrusion. BioScience 69 (5): 368–378.
Tweedley, J.R., C.S. Hallett, R.M. Warwick, K.R. Clarke, and I.C. Potter. 2015. The hypoxia that developed in a microtidal estuary following an extreme storm produced dramatic changes in the benthos. Marine and Freshwater Research 67 (3): 327–341.
Urrutia, R., A. Lara, R. Villalba, D. Christie, C. LeQuesne, and A. Cuq. 2011. Multi-century tree ring reconstruction of annual streamflow for the Maule River watershed in South-Central Chile. Water Resources Research 47: W06527. https://doi.org/10.1029/2010WR009562.
Van Vliet, M.T.H., and J.J.G. Zwolsman. 2008. Impact of summer droughts on the water quality of the Meuse river. Journal of Hydrology 353 (1–2): 1–17. https://doi.org/10.1016/j.jhydrol.2008.01.001.
Vannote, R.L., G.W. Minshall, K.W. Cummins, J.R. Sedell, and C.E. Cushing. 1980. The river continuum concept. Canadian Journal of Fisheries and Aquatic Sciences 37: 130–137.
Vargas, C.A., R. Garreaud, R. Barra, F. Vásquez-Lavin, G.S. Saldías, and O. Parra. 2020. Environmental costs of water transfers. Nature Sustainability 3 (6): 408–409.
Watanabe, K., A. Kasai, E.S. Antonio, K. Suzuki, M. Ueno, and Y. Yamashita. 2014. Influence of salt-wedge intrusion on ecological processes at lower trophic levels in the Yura Estuary, Japan. Estuarine Coastal and Shelf Science 139: 67–77.
Welsh, D.T. 2003. It’s a dirty job but someone has to do it: The role of marine benthic macrofauna in organic matter turnover and nutrient recycling to the water column. Chemistry and Ecology 19 (5): 321–342.
Whitehead, P.G., R.L. Wilby, R.W. Battarbee, M. Kernan, and A.J. Wade. 2009. A review of the potential impacts of climate change on surface water quality. Hydrological Sciences Journal 54 (1): 101–123.
Zwolsman, J.J.G., and A.J. van Bokhoven. 2007. Impact of summer droughts on water quality of the Rhine River – a preview of climate change? Water Science and Technology 56 (4): 45–55. https://doi.org/10.2166/wst.2007.535.
Zuur, A., E. Ieno, and G. Smith. 2007. Analyzing ecological data. Series: Statistics for biology and health series. Springer Science, New York, USA.
Acknowledgements
We express gratitude to Robinson Carrasco and Carol Reyes for all their help in the field sampling and in the laboratory work and to Eduardo Hernández for their invaluable comments that improved this manuscript. P.T-R thanks the National Research and Development Agency (ANID) for funding his doctoral scholarship (21210745). J.L-M. received financial support from FONDAP INCAR 1522A0004 and FONDECYT 1221102. A.G. has been supported by ANID, Fomento a la Vinculación Internacional para Instituciones de Investigación Regionales, grant FOVI210005 and ANID–Millennium Science Initiative Program–Code ICN2019_015 (SECOS). The authors thank the INCA 210005 project at the UCSC for funding the open access publication of this article.
Funding
This research was supported by the Management of Environmental office, Arauco Company, who facilitated the use of the information presented in this study.
Author information
Authors and Affiliations
Contributions
Conceptualization: P.T-R., A.G., J.L-M. Conducting the research: P.T-R., L.B-E., S.F., R.J. Developing methods: P.T-R. Data analysis and interpretation: P.T-R., A.G., L.B-E., S.F., R.J. Preparation of figures and tables: P.T-R., A.G., L.B-E., S.F., R.J. Writing: P.T-R., A.G., L.B-E., S.F.
Corresponding author
Ethics declarations
Competing Interests
The authors declare the following financial interests/personal relationships which may be considered as potential competing interests: P.T-R., L.B-E., S.F., and R.J. report financial support by the Management of Environmental and Occupational Health office, Arauco Company.
Disclaimer
The contents of this paper reflect the views of the author(s) and not necessarily the views of the Universities. The authors are responsible for the facts and the accuracy of the data presented herein. The contents do not necessarily reflect the official views or policies of Arauco Company at the time of publication. This report does not constitute a standard, specification, or regulation.
Additional information
Communicated by Weimin Quan
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Torres-Ramírez, P., Bustos-Espinoza, L., Figueroa, S. et al. Influence of the Hydrological Variability on Water Quality and Benthic Macroinvertebrates in a Chilean Estuary During a Megadrought. Estuaries and Coasts 47, 724–742 (2024). https://doi.org/10.1007/s12237-023-01311-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12237-023-01311-w