Abstract
In the present work, we characterized in detail strain CM-3-T8T, which was isolated from the rhizosphere soil of strawberries in Beijing, China, in order to elucidate its taxonomic position. Cells of strain CM-3-T8T were Gram-negative, non-spore-forming, aerobic, short rod. Growth occurred at 25–37 °C, pH 5.0–10.0, and in the presence of 0–8% (w/v) NaCl. Phylogenetic analysis based on 16S rRNA gene sequences revealed that strain CM-3-T8T formed a stable clade with Lysobacter soli DCY21T and Lysobacter panacisoli CJ29T, with the 16S rRNA gene sequence similarities of 98.91% and 98.50%. The average nucleotide identity and digital DNA-DNA hybridization values between strain SG-8 T and the two reference type strains listed above were 76.3%, 79.6%, and 34.3%, 27%, respectively. The DNA G + C content was 68.4% (mol/mol). The major cellular fatty acids were comprised of C15:0 iso (36.15%), C17:0 iso (8.40%), and C11:0 iso 3OH (8.28%). The major quinone system was ubiquinone Q-8. The major polar lipids were phosphatidylethanolamine (PE), phosphatidylethanolamine (PME), diphosphatidylglycerol (DPG), and aminophospholipid (APL). On the basis of phenotypic, genotypic, and phylogenetic evidence, strain CM-3-T8T (= ACCC 61714 T = JCM 34576 T) represents a new species within the genus Lysobacter, for which the name Lysobacter changpingensis sp. nov. is proposed.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The genus Lysobacter, in the class Gammaproteobacteria and family Xanthomonadaceae, was first described by Christensen and Cook (1978) and emended by Park et al. (2008). It is non-fruiting, Gram-stain-negative, aerobic, gliding nature of bacteria with high DNA G + C contents (61.7–70.7% mol/mol), contain ubiquinone Q-8 as the major respiratory quinone and the major polar lipids are diphosphatidylglycerol (DPG), phosphatidylglycerol (PG), and phosphatidylethanolamine (PE). At present, there are 70 species with a validly published and correct name in the genus Lysobacter recorded on LPSN (https://lpsn.dsmz.de/genus/Lysobacter; Feb 2023). Lysobacter strains are ubiquitously distributed in various environments, most of them were isolated from Antarctic coastal sediment (Liu et al. 2022), soil (Srinivasan et al. 2009; Coil et al. 2014; Gross et al. 2016; Zhang et al. 2019), plant rhizosphere (Xiao et al. 2019), feces (Lee et al. 2022), sludge (Ye et al. 2015), estuary (Sang et al. 2016), spongin (Choi et al. 2018), and freshwater (Siddiqi and Im 2016). In this paper, the authors report a novel bacterial strain CM-3-T8T, which was isolated from the rhizosphere soil of strawberry. Analysis of the phylogenetic and phenotypic characteristics confirmed that strain CM-3-T8T represents a novel species within the genus Lysobacter, for which the name Lysobacter changpingensis sp. nov. is proposed.
Materials and methods
Isolation and ecology
Rhizosphere soil samples of strawberries were collected in the Changping district, Beijing, China (116° 20′ E, 40° 22′ N). For isolation of bacteria, the samples were suspended in sterile water and serially diluted to 10−5, 10−6, and 10−7, then 100 µL from each dilution was spread on TSA plates. The TSA medium contained (per liter): tryptone 15 g, soy peptone 5 g, sodium chloride 5 g, pH 7.0. After 3 days of incubation at 30 °C, a colony was subcultivated on TSA medium and named CM-3-T8T to taxonomic characterization. The strain CM-3-T8T was maintained on glycerol (w/v) and stored at −80 °C.
16S RNA phylogeny
Genomic DNA of CM-3-T8T was extracted using a DNA extraction kit (Biotech) by following the manufacturer’s instructions. PCR amplification of the 16S rRNA gene was performed with the universal primers 27F (5′-GAGTTTGATCCTGGCTCAG-3′) and 1492R (5′-ACGGCTACCTTGTTACGACTT-3′) (Farris and Olson 2007). PCR conditions were an initial denaturation step at 94 °C for 2 min followed by 35 cycles of 95 °C for 30 s denaturing, 55 °C for 30 s annealing, and 72 °C for 30 s followed by a 10 min final extension at 72 °C. Purified PCR products were sequenced by the Sangon Biotech (Shanghai, China; http://www.sangon.com/). We used NCBI’s BLAST search (http://www.ncbi.nlm.nih.gov/blast) and the EzTaxone server (www. ezbiocloud.net) to identify phylogenetic neighbors and calculate pairwise sequence similarities. Then, phylogenetic trees were reconstructed using the software MEGA 7.0 and based on maximum-likelihood (Felsenstein 1981), neighbor-joining (Saitou and Nei 1987), and minimum-evolution (Rzhetsky and Nei 1992) models with bootstrap values under 1000 replications (Mikkel 2016). Distances were calculated according to Kimura’s two-parameter model (1980).
Physiology and chemotaxonomy
For this part of the study, cells are cultured in TSA medium under aerobic conditions, and all data presented are the average of three replicates. Cell morphology and size was observed by a transmission electron microscope at 30 k magnification (Hitachi, Model H-7500, acceleration voltage 80 kV). Gram staining was performed by Beveridge (2001). The optimal pH and temperature for growth of strain CM-3-T8T and the reference strains Lysobacter soli DCY21T and Lysobacter panacisoli CJ29T were determined by incubating the strains on TSA medium at different temperatures (4, 25, 30, 37, 42, and 45 °C), different pH levels (pH 4.0, 5.0, 6.0, 7.0, 8.0, 9.0, 10.0; acetate buffer was used for pH 4.0–7.0 and phosphate buffer was used for pH 7.0–10.0), and different NaCl levels with 0–10.0% (w/v) NaCl (1% increments). All the growth of the bacterium was determined by measuring the A600 (infinite M200PRO, TECAN) of the cultures after 5 days, except for the temperatures of 4 and 10 °C, which was assessed after 10 days. Enzyme activities were assayed using the API ZYM and API 20NE systems. Additional biochemical tests were determined using the API 50CH system and the Bio GN2 microplate according to the manufactures’ instructions.
For assaying the differences of fatty acid composition between the strain CM-3-T8T and the most closely related species, strain CM-3-T8T, Lysobacter soli DCY21T, and Lysobacter panacisoli CJ29T were used. The strains were cultured under aerobic conditions on TSA medium at 30 °C for 48 h. Fatty acid methyl esters were prepared and identified with the MIDI Sherlock Microbial Identification System (Sherlock version 6.1).
The polar lipids were extracted from 1 g freeze-dried cells using methanol/chloroform/saline extraction (2:1:0.8 ratio by vol.), as described by Kates (1972). Two-dimensional chromatography on a silica gel thin-layer chromatography (TLC) plate (10 × 10 cm) was used to separate and identify polar lipids, as described by Raj et al. (2013). Total polar lipids were detected by spraying with 10% ethanolic molybdophosphoric acid solution (Sigma-Aldrich) followed by heating at 150 °C for 10 min, and further characterized by spraying with ninhydrin, molybdenum blue (specific for phosphates), and Dragendorff’s reagent. The quinones were isolated according to the methods of Minnikin et al. (1984) and determined using HPLC (Kroppenstedt 1982).
Genome features
The High Pure PCR Template Preparation kit (Roche) was employed for isolation of genomic DNA for whole-genome sequencing and DNA-DNA hybridization experiments. The genome of strain CM-3-T8T was sequenced at Sistemas Genomicos (Valencia, Spain) using Illumina paired-end sequencing technology. The reads were trimmed using Trimmomatic 0.32 (Bolger et al. 2014). Genome assembly was performed using SPAdes 3.6.1 (Nurk et al. 2013). The average nucleotide identity blast (ANIb) values were calculated as described by Richter and RossellóMóra (2009) using JSpecies (version 1.2.1) and Lee et al. (2016). The G + C content of chromosomal DNA was calculated on the basis of its whole-genome sequence. The estimated DNA-DNA hybridization (dDDH) value was determined among these strains using the Genome-to Genome Distance Calculator (version 2.1) (Auch et al. 2010; Meier-Kolthoff et al. 2013).
Results and discussion
Molecular phylogenetic analysis
The 16S rRNA gene sequence (1408 bp) used NCBI’s BLAST search (http://www.ncbi.nlm.nih.gov/blast) and the EzTaxone server (www.ezbiocloud.net) to identify phylogenetic neighbors and calculate pairwise sequence similarities. Lysobacter soli DCY21T and Lysobacter panacisoli CJ29T exhibited the greatest similarity to the strain CM-3-T8T (98.84% and 98.44% identities). The 16S rRNA gene sequence was deposited in the GenBank/EMBL/DDBJ database under accession number MW295626. The phylogenetic trees (Figs. 1, S1, S2) demonstrated that strain CM-3-T8T belonged to the genus Lysobacter and formed a cluster with strains Lysobacter soli DCY21T and Lysobacter panacisoli CJ29T and other type strains were in different clades dispersedly in the maximum-likelihood, neighbor-joining, and minimum-evolution trees. In conclusion, Lysobacter soli DCY21T and Lysobacter panacisoli CJ29T were chosen as reference strains for further study.
The maximum-likelihood (ML) tree based on partial 16S rRNA gene (1210 bp) sequence comparison showing the relationships between strain CM-3-T8T and related strains of the family Lysobacter; bootstrap values > 50%, based on 1000 replications, are shown at branch points; bar corresponds to 0.005 substitutions per nucleotide position. Xanthomonas campestris strain LMG 568 T was using as the outgroup
Physiology and chemotaxonomy analysis
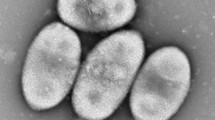
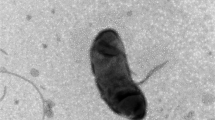
Cells of strain CM-3-T8T were Gram-negative, non-spore-forming, aerobic, short rods (0.4–0.7 μm × 0.8–1.6 μm), commonly observed as single cells under the microscope (Fig. 2). Strain CM-3-T8T can grow at 25–37 °C, pH 5.0–10.0, and in the presence of 0–8% (w/v) NaCl. The results of enzyme activities and other biochemical tests were listed in the species description (Table 1). Although strain CM-3-T8T shared many phenotypic features with closely related taxa Lysobacter soli DCY21T and Lysobacter panacisoli CJ29T, there were some differences among them. Strain CM-3-T8T showed N-acetyl-D-glucosamine, N-acetyl-β-D-mannosamine, L-glutamic acid, and lithium chloride reactions are positive, but L. panacisoli CJ29T and L. soli DCY21T were negative for these characteristics. Whole cell fatty acid analysis revealed that the predominant fatty acids in strain CM-3-T8T were C15:0 iso (36.15%), C17:0 iso (8.40%), and C11:0 iso 3OH (8.28%). These results were in line with other members of the genus Lysobacter. Nevertheless, there were several differences in the proportions of some fatty acids, such as more C15:0 iso and C15:0 anteiso (Table 2). The major polar lipids of strain CM-3-T8T contain phosphatidylethanolamine (PE), phosphatidylethanolamine (PME), diphosphatidylglycerol (DPG), aminophospholipid (APL), small account of phosphatidylmonomethylethanolamine (PL), phosphatidylglycerol (PG), and unknownpolarlipids (Fig. S3). Q-8 was found to be the major quinone, in agreement with other members of the genus Lysobacter (Fig. S4).
Genome features
The DNA G + C content of strain was estimated at 68.4% (mol/mol) according to the draft genome of strain CM-3-T8T, which is in the range of the genus Lysobacter 66.8–72.2% (mol/mol). The estimated DNA-DNA hybridization (dDDH) values for strain CM-3-T8T with strains Lysobacter soli DCY21T and Lysobacter panacisoli CJ29T were 34.3% and 27%, respectively. The average nucleotide identity (ANI) values of CM-3-T8T with the Lysobacter soli DCY21T and Lysobacter panacisoli CJ29T are between 76.3 and 79.6% (Fig. 3). The average nucleotide identity and in silico estimated DNA-DNA reassociation values among strain CM-3-T8T, strains Lysobacter soli DCY21T and Lysobacter panacisoli CJ29T were in all cases below the respective threshold for species differentiation (95–96% for ANI, 70% for dDDH) (Lee et al. 2016), suggesting that strains be proposed as a novel species of the genus Lysobacter. The whole-genome phylogenetic tree was constructed with other eleven publicly available Lysobacter species genomes (Fig. 4), showing the close phylogenetic relationship between closely related type strains Lysobacter panacisoli CJ29T, Lysobacter soli DCY21T and strain CM-3-T8T, suggesting that strain CM-3-T8T was affiliated to the genus Lysobacter. The genome of strain CM-3-T8T was sequenced and compared to two reference genomes of Lysobacter species (Table 3). These genomic features can distinguish strain CM-3-T8T from its closely related strains.
Lysobacter spp. are widely distributed and exhibit remarkable bactericidal activity against a wide range of phytopathogenic fungi, bacteria, and nematodes. The vast majority of Lysobacter spp. adapt to the soil environment; among the 70 species of the genus, 56 species were isolated from soil. These soil types include greenhouse soil with relatively high humidity (Weon et al. 2006), dry soil with strong ultraviolet radiation (Zhang et al. 2011), saline-alkali soil (Xu et al. 2020), oil-contaminated soil (Chaudhary et al. 2017), and alpine forest soil (Margesin et al. 2018), indicating that Lysobacter bacteria have a wide range of adaptability in soil. The strain CM-3-T8T in this study was also isolated from soil. Unlike other published strains of the genus Lysobacter, strain CM-3-T8T can utilize multiple carbon sources, such as N-acetyl-D-glucosamine, N-acetyl-β-D-mannosamine, 1% sodium lactate, L-fucose, L-glutamic acid, guanidine HCl, lithium chloride, and D-fucose, which contributes to its distribution. N-acetyl-D-glucosamine is one of the monomers of chitin, glycosaminoglycans, and glycoproteins and plays an important role in the formation of microbial cell walls (Mobley et al. 1982). Strain CM-3-T8T exhibits the ability to oxidize N-acetyl-D-glucosamine, which may contribute to its antibacterial activity. Many reported Lysobacter spp. have demonstrated significant antagonistic effects against a variety of pathogens. In our future work, we will study and evaluate the biocontrol potential and mechanism of strain CM-3-T8T. We will assess its biocontrol potential under different environmental conditions, including its ability to inhibit various plant pathogens and promote crop growth. Our studies will help to uncover the biocontrol potential and mechanism of CM-3-T8T, providing a theoretical basis and technical support for its application in agricultural production.
Conclusion
In conclusion, the characteristics of the novel species are consistent with the description of the genus Lysobacter according to morphological, biochemical, and chemotaxonomic properties, but there are several differences between CM-3-T8T and other published members of the genus Lysobacter. Phylogenetic and chemotaxonomic analyses demonstrate that strain CM-3-T8T represents a novel species within the genus Lysobacter. The genome sequence of strain CM-3-T8T was deposited in the GenBank/EMBL/DDBJ database under accession number GCA_017308985.
Description of Lysobacter changpingensis sp. nov
Lysobacter changpingensis
(chang.ping.en’sis. N.L. masc. adj. changping of a district in Beijing of China, where the type strain was isolated).
Cells are Gram-negative, non-spore-forming, aerobic, short rods, commonly observed as single cells under the microscope. Lysobacter changpingensis can grow at 25–37 °C (optimum, 30 °C) and at pH 5.0–10.0 (optimum, pH 7.0–9.0). The salt tolerance range for growth is 0–8% (w/v) NaCl. The major cellular fatty acids are comprised of C15:0 iso (36.15%), C17:0 iso (8.40%), and C11:0 iso 3OH (8.28%). Reactions are positive for N-acetyl-D-glucosamine, N-acetyl-β-D-mannosamine, 1% NaCl, D-galactose, L-fucose, 1% sodium lactate, L-glutamic acid, guanidine HCl, D-galacturonic acid, D-glucuronic acid, and lithium chloride in BIOLOG strip; cystinol arylamidase, chymotrypsin, and α-glucosidase in API ZYM strip; D-glucose in API 20NE strip; and D-fucose in API 50CH strip. Resistant to D-fructose-6-PO4, sodium butyrate, β-glucosidase, glucose, geranyl, and 5-keto-gluconate, while weakly sensitive to L-rhamnose, tween 40, trypsin, 4-nitroso-β-D-methyl galactose, gluconate, galactose, and D-lyxose. Strain CM-3-T8T mainly contains phosphatidylethanolamine (PE), phosphatidylethanolamine (PME), diphosphatidylglycerol (DPG), aminophospholipid (APL), small account of phosphatidylmonomethylethanolamine (PL), unknownpolarlipids (L), and phosphatidylglycerol (PG). The major quinone system is ubiquinone Q-8.
The type strain of Lysobacter changpingensis is CM-3-T8T (= ACCC 61731 T = JCM 33722 T), isolated from the rhizosphere of strawberry in Changping district, Beijing, China. The DNA G + C content is 68.24% (mol/mol). The 16S rRNA gene sequence is deposited in the GenBank/EMBL/DDBJ database under accession number MW295626. The genome sequence is deposited in the GenBank/EMBL/DDBJ database under accession number GCA_017308985.
Data availability
All data generated during this study are publicly available from the GenBank database at http://www.ncbi.nlm.nih.gov/blast and http://www.ezbiocloud.net.
References
Auch AF, von JM, Klenk HP, Göker M (2010) Digital DNA-DNA hybridization for microbial species delineation by means of genome-to-genome sequence comparison. Stand Genomic Sci 2(117–134):29. https://doi.org/10.4056/sigs.531120
Beveridge T (2001) Use of the Gram stain in microbiology. Biotech Histochem 76(3):111–118
Bolger AM, Lohse M, Usadel B (2014) Trimmomatic: a flexible trimmer for Illumina sequence data. Bioinformatics 30:2114–2120. https://doi.org/10.1093/bioinformatics/btu170
Chaudhary DK, Lee SD, Kim J (2017) Lysobacter olei sp. nov., isolated from oil-contaminated soil. Int J Syst Evol Microbiol 67:4660–4666. https://doi.org/10.1099/ijsem.0.002348
Choi H, Im WT, Park JS (2018) Lysobacter spongiae sp. nov. isolated from spongin. J Microbiol 56(2):97–103. https://doi.org/10.1007/s12275-018-7462-3
Christensen P, Cook FD (1978) Lysobacter, a new genus of nonfruiting, gliding bacteria with a high base ratio. Int J Syst Bacteriol 28:367–393. https://doi.org/10.1099/00207713-28-3-367
Coil D, Jospin G, Darling AE (2014) A5-MiSeq: an updated pipeline to assemble microbial genomes from Illumina MiSeq data. Bioinformatics 31:587–589. https://doi.org/10.1093/bioinformatics/btu661
Farris MH, Olson JB (2007) Detection of Actinobacteria cultivated from environmental samples reveals bias in universal primers. Lett Appl Microbiol 45(4):376–381. https://doi.org/10.1111/j.1472-765X.2007.02198.x
Felsenstein J (1981) Evolutionary trees from DNA sequences: a maximum likelihood approach. J Mol Evol 17:368–376. https://doi.org/10.1007/BF01734359
Gross H, Trappen S-V, Cleenwerck I, Miess H, Vos PD (2016) Reclassification of Pseudomonas sp. PB-6250t as Lysobacter firmicutimachus sp. nov. Int J Syst Evol Microbiol 66(10):4162–4166. https://doi.org/10.1099/ijsem.0.001329
Kates M (1972) Techniques of lipidology: isolation, analysis and identification of lipids. In: Work TS, Work E (eds) Laboratory techniques in biochemistry and molecular biology, vol 3. Elsevier, Amsterdam, pp 269–610
Kimura MA (1980) Simple method for estimating evolutionary rates of base substitutions through comparative studies of nucleotide sequences. J Mol Evol 16(2):111–120
Kroppenstedt RM (1982) Separation of bacterial menaquinones by HPLC using reverse phase (RP18) and a silver loaded ion exchanger as stationary phases. J Liq Chromatogr 5:2359–2367
Lee I, Ouk KY, Park SC, Chun J (2016) OrthoANI: an improved algorithm and software for calculating average nucleotide identity. Int J Syst Evol Microbiol 66:1100–1103. https://doi.org/10.1099/ijsem.0.000760
Lee SY, Kim PS, Sung H, Hyun DW, Bae JW (2022) Lysobacter ciconiae sp. nov., and Lysobacter avium sp. nov., isolated from the faeces of an Oriental stork. J Microbiol 60:469–477. https://doi.org/10.1007/s12275-022-1647-5
Liu ZY, Jiang PQ, Niu GJ, Wang WJ, Li J (2022) Lysobacter antarcticus sp. nov., an SUF-system-containing bacterium from Antarctic coastal sediment. Int J Syst Evol Microbiol 72(2):1466–5034. https://doi.org/10.1099/ijsem.0.005250
Margesin R, Zhang DC, Albuquerque L, Froufe HJC, Egas C, da Costa MS (2018) Lysobacter silvestris sp. nov., isolated from alpine forest soil, and reclassification of Luteimonas tolerans as Lysobacter tolerans comb. nov. Int J Syst Evol Microbiol 68:1571–1577. https://doi.org/10.1099/ijsem.0.002710
Meier-Kolthoff JP, Auch AF, Klenk HP, Göker M (2013) Genome sequence-based species delimitation with confidence intervals and improved distance functions. BMC Bioinformatics 14(60):1471–2105. https://doi.org/10.1186/1471-2105-14-60
Mikkel S (2016) Adapter Removal v2: rapid adapter trimming, identification, and read merging. BMC Res Notes 9:88. https://doi.org/10.1186/s13104-016-1900-2
Minnikin DE, O’Donnell AG, Goodfellow M, Alderson G, Athalye M, Schaal A, Parlett JH (1984) An integrated procedure for the extraction of bacterial isoprenoid quinones and polar lipids. J Microbiol Methods 2:233–241
Mobley HL, Doyle RJ, Streips UN, Langemeier SO (1982) Transport and incorporation of N-acetyl-D-glucosamine in Bacillus subtilis. J Bacteriol 150(1):8–15. https://doi.org/10.1128/jb.150.1.8-15.1982
Nurk S, Bankevich A, Antipov D, Gurevich A, Korobeynikov A, Lapidus A, Prjibelsky A, Pyshkin A, Sirotkin A, Sirotkin Y (2013) Assembling genomes and mini-metagenomes from highly chimeric reads. Lect N Bioinformat 7821:158–170. https://doi.org/10.1007/978-3-642-37195-0_13
Park JH, Kim R, Aslam Z, Jeon CO, Chung YR (2008) Lysobacter capsici sp. nov., with antimicrobial activity, isolated from the rhizosphere of pepper, and emended description of the genus Lysobacter. Int J Syst Evol Microbiol 58(Pt 2):387–92. https://doi.org/10.1099/ijs.0.65290-0
Raj PS, Ramaprasad EV, Vaseef S, Sasikala C, Ramana C (2013) Rhodobacter viridis sp. nov., a phototrophic bacterium isolated from mud of a stream. Int J Syst Evol Microbiol 63:181–186. https://doi.org/10.1099/ijs.0.038471-0
Richter M, RossellóMóra R (2009) Shifting the genomic gold standard for the prokaryotic species definition. Proc Natl Acad Sci USA 106:19126–19131. https://doi.org/10.1073/pnas.0906412106
Rzhetsky A, Nei M (1992) A simple method for estimating and testing minimum-evolution trees. Mol Biol Evol 9:945–967
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4:406–425. https://doi.org/10.1093/oxfordjournals.molbev.a040454
Sang EJ, Hyo JL, Che OJ (2016) Lysobacter aestuarii sp nov. isolated from estuary sediment. Int J Syst Evol Microbiol 66:1346–1351. https://doi.org/10.1099/ijsem.0.000884
Siddiqi MZ, Im WT (2016) Lysobacter pocheonensis sp. nov., isolated from soil of a ginseng field. Arch Microbiol 198:551–557
Srinivasan S, Kim MK, Sathiyaraj G, Kim HB, Kim YJ, Yang DC (2009) Lysobacter soli sp. nov., isolated from soil of a ginseng field. Int J Syst Evol Microbiol 60(7):1543–1547. https://doi.org/10.1099/ijs.0.016428-0
Weon HY, Kim BY, Baek YK, Yoo SH, Kwon SW, Stackebrandt E, Go SJ (2006) Two novel species, Lysobacter daejeonensis sp. nov. and Lysobacter yangpyeongensis sp. nov., isolated from Korean greenhouse soils. Int J Syst Evol Microbiol 56(Pt 5):947–951. https://doi.org/10.1099/ijs.0.64095-0
Xiao M, Zhou XK, Chen X, Duan YQ, Alkhalifah DHM, Im WT, Hozzein WN, Chen W, Li WJ (2019) Lysobacter tabacisoli sp. nov. isolated from rhizosphere soil of Nicotiana tabacum. Int J Syst Evol 69(7):1875–1880. https://doi.org/10.1099/ijsem.0.003164
Xu L, Huang XX, Fan DL, Sun JQ (2020) Lysobacter alkalisoli sp. nov., a chitin-degrading strain isolated from saline-alkaline soil. Int J Syst Evol Microbiol 70:1273–1281. https://doi.org/10.1099/ijsem.0.003911
Ye XM, Chu CW, Shi C, Zhu JC, He Q, He J (2015) Lysobacter caeni sp. nov., isolated from the sludge of a pesticide manufacturing factory. Int J Syst Evol Microbiol 65(Pt 3):845–850. https://doi.org/10.1099/ijs.0.000024
Zhang L, Bai J, Wang Y, Wu GL, Dai J, Fang CX (2011) Lysobacter korlensis sp. nov. and Lysobacter bugurensis sp. nov., isolated from soil. Int J Syst Evol Microbiol 61(Pt 9):2259–2265. https://doi.org/10.1099/ijs.0.024448-0
Zhang XJ, Yao Q, Wang YH, Yang SZ, Feng GD, Zhu HH (2019) Lysobacter silvisoli sp. nov. isolated from forest soil. Int J Syst Evol Microbiol 69:93–98. https://doi.org/10.1099/ijsem.0.003105
Acknowledgements
The authors are grateful to Hong-Tu Zhu for support with fatty acids detection and polar lipids extracted.
Funding
This research was supported by the Major Science and Technology Special Project of Yunnan Province (202202AE090025), National Nonprofit Institute Research Grant of CAAS (1610132021011; Y2022PT12), and the National Natural Science Foundation of China (31870004).
Author information
Authors and Affiliations
Contributions
D.-J.R., B.-Y.N., F.-B.Z., H.-T.Z., H.-L.W., M.-C.M., and M.G. conceived and supervised the study; H.-L.W. and M.-G. designed the experiments; F.-B.Z., H.-T.Z., and M.-C.M. performed the experiments; D.-J.R. and B.-Y.N. analyzed the data, prepared the figures, and wrote the manuscript; D.-J.R., B.-Y.N., F.-B.Z., H.-T.Z., H.-L.W., M.-C.M., and M.G. edited the manuscript and reviewed the literature. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Niu, BY., Ren, DJ., Zhang, FB. et al. Lysobacter changpingensis sp. nov., a novel species of the genus Lysobacter isolated from a rhizosphere soil of strawberry in China. Folia Microbiol 68, 991–998 (2023). https://doi.org/10.1007/s12223-023-01058-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12223-023-01058-8