Abstract
In estuarine and coastal systems, anthropogenic activities and directional changes in global air temperatures have led to increased water temperatures, as well as increased frequency and severity of episodic hypoxia. These alterations have had population-level effects on aquatic organisms, including changes in species-specific distributions. Because physiology is the transfer function connecting environmental conditions to individual behaviors and eventually to population-level effects, we used individual-based models (IBMs) that incorporate changes in metabolic scope as motivation for movement. Our objective was to investigate the effects of temperature and hypoxia on the distribution of Atlantic croaker and spot in Chesapeake Bay. We compared the predicted monthly fish distributions from the IBMs with apparent fish distributions recorded by the VIMS Juvenile Fish Trawl Survey during 1988–2014. IBMs failed to reproduce accurately the apparent spatial distributions of Atlantic croaker and spot. More specifically, compared with the trawl survey, IBMs predicted larger proportions of these two species would be captured in the lower regions of Chesapeake Bay and smaller proportions in the York and Rappahannock rivers. We postulate that, because similar thermal and oxygen conditions were occupied by fish in the IBMs and in the wild, temperature may not be the most important factor motivating the movement of Atlantic croaker and spot in Chesapeake Bay, and other spatial factors (e.g., prey availability) act in concert with temperature and hypoxia to determine the spatial distributions of Atlantic croaker and spot in Chesapeake Bay. Alternatively, surveys used to estimate species occurrences could be biased if gear vulnerability is affected by temperature or oxygen conditions (or both). Additional research is needed to clarify which condition is true or if a combination of these factors (and others) led to discrepancies between predicted and apparent fish distributions based on trawl survey data.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Directional changes in global air temperatures are increasing water temperatures and altering oxygen conditions, especially in estuarine and coastal systems (IPCC 2014; Laffoley and Baxter 2019). Urbanization and agricultural runoff are also contributing to eutrophication resulting in increases in the frequency and severity of hypoxic events (Diaz and Rosenberg 2008, 2011; Rabalais et al. 2010; Howarth et al. 2011; Breitburg et al. 2018). In fishes, increases in temperature and exposure to hypoxia negatively impact individuals through diminution of aerobic metabolic scope (defined below) (Schurmann and Steffensen 1997; Claireaux and Lagardère 1999; Lapointe et al. 2014; Marcek et al. 2019). Reductions in metabolic scope, in turn, result in alterations to biological processes such as reducing growth rates (Del Toro-Silva et al. 2008; Beuvard et al. 2022). Although a causal link between metabolic scope and fish distributions has not yet been demonstrated, environmental conditions (e.g., temperature and hypoxia) are known to influence the spatial distributions of estuarine and coastal fishes (Perry et al. 2005; Sabatés et al. 2006; Brady and Targett 2013; Buchheister et al. 2013). Because physiology is the transfer function that links environmental conditions to individual fish behaviors and thereby distributions (Pörtner and Farrell 2008; Denny and Helmuth 2009; Chown et al. 2010; Horodysky et al. 2015), one explanation for these shifts in spatial distributions of coastal fish populations is that these shifts reflect the limits of physiological systems to maintain homeostasis (Pörtner and Knust 2007; Hare et al. 2012; Deutsch et al. 2015; Kleypas 2015; Little et al. 2020). The potential link between individual physiology and population-level processes such as shifts in spatial distribution suggests that understanding the ability of physiological systems to maintain homeostasis in the face of directionally changing environmental conditions is critical to effective fisheries management (see McKenzie et al. 2016; Brosset et al. 2021). Variation in individual responses to environmental stimuli should, therefore, be incorporated into population models (Kearney 2006; Fabry et al. 2008; Horodysky et al. 2015; Riebesell and Gattuso 2015; Cooke et al. 2016; Koenigstein et al. 2016; Townhill et al. 2017). Although a variety of models can incorporate physiology into population-level processes (e.g., species distribution models (Gamliel et al. 2020), dynamic energy budget models (Kooijman 2010), bioenergetics models (Kitchell et al. 1977), and individual-based models, IBMs (Sibly et al. 2013)), only IBMs allow the incorporation of individual variation and stochasticity (Huston et al. 1988; Judson 1994; Grimm 1999). We therefore contend that thoroughly developed and tested IBMs are critical to improve our understanding of alterations in population processes and species’ distributions.
Although IBMs have been used to determine population-level responses to changes in environmental conditions (Humston et al. 2000, 2004; Goodwin et al. 2006; Fulford et al. 2011, 2014, 2016; Rose et al. 2013a, 2018a), few studies have simultaneously gathered data on environmental conditions and individual fish movements at high spatial and temporal resolutions (Childs et al. 2008; Fabrizio et al. 2013, 2014). IBMs designed to examine the population dynamics of fishes, therefore, typically employ movement algorithms that are based on the environmental conditions observed where fish are present (Humston et al. 2000) or on bioenergetics models designed to predict fish movements based on conditions that maximize growth (Humston et al. 2004; Rose et al. 2013a, b, 2018a, b). These approaches do not, however, capture the direct effects of environmental conditions on aerobic metabolic scope (defined below). They may, therefore, direct fish to environmental conditions that maximize somatic growth rather than conditions that may maximize other equally critical aerobic processes such as reproduction, maximum sustainable swimming speeds, and rates of recovery from exhaustive exercise.
Environmental conditions can be integrated into population-level processes through the concept of aerobic metabolic scope (hereafter “metabolic scope”). Metabolic scope is the difference between the maximum and the minimum (hereafter “standard”) metabolic rates needed to maintain homeostasis (Fry 1947, 1971) and has been purported to define the state space within which all aerobic activities must occur (Fry 1947, 1971; Horodysky et al. 2015; Claireaux and Chabot 2016). A debate surrounds the broad applicability of metabolic scope as a unifying principle for understanding the relationship between the physiology of ectotherms relative to their environment (Pörtner et al. 2017; Jutfelt et al. 2018). But we contend, as do others (e.g., Claireaux and Lefrançois 2007), that metabolic scope is useful for gauging habitat suitability because it is a measure of the potential energy available to an individual.
Metabolic scope can be reduced due to a decrease in maximum metabolic rate (e.g., through exposure to low dissolved oxygen concentration, hereafter “DO”) or an increase in standard metabolic rate (e.g., through exposure to elevated temperatures; (Fry 1947, 1971; Neill et al. 1994; Pörtner et al. 2010; Horodysky et al. 2015), or both. Decreases in metabolic scope resulting from exposure to suboptimal environmental conditions reduce individual growth and reproductive potential and thus affect population productivity. We argue that this is a sufficiently strong selective pressure for species to evolve the ability to minimize these sublethal effects. This is accomplished by evolution of the ability to avoid areas that result in reductions in metabolic scope (Kelsch and Neill 1990; Neill et al. 1994; Claireaux and Lefrançois 2007; Claireaux and Chabot 2016). Although models that examine the effects of environmental conditions on metabolic scope and the subsequent impacts on the spatial distributions of populations have been developed (Cucco et al. 2012; Marras et al. 2015), there are few examples of these types of modeling efforts, and, to our knowledge, population-level responses to environmental conditions have not been investigated for temperate fishes using IBMs.
Our goal was to compare the performance of IBMs which incorporate environmentally driven reductions in metabolic scope with IBMs that incorporate only random movement (i.e., models where movement is not influenced changes by metabolic scope). To ensure a comprehensive assessment, we implemented three movement submodels:
-
1.
Random walk (i.e., fish movements are completely random)
-
2.
Kinesis (i.e., fish movements are reactionary to temperature and DO but undirected)
-
3.
Restricted-area search ((RAS); i.e., fish movements are reactionary to temperature and DO and are directed)
We assessed each submodels’ performance by how well each reproduced apparent fish distributions.
This approach allowed us to determine if maximizing metabolic scope is a driver of fish distribution while simultaneously accounting for potential differences in results based on model assumptions. Because it is uncoupled from the influence of temperature and hypoxia, the random walk submodel allowed us to evaluate the effectiveness of incorporating physiological responses into the kinesis and restricted-area search submodels. Watkins and Rose (2013, 2014, 2017) provide in-depth review of these and other movement submodels.
We chose adult Atlantic croaker (Micropogonias undulatus) and spot (Leiostomus xanthurus) as our study species because these species are common in Chesapeake Bay and the effects of temperature on metabolic rates and metabolic scope are documented (Horodysky et al. 2011; Marcek et al. 2019). Specifically, Marcek et al. (2019) examined the relationship between metabolic scope and temperature for both species. Although they did not identify an optimal temperature range at which the metabolic scope for either Atlantic croaker or spot was maximized, they were able to determine the shape of the relationship between temperature and metabolic scope for both species over the range of temperatures common in Chesapeake Bay. Furthermore, Marcek et al. (2019) identified dissolved oxygen concentrations at which both Atlantic croaker and spot could no longer sustain standard metabolic rate aerobically (hereafter “Ccrit”), and that would likely elicit an avoidance response.
In addition to the physiological information available for Atlantic croaker and spot, their relative abundance and spatial distributions within Chesapeake Bay have been monitored monthly since 1988 by the Virginia Institute of Marine Science (VIMS) Juvenile Fish Trawl Survey (hereafter “trawl survey”). The survey includes real-time measurements of temperature and DO which allowed us to validate our model results for a period of increasing temperatures and increasing frequency and severity of hypoxic conditions (Hagy et al. 2004; Kemp et al. 2005). Atlantic croaker and spot are demersal species that typically inhabit Chesapeake Bay from May to October and, therefore, are likely exposed to hypoxia during summer. Atlantic croaker and spot depart Chesapeake Bay during fall to spawn on the continental shelf (Haven 1959), although some Atlantic croaker may spawn in the lower Chesapeake Bay (Barbieri et al. 1994).
Because the kinesis and RAS submodels incorporate the physiological abilities and tolerances of individual fish, we hypothesize that compared with the random walk submodel, the kinesis and RAS submodels will result in simulated fish inhabiting areas where metabolic scope is maximal (i.e., “optimal habitat”) and thus should show better agreement with the apparent distributions of Atlantic croaker and spot (derived from the trawl survey data) than the random walk submodel. We also assessed the efficacy of using metabolic scope to predict fish distributions in dynamic environments by comparing (1) environmental conditions where fish were captured by the trawl survey to those occupied by simulated individuals and (2) the apparent and IBM-predicted spatial distributions of Atlantic croaker and spot in Chesapeake Bay.
Methods
Study Area
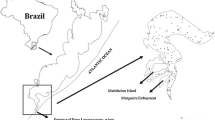
Chesapeake Bay is the largest estuary in North America, encompassing 11,500 km2 with a 164,200 km2 watershed. Although its central channel is relatively deep (20–30 m), the mean depth is only 6.5 m (Kemp et al. 2005). Large inputs of nutrients from urban and agricultural areas throughout its extensive watershed and the Chesapeake Bay’s shallow mean depth result in a highly productive system that provides foraging or nursery habitat (or both) for > 350 species of fish, many of which are resident only during spring and summer (Murdy and Musick 2013). In recent decades, however, warming of Chesapeake Bay and changes in wind patterns have led to prolonged periods of reduced turnover between warm, oxygen-rich surface waters, and cooler bottom waters (i.e., water column stratification) (Scully 2016; Du et al. 2018). This and the simultaneously increasing eutrophication of bay waters have led to increases in the magnitude, duration, and spatial extent of seasonal hypoxic events throughout Chesapeake Bay (Officer et al. 1984; Cooper and Brush 1991; Hagy et al. 2004; Kemp et al. 2005). Because fisheries and environmental data from the VIMS trawl survey are limited to the mainstem of Chesapeake Bay and three of its major subestuaries (the James, York, and Rappahannock rivers) (Fig. 1), our study focused on these areas. Because Atlantic croaker and spot are both demersal species that feed primarily on the benthos (Pihl et al. 1992; Nye et al. 2011), we used water quality measurements (temperature and DO) for areas ≈ 1 m above the substrate in our models.
Individual-Based Model (IBM) of Fish Distribution
A complete, detailed model description, following the overview, design concepts, details (ODD) protocol (Grimm et al. 2006, 2010, 2020), is provided in the Supplementary Material but is briefly described here. We implemented the models in Netlogo 5.3.1 (Railsback and Grimm 2011).
Our purpose was to determine if physiologically mediated movements can be used to predict accurately fish distributions in dynamic environments such as Chesapeake Bay. Specifically, we evaluated if IBMs that incorporate the physiological effects of temperature and DO into movement submodels can better reproduce the apparent spatial distributions of fish relative to a random movement submodel. As such, we compared predictions from IBMs that incorporated the physiological effects of temperature and DO into movement submodels with IBMs that used only random movement (i.e., ignoring the physiological effects of temperature and DO). To determine if the model outputs were realistic, we compare the predicted monthly environmental conditions and locations occupied by simulated fish with monthly observations of these conditions from the trawl survey.
Our IBM included two types of entities: fish (Atlantic croaker or spot) and 1 km2 grid cells. The state variables characterizing these entities are provided in Table 1. Temporal extent and resolution were as follows: a time step represented 1 h, and the model year was defined as May 1 to September 30. Simulations ran from 1988 to 2014. The spatial extent was 162 × 152 grid cells representing the Virginia waters of Chesapeake Bay and its major tributaries.
The key processes of the IBM are (1) simulation of fish movement through random walk, kinesis, or RAS submodels (see Supplemental text and Tables S1-S3 for details), (2) enumerating the cumulative number of time steps during which an individual fish occupied DO < Ccrit, and (3) updating environmental conditions. The first two processes occur every time step (i.e., every hour) for all simulated fish, whereas temperature and DO values in each grid cell were updated every 24 time steps (i.e., daily).
An important design concept of the model is the way in which individuals respond to ambient conditions; the three movement submodels define such responses. In particular, the kinesis and RAS submodels direct subsequent movements in either a reactionary (kinesis) or directed (RAS) manner based on experimentally derived physiological relationships (Marcek et al. 2019). To facilitate comparisons between model predictions and observations, each individual’s position (UTM coordinates), the cumulative number of time steps during which the individual occupied DO < Ccrit, and the ambient DO and temperature conditions were recorded on the first day of each month. We used these data to compare the monthly distributions of individuals as well as the environmental conditions to which they were exposed from each of the three movement submodels to trawl survey observations to determine if physiologically mediated movement predicted the apparent distributions of individuals better than random movement.
Model Performance
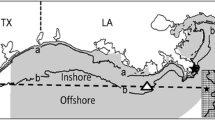
To examine how movement submodels determined the environmental conditions experienced by simulated fish, we examined the bottom temperature and bottom DO in areas occupied by individuals at the time of sampling and the proportion of observations in which individuals occupied DO concentrations < Ccrit. Additionally, we divided the study area into nine regions (Fig. 2) to facilitate comparison of the predicted distributions of individuals from each of the movement submodels. All analyses (described below) were conducted using R version 3.5.1 (R Core Team 2020) or the MIXED procedure in SAS version 9.4 (SAS Institute, Cary, NC).
The regions used to compare output from the simulations to observations from the VIMS Juvenile Fish Trawl Survey. The mainstem of Chesapeake Bay is separated into three regions (B1, B2, and B3), whereas the rivers (James, York, and Rappahannock) are each separated into 2 regions (JA1, JA2, YK1, YK2, RA1, and RA2)
Submodel Comparisons
We used generalized linear models to determine if the mean temperature and DO conditions experienced by simulated Atlantic croaker and spot varied between movement submodels. We used a repeated measures ANOVA, with individual fish as the subject, to account for correlations among observations because there were multiple observations per individual (monthly from June to September). We used separate models for each species where
In these models, Yij represents the mean environmental conditions (either temperature or DO, which we assumed to be normally distributed and modeled separately) experienced by individual i in submodel j (where “submodel” refers to the random walk, kinesis, or RAS submodels); εij is the random, unexplained error associated with individual i in each of the submodels. We investigated multiple covariance structures (including compound symmetry, first-order autoregressive, banded Toeplitz, and unstructured) which were used to account for repeated temperature and DO observations for each individual and determined the covariance structure that best described the correlations and variances using Akaike’s information criterion (AIC) (Burnham and Anderson 2002; Logan 2010).
For each submodel, we calculated the number of time steps spent by fish in areas where DO < Ccrit. This was expressed as the proportion of observations for which a simulated individual was predicted to occupy such areas. These data occurred in the interval [0,1] and were zero-inflated because between 54 and 71% of individuals (across all movement submodels and species) did not occupy areas with DO < Ccrit. As a result, we used zero-inflated beta regressions (Ospina and Ferrari 2010, 2012) to investigate the effect of each movement submodel on the proportion of observations in areas where DO was less than Ccrit. Beta regressions describing the proportion of observations in areas where DO was less than Ccrit followed the form
In these models, the responses are the logit of the probability that the proportion of observations in areas with DO < Ccrit is 0 (α), the logit of the proportion of observations in areas with DO < Ccrit (μ), and the log of the precision parameter (ϕ) for individual i in submodel j. Submodel and εij are defined as above. We implemented zero-inflated beta regressions for Atlantic croaker and spot in the gamlss package in R (Stasinopoulos and Rigby 2007).
Validation of Individual-Based Models
To validate the IBM output, we assessed the goodness-of-fit of mean monthly temperatures and DO conditions occupied by simulated fish from each submodel relative to observations from the VIMS trawl survey (Smith and Rose 1995). Specifically, we regressed the mean monthly environmental conditions occupied by simulated fish against the mean monthly conditions in which fish were captured by the VIMS trawl survey such that
In these regressions, Yij is the mean temperature or DO occupied by simulated fish from the random walk, kinesis, or RAS submodel in month i and year j; μ is the overall mean temperature at which fish were captured by the trawl survey; Xij is the mean temperature or DO at which fish were captured by the trawl survey in month i and year j; and εij is the random, unexplained error associated with month i and year j. We assumed both temperature and DO were normally distributed. We used two statistics to assess the relationship between environmental conditions where Atlantic croaker and spot were captured by the VIMS trawl survey and those conditions our models predicted they would occupy: (1) the coefficient of determination (R2) and (2) the root mean squared error (RMSE). Movement submodels with high R2 and low RMSE provided better agreement between predicted and observed environmental conditions.
In addition to assessing the goodness-of-fit, we compared the mean and 95% confidence intervals for temperature and DO where Atlantic croaker and spot were captured by the VIMS trawl survey to mean and 95% confidence intervals from model predictions. We considered the mean environmental conditions from the IBM output and trawl survey observations to be different if the 95% confidence intervals did not overlap. We note that these are imperfect comparisons due to the repeated nature of the observations for each simulated individual and, therefore, took steps to minimize the effect of temporal correlations among observations for simulated fish. First, each observation from simulated individuals was separated by 720 time steps from the subsequent observation thereby reducing correlations among consecutive (simulated) observations. Second, we accounted for any remaining correlation using a repeated measures framework to model the simulated data (see the “Submodel Comparisons” section). These precautions provided us with observations from which reasonable comparisons could be made.
In an attempt to further validate our IBM results, we also compared the apparent spatial distributions of fish from the trawl survey across all years to those predicted by our submodels. We calculated the mean monthly proportion of individuals predicted to occupy each region of Chesapeake Bay (Fig. 2) for each movement submodel by dividing the number of individuals predicted to occupy a given region by the total number of individuals predicted to occur within the model domain. We similarly divided the number of fish captured by the trawl survey in each region by the total number of individuals captured each month. We qualitatively compared the apparent proportion of individuals occupying each region (based on trawl survey data) to the proportion of individuals predicted to occupy each region by the IBM submodels. We used these comparisons to provide a qualitative measure of the accuracy with which the IBM submodels recreated the apparent spatial distributions of Atlantic croaker and spot when the relationship between environmental conditions and metabolic scope was used as motivation for movement or when fish moved randomly.
Results
Submodel Comparisons
The mean temperature and DO conditions predicted to be occupied by Atlantic croaker and spot differed significantly among movement submodels (temperature: FAtlantic croaker = 70603.6, P < 0.01; Fspot = 96991.1, P < 0.01; DO: FAtlantic croaker = 372.7, P = 0.04; Fspot = 3146.8, P < 0.01). The random walk submodel consistently predicted that fish occupied lower mean temperature and DO conditions relative to those in the kinesis and RAS submodels, regardless of species (Fig. 3). These results indicate that incorporation of physiologically driven movements allowed fish to find and occupy temperatures that increased metabolic scope and to avoid areas of low DO. Furthermore, the RAS submodel consistently predicted that fish occupied higher mean temperature and DO conditions than those in the kinesis submodel (Fig. 3). Simulated fish in the RAS submodel occupied areas that maximized metabolic scope, relative to those in the kinesis or random walk submodels.
The model-estimated mean temperatures (top panel) and dissolved oxygen concentrations (bottom panel) occupied by simulated Atlantic croaker (black circles) and spot (gray squares) in the random walk (RW), kinesis, and restricted-area search (RAS) submodels. Error bars represent the 95% confidence intervals and letters represent statistically significant differences among submodels within a species
Although the predicted mean temperature and DO of habitats occupied by fish in each movement submodel differed statistically, the absolute differences were small (Atlantic croaker ≤ 1.2 °C, spot ≤ 1.3 °C; Atlantic croaker ≤ 0.1 mg DO L−1, spot ≤ 0.2 mg DO L−1), and we consider them not to be biologically meaningful especially when compared with the range of available conditions (12.6–35.0 °C and 0.0–12.6 mg DO L−1). This suggests that submodels incorporating metabolic scope to inform movements resulted in fish occupying habitats that were only marginally different from those predicted to be occupied by the random walk submodel.
Regardless of movement submodel, the zero-inflated beta regressions indicated that less than half of the fish (40–45% for Atlantic croaker and 29–46% for spot) entered areas where DO was less than their Ccrit (Fig. 4). Both the kinesis (tAtlantic croaker = 12.3, P < 0.01; tspot = 28.0, P < 0.01) and RAS (tAtlantic croaker = 33.5, P < 0.01; tspot = 124.0, P < 0.01) submodels predicted significantly greater avoidance of low DO by both species than the random walk submodel. It is likely that the directed nature of movement in the RAS submodel, where individuals move to preferred conditions (i.e., highest available temperatures < 32 °C and DO > Ccrit; see Supplement for details), resulted in the RAS submodel predicting a greater proportion of individuals avoiding low DO than the kinesis and random walk submodels (Fig. 4). The RAS submodel, however, also predicted that Atlantic croaker and spot would reside in low DO areas for longer periods than did the random walk submodel (tAtlantic croaker = 22.6, P < 0.01; tspot = 27.2, P < 0.01). In contrast, the kinesis submodel predicted that fishes entering areas of low DO would spend less time in those areas than the random walk submodel (tAtlantic croaker = -84.2, P < 0.01; tspot = − 107.7, P < 0.01). On average, only 1.7% of observations were from areas where DO was < Ccrit in the kinesis submodel, whereas 2.7% and 3.1% of observations were from areas where DO was < Ccrit in the random walk and RAS submodels, respectively (Fig. 4). For simulated spot, the mean percentage of observations in areas where DO was < Ccrit was 1.6% for the kinesis submodel, 2.9% for the random walk submodel, and 3.5% for the RAS submodel (Fig. 4). These results indicate that the kinesis submodel (reactionary movement) better predicted that simulated individuals encountering hypoxic conditions moved away from this negative stimulus. Additionally, because fishes spent less time in hypoxic areas in the random walk submodel relative to the RAS submodel, we conclude that the gradient response we used in the RAS submodel resulted in fish becoming trapped in areas of local temperature optima despite suboptimal DO conditions. In summer, local temperature optima may be associated with low DO, leading to the prediction that simulated fish spent a greater proportion of time at suboptimal DO.
The model-estimated mean percentage of individuals experiencing dissolved oxygen conditions < Ccrit (top panel) and the percentage of observations where dissolve oxygen was < Ccrit (bottom panel) for individuals entering areas of low dissolved oxygen for the random walk (RW), kinesis, and restricted-area search (RAS) submodels. Simulated Atlantic croaker is represented with black circles and spot with gray squares. Error bars represent the 95% confidence intervals and letters represent statistically significant differences among submodels within a species
Validation of Individual-Based Models
The distributions of fish predicted by the three movement submodels were generally consistent with the apparent distributions of fish deduced from trawl survey catches. The kinesis submodel predicted that simulated fish are better able to avoid hypoxic areas than did the random walk or RAS submodels. We consider that this type of movement (reactionary to local conditions rather than directed or random) is likely the most realistic of the three used in our simulations. We, therefore, chose to validate the results of the kinesis submodel by comparing them to the apparent distributions of Atlantic croaker, and spot in Chesapeake Bay. Regressions indicated a strong positive relationship between temperatures predicted to be occupied by Atlantic croaker and those in which Atlantic croaker were captured; the RMSE was less than 1 ℃, regardless of month (Fig. 5). Likewise, the DO predicted to be occupied by simulated Atlantic croaker was also strongly and positively related to DO conditions where fish were captured; the RMSE was less than 1 mg L−1 regardless of month. Notably, however, the kinesis model predicted that fish would occupy lower DO conditions in the lower Rappahannock River in July and August than their apparent distributions (based on the VIMS trawl survey) (Fig. 6). Similar relationships were found between the environmental conditions predicted to be occupied by spot in the kinesis submodel and their apparent distributions (Figs. S5 and S6).
The mean monthly temperatures (June to September) occupied by simulated Atlantic croaker (kinesis) within each region (B1, B2, B3, JA1, JA2, YK1, YK2, RA1, and RA2) and year (1988–2014) regressed against the mean temperatures in which spot was captured by the trawl survey. Solid lines indicate the regression line, whereas dashed lines represent a 1:1 relationship. The left panel is for all months combined, whereas the right panels show the relationship for each month included in the simulations
The mean monthly DO concentrations (June to September) occupied by simulated Atlantic croaker (kinesis) within each region (B1, B2, B3, JA1, JA2, YK1, YK2, RA1, and RA2) and year (1988–2014) regressed against the mean DO concentrations in which spot were captured by the trawl survey. Solid lines indicate the regression line, whereas dashed lines represent a 1:1 relationship. The left panel is for all months combined, whereas the right panels show the relationship for each month included in the simulations
The mean temperature occupied by simulated fish in the kinesis model indicated that Atlantic croaker should be concentrated in areas of higher mean temperature than the apparent distributions (Fig. 7); this prediction was driven by the potential for increased metabolic scope at higher temperatures. In contrast, the kinesis model predicted spot should occupy areas of lower mean temperature (and therefore lower metabolic scope) than the apparent distributions suggest (Fig. 7). For both species, the kinesis model predicted that individuals would occupy areas of higher mean DO than the apparent distributions (Fig. 7). The kinesis model suggested that both species are less hypoxia tolerant than the trawl survey observations suggest. As with submodel comparisons, however, the absolute differences between the mean temperature and DO predicted to be occupied and those apparently occupied were small (temperature: Atlantic croaker = 1.3 °C, spot = 0.5 °C; DO: Atlantic croaker = 1.0 mg L−1, spot = 0.7 mg L−1), and we consider them to be not biologically relevant.
The model-estimated mean temperatures (top panel) and dissolved oxygen concentrations (bottom panel) occupied by simulated Atlantic croaker (black circles) and spot (gray squares) in the kinesis submodel compared with mean temperature and dissolved oxygen concentrations calculated from trawl survey observations. Error bars represent the 95% confidence intervals
Geographic regions of Chesapeake Bay predicted to be occupied and those apparently occupied by Atlantic croaker and spot differed markedly (Figs. 8 and 9, respectively). For Atlantic croaker, the kinesis submodel predicted a greater proportion of the population would occupy the lower bay (region B1) throughout the model year (June to September), the middle bay (region B2) from June to July, and the upper James River (JA2) in August and September. This submodel predicted a smaller proportion of the population would be found throughout the York and Rappahannock rivers (YK1, YK2, RA1, and RA2) in June, with differences generally decreasing throughout the model year. The exception is the lower York River (YK1) where predicted differences remained substantial throughout the model year (June to September) (Fig. 8). Differences in the predicted and apparent distributions of spot were similar to those for Atlantic croaker, except that the predicted proportion of the population occupying the Rappahannock River (RA1 and RA2) throughout the model year (June to September) was substantially lower than the apparent distributions (based on VIMS trawl survey catches) suggest (Fig. 9).
The mean proportion of Atlantic croaker found in each region of the study area for each month of the simulation. The proportion of individuals from the kinesis submodel is indicated with light gray and the VIMS Juvenile Fish Trawl Survey (Survey) in dark gray. Error bars represent the standard deviation
Discussion
Although previous studies (e.g., Cucco et al. 2012; Marras et al. 2015) examined metabolic scope as a physiological driver of the spatial distributions of fish, to our knowledge, this is the first study to incorporate metabolic scope into fish movement as a means of predicting distributions in an individual-based modeling (IBM) framework. Our IBMs failed to reproduce effectively the apparent spatial distributions of Atlantic croaker and spot regardless of submodel used to inform movement, even though environmental conditions predicted to be experienced were generally similar to those that appear to be occupied by fish based on trawl survey observations.
Movement submodels using either a reactionary (i.e., kinesis) or gradient response (i.e., RAS) approach predicted that fish would occupy habitats with relatively warm mean temperatures and would therefore realize increased metabolic scope. Compared with the outcome of submodels that impose random movements, our results show that the prediction of fish movements in a dynamic environment benefitted from the incorporation of physiological responses. The improvement, however, appears to be minimal and is likely not biologically significant. We suggest, however, that this minimal improvement is due to a lack of contrast in bottom-water temperatures in Chesapeake Bay during summer. Because the predicted movements of individuals in the kinesis and RAS submodels were primarily driven by temperature, low variation in bottom-water temperatures resulted in predicted movements with consistent directional persistence (i.e., kinesis), or that were largely undirected (i.e., when temperatures are similar throughout large portions of the environment, the RAS algorithm randomly selects the grid cell into which an individual moves to in a given time step).
Although the RAS submodel effectively reduced the predicted proportion of the population experiencing low DO across all model years, individuals that were predicted to experience low DO were simultaneously predicted to reside in these areas longer than those in the kinesis and random walk submodels. Our results are, therefore, consistent with the known sensitivity of submodels using a gradient response to complex environments (Humston et al. 2004; Watkins and Rose 2013, 2014, 2017). In this case, the gradient response to temperature predicted that fish would aggregate at local temperature optima, which in summer may also be associated with low DO.
In the Virginia waters of Chesapeake Bay, reductions in DO are most severe in July and August in the lower Rappahannock River and are mild and periodic in the lower York River (Tuckey and Fabrizio 2016). The proportion of individuals observed in the York and Rappahannock rivers by the VIMS trawl survey catches was greater than that predicted in our models, suggesting that Atlantic croaker and spot experience lower mean DO conditions than our models predict. The differences in mean temperature and DO concentrations predicted to be occupied, and those apparently occupied were, however, small.
Although differences in environmental conditions predicted to be occupied and those apparently occupied may not be substantive or biologically relevant, the spatial distributions of individuals were markedly different between predictions from our IBM models and apparent distributions based on the trawl survey. To account for these discrepancies, we identified three plausible hypotheses which may act alone or in concert. First, by incorporating the effects of temperature on metabolic scope with a hypoxia avoidance response as motivation for movement, we ignored other important drivers of movement. Thus, our movement models were likely overly simplistic and led to inaccuracies in predicted spatial distributions. Second, the trawl survey observations, against which we compared our predicted distributions, may have been biased by environmentally induced changes in catchability. For example, fish were less likely to avoid capture when occupying areas with suboptimal environmental conditions (e.g., low DO). Lastly, the spatial and temporal resolution of environmental conditions and movement was too coarse and did not allow fish to move in a biologically relevant manner resulting in discrepancies in spatial distribution between model predictions and trawl survey observations.
In support of the first hypothesis, the effect of temperature on the metabolic scope of fishes is well-documented (Claireaux and Lagardère 1999; Claireaux et al. 2000; Lefrançois and Claireaux 2003; Claireaux and Lefrançois 2007; Horodysky et al. 2011; Clark et al. 2013; Lapointe et al. 2014), and there is ample evidence that the spatial distributions of fishes and other marine ectotherms are affected by temperature (Murawski 1993; Perry et al. 2005; Pörtner and Knust 2007; Nye et al. 2009; Cucco et al. 2012; Deutsch et al. 2015; Kleypas 2015). Despite these established relationships, our simulations suggested that between May and September, temperature may not be the most important factor motivating the movement of Atlantic croaker and spot in Chesapeake Bay. As such, the predicted fish distributions reflected the generally optimal thermal conditions throughout the Virginia waters of Chesapeake Bay and its subestuaries. The relationship between temperature and metabolic scope of Atlantic croaker and spot and the subsequent discrepancies in predicted and apparent fish distributions also point to the lack of information regarding the interactive effects of temperature and DO on metabolic scope which, if included here, may have resulted in fish more frequently avoiding warmer conditions with lower DO if cooler, higher DO areas resulted in a relatively large metabolic scope. Although we incorporated hypoxia avoidance into the kinesis and RAS submodels, the ability to incorporate DO more explicitly into the movement algorithms may have resulted in predicted fish distributions better matching the apparent fish distributions based on trawl survey observations (see Cucco et al. 2012). Interestingly, in opposition to this hypothesis, Schlenger et al. (2022) found little interannual variation in optimal habitat volume for juvenile Atlantic croaker using 3-dimensional habitat model of Chesapeake Bay. This lack of variation in habitat volume was attributed to the broad physiological tolerances of this species to temperature, DO, and salinity and suggests that abiotic conditions may not be the most important factors in determining the spatial distributions of Atlantic croaker and spot in Chesapeake Bay during summer. Although we did not consider salinity in our models, it can affect species distributions in estuaries (Childs et al. 2008; Lauchlan and Nagelkerken 2020). Its effect on metabolic rate is, however, less clear (Ern et al. 2014) and requires additional research.
Although abiotic environmental gradients are important drivers of fish movements and distributions, we contend that prey availability is equally so, particularly when abiotic conditions allow a large metabolic scope. More specifically, we argue that prey availability influences the use of subestuaries (specifically the York and Rappahannock rivers) by Atlantic croaker and spot. Indices of benthic habitat quality in Virginia indicate that benthic habitats in the York and Rappahannock rivers are more stressed than those in the main stem of Chesapeake Bay and the James River (Diaz et al. 2003). In the York River, sediment instability is a major stressor of the benthic community (Dellapenna et al. 2001), maintaining early successional macrobenthic communities (Schaffner et al. 2001) which are often dominated by polychaetes in temperate areas (Lu and Wu 2000). Polychaetes are also one of the most common prey organisms found in the diets of Atlantic croaker (Nye et al. 2011) and spot (Pihl et al. 1992), which suggests that early successional benthic communities may provide an abundance of prey for these sciaenids. Unlike the York River, the primary stressor of the benthic community in the Rappahannock River is severe seasonal hypoxia (Llansó 1992) which is known to alter the behavior of benthic organisms, resulting in decreased burrowing depths and the extension of molluscan siphons into the water column. These behaviors increase the susceptibility of benthic organisms to predation by mobile predators such as Atlantic croaker and spot (Diaz and Rosenberg 1995; Taylor and Eggleston 2000; Seitz et al. 2003; Long and Seitz 2008). Furthermore, Atlantic croaker are known to congregate at the edges of hypoxic zones in the Gulf of Mexico (Craig and Crowder 2005; Craig 2012), presumably to forage. Both Atlantic croaker and spot exploit hypoxic areas in Chesapeake Bay to feed on stressed benthos (Pihl et al. 1991, 1992; Long and Seitz 2008). These trophic interactions may account for the greater proportion of Atlantic croaker and spot observed in the wild in the York and Rappahannock rivers compared with the proportions predicted by the IBM model.
In support of the second hypothesis, foraging forays or movements that result in exposure to hypoxic conditions may have negative physiological consequences such as reduced swimming speeds and visual impairments in fish (Robinson et al. 2013; McCormick and Levin 2017) that may lead to increases in catchability. It is plausible that low DO increases catchability as the detection and avoidance of trawl gear become more difficult in such conditions. For example, maximum metabolic rate decreases with decreasing DO (Norin and Clark 2016) and may lead to prioritization of some aerobic processes over others or reductions in critical swimming speeds (Ucrit; a measure of the prolonged swimming performance of fish (Brett 1964; Nelson 1989)) or both (Schurmann and Steffensen 1994; Kraskura and Nelson 2018). Indeed, reductions in swimming activity and Ucrit with hypoxic exposure have been demonstrated for multiple fishes including cod (Schurmann and Steffensen 1994), dogfish (Metcalfe and Butler 1984), sole (Dalla Via et al. 1998), and striped bass (Kraskura and Nelson 2018). Although such data for Atlantic croaker and spot are lacking, we contend that capture vulnerabilities of Atlantic croaker and spot are likely higher in areas of low DO. In laboratory experiments, decreases in Ucrit related to hypoxia exposure increased the catchability of zebrafish (Danio rerio) by trawl nets (Thambithurai et al. 2019) — but we know of no field-based data corroborating these results. We note that catchability of fish is determined by availability, vulnerability, and selectivity, and the experiment conducted by Thambithurai et al. (2019) demonstrated changes in vulnerability only. Several studies have, however, shown strong evidence for increases in availability of fishes through aggregation caused by habitat compression. Specifically, hypoxia reduces the amount of available habitat for fish, leading to their aggregation at the vertical or horizontal edges (or both) of hypoxic areas (Eby and Crowder 2002; Craig and Crowder 2005; Hazen et al. 2009; Ludsin et al. 2009; Zhang et al. 2009; Craig 2012). This results in an increased availability of these individuals to fishing gears. Indeed, several studies have demonstrated increases in catch rates associated with aggregations that form in response to the formation of hypoxic areas (Breitburg 2002; Langseth et al. 2014; Kraus et al. 2015; Stone et al. 2020). In Chesapeake Bay, catch rates of Atlantic croaker and spot are substantially reduced in low DO (< 4 mg/L) conditions (Buchheister et al. 2013) suggesting that if fish are present in such conditions, they are able to avoid capture. It is not clear if intermittent exposures to hypoxic conditions in Chesapeake Bay affect vulnerability of fish to capture, and it is possible that increased vulnerability results only from long-term chronic exposure. There is also the additional layer of complexity that gear vulnerability of populations may be changing (in concert with increased water temperatures and the increased frequency and severity of episodic hypoxia) due to fisheries-induced evolution (Hollins et al. 2018).
In support of the final hypothesis, fish in these IBMs moved on an hourly time step and environmental conditions changed daily (i.e., every 24 time steps in the models). In addition, grid cells were 1 km2. These spatial and temporal resolutions are relatively coarse when considering individual fish reacting to environmental conditions, which likely occurs at spatial scales of < 1 m and on the order of seconds. Based on this information, our assumption that fish move in a consistent direction and at a consistent speed for an hour is likely unrealistic and may have resulted in some of the discrepancies in predicted and apparent fish distributions. Additionally, the coarse spatial scale of the model means that relatively few grid cells make up the width of the subestuaries at any given point. As the number of grid cells to which individuals can move is reduced, it becomes less likely that simulated fish are able to move with directional persistence and more likely that the time required for individuals to move into areas of more favorable conditions is increased. This may account for apparent higher abundance of fish in areas of more favorable conditions than predicted by the IBMs. Despite these limitations, we contend that these IBMs demonstrate the importance of incorporating physiological abilities and tolerances into population models and the utility of using metabolic scope in this context.
Additional research examining the behavioral responses of fish to environmental conditions is necessary to determine how catchability and prey availability, as well as other factors (e.g., spatial and temporal resolution), and explain the discrepancies between our models’ predictions and survey observations. Such knowledge could be gained by tracking individual fish movements using acoustic telemetry and by simultaneously sampling environmental conditions experienced by telemetered fish in near real time (Szedlmayer and Able 1993; Almeida 1996; Taverny et al. 2002; Kelly et al. 2007; Childs et al. 2008; Fabrizio et al. 2013, 2014). There are several major challenges to this type of study, such as the time and cost of monitoring individuals and the environmental conditions they experience at high spatial and temporal resolutions, and simultaneously assessing prey availability. For benthivores like Atlantic croaker and spot, an assessment of prey abundance requires sampling the benthos to determine prey density. This would, however, also require quantification of prey availability and vulnerability from the fishes’ perspective, which may or may not match availability and vulnerability to sampling gear. Simultaneously, sampled movement and environmental data (biotic and abiotic) may yield important information about potential drivers of individual fish movement at spatial and temporal resolutions that may be more meaningful to fish. These data could be used to create a detailed model of fish behavior in response to abiotic and biotic conditions through linked individual-based and statistical inferential models (Zhang et al. 2017). These movement models could provide a more accurate assessment of environmental conditions that are important in driving individual movement and ultimately the spatial distribution of the population. Understanding individual movement and its effects on population distributions in response to environmental conditions is increasingly important in this time of rapid environmental change. Such understanding can increase the ability of researchers to assess accurately the status of exploited populations through the use of habitat-based standardization, a technique which better represents true fish abundances than assessments that ignore environmental effects on population processes (Bigelow et al. 2002; Hinton and Maunder 2004; Bigelow and Maunder 2007).
Data Availability
The data that support the findings of this study are available from the corresponding author, BJM, upon reasonable request.
References
Almeida, P.R. 1996. Estuarine movement patterns of adult thin-lipped grey mullet, Liza ramada (Risso) (Pisces, Mugilidae), observed by ultrasonic tracking. Journal of Experimental Marine Biology and Ecology 202: 137–150. https://doi.org/10.1016/0022-0981(95)00162-X.
Barbieri, L.R., M.E. Chittenden Jr., and S.K. Lowerre-Barbieri. 1994. Maturity, spawning, and ovarian cycle of Atlantic croaker, Micropogonias undulatus, in the Chesapeake Bay and adjacent coastal waters. Fishery Bulletin 92: 671–685.
Beuvard, C., A.K.D. Imsland, and H. Thorarensen. 2022. The effect of temperature on growth performance and aerobic metabolic scope in Arctic charr, Salvelinus alpinus (L.). Journal of Thermal Biology 104: 103117.
Bigelow, K.A., J. Hampton, and N. Miyabe. 2002. Application of a habitat-based model to estimate effective longline fishing effort and relative abundance of Pacific bigeye tuna ( Thunnus obesus): Habitat-based model to estimate effective longline fishing effort. Fisheries Oceanography 11: 143–155. https://doi.org/10.1046/j.1365-2419.2002.00196.x.
Bigelow, K.A., and M.N. Maunder. 2007. Does habitat or depth influence catch rates of pelagic species? Canadian Journal of Fisheries and Aquatic Sciences 64: 1581–1594. https://doi.org/10.1139/F07-115.
Brady, D., and T. Targett. 2013. Movement of juvenile weakfish Cynoscion regalis and spot Leiostomus xanthurus in relation to diel-cycling hypoxia in an estuarine tidal tributary. Marine Ecology Progress Series 491: 199–219. https://doi.org/10.3354/meps10466.
Breitburg, D. 2002. Effects of hypoxia, and the balance between hypoxia and enrichment, on coastal fishes and fisheries. Estuaries 25: 767–781. https://doi.org/10.1007/BF02804904.
Breitburg, D., L.A. Levin, A. Oschlies, M. Grégoire, F.P. Chavez, D.J. Conley, V. Garçon, et al. 2018. Declining oxygen in the global ocean and coastal waters. Science 359. https://doi.org/10.1126/science.aam7240.
Brett, R.J. 1964. The respiratory metabolism and swimming performance of young sockeye salmon. Journal of the Fisheries Research Board of Canada 21: 1183–1226.
Brosset, P., S.J. Cooke, Q. Schull, V.M. Trenkel, P. Soudant, and C. Lebigre. 2021. Physiological biomarkers and fisheries management. Reviews in Fish Biology and Fisheries 31: 797–819. https://doi.org/10.1007/s11160-021-09677-5.
Buchheister, A., C.F. Bonzek, J. Gartland, and R.J. Latour. 2013. Patterns and drivers of the demersal fish community of Chesapeake bay. Marine Ecology Progress Series 481: 161–180. https://doi.org/10.3354/meps10253.
Burnham, K.P., and D.R. Anderson. 2002. Model selection and multimodel inference: A practical information-theoretic approach, 2nd ed. New York: Springer-Verlag.
Childs, A.R., P.D. Cowley, T.F. Næsje, A.J. Booth, W.M. Potts, E.B. Thorstad, and F. Økland. 2008. Do environmental factors influence the movement of estuarine fish? A case study using acoustic telemetry. Estuarine, Coastal and Shelf Science 78: 227–236. https://doi.org/10.1016/j.ecss.2007.12.003.
Chown, S.L., A.A. Hoffmann, T.N. Kristensen, M.J. Angilletta, N.C. Stenseth, and C. Pertoldi. 2010. Adapting to climate change: A perspective from evolutionary physiology. Climate Research 43: 3–15. https://doi.org/10.3354/cr00879.
Claireaux, G., and D. Chabot. 2016. Responses by fishes to environmental hypoxia: Integration through Fry’s concept of aerobic metabolic scope: Hypoxia and fry’s paradigm of aerobic scope. Journal of Fish Biology 88: 232–251. https://doi.org/10.1111/jfb.12833.
Claireaux, G., and J.-P. Lagardère. 1999. Influence of temperature, oxygen and salinity on the metabolism of the European sea bass. Journal of Sea Research 42: 157–168. https://doi.org/10.1016/S1385-1101(99)00019-2.
Claireaux, G., and C. Lefrançois. 2007. Linking environmental variability and fish performance: Integration through the concept of scope for activity. Philosophical Transactions of the Royal Society b: Biological Sciences 362: 2031–2041. https://doi.org/10.1098/rstb.2007.2099.
Claireaux, G., D.M. Webber, J.-P. Lagardère, and S.R. Kerr. 2000. Influence of water temperature and oxygenation on the aerobic metabolic scope of Atlantic cod (Gadus morhua). Journal of Sea Research 44: 257–265.
Clark, T.D., E. Sandblom, and F. Jutfelt. 2013. Aerobic scope measurements of fishes in an era of climate change: Respirometry, relevance and recommendations. Journal of Experimental Biology 216: 2771–2782. https://doi.org/10.1242/jeb.084251.
Cooke, S.J., E.G. Martins, D.P. Struthers, L.F.G. Gutowsky, M. Power, S.E. Doka, J.M. Dettmers, et al. 2016. A moving target—incorporating knowledge of the spatial ecology of fish into the assessment and management of freshwater fish populations. Environmental Monitoring and Assessment 188. https://doi.org/10.1007/s10661-016-5228-0.
Cooper, S.R., and G.S. Brush. 1991. Long-term history of Chesapeake Bay anoxia. Science 254: 992–996.
Craig, J.K. 2012. Aggregation on the edge: Effects of hypoxia avoidance on the spatial distribution of brown shrimp and demersal fishes in the Northern Gulf of Mexico. Marine Ecology Progress Series 445: 75–95. https://doi.org/10.3354/meps09437.
Craig, J.K., and L.B. Crowder. 2005. Hypoxia-induced habitat shifts and energetic consequences in Atlantic croaker and brown shrimp on the Gulf of Mexico shelf. Marine Ecology Progress Series 294: 79–94. https://doi.org/10.3354/meps294079.
Cucco, A., M. Sinerchia, C. Lefrançois, P. Magni, M. Ghezzo, G. Umgiesser, A. Perilli, and P. Domenici. 2012. A metabolic scope based model of fish response to environmental changes. Ecological Modelling 237–238: 132–141. https://doi.org/10.1016/j.ecolmodel.2012.04.019.
Dalla Via, J., G. Van Den Thillart, O. Cattani, and P. Cortesi. 1998. Behavioural responses and biochemical correlates in Solea solea to gradual hypoxic exposure. Canadian Journal of Zoology 76: 2108–2113. https://doi.org/10.1139/z98-141.
Del Toro-Silva, F.M., J.M. Miller, J.C. Taylor, and T.A. Ellis. 2008. Influence of oxygen and temperature on growth and metabolic performance of Paralichthys lethostigma (Pleuronectiformes: Paralichthyidae). Journal of Experimental Marine Biology and Ecology 358: 113–123. https://doi.org/10.1016/j.jembe.2008.01.019.
Dellapenna, T.M., S.A. Kuehl, and L. Pitts. 2001. Transient, longitudinal, sedimentary furrows in the York River subestuary, Chesapeake Bay: Furrow evolution and effects on seabed mixing and sediment transport. Estuaries 24: 215–227. https://doi.org/10.2307/1352946.
Denny, M., and B. Helmuth. 2009. Confronting the physiological bottleneck: A challenge from ecomechanics. Integrative and Comparative Biology 49: 197–201. https://doi.org/10.1093/icb/icp070.
Deutsch, C., A. Ferrel, B. Seibel, H.O. Pörtner, and R.B. Huey. 2015. Climate change tightens a metabolic constraint on marine habitats. Science 348: 1132–1135. https://doi.org/10.1126/science.aaa1605.
Diaz, R.J., and R. Rosenberg. 1995. Marine benthic hypoxia: A review of its ecological effects and the behavioural responses of benthic macrofauna. Oceanography and Marine Biology: An Annual Review. 33: 245–303.
Diaz, Robert J., G.R. Cutter, and D.M. Dauer. 2003. A comparison of two methods for estimating the status of benthic habitat quality in the Virginia Chesapeake Bay. Journal of Experimental Marine Biology and Ecology 285–286: 371–381. https://doi.org/10.1016/S0022-0981(02)00538-5.
Diaz, Robert J., and R. Rosenberg. 2008. Spreading dead zones and consequences for marine ecosystems. Science 321: 926–929. https://doi.org/10.1126/science.1156401.
Diaz, Robert J., and R. Rosenberg. 2011. Introduction to environmental and economic consequences of hypoxia. International Journal of Water Resources Development 27: 71–82. https://doi.org/10.1080/07900627.2010.531379.
Du, J., J. Shen, K. Park, Y.P. Wang, and X. Yu. 2018. Worsened physical condition due to climate change contributes to the increasing hypoxia in Chesapeake Bay. Science of the Total Environment 630: 707–717. Elsevier B.V. https://doi.org/10.1016/j.scitotenv.2018.02.265.
Eby, L.A., and L.B. Crowder. 2002. Hypoxia-based habitat compression in the Neuse River Estuary: Context-dependent shifts in behavioral avoidance thresholds. Canadian Journal of Fisheries and Aquatic Sciences 59: 952–965. https://doi.org/10.1139/f02-067.
Ern, R., D.T.T. Huong, N.V. Cong, M. Bayley, and T. Wang. 2014. Effect of salinity on oxygen consumption in fishes: A review. Journal of Fish Biology 84: 1210–1220. https://doi.org/10.1111/jfb.12330.
Fabrizio, M.C., J.P. Manderson, and J.P. Pessutti. 2013. Habitat associations and dispersal of black sea bass from a mid-Atlantic Bight reef. Marine Ecology Progress Series 482: 241–253. https://doi.org/10.3354/meps10302.
Fabrizio, M.C., J.P. Manderson, and J.P. Pessutti. 2014. Home range and seasonal movements of Black Sea Bass (Centropristis striata) during their inshore residency at a reef in the mid-Atlantic Bight. Fishery Bulletin 112: 82–97. https://doi.org/10.7755/FB.112.1.5.
Fabry, V.J., B.A. Seibel, R.A. Feely, and J.C. Orr. 2008. Impacts of ocean acidification on marine fauna and ecosystem processes. ICES Journal of Marine Science 65: 414–432. https://doi.org/10.2307/j.ctv8jnzw1.25.
Fry, F.E.J. 1947. Effect of environment on animal activity. University of Toronto Studies Biological Series 55: 1–62.
Fry, F.E.J. 1971. The effect of environmental factors on the physiology of fish. In Fish Physiology, ed. W. S. Hoar and D. J. Randall, VI:1–98. New York, New York: Academic Press.
Fulford, R. S., M.S. Peterson, and P.O. Grammer. 2011. An ecological model of the habitat mosaic in estuarine nursery areas: part I-interaction of dispersal theory and habitat variability in describing juvenile fish distributions. Ecological Modelling 222: 3203–3215. Elsevier B.V. https://doi.org/10.1016/j.ecolmodel.2011.07.001.
Fulford, R.S., M.S. Peterson, W. Wu, and P.O. Grammer. 2014. An ecological model of the habitat mosaic in estuarine nursery areas: Part II-projecting effects of sea level rise on fish production. Ecological Modelling 273: 96–108. https://doi.org/10.1016/j.ecolmodel.2013.10.032.
Fulford, R.S., M. Russell, and J.E. Rogers. 2016. Habitat restoration from an ecosystem goods and services perspective: application of a spatially explicit individual-based model. Estuaries and Coasts 39: 1801–1815. https://doi.org/10.1007/s12237-016-0100-6.
Gamliel, I., Y. Buba, T. Guy-Haim, T. Garval, D. Willett, G. Rilov, and J. Belmaker. 2020. Incorporating physiology into species distribution models moderates the projected impact of warming on selected Mediterranean marine species. Ecography 43: 1–17. https://doi.org/10.1111/ecog.04423.
Goodwin, R.A., J.M. Nestler, J.J. Anderson, L.J. Weber, and D.P. Loucks. 2006. Forecasting 3-D fish movement behavior using a Eulerian-Lagrangian-agent method (ELAM). Ecological Modelling 192: 197–223. https://doi.org/10.1016/j.ecolmodel.2005.08.004.
Grimm, V. 1999. Ten years of individual-based modelling in ecology: What have we learned and what could we learn in the future? Ecological Modelling 115: 129–148. https://doi.org/10.1016/S0304-3800(98)00188-4.
Grimm, V., U. Berger, F. Bastiansen, S. Eliassen, V. Ginot, J. Giske, J. Goss-Custard, et al. 2006. A standard protocol for describing individual-based and agent-based models. Ecological Modelling 198: 115–126. https://doi.org/10.1016/j.ecolmodel.2006.04.023.
Grimm, V., U. Berger, D.L. DeAngelis, J.G. Polhill, J. Giske, and S.F. Railsback. 2010. The ODD protocol: a review and first update. Ecological Modelling 221: 2760–2768. Elsevier B.V. https://doi.org/10.1016/j.ecolmodel.2010.08.019.
Grimm, V., S.F. Railsback, C.E. Vincenot, U. Berger, C. Gallagher, D.L. Deangelis, B. Edmonds, et al. 2020. The ODD protocol for describing agent-based and other simulation models: a second update to improve clarity, replication, and structural realism. Journal of Artificial Societies and Social Simulation 23. https://doi.org/10.18564/jasss.4259.
Hagy, J.D., W.R. Boynton, C.W. Keefe, and K.V. Wood. 2004. Hypoxia in Chesapeake Bay, 1950–2001: Long-term change in relation to nutrient loading and river flow. Estuaries 27: 634–658. https://doi.org/10.1007/BF02907650.
Hare, J.A., M.J. Wuenschel, and M.E. Kimball. 2012. Projecting range limits with coupled thermal tolerance - climate change models: an example based on gray snapper (Lutjanus griseus) along the U.S. East Coast. Edited by Myron Peck. PLoS One 7: e52294. https://doi.org/10.1371/journal.pone.0052294.
Haven, D.S. 1959. Migration of the Croaker, Micropogonias undulatus. Copeia 1959: 25. https://doi.org/10.2307/1440095.
Hazen, E., J. Craig, C. Good, and L. Crowder. 2009. Vertical distribution of fish biomass in hypoxic waters on the Gulf of Mexico shelf. Marine Ecology Progress Series 375: 195–207. https://doi.org/10.3354/meps07791.
Hinton, M.G., and M.N. Maunder. 2004. Methods for standardizing CPUE and how to select among them. International Commission for the Conservation of Atlantic Tunas, Collective Volume of Science Papers 56: 169–177.
Hollins, J., D. Thambithurai, B. Koeck, A. Crespel, D.M. Bailey, S.J. Cooke, J. Lindström, K.J. Parsons, and S.S. Killen. 2018. A physiological perspective on fisheries-induced evolution. Evolutionary Applications 11: 561–576. https://doi.org/10.1111/eva.12597.
Horodysky, A.Z., R.W. Brill, P.G. Bushnell, J.A. Musick, and R.J. Latour. 2011. Comparative metabolic rates of common western North Atlantic Ocean sciaenid fishes. Journal of Fish Biology 79: 235–255. https://doi.org/10.1111/j.1095-8649.2011.03017.x.
Horodysky, A.Z., S.J. Cooke, and R.W. Brill. 2015. Physiology in the service of fisheries science: Why thinking mechanistically matters. Reviews in Fish Biology and Fisheries 25: 425–447. https://doi.org/10.1007/s11160-015-9393-y.
Howarth, R., F. Chan, D.J. Conley, J. Garnier, S.C. Doney, R. Marino, and G. Billen. 2011. Coupled biogeochemical cycles: Eutrophication and hypoxia in temperate estuaries and coastal marine ecosystems. Frontiers in Ecology and the Environment 9: 18–26. https://doi.org/10.1890/100008.
Humston, R., J.S. Ault, M. Lutcavage, and D.B. Olson. 2000. Schooling and migration of large pelagic fishes relative to environmental cues. Fisheries Oceanography 9: 136–146. https://doi.org/10.1046/j.1365-2419.2000.00132.x.
Humston, R., D.B. Olson, and J.S. Ault. 2004. Behavioral assumptions in models of fish movement and their influence on population dynamics. Transactions of the American Fisheries Society 133: 1304–1328. https://doi.org/10.1577/t03-040.1.
Huston, M., D. DeAngelis, and W. Post. 1988. New models unify computer be explained by interactions among individual organisms. BioScience 38: 682–691.
IPCC. 2014. Climate change 2014: synthesis report. Contribution of working groups I, II and III to the Fifth assessment report of the intergovernmental panel on climate change [Core Writing Team, R.K. Pachuari and L.A. Meyers (eds.)]. Geneva, Switzerland.
Judson, O.P. 1994. The rise of the individual-based model in ecology. Trends in Ecology and Evolution 9: 9–14. https://doi.org/10.1016/0169-5347(94)90225-9.
Jutfelt, F., T. Norin, R. Ern, J. Overgaard, T. Wang, D.J. McKenzie, S. Lefevre, et al. 2018. Oxygen- and capacity-limited thermal tolerance: blurring ecology and physiology. Journal of Experimental Biology 221: jeb169615. https://doi.org/10.1242/jeb.169615.
Kearney, M. 2006. Habitat, environment and niche: What are we modelling? Oikos 115: 186–191. https://doi.org/10.1111/j.2006.0030-1299.14908.x.
Kelly, J.T., A.P. Klimley, and C.E. Crocker. 2007. Movements of green sturgeon, Acipenser medirostris, in the San Francisco Bay estuary, California. Environmental Biology of Fishes 79: 281–295. https://doi.org/10.1007/s10641-006-0036-y.
Kelsch, S.W., and W.H. Neill. 1990. Temperature preference versus acclimation in fishes: selection for changing metabolic optima. Transactions of the American Fisheries Society1 119: 601–610. https://doi.org/10.1577/1548-8659(1990)119<0601.
Kemp, W.M., W.R. Boynton, J.E. Adolf, D.F. Boesch, W.C. Boicourt, G. Brush, J.C. Cornwell, et al. 2005. Eutrophication of Chesapeake Bay: Historical trends and ecological interactions. Marine Ecology Progress Series 303: 1–29. https://doi.org/10.3354/meps303001.
Kitchell, J.F., D.J. Stewart, and D. Weininger. 1977. Applications of a bioenergetics model to Yellow Perch (Perca flavescens) and Walleye (Stizostedion vitreum vitreum). Journal of the Fisheries Research Board of Canada 34: 1922–1935. https://doi.org/10.1139/f77-258.
Kleypas, J. 2015. Invisible barriers to dispersal. Science 348: 1086–1087. https://doi.org/10.1126/science.aab4122.
Koenigstein, S., F.C. Mark, S. Gößling-Reisemann, H. Reuter, and H.O. Poertner. 2016. Modelling climate change impacts on marine fish populations: Process-based integration of ocean warming, acidification and other environmental drivers. Fish and Fisheries 17: 972–1004. https://doi.org/10.1111/faf.12155.
Kooijman, S.A.L.M. 2010. Dynamic energy budget theory. Cambridge: Cambridge University Press.
Kraskura, K., and J.A. Nelson. 2018. Hypoxia and sprint swimming performance of juvenile striped bass, Morone saxatilis. Physiological and Biochemical Zoology 91: 682–690. https://doi.org/10.1086/694933.
Kraus, R.T., C.T. Knight, T.M. Farmer, A.M. Gorman, P.D. Collingsworth, G.J. Warren, P.M. Kocovsky, and J.D. Conroy. 2015. Dynamic hypoxic zones in Lake Erie compress fish habitat, altering vulnerability to fishing gears. Canadian Journal of Fisheries and Aquatic Sciences 72: 797–806. https://doi.org/10.1139/cjfas-2014-0517.
Laffoley, D., and J.M. Baxter, ed. 2019. Ocean deoxygenation: everyone’s problem. Causes, impacts, consequences and solutions. IUCN, International Union for Conservation of Nature. https://doi.org/10.2305/IUCN.CH.2019.13.en.
Langseth, B.J., K.M. Purcell, J.K. Craig, A.M. Schueller, J.W. Smith, K.W. Shertzer, S. Creekmore, K.A. Rose, and K. Fennel. 2014. Effect of changes in dissolved oxygen concentrations on the spatial dynamics of the gulf menhaden fishery in the northern Gulf of Mexico. Marine and Coastal Fisheries 6: 223–234. https://doi.org/10.1080/19425120.2014.949017.
Lapointe, D., W. Vogelbein, M. Fabrizio, D. Gauthier, and R. Brill. 2014. Temperature, hypoxia, and mycobacteriosis: Effects on adult striped bass Morone saxatilis metabolic performance. Diseases of Aquatic Organisms 108: 113–127. https://doi.org/10.3354/dao02693.
Lauchlan, S.S., and I. Nagelkerken. 2020. Species range shifts along multistressor mosaics in estuarine environments under future climate. Fish and Fisheries 32–46. https://doi.org/10.1111/faf.12412.
Lefrançois, C., and G. Claireaux. 2003. Influence of ambient oxygenation and temperature on metabolic scope and scope for heart rate in the common sole Solea solea. Marine Ecology Progress Series 259: 273–284. https://doi.org/10.3354/meps259273.
Little, A.G., I. Loughland, and F. Seebacher. 2020. What do warming waters mean for fish physiology and fisheries? Journal of Fish Biology 328–340. https://doi.org/10.1111/jfb.14402.
Llansó, R.J. 1992. Effects of hypoxia on estuarine benthos: The Lower Rappahannock River (Chesapeake Bay), a case study. Estuarine, Coastal and Shelf Science 35: 491–515.
Logan, M. 2010. Biostatistical design and analysis using R: A practical guide. West Sussex, United Kingdom: Wiley.
Long, W.C., and R.D. Seitz. 2008. Trophic interactions under stress: Hypoxia enhances foraging in an estuarine food web. Marine Ecology Progress Series 362: 59–68. https://doi.org/10.3354/meps07395.
Lu, L., and R.S.S. Wu. 2000. An experimental study on recolonization and succession of marine macrobenthos in defaunated sediment. Marine Biology 136: 291–302. https://doi.org/10.1007/s002270050687.
Ludsin, S.A., X. Zhang, S.B. Brandt, M.R. Roman, W.C. Boicourt, D.M. Mason, and M. Costantini. 2009. Hypoxia-avoidance by planktivorous fish in Chesapeake Bay: Implications for food web interactions and fish recruitment. Journal of Experimental Marine Biology and Ecology 381: 121–131. https://doi.org/10.1016/j.jembe.2009.07.016.
Marcek, B.J., R.W. Brill, and M.C. Fabrizio. 2019. Metabolic scope and hypoxia tolerance of Atlantic croaker (Micropogonias undulatus Linnaeus, 1766) and spot (Leiostomus xanthurus Lacepède, 1802), with insights into the effects of acute temperature change. Journal of Experimental Marine Biology and Ecology 516: 150–158. https://doi.org/10.1016/j.jembe.2019.04.007.
Marras, S., A. Cucco, F. Antognarelli, E. Azzurro, M. Milazzo, M. Bariche, M. Butenschön, et al. 2015. Predicting future thermal habitat suitability of competing native and invasive fish species: from metabolic scope to oceanographic modelling. Conservation Physiology 3: cou059. https://doi.org/10.1093/conphys/cou059.
McCormick, L.R., and L.A. Levin. 2017. Physiological and ecological implications of ocean deoxygenation for vision in marine organisms. Philosophical Transactions of the Royal Society a: Mathematical, Physical and Engineering Sciences 375: 20160322. https://doi.org/10.1098/rsta.2016.0322.
McKenzie, D.J., M. Axelsson, D. Chabot, G. Claireaux, S.J. Cooke, R.A. Corner, G. De Boeck, et al. 2016. Conservation physiology of marine fishes: state of the art and prospects for policy. Conservation Physiology 4: cow046. https://doi.org/10.1093/conphys/cow046.
Metcalfe, J.D., and P.J. Butler. 1984. Changes in activity and ventilation in response to hypoxia in unrestrained, unoperated dogfish (Scyliorhinus canicula L.). Journal of Experimental Biology 108: 411–418.
Murawski, S.A. 1993. Climate change and marine fish distributions: Forecasting from historical analogy. Transactions of the American Fisheries Society 122: 647–658. https://doi.org/10.1577/1548-8659(1993)122<0647:ccamfd>2.3.co;2
Murdy, E.O., and J.A. Musick. 2013. Field guide to fishes of the Chesapeake Bay. Baltimore, Maryland: Johns Hopkins University Press.
Neill, W.H., J.M. Miller, H.W. Van Der Veer, and K.O. Winemiller. 1994. Ecophysiology of marine fish recruitment: A conceptual framework for understanding interannual variability. Netherlands Journal of Sea Research 32: 135–152.
Nelson, J.A. 1989. Critical swimming speeds of yellow perch Perca flavescens: Comparison of populations from a naturally acidic and circumneutral lake in acid and neutral water. Journal of Experimental Biology 145: 239–254.
Norin, T., and T.D. Clark. 2016. Measurement and relevance of maximum metabolic rate in fishes. Journal of Fish Biology 88: 122–151. https://doi.org/10.1111/jfb.12796.
Nye, J.A., J.S. Link, J.A. Hare, and W.J. Overholtz. 2009. Changing spatial distribution of fish stocks in relation to climate and population size on the Northeast United States continental shelf. Marine Ecology Progress Series 393: 111–129. https://doi.org/10.3354/meps08220.
Nye, J.A., D.A. Loewensteiner, and T.J. Miller. 2011. Annual, seasonal, and regional variability in diet of Atlantic croaker (Micropogonias undulatus) in Chesapeake Bay. Estuaries and Coasts 34: 691–700. https://doi.org/10.1007/s12237-010-9348-4.
Officer, C.B., R.B. Biggs, J.L. Taft, L.E. Cronin, M.A. Tyler, and W.R. Boynton. 1984. Chesapeake Bay anoxia: Origin, development, and significance. Science 223: 22–27. https://doi.org/10.1126/science.223.4631.22.
Ospina, R., and S.L.P. Ferrari. 2010. Inflated beta distributions. Statistical Papers 51: 111–126. https://doi.org/10.1007/s00362-008-0125-4.
Ospina, R., and S.L.P. Ferrari. 2012. A general class of zero-or-one inflated beta regression models. Computational Statistics and Data Analysis 56: 1609–1623. Elsevier B.V. https://doi.org/10.1016/j.csda.2011.10.005.
Perry, A.L., P.J. Low, J.R. Ellis, and J.D. Reynolds. 2005. Ecology: Climate change and distribution shifts in marine fishes. Science 308: 1912–1915. https://doi.org/10.1126/science.1111322.
Pihl, L., S.P. Baden, and R.J. Diaz. 1991. Effects of periodic hypoxia on distribution of demersal fishes and crustaceans. Marine Biology 108: 349–360.
Pihl, L., S.P. Baden, R.J. Diaz, and L.C. Schaffner. 1992. Hypoxia-induced structural changes in the diet of bottom-feeding fish and Crustacea. Marine Biology 112: 349–361. https://doi.org/10.1055/s-2003-44271.
Pörtner, H.O., C. Bock, and F.C. Mark. 2017. Oxygen- and capacity-limited thermal tolerance: Bridging ecology and physiology. Journal of Experimental Biology 220: 2685–2696. https://doi.org/10.1242/jeb.134585.
Pörtner, H.O., and A.P. Farrell. 2008. Physiology and climate change. Science 322: 690–692.
Pörtner, H.O., and R. Knust. 2007. Climate change affects marine fishes through the oxygen limitation of thermal tolerance. Science 315: 95–97. https://doi.org/10.1126/science.1135471.
Pörtner, H.O., P.M. Schulte, C.M. Wood, and F. Schiemer. 2010. Niche dimensions in fishes: An integrative view. Physiological and Biochemical Zoology 83: 808–826. https://doi.org/10.1086/655977.
R Core Team. 2020. R: a language and environment for statistical computing. In R Foundation for Statistical Computing. Vienna, Austria.
Rabalais, N.N., R.J. Díaz, L.A. Levin, R.E. Turner, D. Gilbert, and J. Zhang. 2010. Dynamics and distribution of natural and human-caused hypoxia. Biogeosciences 7: 585–619. https://doi.org/10.5194/bg-7-585-2010.
Railsback, S.F., and V. Grimm. 2011. Agent-based and individual-based modeling: A practical introduction. Princeton, New Jersey: Princeton University Press.
Riebesell, U., and J.P. Gattuso. 2015. Lessons learned from ocean acidification research. Nature Climate Change 5. Nature Publishing Group 12–14. https://doi.org/10.1038/nclimate2456.
Robinson, E., A. Jerrett, S. Black, and W. Davison. 2013. Hypoxia impairs visual acuity in snapper (Pagrus auratus). Journal of Comparative Physiology A 199: 611–617. https://doi.org/10.1007/s00359-013-0809-7.
Rose, K.A., S. Creekmore, D. Justić, P. Thomas, J.K. Craig, R.M. Neilan, L. Wang, Md.S. Rahman, and D. Kidwell. 2018a. Modeling the population effects of hypoxia on Atlantic croaker (Micropogonias undulatus) in the Northwestern Gulf of Mexico: Part 2—realistic hypoxia and eutrophication. Estuaries and Coasts 41: 255–279. https://doi.org/10.1007/s12237-017-0267-5.
Rose, K.A., S. Creekmore, P. Thomas, J.K. Craig, Md.S. Rahman, and R.M. Neilan. 2018b. Modeling the population effects of hypoxia on Atlantic croaker (Micropogonias undulatus) in the Northwestern Gulf of Mexico: Part 1—model description and idealized hypoxia. Estuaries and Coasts 41: 233–254. https://doi.org/10.1007/s12237-017-0266-6.
Rose, K.A., W.J. Kimmerer, K.P. Edwards, and W.A. Bennett. 2013a. Individual-based modeling of delta smelt population dynamics in the Upper San Francisco estuary: II. Alternative baselines and good versus bad years. Transactions of the American Fisheries Society 142: 1260–1272. https://doi.org/10.1080/00028487.2013.799519.
Rose, K.A., W.J. Kimmerer, K.P. Edwards, and W.A. Bennett. 2013b. Individual-based modeling of delta smelt population dynamics in the Upper San Francisco estuary: I. Model description and baseline results. Transactions of the American Fisheries Society 142: 1238–1259. https://doi.org/10.1080/00028487.2013.799518.
Sabatés, A., P. Martín, J. Lloret, and V. Raya. 2006. Sea warming and fish distribution: The case of the small pelagic fish, Sardinella aurita, in the western Mediterranean. Global Change Biology 12: 2209–2219. https://doi.org/10.1111/j.1365-2486.2006.01246.x.
Schaffner, L.C., T.M. Dellapenna, E.K. Hinchey, C.T. Friedrichs, M.T. Neubauer, M.E. Smith, and S.A. Kuehl. 2001. Physical energy regimes, seabed dynamics, and organism-sediment interactions along an estuarine gradient. In Organism-Sediment Interactions, ed. J.Y. Aller, S.A. Woodin, and R.C. Aller, 159–179. Columbia, South Carolina: University of South Carolina Press.
Schlenger, A., E.W. North, Y. Li, M. Li, and W.M. Kemp. 2022. Applying a three-dimensional habitat volume model to estimate sensitivity of Chesapeake Bay living resources to environmental change: A proof-of-concept exercise. Estuaries and Coasts 45: 393–412. https://doi.org/10.1007/s12237-021-00967-6.
Schurmann, H., and J.F. Steffensen. 1994. Spontaneous swimming activity of Atlantic Cod Gadus morhua exposed to graded hypoxia at three temperatures. Journal of Experimental Biology 197: 129–142.
Schurmann, H., and J.F. Steffensen. 1997. Effects of temperature, hypoxia and activity on the metabolism of juvenile Atlantic cod. Journal of Fish Biology 50: 1166–1180.
Scully, M.E. 2016. The contribution of physical processes to inter-annual variations of hypoxia in Chesapeake Bay: A 30-yr modeling study. Limnology and Oceanography 61: 2243–2260. https://doi.org/10.1002/lno.10372.
Seitz, R.D., L.S. Marshall, A.H. Hines, and K.L. Clark. 2003. Effects of hypoxia on predator-prey dynamics of the blue crab Callinectes sapidus and the Baltic clam Macoma balthica in Chesapeake Bay. Marine Ecology Progress Series 257: 179–188. https://doi.org/10.3354/meps257179.
Sibly, R.M., V. Grimm, B.T. Martin, A.S.A. Johnston, K. Kulakowska, C.J. Topping, P. Calow, J. Nabe-Nielsen, P. Thorbek, and D.L. Deangelis. 2013. Representing the acquisition and use of energy by individuals in agent-based models of animal populations. Methods in Ecology and Evolution 4: 151–161. https://doi.org/10.1111/2041-210x.12002.
Smith, E.P., and K.A. Rose. 1995. Model goodness-of-fit analysis using regression and related techniques. Ecological Modelling 77: 49–64. https://doi.org/10.1016/0304-3800(93)E0074-D.
Stasinopoulos, D.M, and R.A. Rigby. 2007. Generalized additive models for location scale and shape (GAMLSS) in R. Journal of Statistical Software 23: 1–46. https://doi.org/10.18637/jss.v023.i07.
Stone, J.P., K.L. Pangle, S.A. Pothoven, H.A. Vanderploeg, S.B. Brandt, T.O. Höök, T.H. Johengen, and S.A. Ludsin. 2020. Hypoxia’s impact on pelagic fish populations in Lake Erie: A tale of two planktivores. Canadian Journal of Fisheries and Aquatic Sciences 77: 1131–1148. https://doi.org/10.1139/cjfas-2019-0265.
Szedlmayer, S.T., and K.W. Able. 1993. Ultrasonic telemetry of age-0 summer flounder, Paralichthys dentatus, movements in a Southern New Jersey estuary. Copeia 1993: 728. https://doi.org/10.2307/1447234.
Taverny, C., M. Lepage, S. Piefort, P. Dumont, and E. Rochard. 2002. Habitat selection by juvenile European sturgeon Acipenser sturio in the Gironde estuary (France). Journal of Applied Ichthyology 18: 536–541. https://doi.org/10.1046/j.1439-0426.2002.00414.x.
Taylor, D., and D. Eggleston. 2000. Effects of hypoxia on an estuarine predator-prey interaction:Foraging behavior and mutual interference in the blue crab Callinectes sapidus and the infaunal clam prey Mya arenaria. Marine Ecology Progress Series 196: 221–237. https://doi.org/10.3354/meps196221.
Thambithurai, D., A. Crespel, T. Norin, A. Rácz, J. Lindström, K.J. Parsons, and S.S. Killen. 2019. Hypoxia alters vulnerability to capture and the potential for trait-based selection in a scaled-down trawl fishery. Conservation Physiology 7: 1–12. https://doi.org/10.1093/conphys/coz082.
Townhill, B.L., J.K. Pinnegar, D.A. Righton, and J.D. Metcalfe. 2017. Fisheries, low oxygen and climate change: How much do we really know? Journal of Fish Biology 90: 723–750. https://doi.org/10.1111/jfb.13203.
Tuckey, T.D., and M.C. Fabrizio. 2016. Variability in fish tissue proximate composition is consistent with indirect effects of hypoxia in Chesapeake Bay tributaries. Marine and Coastal Fisheries 8: 1–15. https://doi.org/10.1080/19425120.2015.1103824.
Watkins, K.S., and K.A. Rose. 2013. Evaluating the performance of individual-based animal movement models in novel environments. Ecological Modelling 250: 214–234. Elsevier B.V. https://doi.org/10.1016/j.ecolmodel.2012.11.011.
Watkins, K.S., and K.A. Rose. 2014. The effects of spatial and temporal resolution in simulating fish movement in individual-based models. Transactions of the American Fisheries Society 143: 1143–1160. https://doi.org/10.1080/00028487.2014.911208.
Watkins, K.S., and K.A. Rose. 2017. Simulating individual-based movement in dynamic environments. Ecological Modelling 356: 59–72. https://doi.org/10.1016/j.ecolmodel.2017.03.025.
Zhang, H., S.A. Ludsin, D.M. Mason, A.T. Adamack, S.B. Brandt, X. Zhang, D.G. Kimmel, M.R. Roman, and W.C. Boicourt. 2009. Hypoxia-driven changes in the behavior and spatial distribution of pelagic fish and mesozooplankton in the northern Gulf of Mexico. Journal of Experimental Marine Biology and Ecology 381: S80–S91. https://doi.org/10.1016/j.jembe.2009.07.014.
Zhang, J., T.E. Dennis, T.J. Landers, E. Bell, and G.L. W. Perry. 2017. Linking individual-based and statistical inferential models in movement ecology: a case study with black petrels (Procellaria parkinsoni). Ecological Modelling 360: 425–436. Elsevier B.V. https://doi.org/10.1016/j.ecolmodel.2017.07.017.
Acknowledgements
We thank the staff of the VIMS Juvenile Fish Trawl Survey and the Chesapeake Bay Program for the use of their data. The authors acknowledge William & Mary Research Computing for providing computational resources and/or technical support that contributed to the results reported within this paper (https://www.wm.edu/it/rc).
Funding
Financial support was provided by the National Marine Fisheries Service-Sea Grant Population and Ecosystem Dynamics Fellowship (grant number V718650). The findings and conclusions in this article are those of the authors and do not necessarily represent the views of the U.S. Fish and Wildlife Service.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by James Allen Nelson
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Marcek, B.J., Humston, R., Fabrizio, M.C. et al. Modeling the Distribution of Atlantic Croaker and Spot in a Dynamic Seascape Using Metabolic Scope. Estuaries and Coasts 47, 258–275 (2024). https://doi.org/10.1007/s12237-023-01240-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12237-023-01240-8