Abstract
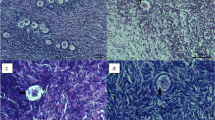
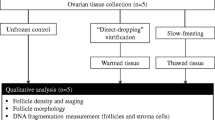
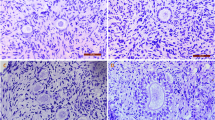
This study was tailored to compare the cryopreservation of the human ovarian cortex using closed metal container vitrification or the slow-freezing technique. Superficial ovarian cortical tissue biopsies were collected from 12 participants who underwent gynaecological videolaparoscopy. The fragmented samples were allocated to three experimental conditions: (a) fresh ovarian tissue, (b) slow-freezing, and (c) vitrification with a metal closed container. After thawing or rewarming, cellular morphological analyses were performed to determine tissue viability. The cellular response to thermal stress was measured by a putative increase in the immune quantification of the heat shock protein 70 kDa (heat shock protein 70 kDa response — HSR) after a heat challenge (2 h exposure at 42 °C). Both the total number of intact follicles and the frequency of primordial follicles were higher in fresh ovarian tissue than in the preserved samples, regardless of the technique employed. There was a trend towards an increase in the absolute number of intact follicles in the tissue preserved by vitrification. After cryopreservation, a higher HSR was obtained after slow-freezing. These results indicate that both cryopreservation techniques present advantages and may be used as alternatives to ovarian tissue cryopreservation.




Similar content being viewed by others
References
Almodin CG et al (2004) Recovery of fertility after grafting of cryopreserved germinative tissue in female rabbits following radiotherapy. Hum Reprod 19:1287–1293. https://doi.org/10.1093/humrep/deh246
Amorim CA, Curaba M, Van Langendonckt A, Dolmans MM, Donnez J (2011) Vitrification as an alternative means of cryopreserving ovarian tissue. Reprod Biomed Online 23:160–186. https://doi.org/10.1016/j.rbmo.2011.04.005
Andersen CY, Bollerup AC, Kristensen SG (2018) Defining quality assurance and quality control measures in connection with ovarian tissue cryopreservation and transplantation: a call to action. Hum Reprod 33:1201–1204. https://doi.org/10.1093/humrep/dey105
Aquino D, Danielli L, Rigon P, Lothhammer N, Frantz N, Bös-Mikich A (2014) Ovarian Tissue Vitrification: the Use of a Novel Metal Closed System for Clinical Grade Cryopreservation. JBRA Assist Reprod 18:12–15
Bielanski A, Bergeron H, Lau PC, Devenish J (2003) Microbial contamination of embryos and semen during long term banking in liquid nitrogen. Cryobiol 46:146–152. https://doi.org/10.1016/s0011-2240(03)00020-8
Bielanski A, Nadin-Davis S, Sapp T, Lutze-Wallace C (2000) Viral contamination of embryos cryopreserved in liquid nitrogen. Cryobiol 40:110–116. https://doi.org/10.1006/cryo.1999.2227
Bös-Mikich A, Aquino D, Danielli L, Rigon P, Lothhammer N, Frantz N (2013) Metal cryovial: a new, promising system for clinical grade vitrification of ovarian tissue. Hum Reprod 28:112–115
Bos-Mikich A, Marques L, Rodrigues JL, Lothhammer N, Frantz N (2012) The use of a metal container for vitrification of mouse ovaries as a clinical grade model for human ovarian tissue cryopreservation after different times and temperatures of transport. J Assist Reprod Genet 29:1267–1271. https://doi.org/10.1007/s10815-012-9867-y
Chen SU, Chien CL, Wu MY, Chen TH, Lai SM, Lin CW, Yang YS (2006) Novel direct cover vitrification for cryopreservation of ovarian tissues increases follicle viability and pregnancy capability in mice. Hum Reprod 21:2794–2800. https://doi.org/10.1093/humrep/del210
Chibelean CB, Petca RC, Radu DC, Petca A (2020) State of the art in fertility preservation for female patients prior to oncologic therapies Medicina (Kaunas) 56 https://doi.org/10.3390/medicina56020089
Dalman A, Deheshkar Gooneh Farahani NS, Totonchi M, Pirjani R, Ebrahimi B, Rezazadeh Valojerdi M (2017) Slow-freezing versus vitrification technique for human ovarian tissue cryopreservation: an evaluation of histological changes. WNT Signaling Pathway and Apoptotic Genes Expression Cryobiology 79:29–36. https://doi.org/10.1016/j.cryobiol.2017.09.007
de Lemos Muller CH et al. (2018) Heat-induced extracellular HSP72 release is blunted in elderly diabetic people compared with healthy middle-aged and older adults, but it is partially restored by resistance training Exp Gerontol 111:180–187:S0531–5565(18)30299–7 (pii) https://doi.org/10.1016/j.exger.2018.07.014
Demeestere I, Simon P, Emiliani S, Delbaere A, Englert Y (2007) Fertility preservation: successful transplantation of cryopreserved ovarian tissue in a young patient previously treated for Hodgkin’s disease. Oncologist 12:1437–1442. https://doi.org/10.1634/theoncologist.12-12-1437
Donnez J et al (2004) Livebirth after orthotopic transplantation of cryopreserved ovarian tissue. Lancet 364:1405–1410. https://doi.org/10.1016/S0140-6736(04)17222-X
Donnez J, Dolmans MM (2010) Cryopreservation and transplantation of ovarian tissue. Clin Obstet Gynecol 53:787–796. https://doi.org/10.1097/GRF.0b013e3181f97a55
Donnez J, Dolmans MM (2013) Fertility Preservation in Women. Nat Rev Endocrinol 9:735–749. https://doi.org/10.1038/nrendo.2013.205
Donnez J, Dolmans MM (2017) The ovary: from conception to death. Fertil Steril 108:594–595. https://doi.org/10.1016/j.fertnstert.2017.08.031
Fabbri R et al (2016) Morphological, ultrastructural and functional imaging of frozen/thawed and vitrified/warmed human ovarian tissue retrieved from oncological patients. Hum Reprod 31:1838–1849
Gougeon A (1996) Regulation of ovarian follicular development in primates: facts and hypotheses. Endocr Rev 17:121–155. https://doi.org/10.1210/edrv-17-2-121
Gosden R (2011) Cryopreservation: a cold look at technology for fertility preservation. Fertil Steril 96:264–268. https://doi.org/10.1016/j.fertnstert.2011.06.029
Haino T et al (2018) Determination of follicular localisation in human ovarian cortex for vitrification J Adolesc Young Adult. Oncol 7:46–53. https://doi.org/10.1089/jayao.2017.0028
Herraiz S, Novella-Maestre E, Rodríguez B, Díaz C, Sánchez-Serrano M, Mirabet V, Pellicer A (2014) Improving ovarian tissue cryopreservation for oncologic patients: slow-freezing versus vitrification effect of different procedures and devices. Fertil Steril 101:775–784. https://doi.org/10.1016/j.fertnstert.2013.11.016
Hovatta O et al (1996) Cryopreservation of human ovarian tissue using dimethylsulphoxide and propanediol-sucrose as cryoprotectants. Hum Reprod 11:1268–1272. https://doi.org/10.1093/oxfordjournals.humrep.a019370
Huang L, Mo Y, Wang W, Li Y, Zhang Q, Yang D (2008) Cryopreservation of human ovarian tissue by solid-surface vitrification. Eur J Obstet Gynecol Reprod Biol 139:193–198. https://doi.org/10.1016/j.ejogrb.2008.03.002
Jensen AK, Macklon KT, Fedder J, Ernst E, Humaidan P, Andersen CY (2017) 86 Successful births and 9 ongoing pregnancies worldwide in women transplanted with frozen–thawed ovarian tissue: focus on birth and perinatal outcome in 40 of these children. J Assist Reprod Genet 34:325–336. https://doi.org/10.1007/s10815-016-0843-9
Klein D, Kern RM, Sokol RZ (1995) A method for quantification and correction of proteins after transfer to immobilisation membranes. Biochem Mol Biol Int 36:59–66
Klocke S, Bündgen N, Köster F, Eichenlaub-Ritter U, Griesinger G (2015) Slow-freezing versus vitrification for human ovarian tissue cryopreservation. Arch Gynecol Obstet 291:419–426. https://doi.org/10.1007/s00404-014-3390-6
Krause M, Gerchman F, Friedman R (2020) Coronavirus infection (SARS-CoV-2) in obesity and diabetes comorbidities: is heat shock response determinant for the disease complications? Diabetol Metab Syndr 12:63. https://doi.org/10.1186/s13098-020-00572-w572[pii]
ESHRE Working Group on Oocyte Cryopreservation in Europe, Shenfield F, de Mouzon J, Scaravelli G, Kupka M, Ferraretti AP, Prados FJ, Goossens V (2017) Oocyte and ovarian tissue cryopreservation in European countries: statutory background, practice, storage and use Hum Reprod Open 2017:hox003 https://doi.org/10.1093/hropen/hox003
Krause M, Heck TG, Bittencourt A, Scomazzon SP, Newsholme P, Curi R, Homem de Bittencourt PI, Jr. (2015) The chaperone balance hypothesis: the importance of the extracellular to intracellular HSP70 ratio to inflammation-driven type 2 diabetes, the effect of exercise, and the implications for clinical management Mediators Inflamm 2015:249205 https://doi.org/10.1155/2015/249205
Krause MS et al (2007) MRP1/GS-X pump ATPase expression: is this the explanation for the cytoprotection of the heart against oxidative stress-induced redox imbalance in comparison to skeletal muscle cells? Cell Biochem Funct 25:23–32. https://doi.org/10.1002/cbf.1343
Madden LA, Sandström ME, Lovell RJ, McNaughton L (2008) Inducible heat shock protein 70 and its role in preconditioning and exercise. Amino Acids 34:511–516. https://doi.org/10.1007/s00726-007-0004-7
Maffei S, Pennarossa G, Brevini TA, Arav A, Gandolfi F (2014) Beneficial effect of directional freezing on in vitro viability of cryopreserved sheep whole ovaries and ovarian cortical slices. Hum Reprod 29:114–124. https://doi.org/10.1093/humrep/det377
Massignam E, Ferreita M, Sanguinet E, Dupont A, Klamt F, Frantz N, Bos-Mikich A (2018) Antioxidant Defence Capacity of Ovarian Tissue after Vitrification in a Metal Closed System. JBRA Assist Reprod 22:199–204
Massignam ET, Ferreira M, Sanguinet E, Dupont Á, Klamt F, Frantz N, Bos-Mikich A (2018) Antioxidant Defense Capacity of Ovarian Tissue after Vitrification in a Metal Closed System. JBRA Assist Reprod 22:199–204. https://doi.org/10.5935/1518-0557.20180044
Meirow D et al. (2005) Pregnancy after transplantation of cryopreserved ovarian tissue in a patient with ovarian failure after chemotherapy. In: N Engl J Med 353. vol 3. United States, pp 318–321. https://doi.org/10.1056/NEJMc055237
Muller CHLM Jr, Grigolo GB, Schroeder HT, Rodrigues-Krause JC, Krause M (2019) Exercise training for the elderly: inflammaging and the central role for HSP70. J Sci Sport and Exercise 1:10–25
Muraro A et al (2017) Precision medicine in allergic disease-food allergy drug allergy and anaphylaxis-PRACTALL document of the European Academy of Allergy and Clinical Immunology and the American Academy of Allergy Asthma & Immunology. Allergy 72:1006–1021. https://doi.org/10.1111/all.13132
Nikishin DA et al (2018) Selection of stable expressed reference genes in native and vitrified/thawed human ovarian tissue for analysis by qRT-PCR and Western blot. J Assist Reprod Genet 35:1851–1860. https://doi.org/10.1007/s10815-018-1263-9
Noble EG, Milne KJ, Melling CW (2008) Heat shock proteins and exercise: a primer. Appl Physiol Nutr Metab 33:1050–1065. https://doi.org/10.1139/H08-069
Oktay K, Bedoschi G, Pacheco F, Turan V, Emirdar V (2016) First pregnancies live birth and in vitro fertilization outcomes after transplantation of frozen-banked ovarian tissue with a human extracellular matrix scaffold using robot-assisted minimally invasive surgery. Am J Obstet Gynecol 214(94):e1-94e9. https://doi.org/10.1016/j.ajog.2015.10.001
Oktay K, Karlikaya G (2000) Ovarian function after transplantation of frozen, banked autologous ovarian tissue. N Engl J Med 342:1919. https://doi.org/10.1056/NEJM200006223422516
Oktem O, Alper E, Balaban B, Palaoglu E, Peker K, Karakaya C, Urman B (2011) Vitrified human ovaries have fewer primordial follicles and produce less antiMüllerian hormone than slow-frozen ovaries. Fertil Steril 95:2661–4.e1. https://doi.org/10.1016/j.fertnstert.2010.12.057
Rall WF (1987) Factors affecting the survival of mouse embryos cryopreserved by vitrification. Cryobiology 24:387–402. https://doi.org/10.1016/0011-2240(87)90042-3
Rivas Leonel EC, Lucci CM, Amorim CA (2019) Cryopreservation of Human Ovarian Tissue: a Review. Transfus Med Hemother 46:173–181. https://doi.org/10.1159/000499054 (tmh-0046–0173 (pii ))
Rosendahl M et al (2011) Cryopreservation of ovarian tissue for a decade in Denmark: a view of the technique. Reprod Biomed Online 22:162–171. https://doi.org/10.1016/j.rbmo.2010.10.015
Sanada Y et al (2019) A Japanese nationwide survey on the cryopreservation of embryos oocytes and ovarian tissue for cancer patients. J J Obstet Gynaecol Res 45:2021–2028. https://doi.org/10.1111/jog.14073
Sheikhi M, Hultenby K, Niklasson B, Lundqvist M, Hovatta O (2011) Clinical grade vitrification of human ovarian tissue: an ultrastructural analysis of follicles and stroma in vitrified tissue. Hum Reprod 26:594–603. https://doi.org/10.1093/humrep/deq357
Shi Q, Xie Y, Wang Y, Li S (2017) Vitrification versus Slow-Freezing for Human Ovarian Tissue Cryopreservation: a Systematic Review and Meta-Anlaysis. Sci Rep 7:8538. https://doi.org/10.1038/s41598-017-09005-7
Silber S (2016) How Ovarian Transplantation Works and How Resting Follicle Recruitment Occurs: a Review of Results Reported from One Center. Womens Health (lond) 12:217–227. https://doi.org/10.2217/whe.15.108
Suzuki N et al (2015) Successful fertility preservation following ovarian tissue vitrification in patients with primary ovarian insufficiency. Hum Reprod 30:608–615. https://doi.org/10.1093/humrep/deu353
Takae S et al (2019) Fertility Preservation for Child and Adolescent Cancer Patients in Asian Countries. Front Endocrinol (lausanne) 10:655. https://doi.org/10.3389/fendo.2019.00655
Takae S, Suzuki N (2019) Current state and future possibilities of ovarian tissue transplantation Reprod. Med Biol 18:217–224. https://doi.org/10.1002/rmb2.12268
Tammiste T et al (2019) A case report and follow-up of the first live birth after heterotopic transplantation of cryopreserved ovarian tissue in Eastern Europe. BMC Womens Health 19:65. https://doi.org/10.1186/s12905-019-0764-8
von Wolff M, Andersen CY, Woodruff TK, Nawroth F (2019) FertiPROTEKT, oncofertility consortium and the Danish fertility-preservation networks—what can we learn from their experiences? Clin Med Insights Reprod Health 13:1179558119845865. https://doi.org/10.1177/1179558119845865
Xiao Z, Zhang Y, Fan W (2017) Cryopreservation of human ovarian tissue using the silver closed vitrification system. J Assist Reprod Genet 34:1435–1444. https://doi.org/10.1007/s10815-017-1004-5
Zhou XH, Wu YJ, Shi J, Xia YX, Zheng SS (2010) Cryopreservation of human ovarian tissue: comparison of novel direct cover vitrification and conventional vitrification. Cryobiology 60:101–105. https://doi.org/10.1016/j.cryobiol.2009.02.006
Acknowledgements
The authors would like to thank all the patients enrolled in the study. We thank the Federal University of Rio Grande do Sul (UFRGS), Department of Physiology, for supporting this work.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors. MK was supported by a research fellowship from CNPq (process# 302959/2020–3). This work was partially supported by FAPERGS. MK was responsible for the grant support from FAPERGS (Edital FAPERGS/Decit/SCTIE/MS/CNPq/SESRS n. 03/2017–PPSUS #17/2551–0001424-3).
Author information
Authors and Affiliations
Contributions
SG, MK, MFJ, and ABM conceptualised and designed the study. SG performed the tissue collection. LSK, GBG, NF, and LRS conducted the histological and viability analyses. SG, MK, MFJ, and ABM analysed the results. SG and MK drafted the manuscript. MFJ and ABM critically revised the text. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Galbinski, S., Kowalewski, L.S., Grigolo, G.B. et al. Comparison between two cryopreservation techniques of human ovarian cortex: morphological aspects and the heat shock response (HSR). Cell Stress and Chaperones 27, 97–106 (2022). https://doi.org/10.1007/s12192-022-01252-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12192-022-01252-6