Abstract
The published data indicate that the bacterium Clavibacter michiganensis subsp. sepedonicus (Cms) shows a notably low degree of intraspecific variation, and most of the molecular diagnostic methods could differentiate among subspecies groups but lacked the resolution to detect genetic diversity within subspecies. The present study describes effective differentiation of 50 isolates of C. michiganensis subsp. sepedonicus by the PCR MP method using five restriction enzymes (ApaI, PstI, BamHI, XmaI, HindIII). By analysing the electrophoretic patterns obtained by the PCR MP method performed with the applied restriction enzymes, each of the 50 tested isolates could be identified and distinguished from the remaining isolates. The PCR MP method of bacterial strain identification can be used in epidemiological studies, particularly for excluding the isolates of C. michiganensis subsp. sepedonicus as a source of primary infection.
Resumen
Los datos publicados indican que la bacteria Clavibacter michiganensis subsp. sepedonicus (Cms) muestra un grado notablemente bajo de variación intraespecífica y la mayoría de los métodos de diagnóstico molecular podrían diferenciar entre los grupos de subespecies pero carecían de la resolución para detectar la diversidad genética dentro de las subespecies. El presente estudio describe la diferenciación de 50 aislamientos de C. michiganensis subsp. sepedonicus por el método PCR MP usando cinco enzimas de restricción (ApaI, PstI, BamHI, XmaI, HindIII). Al analizar los patrones electroforéticos obtenidos por el método de PCR MP realizado con las enzimas de restricción aplicadas, cada uno de los 50 aislamientos probados se pudo identificar y distinguir de los demás aislamientos. El método PCR MP de identificación de cepas bacterianas se puede utilizar en estudios epidemiológicos, particularmente para excluir los aislamientos de C. michiganensis subsp. sepedonicus como fuente de infección primaria.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Bacterial potato ring rot caused by C. michiganensis subsp. sepedonicus (Cms) (Spieckermann & Kotthoff) is a highly important quarantine disease in the potato industry, primarily in economic terms (Van der Wolf et al. 2005). This disease causes large economic losses, both directly and indirectly. The direct loses occur during potato cultivation and storage, the indirect losses are caused by the denied access to the current export markets and restricted or denied access to new markets. Zero tolerance for ring rot is obligatory in all certified EPPO potato seed production areas, both in the European Union and in North America (Smith et al. 1997; Commission Directive 2006/56/EC of 12 June 2006). The routine detection techniques, such as immunofluorescence microscopy (IF) (De Boer and McCann 1990), molecular methods based on PCR reaction (Pastrik 2000; Gudmestad et al. 2009), hybridization (Drennan et al. 1993), and pathogenicity tests on eggplants (Black Beauty) (Olsson 1976) do not have sufficient discriminatory power to efficiently differentiate Cms isolates. The close intraspecific affinity of Cms isolates was observed in morphology, biochemistry, protein, plasmid profiles and in DNA hybridization (Carlson and Vidaver 1982; Davis et al. 1984; Mogen et al. 1987; Mogen et al. 1985; Snieszko and Bonde 1943; Vantomme et al. 1987). RFLP PCR of three housekeeping gene fragments (recA, rpoB, and rpoD) differentiated between C. michiganensis subspecies however, no differences were demonstrated within C. michiganensis subsp. sepedonicus (Waleron et al. 2011). The cluster analysis of the combined sets of BOX-, ERIC- and Rep-PCR experiments used by a group of researchers from the USA showed that subspecies C. michiganensis can be divided into five different groups (Louws et al. 1998). Genetic similarities between the obtained amplification profiles of Rep-PCR between the five subspecies of Clavibacter were lower than 40%. Therefore, the rep-PCR technique uniquely distinguished the subspecies. However, the electrophoretic patterns obtained for the isolates of each C. michiganensis subspecies were similar and the isolates of the subspecies Clavibacter michiganensis subsp. sepedonicus showed over 94.7% similarity (Louws et al. 1998; Smith et al. 2001). It has been indicated that each subspecies of C. michiganensis had distinct Rep-PCR pattern and that isolates within individual subspecies had almost identical Rep-PCR patterns. Therefore, Rep-PCR is effective for the differentiation between the subspecies, not within the subspecies (Carlson and Vidaver 1982; Louws 1998). Most of the molecular methods did not differentiate the Cms isolates, even those coming from different geographical regions (Lee et al. 1997; Palomo et al. 2000; Smith et al. 2001; Waleron et al. 2011). The analysis of 16S and 23S rRNA coding sequences showed a close phylogenetic relationship between the five subspecies of C. michiganensis (Fessehaie et al. 2003; Li and De Boer 1995a, b; Mirza et al. 1993). However, based on the whole-genome and multi-locus sequence analyses it was suggested that the subspecies of Clavibacter michiganensis should be raised to species status (Li et al. 2018). Using molecular methods, the Cms isolates were classified into virulent and avirulent groups (Lee et al. 1997).
CHEF analysis, detected differences between virulent and non-virulent strains, which were correlated with alterations in the genomic regions responsible for hypersensitivity response and cellulase synthesis (Brown et al. 2002). However, there was no correlation between the morphology of bacterial colonies and pathogenicity (Brown et al. 2002). The effect of mucoids on the degree of differentiation of Cms isolates has not been observed either (Fousek and Mraz 2003; Fousek et al. 2002).
Thus, there was a consensus concerning low genetic variation among Cms isolates which has been confirmed by numerous scientific teams using various genotyping techniques (Carlson and Vidaver 1982; Davis et al. 1984; Lee et al. 1997; Li and De Boer 1995a, b; Pastrik and Rainey 1999; Louws et al. 1998; Smith et al. 2001).
The PCR melting profiles (PCR MP) method, has been confirmed efficient in the differentiation of the plant pathogen Pseudomonas syringae pv. lachrymans (Olczak-Woltman et al. 2007) and other Pseudomonas species pathogenic to plants (Bultreys and Kałużna 2010; Kałużna et al. 2016 a; Kałużna et al. 2010; Kałużna et al. 2016,b). Previously, the PCR MP method was used to differentiate clinical bacterial strains in epidemiological studies (Krawczyk et al. 2007; Stojowska et al. 2009) and demonstrated discriminatory power at least equivalent to the discriminatory power of pulsed field gel electrophoresis (PFGE), which was considered to be the gold standard (Krawczyk et al. 2007; Krawczyk et al. 2009). The ability to analyse genomes with unknown nucleotide sequence and the ease of analysing results on agarose gels are the main advantages of the PCR MP method (Masny and Płucienniczak 2003). There were also reports on applicability of PCR MP to Cms isolate differentiation (Żaczek et al. 2018).
There is little published data regarding the genetic diversity among Cms isolates recovered from potato tubers in Poland. Gathering information regarding the genetic diversity within the populations of Cms in our country and worldwide would help to study the epidemiology of these bacteria and to define its economic threat to potato production.
The objective of this study was to determine the discriminatory power of the PCR MP method in genotyping subspecies of Cms isolates.
Materials and Methods
Origin and Growth Conditions of Cms Strains
This study presents the results of genotyping 50 isolates of Cms bacteria. Year of isolation, place of origin and morphology of the bacterial colonies are shown in Table 1. Isolates were organized into three groups, depending on the degree of pathogenicity to eggplant. Degree of pathogenicity of Cms isolate was assessed in a biological assay based on the five-stage symptom scale and appendage proposed by Stead and Janse (2000). In this test, the rate of their growth in plants and the production of virulence factors inducing wilt in controlled conditions were used as the basis for isolation of isolates. The first group of isolates were characterized by the highest degree of pathogenicity (group A), the second group were medium pathogenic isolates (group B) and the third group were isolates exhibiting the lowest degree of pathogenicity (group C). The third group contained the reference strain most frequently used for research in Polish laboratories (Ref BPRIOR 527). All tested Cms isolates were derived from sixteen Voivodship Inspectorates of Plant Health and Seed Inspection (WIORiN). These isolates were derived from the Polish bacterial collection of IHAR-PIB in Radzików collected in the years 2005–2010. Two foreign isolates were also included in the study (NCPPB 3324 - an isolate from Belgium and NCPPB 3917 – an isolate from Canada).
These isolates were grown on yeast - peptone glucose agar (YPGA) and yeast extract mineral salt medium (YGM) at 22 °C for 5 days. All bacteria were stored in glycerol (70% final concertation) at −80 °C.
Identification Tests
PCR with Species-Specific Primers
To identify the Cms bacterial isolates, PCR reactions were performed according to the protocol by (Pastrik and Rainey 1999), with specific primers PSA-1 (5’-CTCCTTGTGGGGTGGGAAAA-3′) and PSA-R (5’-TACTGAGATGTTTCACTTCCCC-3′) targeting a portion of the ITS region with the expected amplicon size of 502 bp. The PCR reaction was performed in total volume of 15 μL: 1x reaction buffer (20 mM Tris-HCl, pH 8.4; 50 mM KCl) 1.5 mM MgCl2, 100 μM of each dNTP; 0.2 μM of each primer (PSA-1 and PSA-R), 1 U Taq DNA polymerase (Fermentas) and 1 ng of DNA. The following PCR conditions were used: initial denaturation at 94 °C for 2 min 30 s, followed by 10 reaction cycles of 95 °C for 1 min, 65 °C for 1 min and 72 °C for 1 min. The initial 10 cycles were followed by 25 more cycles of amplification: 95 °C for 30 s, 62 °C for 30 s, 72 °C for 1 min and incubation in 72 °C for 5 min after the last cycle.
DNA Extraction
The extraction of bacterial DNA was performed with the Easy-DNA TM Kit (Invitrogen, Netherlands) following the protocol provided by the manufacturer. Concentration and the purity of DNA was determined spectrophotometrically by measuring the absorbance at 260 nm and 280 nm using NanoDrop ND-1000 (Thermo Scientific).
PCR MP Procedure
Five restriction endonucleases (ApaI, PstI, BamHI, XmaI, HindIII) were used to obtain a variable level of discrimination of the isolates in PCR MP. For all restriction enzymes applied the number of the created genomic DNA restriction fragments could be predicted by in silico restriction digestion of the C. michiganensis subsp. sepedonicus reference genomes (NCBI Reference Sequence: NC_010407.1 and C. michiganensis subsp. sepedonicus strain CFIA-Cs3N NCBI Reference Sequence: NZ_MZMM01000001.1) using free software (tools.neb.com /REBsites/).
Prior to the PCR amplification, the genetic material was digested with five restriction enzymes in separate reactions in their recommended buffers: ApaI (Buffer B, Fermentas), PstI (Buffer O, Fermentas), XmaI (Buffer Tango, Thermo Scientific), HindIII (Buffer R, Thermo Scientific) and BamHI (Buffer BamHI Lsp 1109I, Thermo Scientific). The digestion of 200 ng of bacterial DNA was performed in total volume of 10 μL by adding 1 μL of restriction buffer to the reaction and 0.2 μL (10 U/μL) of endonuclease. The digestion was performed from 4 to 16 h at 16–37 °C depending on the restriction enzyme used. After incubation, a ligation mix was prepared in a total volume of 15 μL as follows: 1 μl of 20 μM adapters matching the sticky ends of the DNA fragments produced by the restriction enzyme used 3 μL of 5x Ligase Buffer (Invitrogen), and 0.2 μL of DNA ligase (5 U /μL, Invitrogen). The samples were incubated 3 min. at 37 °C and 3 h at 16 °C. The PCR was performed in a volume of 15 μL. The reaction mixture contained 1 μL of ligation solution, 1.5 μl 10x Dream Taq Green Buffer (Fermentas), 0.3 μL dNTP (10 mM), 0.08 μL Dream Taq DNA Polymerase 5 U/μL (Fermentas) and 1.5 μL (10 μM) primer (Table 2). Denaturation temperature ranges were optimized for each enzyme separately in the thermocycler with gradient functions (Mastercycler ep Gradient S; Eppendorf). It has to be emphasized that a number of gradients has been tested and only those with the highest level of electrophoretic pattern reproducibility and an analysable number of bands produced in PCR MP were chosen for further analyses. From each gradient single denaturation temperature was selected for each restriction endonuclease: ApaI - 93.3 °C, PstI - 92.7 °C, BamHI - 91.8 °C and 92.8 °C, XmaI -94 °C, HindIII - 95 °C, in which the number of the obtained bands was analysable and the electrophoretic band pattern was reproducible in at least two independent experiments performed on DNA extracted from two independent cultures of a Cms isolate.
The ranges of denaturation temperature gradients applied for PCR MP were: 90 °C - 95 °C for ApaI, 90 °C - 94 °C for PstI, 92.6 °C - 94.1 °C for XmaI 92.6- 94.1. The PCR MP reactions for the enzyme BamHI were performed for two denaturation temperature ranges: 90 °C - 92 °C and 91 °C - 93 °C. PCR reactions were carried out as follows for the ApaI enzyme: initial elongation of 72 °C – 5 min, 30 cycles (denaturation in the gradient 90 °C - 95 °C – 40 s, annealing 55 °C – 40 s, elongation 68 °C – 1 min 30 s) and a final elongation of 72 °C – 30 min; for the PstI enzyme initial elongation of 72 °C – 5 min, 30 cycles (denaturation in the gradient 90 °C - 94 °C – 40 s, annealing 55 °C – 40 s, elongation 68 °C – 1 min 30 s) and a final elongation of 72 °C – 30 min; for the XmaI enzyme initial elongation of 72 °C – 5 min, 27 cycles (denaturation in the gradient of 92.6 °C - 94.1 °C – 40 s, annealing 55 °C – 40 s, elongation 68 °C – 1 min30 s) and a final elongation of 72 °C – 30 s; for the BamHI enzyme initial elongation of 72 °C – 5 min, 30 cycles (denaturation in the gradient of 90 °C - 92 °C or 91 °C - 93 °C – 40 s, annealing 55 °C – 40 s, elongation 68 °C – 1 min 30 s), and a final elongation of 72° - 30 min.
For HindIII enzyme PCR reactions were conducted as follows: initial elongation 72 °C - 5 min, denaturation at 95 °C - 2 min, 23 cycles (denaturation and annealing: 95 °C - 1 min, elongation: 72 °C – 2 min).
For the PCR MP products, 6 μL or 15 μL were electrophoresed on a 2% agarose gel (24 × 20 cm) with TBE buffer and stained in ethidium bromide (10 mg/mL aqueous solution) for 5 min. The electrophoresis was performed at 120 V for 4 h and 45 min. The images of the gels were analysed using a Kodak Gel Logic 200 Imagine System (Eastman Kodak Company, Rochester, NY 14650, USA).
The images of the agarose gels with PCR MP profiles were analysed using BioNumerics v6.6 software (Applied Maths, Belgium). The similarity of the profiles obtained for the reactions with individual restriction enzymes were determined using the Dice correlation coefficient (using optimization and tolerance parameters equal to 1% and 1%, respectively). The dendrograms were prepared using the UPGMA clustering algorithm. Additionally a summary analysis was performed for all of the tests based on a similarity analysis for averaged data values from individual experiments. The dendrogram was prepared using the UPGMA clustering algorithm, as well.
PCR Product Cloning and Sequencing
The PCR products obtained from DNA of Cms isolate CL 511 using previously described PCR MP for HindIII were separated in a 6% polyacrylamide gel. Two fragments of DNA (approximately 600 bp and approximately 1000 bp) were excised from the gel and eluted (Dybczynski and Plucienniczak 1988). The extracted DNA fragments were phosphorylated with T4 polynucleotide kinase (BioLabs), subsequently the enzyme was heat inactivated, the reactions were carried out according to the manufacturer’s recommendations. Phosphorylated DNA fragments were ligated to the blunt ended linear vector pUC19, previously digested with SmaI (BioLabs) and dephosphorylated with calf intestinal alkaline phosphatase CIAP (Promega). The ligation was carried out with the T4 DNA ligase (Biolabs) according to the manufacturer’s recommendations. Ligation products were transformed into E.coli NEB Turbo Competent (BioLabs) following the recommendations from the user’s manual, and bacteria were plated on LB/ampicillin plates. The positive clones were identified by colony PCR with universal M13 forward and reverse primers. Overnight bacterial cell cultures of positive clones were grown in the liquid LB medium with ampicillin. Plasmids were isolated using Plasmid Mini kit (A&A Biotechnology) and subjected to Sanger sequencing with M13 forward and reverse primers using 3130 Genetic Analyzer (Applied Biosystems).
Results
Specific PCR for Cms Bacteria
Specific primers PSA-1 and PSA-R were used for all the tested isolates to obtain the 502 bp amplification product - a characteristic fragment for C. michiganensis subsp. sepedonicus used for Cms identification according to Commission Directive 2006/56 / EC of 12 June 2006.
Genetic Polymorphism Obtained by PCR MP
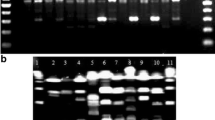
The results of all the PCR analyses performed for all the restriction enzymes and the conditions of the denaturation temperature are shown as electrophoretic patterns in Figs. 1, 2, 3, 4, 5 and 6.
The analysis of the genetic diversity based on the results obtained for the single enzyme versions of the PCR MP showed a relatively low discrimination power using the ApaI, HindIII and PstI enzymes. Each of those enzymes allowed to obtain four distinct electrophoretic patterns for the Polish C. michiganensis subsp. sepedonicus isolates and the reference strains ApaI, HindIII and PstI enzymes (Figs. 1, 2 and 3). ApaI PCR MP enzyme enabled us to distinguish two primary clusters grouping the majority of the studied isolates (internally indistinguishable by the adopted research methodology) and showing a 92% similarity level of the profiles. Analysis using the HindIII enzyme did not allow differentiation of the majority of the tested isolates (identical profiles were obtained for 46 isolates) however, four distinct clusters were identified. Analysis using the PstI enzyme enabled distinguishing two primary clusters grouping the majority of the studied isolates (internally indistinguishable by the adopted research methodology) showing a 94% similarity level of the profiles. Higher discriminating power was demonstrated by analysis using the BamHI enzyme; in denaturation temperature gradient 90 °C - 92 °C in single temperature of 91.8°C at least 9 unique electrophoretic patterns were obtained (Fig. 4). The increase of the denaturation temperature to the level of 91 °C - 93 °C additionally increased the discriminating power of this method and in single denaturation temperature of 92.8 °C 12 unique electrophoretic patterns or clusters were obtained (Fig. 5). The highest discriminatory power of the PCR MP differentiation of the isolates was achieved using the XmaI enzyme - 26 distinct electrophoretic patterns were obtained (Fig. 6). The dendrogram showing a summary analysis of the average PCR MP results obtained for all five enzymes is shown in Fig. 7. Only the dendrogram based on the results of analyses with all five restriction endonucleases based PCR MP allowed discrimination to the level of single, unique isolates Fig. 7.
Bioinformatic Restriction Fragment Analysis
The results of the restriction fragment analysis of the Cms genomes are presented in Table 3. Theoretically predicted restriction fragments, of size below 4000 bp, obtained from the reference genomes digested with HindIII were as follows: NCBI Reference Sequence NC_010407.1 - two fragments of the same size 3428 bp, 1728 bp, 945 bp and 608 bp, C. michiganensis subsp. sepedonicus strain CFIA-Cs3N (NZ_MZMM01000001.1) – 3428 bp (two fragments of the same size), 1728 bp and 608 bp.
Discussion
The bacterial species differentiation methods used so far for Clavibacter michiganensis subsp. sepedonicus isolates: rep-PCR (Louws et al. 1998; Fousek et al. 2002), RFLP (Mogen et al. 1990), AFLP (De León et al. 2009), RAPD PCR (Pastrik and Rainey 1999), PFGE (Milijašević-Marčić et al. 2012) and genetic marker based methods (Fessehaie et al. 2003) did not assure satisfactory discrimination between the isolates of the pathogen. Therefore, we decided to apply a method of bacterial species differentiation based on a different principle than rep-PCR, PFGE and RAPD PCR for discriminating between C. michiganensis subsp. sepedonicus isolates. The principle of the PCR MP is worth explaining since it differs from the principle of standard PCR techniques. In the PCR MP procedure used entire genome is converted into restriction fragments and to all those fragments artificial oligonucleotides are ligated. Thus, entire genome is converted into fragments which have primer binding sites on both ends and each of this fragments could be potentially amplified by PCR using single primer – such amplifiable fragments are called amplicons. It is not the presence of the primer binding site which determines amplification of the amplicon in PCR MP. In every PCR technique the essential step is denaturation of dsDNA in order to obtain ssDNA to which the primers can bind. The melting temperature of the DNA fragments depends on: the GC composition of those fragments and the sequence of those fragments. Two fragments of exactly the same length can have distinct melting temperature such as in the case of two PCR primers of the same length and different nucleotide sequence. In PCR MP the denaturation temperature is decreased below 95 °C and only less thermally stable DNA fragments become ssDNA and only those fragments can be amplified by PCR because primers can bind only to ssDNA (Masny and Płucienniczak 2003). In PCR MP gradients of denaturation temperature are used in PCR to establish the temperature in which the number of obtained PCR products is small enough to be analyzable by electrophoresis (Masny and Płucienniczak 2003) in contrast to most PCR methods in which the denaturation temperature is 94- 95 °C and the primer annealing and primers sequences determine which target sequences are amplified. Each DNA fragment amplified in PCR MP is characterized by two features: the size (position on the gel after electrophoresis) and the lowest denaturation temperature in which the fragment is amplified. Thus, DNA fragments of the same length and distinct sequence can be differentiated by PCR MP by the difference in the lowest denaturation temperature in which those fragments are amplified (Krystynowicz et al. 2005). C. michiganensis subsp. sepedonicus has a very high percentage of GC in its genome, and the frequency of restriction sites in a genome of a bacterial species depends on the percentage of GC pairs (Masny and Płucienniczak 2003). Identifying a restriction enzyme enabling the differentiation of C. michiganensis subsp. sepedonicus by the MP PCR method posed a problem caused by a very high (over 72%) GC content of the Cms genome (Gartemann et al. 2008). Therefore, a series of restriction enzymes and oligonucleotide adapters were tested. In each denaturation temperature gradient single denaturation temperature was used for PCR MP in our study. Selection of such single denaturation temperature in which the obtained electrophoretic pattern is reproducible in at least two independent repeats of the PCR MP procedure, performed starting from the DNA extraction from the Cms bacterial culture, has to be done experimentally in series of experiments.
The electrophoretic patterns obtained in PCR MP for the BamHI enzyme are good example of the impact of the denaturation temperature on the selective amplification of the discriminatory bands. Approximately 5.900 restriction fragments are created by BamHI digestion of Cms genome Table 3. Applying gradient 90- 92 °C allowed us to amplify limited number of DNA particles out of 5.900 restriction fragments and obtain at least nine distinct clusters or unique electrophoretic patterns (Fig. 4) and in the gradient 91- 93 °C twelve distinct clusters or unique electrophoretic patterns were obtained (Fig. 5). In denaturation temperature 91.8 °C (Fig. 4) less bands were present in the electrophoretic patterns and in Tm 92.8 °C (Fig. 5) more bands were present. We have not performed direct evaluation of PCR MP against another genotyping or DNA fingerprinting method. However, the restriction endonuclease BamHI was used in PCR MP by another research group investigating C. michiganensis subsp. sepedonicus, also (Żaczek et al. 2018). The level of discrimination obtained by PCR MP in the latter study was higher (11 clusters in a set of 93 isolates) than the discrimination level achieved by application of VNTR analysis (7 clusters in the set of 93 isolates) which was considered, by the authors, efficient discrimination (Żaczek et al. 2018). We obtained at least 9 unique electrophoretic patterns in BamHI PCR MP in single denaturation temperature of 91.8 °C in the gradient 90 °C – 92 °C. Using in another denaturation temperature gradient 91 °C – 93 °C, in temperature 92.8 °C, 12 unique electrophoretic patterns or clusters were obtained. Thus, both sets of experiments performed by us and those performed by Żaczek et al. 2018 proved that BamHI PCR MP allowed discrimination of C. michiganensis subsp. sepedonicus into clusters. We and Żaczek et al. 2018 used distinct oligonucleotides sets and temperature gradients yet we obtained similar results regarding the achieved discriminatory power of the BamHI PCR MP assay. Other enzymes used in the present study enabled obtaining electrophoretic patterns that differentiated the isolates and/or groups of isolates, as well. For BamH1 PCR MP in denaturation temperature 92.8 °C 12 distinct electrophoretic patterns were obtained for 50 isolates analyzed and for the same group of isolates 26 distinct electrophoretic patterns were obtained in XmaI PCR MP which corresponds to approximately 2 fold higher discriminatory power of XmaI PCR MP compared to BamHI PCR MP. All electrophoretic patterns obtained in PCR MP performed for all five restriction enzymes applied were used to construct a dendrogram (Fig. 7). It should be noted that the dendrograms for the PCR MP results served as a tool to visualize the degree of genetic differentiation of the isolates and as a tool for epidemiological studies, exclusively. Differences in the electrophoretic patterns obtained for all variants of PCR MP produced a unique combination of electrophoretic patterns for each of the 50 Cms isolates. This finding means that the ability to differentiate a single isolate within a group of isolates has been achieved which, to the best of our knowledge, has been reported for C. michiganensis subsp. sepedonicus isolates by the first time. It should also be noted that the degree of differentiation was low, and for most of the isolates, the degree of similarity was 95% or higher. However, groups (clusters) of C. michiganensis subsp. sepedonicus isolates could be distinguished with every restriction nuclease applied to PCR MP just the discriminatory power varied depending on the restriction endonuclease used.
Of course, the summary analysis was based on the combination of results of series individual electrophoreses, which increases the noise that can sometimes significantly affect the quality of the results. However, this problem applies to many bacterial genotyping techniques, such as the gold standard in typing many pathogens - PFGE, which was also used in the C. michiganensis subsp. sepedonicus genotyping apart from BOX-, ERIC- or Rep-PCR techniques.
We have determined that the oligonucleotides used for HindIII PCR MP of Enetrobacteriaceae (Masny and Płucienniczak 2003) allowed reproducible, theoretically predictable differentiation of C. michiganensis subsp. sepedonicus isolates (Fig. 2). Other authors described HindIII enzyme as unsuitable for PCR MP analysis of Cms strains (Żaczek et al. 2018). The PCR MP results obtained with HindIII enzyme were exceptional because in the size range between 100 bp and 4000 bp maximum of four PCR products were amplified and observed as discrete bands after electrophoresis (Fig. 3). According to our analysis, except for HindIII, all restriction enzymes used in PCR MP produced hundreds or thousands (from 1282 to 5994) of fragments in the size range between 6 and 4000 bp (Table 3) and HindIII produced just 5 DNA fragments in this size range including two same size fragments (Table 3). Therefore, in case of HindIII PCR MP reduction of denaturation temperature was not necessary to obtain limited number of PCR products analysable by gel electrophoresis. The results of the bioinformatic analysis, in-silico digestion of the genomes of the reference strains of C. michiganensis subsp. sepedonicus, matched the results we obtained; most strains we investigated produced electrophoretic patterns with four bands in HindIII PCR MP (Fig. 2) whose size matched the theoretically predicted restriction fragments obtained from the reference genome (NCBI Reference Sequence: NC_010407.1): 3428 bp (two fragments of the same size), 1728 bp, 945 bp and 608 bp. In-silico restriction digestion with HindIII revealed that only 28 fragments were created by the enzyme from entire NCBI Reference Sequence: NC_010407.1. Thus, the theoretical predictions matched our results. We analysed another full genome sequence of C. michiganensis subsp. sepedonicus strain CFIA-Cs3N (NZ_MZMM01000001.1) and in this genome 26 fragments in total were created and the following DNA fragments shorter than 4000 bp were obtained: 3428 (two fragments of the same size), 1728 bp and 608 bp. Thus, three PCR products would be visible on the electrophoretic pattern matching the sizes of PCR products obtained for isolates 25 and 8076 (Fig. 2). Concordant results of the in silico analyses and the results of the experiments confirm that the differences in the electrophoretic patterns correspond to differences in the genomic sequences. We cloned and sequenced two (approximately 600 bp an approximately 1000 bp) out of four PCR products obtained by PCR MP after digestion of isolate CL 511 with HindIII and the obtained sequences were identical to the 608 bp sequence region 1,791,286–1,791,893 and 945 bp region 3,116,062–3,117,006 of the Clavibacter michiganensis subsp. sepedonicus complete genome (NCBI Reference Sequence: NC_010407.1). Thus both the sizes of PCR MP products and the sequences were in agreement with the predictions based on the available genomic data. The most unique electrophoretic patterns were obtained for isolates CL 622 and 4 K in HindIII PCR MP (Fig. 2). Thus, the genomes of those isolates differ from the reference genomes described above and could be interesting objects for further analyses. Despite the low number of electrophoretic bands produced HindIII PCR MP proved to be useful in detection of polymorphisms in the Cms reference genomes and the genomes of the investigated Cms isolates.
In the presented analysis, the lowest degree of similarity between the groups of isolates was 82.2%, which is still a high level of similarity. In our study, PCR MP allowed the differentiation of the isolates of C. michiganensis subsp. sepedonicus both from the same and distant geographical regions. Two of the least similar isolates, P1 and CL 622, were isolated in the same region of Poland (Wielkopolska) in 2006 and 2010, respectively. The ability to differentiate isolates from the same region or different regions has important practical consequences because it allows us to conclude that a developed set of PCR MP tests might be applied in epidemiological studies. Other authors also considered PCR MP a tool suitable for epidemiological analyses of C. michiganensis subsp. sepedonicus (Żaczek et al. 2018). Using the developed PCR MP tests with a set of five distinct restriction enzymes, we could determine whether any two isolates were genetically distinct or not in contrast to differentiation of C. michiganensis subsp. sepedonicus isolates by single restriction enzyme PCR MP analysis combined with VNTR analysis allowing discrimination of isolate clusters, only (Żaczek et al. 2018). The lack of differences does not determine the identity of the compared isolates, but the occurrence of the difference confirms the distinctness, which is important when a proof is necessary that the source of infection in a given region was not an isolate from another region. Reproducibility is always an important issue in bacterial isolate differentiation methods. Larger number of obtained bands in an electrophoretic profile increases the noise of the analysis and contributes to the possibility of ambiguous interpretation of the patterns of bands, which is known phenomenon common for the DNA fingerprinting methods (Koopman 2005). Reproducibility of PCR MP in C. michiganensis subsp. sepedonicus isolate differentiation was confirmed by other authors (Żaczek et al. 2018).
Based on the obtained results, we may claim that the designed PCR MP tests are currently the most efficient PCR based method for genetic differentiation isolates of C. michiganensis subsp. sepedonicus. In addition, despite the very low diversity among Cms individual isolates, these can be unambiguously identified within this subspecies. It is probable that PCR MP can allow the identification of genetic markers, enabling the differentiation of Cms isolates by cloning and sequencing of the isolate differentiating DNA bands. The differentiation of Cms isolates and the detection of unique DNA polymorphisms in their genome can be highly important in epidemiological studies, in which it would be possible to track the spread of a particular strain. Integrated diagnostics based on the combination of biochemical and genetic features would help in achieving better diagnosis of quarantined plant organisms.
References
Brown, S.E., A.A. Reilley, D.L. Knudson, and C.A. Ishimaru. 2002. Genomic fingerprinting of virulent and avirulent strains of Clavibacter michiganensis subspecies sepedonicus. Current Microbiology 44: 112–119.
Bultreys, A., and M. Kałużna. 2010. Bacterial cankers caused by Pseudomonas syringae on stone fruit species with special emphasis on the pathovars syringae and monsprunorum race 1 nad race 2. Journal of Plant of Pathology 92 (1): 21–33.
Carlson, R.R., and A.K. Vidaver. 1982. Taxonomy of Corynebacterium plant pathogens,including a new pathogen of wheat, based on polyacrylamide gel electrophoresis of cellular proteins. International Journal of Systematic Bacteriology 32: 315–326.
Commission Directive 2006/56 / EC of 12 June. 2006. Amending the Annexes to Council Directive 93/85 / EEC on the control of potato ring rot.
Davis, M.J., A.G. Gillaspie Jr., A.K. Vidaver, and R.W. Harris. 1984. Clavibacter: A new genus containing some phytopathogenic coryneform bacteria, including Clavibacter xyli subsp. xyli sp. nov., subsp. nov. and Clavibacter xyli subsp. cynodontis subsp. nov., pathogens that cause ratoon stunting disease of sugarcane and Bermudagrass stunting disease. International Journal of Systematic Bacteriology 34: 107–117.
De Boer, S.H., and M. McCann. 1990. Detection of Corynebacterium sepedonicum in potato cultivars with different propensities to express ring rot symptoms. Am. Potato J. 67: 685–694.
De León, L., L. Rodríguez, A. Llop, P. López, and F. Siverio. 2009. Comparative study of genetic diversity of Clavibacter michiganensis subsp. michiganensis isolates from the Canary Islands by RAPD-PCR, BOX-PCR and AFLP. Plant Pathology 58: 862–871.
Drennan, J., A.A.G. Westra, S.A. Slack, L.M. Delserone, A. Collmer, N.C. Gumestas, and A.E. Oleson. 1993. Comparison of DNA probe and ELISA for the detection of Clavibacter michiganensis subsp. sepedonicus in field – Grown potatoes. Plant Disease 77: 1243–1247.
Dybczynski, I., and A. Plucienniczak. 1988. A protocol for DNA fragment extraction from polyacrylamide gels. Biotechniques 6 (10): 924–926.
Fessehaie, A., S.H. De Boer, and C.A. Lévesque. 2003. An oligonucleotide array for the identification and differentiation of bacteria pathogenic on potato. Phytopathology 93 (3): 262–269.
Fousek, J., and I. Mraz. 2003. Determination of genetic differences between fluidal and nonfluidal variants of Clavibacter michiganensis subsp. sepedonicus using rep-PCR Technigue. Folia Microbiologica 48 (5): 682–686.
Fousek, J., I. Mraz, and K. Pietrzik. 2002. Comparison of genetic variability between Czech and foreign isolates of Phytopathogenic Bacteria Clavibacter michiganensis subsp. sepedonicus by rep-PCR technique. Folia Microbiologica 47 (4): 450–454.
Gartemann KH, Abt B, Bekel T, Burger A, Engemann J, Flügel M, Gaigalat L, Goesmann A, Gräfen I, Kalinowski J, Kaup O, Kirchner O, Krause L, Linke B, McHardy A, Meyer F, Pohle S, Rückert C, Schneiker S, Zellermann EM, Pühler A, Eichenlaub R, Kaiser O, Bartels D. 2008. The genome sequence of the tomato-pathogenic actinomycete Clavibacter michiganensis subsp. michiganensis NCPPB382 reveals a large island involved in pathogenicity. Journal of Bacteriology 2008 Mar;190(6): 2138–2149. https://doi.org/10.1128/JB.01595-07.
Gudmestad, N.C., I. Mallik, J.S. Pasche, N.R. Anderson, and K. Kinzer. 2009. A real-time PCR assay for the detection of Clavibacter sequence. Plant Disease 93: 649–659.
Kałużna, M., P. Albuquerque, F. Tavares, P. Sobiczewski, and J. Pulawska. 2016. a. Development of SCAR markers for rapid and specific detection of Pseudomonas syringae pv. morsprunorum races 1 and 2, using conventional and real-time PCR. Applied Microbiology and Biotechnology 100 (8): 3693–3711.
Kałużna, M., J. Pulawska, and P. Sobiczewski. 2010. The use of PCR melting profile for typing of Pseudomonas syringae isolates from stone fruit trees. Europne Journal of Plant Pathology 126 (4): 437–443.
Kałużna, M., A. Willems, J.F. Pothier, M. Ruinelli, P. Sobiczewski, and J. Pulawska. 2016b. Pseudomonas cerasi sp nov (non griffin, 1911) isolated from diseased tissue of cherry. Systematic and Applied Microbiology 39 (6): 370–377.
Krawczyk, B., J. Leibner Ciszak, A. Mielech, M. Nowak, and J. Kur. 2009. PCR melting profile (PCR MP) - a new tool for differentiation of Candida albicans strains. BMC Infectious Diseases 9: 177.
Krawczyk, B., J. Leibner, W. Barańska-Rybak, A. Samet, R. Nowicki, and J. Kur. 2007. ADSRRS-fingerprinting and PCR MP techniques for studies of intraspecies genetic relatedness in Staphylococcus aureus. Journal of Microbiological Methods 71: 114–122.
Lee, I.M., I.M. Bartoszyk, D.E. Gundersen-Rindal, and R.E. Davis. 1997. Phylogeny and classification of bacteria in the genera Clavibacter and Rathayibacter on the basis of 16S rRNA gene sequence analyses. Applied and Environmental Microbiology 63: 2631–2636.
Li, X., and S.H. De Boer. 1995a. Comparison of 16S ribosomal RNA genes in Clavibacter michiganensis subspecies with other coryneform. Canadian Journal of Microbiology 41: 925–929.
Li, X., and S.H. De Boer. 1995b. Selection of polymerase chain reaction primers from an RNA intergenic spacer region for specific detection of Clavibacter michiganensis subsp. sepedonicus. Phytopathology 85: 837–842.
Li, X., J. Tambong, K.X. Yuan, W. Chen, H. Xu, C.A. Lévesque, and S.H. De Boer. 2018. Re-classification of Clavibacter michiganensis subspecies on the basis of whole-genome and multi-locus sequence analyses. International Journal of Systematic and Evolutionary Microbiology 68: 234–240.
Louws, F.J., J. Bell, C.M. Medina-Mora, C.D. Smart, D. Opgennorth, C.A. Ishimary, M.K. Hausbeck, F.J. de Bruijn, and D.W. Fulbright. 1998. Rep - PCR – Mediated genomic fingerprinting: A rapid and effective method to identify Clavibacter michiganensis. Phytopathology 88: 862–868.
Koopman, W.J. 2005 Apr. Phylogenetic signal in AFLP data sets. Systematic Biology 54 (2): 197–217.
Krystynowicz A., Koziołkiewicz M., Wiktorowska-Jezierska A., Bielecki S., Klemenska E., Masny A., Płucienniczak, A. 2005. Molecular basis of cellulose biosynthesis disappearance in submerged culture of Acetobacter xylinum. Acta Biochimica Polonica 52: 691–698).
Masny, A., and A. Płucienniczak. 2003. Ligation mediated PCR performed at low denaturation temperatures - PCR melting profiles. Nucl. Acids Res. 31: 114.
Milijašević-Marčić S., Gartemann K.H., Frohwitter J., Eichenlaub R., Todorović B., Rekanović E., Potočnik I. 2012. European Journal Plant Pathology 2012 Apr; 134: (4) 697–711. https://doi.org/10.1007/s10658-012-0046-x.
Mirza, M.S., J.L.W. Rademaker, J.D. Janse, and A.D.L. Akkermans. 1993. Specific 16S ribosomal RNA targeted oligonucleotide probe against Clavibacter michiganensis subsp. sepedonicus. Canadian Journal of Microbiology 39 (11): 1029–1034.
Mogen, B.D., H.R. Olson, R.B. Sparks, N.C. Gudmestad, and A.E. Oleson. 1990. Genetic variation in strains of Clavibacter michiganense subsp. sepedonicum: Polymorphisms in restriction fragments containing a highly repeated sequence. Phytopathology 80: 90–93.
Mogen, B.D., N.C. Gudmestad, and A.E. Oleson. 1987. Evaluation of a cloned sequence from Corynebacterium sepedonicum for use as a pathogen - specific DNA hybridization probe. Phytopathology 77: 1693.
Mogen, B.D., A.E. Oleson, and G.A. Secor. 1985. Presence of a unique plasmid common to several isolates of Corynebacterium sepedonicum. Phytopathology 75: 1378.
Olczak-Woltman, H., A. Masny, G. Bartoszewski, A. Płucienniczak, and K. Niemirowicz-Szczytt. 2007. Genetic diversity of Pseudomonas syringae pv. lachrymans strains isolated from cucumber leaves collected in Poland. Plant Pathology 56 (3): 373–382.
Olsson K. 1976. Experience of ring rot caused by Corynebacterium sepedonicum (Spieck. Et Kotth.) Skapt. Et Burkh. In Sweden. Particularly detection of the disease in its latent form. EPPO bulletin [Olsson, K. : Experience of brown rot caused by Pseudomonas solanacearum (Smith) Smith in Sweden] 6 (4): 209–219.
Palomo, J.L., E. Velasquez, P.F. Mateos, P. Garcia-Benavides, and E. Martinez-Molina. 2000. Rapid identification of Clavibacter michiganensis subspecies sepedonicus based on the stable low molecular weight RNA (LMW RNA) profiles. European Journal of Plant Pathology 106: 789–793.
Pastrik, K.H. 2000. Detection of Clavibacter michiganensis subsp. sepedonicus in potato tubers by multiplex PCR with coamplification of host DNA. European Journal of Plant Pathology 106: 155–165.
Pastrik, K.H., and F.A. Rainey. 1999. Identification and differentiation of Clavibacter michiganensis subspecies by polymerase chain reaction-based techniques. Journal of Phytopathology 147: 687–693.
Smith J.M., Mc Namara D.G., Scott P.R., Holderness M., Burger B. 1997. Clavibacter michiganensis subsp. sepedonicus. In: Quarantine Pests for Europe, CAB International (ed.), Univ. Press, Cambridge UK: 986–990.
Smith, N.C., J. Hennessy, and D.E. Stead. 2001. Repetitive sequence – Derived PCR profiling using the BOX-A1R primer for rapid identification of the plant pathogen Clavibacter michiganensis subspecies sepedonicus. European Journal of Plant Pathology 107: 739–748.
Snieszko, S.F., and R. Bonde. 1943. Studies on the morphology, physiology, serology, longevity and pathogenicity of Corynebacterium sepedonicum. Phytopathology 33: 1032–1044.
Stead D. and Janse J. D. 2000. Protocol for the diagnosis of quarantine organism Clavibacter michiganensis subsp. sepedonicus.
Stojowska, K., B. Krawczyk, S. Kałuzewski, and J. Kur. 2009. Retrospective analysis of the genetic diversity of Klebsiella oxytoca isolated in Poland over a 50-year period. European Journal of Clinical Microbiology & Infectious Diseases 10: 1263–1266. https://doi.org/10.1007/s10096-009-0768-7.
Van der Wolf, J.M., J.R. Van Beckhoven, A. Hukkanen, R. Karjalainen, and P. Mủller. 2005. Fate of the casual organism of Clavibacter michiganensis spp. sepedonicus bacteria ring rot of potato, in weeds and field crops. J. Phytopathology 153: 358–365.
Vantomme, R., M. Baetens, E. De Bruyne, J. Van Vaerenbergh, and J. De Ley. 1987. Characterization of Corynebacterium sepedonicum by API ZYM and SDS protein polyacrylamide gel electrophoresis. Verh. Fac. Landbouwwet. Gent. 52: 1089–1093.
Waleron, M., K. Waleron, J. Kamasa, W. Przewodowski, and E. Łojkowska. 2011. Polymorphism analysis of housekeeping genes for identification and differentiation of Clavibacter michiganensis subspecies. European Journal of Plant Pathology 131: 341–354.
Żaczek, A., A. Sokołowska, K. Struś, and P. Parniewski. 2018. Differentiation of Clavibacter michiganensis subsp. sepedonicus using PCR MP (PCR melting profile) and VNTR (variable number of tandem repeat) methods. Letters in Applied Microbiology 68: 24–30. https://doi.org/10.1111/lam.13081.
Acknowledgments
We thank Edyta Sobczak (Plant Breeding and Acclimatization Institute - National Research Institute, Poland) for preparing the Cms isolates for analysis.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Węgierek-Maciejewska, A., Masny, A., Wołkowicz, T. et al. PCR MP Method for Differentiation of Clavibacter michiganensis Subsp. sepedonicus Isolates. Am. J. Potato Res. 96, 588–604 (2019). https://doi.org/10.1007/s12230-019-09752-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12230-019-09752-5