Abstract
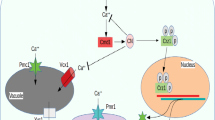
Cadmium (Cd) is a toxic heavy metal that induces irregularity in numerous lipid metabolic pathways. Saccharomyces cerevisiae, a model to study lipid metabolism, has been used to establish the molecular basis of cellular responses to Cd toxicity in relation to essential minerals and lipid homeostasis. Multiple pathways sense these environmental stresses and trigger the mineral imbalances specifically calcium (Ca) and zinc (Zn). This review is aimed to elucidate the role of Cd toxicity in yeast, in three different perspectives: (1) elucidate stress response and its adaptation to Cd, (2) understand the physiological role of a macromolecule such as lipids, and (3) study the stress rescue mechanism. Here, we explored the impact of Cd interference on the essential minerals such as Zn and Ca and their influence on endoplasmic reticulum stress and lipid metabolism. Cd toxicity contributes to lipid droplet synthesis by activating OLE1 that is essential to alleviate lipotoxicity. In this review, we expanded our current findings about the effect of Cd on lipid metabolism of budding yeast.





Similar content being viewed by others
References
Abbà S, Vallino M, Daghino S, Di Vietro L, Borriello R, Perotto S (2011) A PLAC8-containing protein from an endomycorrhizal fungus confers cadmium resistance to yeast cells by interacting with Mlh3p. Nucleic Acids Res 39(17):7548–7563. https://doi.org/10.1093/nar/gkr336
Abhishek A, Benita S, Kumari M, Ganesan D, Paul E, Sasikumar P, Mahesh A, Yuvaraj S, Ramprasath T, Selvam GS (2017) Molecular analysis of oxalate induced endoplasmic reticulum stress mediated apoptosis in the pathogenesis of kidney stone disease. J Physiol Biochem 73:561–573. https://doi.org/10.1007/s13105-017-0587-8
Adle DJ, Sinani D, Kim H, Lee J (2007) A cadmium-transporting P1B-type ATPase in yeast Saccharomyces cerevisiae. J Biol Chem 282:947–955. https://doi.org/10.1074/jbc.M609535200
Aguilar PS, De Mendoza D (2006) Control of fatty acid desaturation: a mechanism conserved from bacteria to humans. Mol Microbiol:62. https://doi.org/10.1111/j.1365-2958.2006.05484.x
Al Kaddissi S, Legeay A, Elia AC, Gonzalez P, Floriani M, Cavalie I, Massabuau J-C, Gilbin R, Simon O (2014) Mitochondrial gene expression, antioxidant responses, and histopathology after cadmium exposure. Environ Toxicol 29:893–907. https://doi.org/10.1002/tox.21817
Anand P, Cermelli S, Li Z, Kassan A, Bosch M, Sigua R, Huang L, Ouellette AJ, Pol A, Welte MA, Gross SP (2012) A novel role for lipid droplets in the organismal antibacterial response. eLife 1:e00003. https://doi.org/10.7554/eLife.00003
Arruda AP, Hotamisligil GS (2015) Calcium homeostasis and organelle function in the pathogenesis of obesity and diabetes. Cell Metab 22:381–397. https://doi.org/10.1016/j.cmet.2015.06.010
Athenstaedt K, Daum G (2005) Tgl4p and Tgl5p, two triacylglycerol lipases of the yeast Saccharomyces cerevisiae are localized to lipid particles. J Biol Chem 280:37301–37309. https://doi.org/10.1074/jbc.M507261200
Bailey AP, Koster G, Guillermier C, Hirst EMA, MacRae JI, Lechene CP, Postle AD, Gould AP (2015) Antioxidant role for lipid droplets in a stem cell niche of Drosophila. Cell 163:340–353. https://doi.org/10.1016/j.cell.2015.09.020
Ballatori N (2002) Transport of toxic metals by molecular mimicry. Environ Health Perspect 110:689–694. https://doi.org/10.1289/ehp.02110s5689
Beyersmann D, Hartwig A (2008) Carcinogenic metal compounds: recent insight into molecular and cellular mechanisms. Arch Toxicol82:493–512. doi: https://doi.org/10.1007/s00204-008-0313-y
Bleackley MR, MacGillivray RTA (2011) Transition metal homeostasis: from yeast to human disease. BioMetals 24:785–809. https://doi.org/10.1007/s10534-011-9451-4
Bridges CC, Zalups RK (2005) Molecular and ionic mimicry and the transport of toxic metals. Toxicol Appl Pharmacol 204:274–308. https://doi.org/10.1016/j.taap.2004.09.007
Brzóska MM, Moniuszko-Jakoniuk J (1998) The influence of calcium content in diet on cumulation and toxicity of cadmium in the organism. Arch Toxicol 72:63–73
Brzóska MM, Moniuszko-Jakoniuk J (2001) Interactions between cadmium and zinc in the organism. Food Chem Toxicol 39:967–980
Buschini A, Poli P, Rossi C (2003) Saccharomyces cerevisiae as an eukaryotic cell model to assess cytotoxicity and genotoxicity of three anticancer anthraquinones. Mutagenesis 18:25–36
Caetano SM, Menezes R, Amaral C, Rodrigues-Pousada C, Pimentel C (2015) Repression of the low affinity iron transporter gene FET4: A novel mechanism against cadmium toxicity orchestrated by yap1 via rox1. J Biol Chem 290(30):18584–18595. https://doi.org/10.1074/jbc.M114.600742
Carman GM, Han G-S (2007) Regulation of phospholipid synthesis in Saccharomyces cerevisiae by zinc depletion. Biochim Biophys Acta 1771:322–330. https://doi.org/10.1016/J.BBALIP.2006.05.006
Carmona-Gutierrez D, Ruckenstuhl C, Bauer MA, Eisenberg T, Bü Ttner S, Madeo F (2010) Cell death in yeast: growing applications of a dying buddy. Cell Death Differ 17:733–734. https://doi.org/10.1038/cdd.2010.10
Chang N, Yao S, Chen D, Zhang L, Huang J, Zhang L (2018) The Hog1 positive regulated YCT1 gene expression under cadmium tolerance of budding yeast. FEMS Microbiol Lett 365(17) doi: https://doi.org/10.1093/femsle/fny170
Chellappa R, Kandasamy P, Oh C-S, Jiang Y, Vemula M, Martin CE (2001) The membrane proteins, Spt23p and Mga2p, play distinct roles in the activation of Saccharomyces cerevisiae OLE1 gene expression. J Biol Chem 276:43548–43556. https://doi.org/10.1074/jbc.M107845200
Chin-Chan M, Navarro-Yepes J, Quintanilla-Vega B (2015) Environmental pollutants as risk factors for neurodegenerative disorders: Alzheimer and Parkinson diseases. Front Cell Neurosci 9:124. https://doi.org/10.3389/fncel.2015.00124
Choong G, Liu Y, Templeton DM (2014) Interplay of calcium and cadmium in mediating cadmium toxicity. Chem Biol Interact 211:54–65. https://doi.org/10.1016/j.cbi.2014.01.007
Cobbett C, Goldsbrough P (2002) Phytochelatins and metallothioneins: roles in heavy metal detoxification and homeostasis. Annu Rev Plant Biol 53:159–182. https://doi.org/10.1146/annurev.arplant.53.100301.135154
Cohen Y, Megyeri M, Chen OCW, Condomitti G, Riezman I, Loizides-Mangold U, Abdul-Sada A, Rimon N, Riezman H, Platt FM, Futerman AH, Schuldiner M (2013) The yeast P5 type ATPase, Spf1, regulates manganese transport into the endoplasmic reticulum. PLoS One 8:e85519. https://doi.org/10.1371/journal.pone.0085519
Cronin SR, Rao R, Hampton RY (2002) Cod1p/Spf1p is a P-type ATPase involved in ER function and Ca2+ homeostasis. J Cell Biol 157:1017–1028. https://doi.org/10.1083/jcb.200203052
Cutler RG, Kelly J, Storie K, Pedersen WA, Tammara A, Hatanpaa K, Troncoso JC, Mattson MP (2004) Involvement of oxidative stress-induced abnormalities in ceramide and cholesterol metabolism in brain aging and Alzheimer’s disease. Proc Natl Acad Sci U S A 101:2070–2075. https://doi.org/10.1073/pnas.0305799101
Ehrensberger KM, Bird AJ (2011) Hammering out details: regulating metal levels in eukaryotes. Trends Biochem Sci 36:524–531. https://doi.org/10.1016/j.tibs.2011.07.002
Eide DJ (2009) Homeostatic and adaptive responses to zinc deficiency in Saccharomyces cerevisiae. J Biol Chem 284:18565–18569. https://doi.org/10.1074/JBC.R900014200
Eraso P, Martinez-Burgos M, Falcon-Perez JM, Portillo F, Mazon MJ (2004) Ycf1-dependent cadmium detoxification by yeast requires phosphorylation of residues Ser(908) and Thr(911). FEBS Lett 577:322–326. https://doi.org/10.1016/j.febslet.2004.10.030
Fang Z, Chen Z, Wang S, Shi P, Shen Y, Zhang Y, Xiao J, Huang Z (2016) Overexpression of OLE1 enhances cytoplasmic membrane stability and confers resistance to cadmium in Saccharomyces cerevisiae. Appl Environ Microbiol 83:AEM.02319–AEM.02316. https://doi.org/10.1128/AEM.02319-16
Fauchon M, Lagniel G, Aude J-C, Lombardia L, Soularue P, Petat C, Marguerie G, Sentenac A, Werner M, Labarre J (2002) Sulfur sparing in the yeast proteome in response to sulfur demand. Mol Cell 9:713–723. https://doi.org/10.1016/S1097-2765(02)00500-2
Fei W, Wang H, Fu X, Bielby C, Yang H (2009) Conditions of endoplasmic reticulum stress stimulate lipid droplet formation in Saccharomyces cerevisiae. Biochem J 424:61–67. https://doi.org/10.1042/BJ20090785
Fu S, Yang L, Li P, Hofmann O, Dicker L, Hide W, Lin X, Watkins SM, Ivanov AR, Hotamisligil GS (2011) Aberrant lipid metabolism disrupts calcium homeostasis causing liver endoplasmic reticulum stress in obesity. Nature 473:528–531. https://doi.org/10.1038/nature09968
Fu S, Watkins SM, Hotamisligil GS (2012) The role of endoplasmic reticulum in hepatic lipid homeostasis and stress signaling. Cell Metab 15:623–634. https://doi.org/10.1016/j.cmet.2012.03.007
Furukawa S, Fujita T, Shimabukuro M, Iwaki M, Yamada Y, Nakajima Y, Nakayama O, Makishima M, Matsuda M, Shimomura I (2004) Increased oxidative stress in obesity and its impact on metabolic syndrome. J Clin Invest 114:1752–1761. https://doi.org/10.1172/JCI21625
Garbarino J, Sturley SL (2005) Homoeostatic systems for sterols and other lipids. Biochem Soc Trans 33:1182–1185. https://doi.org/10.1042/BST20051182
Garbarino J, Padamsee M, Wilcox L, Oelkers PM, D’Ambrosio D, Ruggles KV, Ramsey N, Jabado O, Turkish A, Sturley SL (2009) Sterol and diacylglycerol acyltransferase deficiency triggers fatty acid-mediated cell death. J Biol Chem 284:30994–31005. https://doi.org/10.1074/jbc.M109.050443
Gardarin A, Chédin S, Lagniel G, Aude J-C, Godat E, Catty P, Labarre J (2010) Endoplasmic reticulum is a major target of cadmium toxicity in yeast. Mol Microbiol 76:1034–1048. https://doi.org/10.1111/j.1365-2958.2010.07166.x
Gasch AP, Spellman PT, Kao CM, Carmel-Harel O, Eisen MB, Storz G, Botstein D, Brown PO (2000) Genomic expression programs in the response of yeast cells to environmental changes. Mol Biol Cell 11:4241–4257. https://doi.org/10.1091/mbc.11.12.4241
Gitan RS, Luo H, Rodgers J, Broderius M, Eide D (1998) Zinc-induced inactivation of the yeast ZRT1 zinc transporter occurs through endocytosis and vacuolar degradation. J Biol Chem 273:28617–28624. https://doi.org/10.1074/jbc.273.44.28617
Gitan RS, Shababi M, Kramer M, Eide DJ (2003) A cytosolic domain of the yeast Zrt1 zinc transporter is required for its post-translational inactivation in response to zinc and cadmium. J Biol Chem 278:39558–39564. https://doi.org/10.1074/jbc.M302760200
Gomes DS, Fragoso LC, Riger CJ, Panek AD, Eleutherio EC (2002) Regulation of cadmium uptake by Saccharomyces cerevisiae. Biochim Biophys Acta 1573:21–25. https://doi.org/10.1016/s0304-4165(02)00324-0
Groenendyk J, Sreenivasaiah PK, Kim DH, Agellon LB, Michalak M (2010) Biology of endoplasmic reticulum stress in the heart. Circ Res 107:1185–1197. https://doi.org/10.1161/CIRCRESAHA.110.227033
Guo L, Ganguly A, Sun L, Suo F, Du LL, Russell P (2016) Global fitness profiling identifies arsenic and cadmium tolerance mechanisms in fission Yeast. G3 (Bethesda) 6(10):3317–3333
Han GS, Johnston CN, Chen X, Athenstaedt K, Daum G, Carman GM (2001) Regulation of the Saccharomyces cerevisiae DPP1-encoded diacylglycerol pyrophosphate phosphatase by zinc. J Biol Chem 276:10126–10133. https://doi.org/10.1074/jbc.M011421200
Han S, Ha K, Jeon J, Kim H, Lee K, Kim D (2015) Impact of cadmium exposure on the association between lipopolysaccharide and metabolic syndrome. Int J Environ Res Public Health 12:11396–11409. https://doi.org/10.3390/ijerph120911396
Hechtenberg S, Beyersmann D (1991) Inhibition of sarcoplasmic reticulum Ca (2+)-ATPase activity by cadmium, lead and mercury. Enzyme 45:109–115
Henry SA, Kohlwein SD, Carman GM (2012) Metabolism and regulation of glycerolipids in the yeast Saccharomyces cerevisiae. Genetics 190:317–349. https://doi.org/10.1534/genetics.111.130286
Holland WL, Brozinick JT, Wang L-P, Hawkins ED, Sargent KM, Liu Y, Narra K, Hoehn KL, Knotts TA, Siesky A, Nelson DH, Karathanasis SK, Fontenot GK, Birnbaum MJ, Summers SA (2007) Inhibition of ceramide synthesis ameliorates glucocorticoid-, saturated-fat-, and obesity-induced insulin resistance. Cell Metab 5:167–179. https://doi.org/10.1016/j.cmet.2007.01.002
Horvath SE, Wagner A, Steyrer E, Daum G (2011) Metabolic link between phosphatidylethanolamine and triacylglycerol metabolism in the yeast Saccharomyces cerevisiae. Biochim Biophys Acta 1811:1030–1037. https://doi.org/10.1016/j.bbalip.2011.08.007
Hosiner D, Gerber S, Lichtenberg-Fraté H, Glaser W, Schüller C, Klipp E (2014) Impact of acute metal stress in Saccharomyces cerevisiae. PLoS One 9:1–14. https://doi.org/10.1371/journal.pone.0083330
Huh W-K, Falvo JV, Gerke LC, Carroll AS, Howson RW, Weissman JS, O’Shea EK (2003) Global analysis of protein localization in budding yeast. Nature 425:686–691. https://doi.org/10.1038/nature02026
IARC (1993) Cadmium and cadmium compounds. IARC Monogr Eval Carcinog Risks Hum 58:119–237
Jacobs RL, Zhao Y, Koonen DPY, Sletten T, Su B, Lingrell S, Cao G, Peake DA, Kuo M-S, Proctor SD, Kennedy BP, Dyck JRB, Vance DE (2010) Impaired de Novo choline synthesis explains why phosphatidylethanolamine N-methyltransferase-deficient mice are protected from diet-induced obesity. J Biol Chem 285:22403–22413. https://doi.org/10.1074/jbc.M110.108514
Järup L, Åkesson A (2009) Current status of cadmium as an environmental health problem. Toxicol Appl Pharmacol 238:201–208. https://doi.org/10.1016/j.taap.2009.04.020
Jensen LJ, Kuhn M, Stark M, Chaffron S, Creevey C, Muller J, Doerks T, Julien P, Roth A, Simonovic M, Bork P, von Mering C (2009) STRING 8—a global view on proteins and their functional interactions in 630 organisms. Nucleic Acids Res 37:D412–D416. https://doi.org/10.1093/nar/gkn760
Jin YH, Dunlap PE, McBride SJ, Al-Refai H, Bushel PR, Freedman JH (2008) Global transcriptome and deletome profiles of yeast exposed to transition metals. PLoS Genet 4:e1000053. https://doi.org/10.1371/journal.pgen.1000053
Jomova K, Valko M (2011) Advances in metal-induced oxidative stress and human disease. Toxicol 283:65–87. https://doi.org/10.1016/j.tox.2011.03.001
Jonikas MC, Collins SR, Denic V, Oh E, Quan EM, Schmid V, Weibezahn J, Schwappach B, Walter P, Weissman JS, Schuldiner M (2009) Comprehensive characterization of genes required for protein folding in the endoplasmic reticulum. Science 323:1693–1697. https://doi.org/10.1126/science.1167983
Kamphorst JJ, Cross JR, Fan J, de Stanchina E, Mathew R, White EP, Thompson CB, Rabinowitz JD (2013) Hypoxic and Ras-transformed cells support growth by scavenging unsaturated fatty acids from lysophospholipids. Proc Natl Acad Sci U S A 110:8882–8887. https://doi.org/10.1073/pnas.1307237110
Kim Y-C, Ntambi JM (1999) Regulation of stearoyl-CoA desaturase genes: role in cellular metabolism and preadipocyte differentiation. Biochem Biophys Res Commun 266:1–4. https://doi.org/10.1006/bbrc.1999.1704
Kim SC, Cho MK, Kim SG (2003) Cadmium-induced non-apoptotic cell death mediated by oxidative stress under the condition of sulfhydryl deficiency. Toxicol Lett 144(3):325–336. https://doi.org/10.1016/s0378-4274(03)00233-9
Koch B, Schmidt C, Daum G (2014) Storage lipids of yeasts: a survey of nonpolar lipid metabolism in Saccharomyces cerevisiae, Pichia pastoris, and Yarrowia lipolytica. FEMS Microbiol Rev 38:892–915. doi: https://doi.org/10.1111/1574-6976.12069
Kroemer G, Marino G, Levine B (2010) Autophagy and the integrated stress response. Mol Cell 40:280–293. https://doi.org/10.1016/j.molcel.2010.09.023
Krumpe K, Frumkin I, Herzig Y, Rimon N, Özbalci C, Brügger B, Rapaport D, Schuldiner M (2012) Ergosterol content specifies targeting of tail-anchored proteins to mitochondrial outer membranes. Mol Biol Cell 23:3927–3935. https://doi.org/10.1091/mbc.E11-12-0994
Kudo N, Nakagawa Y, Waku K, Kawashima Y, Kozuka H (1991) Prevention by zinc of cadmium inhibition of stearoyl-CoA desaturase in rat liver. Toxicol 68:133–142. https://doi.org/10.1016/0300-483X(91)90016-T
Kwast KE, Burke PV, Staahl BT, Poyton RO (1999) Oxygen sensing in yeast: evidence for the involvement of the respiratory chain in regulating the transcription of a subset of hypoxic genes. Proc Natl Acad Sci U S A 96:5446–5451
Lee J, Godon C, Lagniel G, Spector D, Garin J, Labarre J, Toledano MB (1999) Yap1 and Skn7 control two specialized oxidative stress response regulons in yeast. J Biol Chem 274:16040–1604610. https://doi.org/10.1074/jbc.274.23.16040
Ligr M, Madeo F, Fröhlich E, Hilt W, Fröhlich K-U, Wolf DH (1998) Mammalian Bax triggers apoptotic changes in yeast. FEBS Lett 438:61–65. https://doi.org/10.1016/S0014-5793(98)01227-7
Listenberger LL, Ory DS, Schaffer JE (2001) Palmitate-induced apoptosis can occur through a ceramide-independent pathway. J Biol Chem 276:14890–14895. https://doi.org/10.1074/jbc.M010286200
Listenberger LL, Han X, Lewis SE, Cases S, Farese RV, Ory DS, Schaffer JE (2003) Triglyceride accumulation protects against fatty acid-induced lipotoxicity. Proc Natl Acad Sci U S A 100:3077–3082. https://doi.org/10.1073/pnas.0630588100
Liu D, Yang J, Li Y, Zhang M, Wang L (2013) Cd-induced apoptosis through the mitochondrial pathway in the hepatopancreas of the freshwater crab Sinopotamon henanense. PLoS One 8:e68770. https://doi.org/10.1371/journal.pone.0068770
Low C, Liew L, Pervaiz S, Yang H (2005) Apoptosis and lipoapoptosis in the fission yeast. FEMS Yeast Res 5:1199–1206. https://doi.org/10.1016/j.femsyr.2005.07.004
Luevano J, Damodaran C (2014) A review of molecular events of cadmium-induced carcinogenesis. J Environ Pathol Toxicol Oncol 33:183–194
Malhotra JD, Kaufman RJ (2011) ER stress and its functional link to mitochondria: role in cell survival and death. Cold Spring Harb Perspect Biol 3:a004424–a004424. https://doi.org/10.1101/cshperspect.a004424
Manon S (2004) Utilization of yeast to investigate the role of lipid oxidation in cell death. Antioxid Redox Signal 6:259–267. https://doi.org/10.1089/152308604322899323
Martin CE, Oh C-S, Jiang Y (2007) Regulation of long chain unsaturated fatty acid synthesis in yeast. Biochim Biophys Acta 1771:271–285. https://doi.org/10.1016/j.bbalip.2006.06.010
Mazzola D, Pimentel C, Caetano S, Amaral C, Menezes R, Santos CN, Eleutherio E, Rodrigues-Pousada C (2015) Inhibition of Yap2 activity by MAPKAP kinase Rck1 affects yeast tolerance to cadmium. FEBS Lett 589(19 Pt B):2841–2849. https://doi.org/10.1016/j.febslet.2015.07.049
Mendoza-Cózatl D, Loza-Tavera H, Hernández-Navarro A, Moreno-Sánchez R (2005) Sulfur assimilation and glutathione metabolism under cadmium stress in yeast, protists and plants. FEMS Microbiol Rev 29:653–671. https://doi.org/10.1016/j.femsre.2004.09.004
Mitsuhashi S, Ohkuma A, Talim B, Karahashi M, Koumura T, Aoyama C, Kurihara M, Quinlivan R, Sewry C, Mitsuhashi H, Goto K, Koksal B, Kale G, Ikeda K, Taguchi R, Noguchi S, Hayashi YK, Nonaka I, Sher RB, Sugimoto H, Nakagawa Y, Cox GA, Topaloglu H, Nishino I (2011) A congenital muscular dystrophy with mitochondrial structural abnormalities caused by defective de novo phosphatidylcholine biosynthesis. Am J Hum Genet 88:845–851. https://doi.org/10.1016/j.ajhg.2011.05.010
Murata Y, Watanabe T, Sato M, Momose Y, Nakahara T, Oka S, Iwahashi H (2003) Dimethyl sulfoxide exposure facilitates phospholipid biosynthesis and cellular membrane proliferation in yeast cells. J Biol Chem 278:33185–33193. https://doi.org/10.1074/jbc.M300450200
Muthukumar K, Nachiappan V (2013) Phosphatidylethanolamine from phosphatidylserine decarboxylase 2 is essential for autophagy under cadmium stress in Saccharomyces cerevisiae. Cell Biochem Biophys 67:1353–1363. https://doi.org/10.1007/s12013-013-9667-8
Muthukumar K, Rajakumar S, Sarkar MN, Nachiappan V (2011) Glutathione peroxidase 3 of Saccharomyces cerevisiae protects phospholipids during cadmium-induced oxidative stress. Antonie Van Leeuwenhoek 99:761–771. https://doi.org/10.1007/s10482-011-9550-9
Nair AR, Degheselle O, Smeets K, Van Kerkhove E, Cuypers A (2013) Cadmium-induced pathologies: where is the oxidative balance lost (or not)? Int J Mol Sci 14:6116–6143. https://doi.org/10.3390/ijms14036116
Nakatogawa H, Ichimura Y, Ohsumi Y (2007) Atg8, a ubiquitin-like protein required for autophagosome formation, mediates membrane tethering and hemifusion. Cell 130:165–178. https://doi.org/10.1016/j.cell.2007.05.021
Nargund AM, Avery SV, Houghton JE (2008) Cadmium induces a heterogeneous and caspase-dependent apoptotic response in Saccharomyces cerevisiae. Apoptosis 13:811–82110. https://doi.org/10.1007/s10495-008-0215-8
Nebauer R, Rosenberger S, Daum G (2007) Phosphatidylethanolamine, a limiting factor of autophagy in yeast strains bearing a defect in the carboxypeptidase Y pathway of vacuolar targeting. J Biol Chem 282:16736–16743. https://doi.org/10.1074/jbc.M611345200
Nordberg GF (2009) Historical perspectives on cadmium toxicology. Toxicol Appl Pharmacol 238:192–200. https://doi.org/10.1016/j.taap.2009.03.015
Ozcan U, Cao Q, Yilmaz E, Lee A-H, Iwakoshi NN, Ozdelen E, Tuncman G, Görgün C, Glimcher LH, Hotamisligil GS (2004) Endoplasmic reticulum stress links obesity, insulin action, and type 2 diabetes. Science 306:457–461. https://doi.org/10.1126/science.1103160
Pereira C, Miguel Martins L, Saraiva L (2014) LRRK2, but not pathogenic mutants, protects against H2O2 stress depending on mitochondrial function and endocytosis in a yeast model. Biochim Biophys Acta 1840:2025–2031. https://doi.org/10.1016/J.BBAGEN.2014.02.015
Petschnigg J, Wolinski H, Kolb D, Zellnig G, Kurat CF, Natter K, Kohlwein SD (2009) Good fat, essential cellular requirements for triacylglycerol synthesis to maintain membrane homeostasis in yeast. J Biol Chem 284:30981–30993. https://doi.org/10.1074/jbc.M109.024752
Pierron F, Baudrimont M, Bossy A, Bourdineaud J, Brethes D, Elie P, Massabuau J (2007) Impairment of lipid storage by cadmium in the European eel (Anguilla anguilla). Aquat Toxicol 81:304–311. https://doi.org/10.1016/j.aquatox.2006.12.014
Pizzino G, Irrera N, Bitto A, Pallio G, Mannino F, Arcoraci V, Aliquò F, Minutoli L, De Ponte C, D’andrea P, Squadrito F, Altavilla D (2017) Cadmium-induced oxidative stress impairs glycemic control in adolescents. Oxidative Med Cell Longev 2017:1–6. https://doi.org/10.1155/2017/6341671
Rahoui S, Chaoui A, El Ferjani E (2010) Membrane damage and solute leakage from germinating pea seed under cadmium stress. J Hazard Mater 178:1128–1131. https://doi.org/10.1016/J.JHAZMAT.2010.01.115
Rajakumar S, Nachiappan V (2017) Lipid droplets alleviate cadmium induced cytotoxicity in Saccharomyces cerevisiae. Toxicol Res 6:30–41. https://doi.org/10.1039/C6TX00187D
Rajakumar S, Bhanupriya N, Ravi C, Nachiappan V (2016a) Endoplasmic reticulum stress and calcium imbalance are involved in cadmium-induced lipid aberrancy in Saccharomyces cerevisiae. Cell Stress Chaperones 21:895–906. https://doi.org/10.1007/s12192-016-0714-4
Rajakumar S, Ravi C, Nachiappan V (2016b) Defect of zinc transporter ZRT1 ameliorates cadmium induced lipid accumulation in Saccharomyces cerevisiae. Metallomics 8:453–460. https://doi.org/10.1039/C6MT00005C
Lisa M Regalla and Thomas J Lyons (2005) Zinc in yeast: mechanisms involved in homeostasis. Topics in Current Genetics, Vol: 14, Springer, Berlin. https://doi.org/10.1007/4735_98
Rogalska J, Brzóska MM, Roszczenko A, Moniuszko-Jakoniuk J (2009) Enhanced zinc consumption prevents cadmium-induced alterations in lipid metabolism in male rats. Chem Biol Interact 177:142–152. https://doi.org/10.1016/j.cbi.2008.09.011
Ruta LL, Popa VC, Nicolau L, Danet AF, Iordache V, Neagoe AD, Farcasanu IC (2014) Calcium signaling mediates the response to cadmium toxicity in Saccharomyces cerevisiae cells. FEBS Lett 588:3202–3212. https://doi.org/10.1016/j.febslet.2014.07.001
Schrey P, Wittsiepe J, Budde U, Heinzow B, Idel H, Wilhelm M, Wilhelm M (2000) Dietary intake of lead, cadmium, copper and zinc by children from the German North Sea island Amrum. Int J Hyg Environ Health 203:1–9. https://doi.org/10.1078/S1438-4639(04)70001-2
Schröder M, Kaufman RJ (2005) ER stress and the unfolded protein response. Mutat Res 569:29–63. https://doi.org/10.1016/j.mrfmmm.2004.06.056
Schuldiner M, Collins SR, Thompson NJ, Denic V, Bhamidipati A, Punna T, Ihmels J, Andrews B, Boone C, Greenblatt JF, Weissman JS, Krogan NJ (2005) Exploration of the function and organization of the yeast early secretory pathway through an epistatic miniarray profile. Cell 123:507–519. https://doi.org/10.1016/j.cell.2005.08.031
Sharon A, Finkelstein A, Shlezinger N, Hatam I (2009) Fungal apoptosis: function, genes and gene functions. FEMS Microbiol Rev 33:833–854. https://doi.org/10.1111/j.1574-6976.2009.00180.x
Sheibani S, Richard VR, Beach A, Leonov A, Feldman R, Mattie S, Khelghatybana L, Piano A, Greenwood M, Vali H, Titorenko VI (2014) Macromitophagy, neutral lipids synthesis, and peroxisomal fatty acid oxidation protect yeast from “liponecrosis”, a previously unknown form of programmed cell death. Cell Cycle 13:138–147. https://doi.org/10.4161/cc.26885
Shivapurkar N, Reddy J, Chaudhary PM, Gazdar AF (2003) Apoptosis and lung cancer: a review. J Cell Biochem 88:885–898. https://doi.org/10.1002/jcb.10440
Singh N, Yadav KK, Rajasekharan R (2016) ZAP1-mediated modulation of triacylglycerol levels in yeast by transcriptional control of mitochondrial fatty acid biosynthesis. Mol Microbiol 100:55–75. https://doi.org/10.1111/mmi.13298
Smith N, Wei W, Zhao M, Qin X, Seravalli J, Kim H, Lee J (2016) Cadmium and secondary structure-dependent function of a degron in the Pca1p cadmium exporter. J Biol Chem 291:12420–12431
Sohn MJ, Yoo SJ, Oh D-B, Kwon O, Lee SY, Sibirny AA, Kang HA (2014) Novel cysteine-centered sulfur metabolic pathway in the thermotolerant methylotrophic yeast Hansenula polymorpha. PLoS One 9:e100725. https://doi.org/10.1371/journal.pone.0100725
Soto-Cardalda A, Fakas S, Pascual F, Choi H-S, Carman GM (2012) Phosphatidate phosphatase plays role in zinc-mediated regulation of phospholipid synthesis in yeast. J Biol Chem 287:968–977. https://doi.org/10.1074/jbc.M111.313130
Surma MA, Klose C, Peng D, Shales M, Mrejen C, Stefanko A, Braberg H, Gordon DE, Vorkel D, Ejsing CS, Farese R, Simons K, Krogan NJ, Ernst R (2013) A lipid E-MAP identifies Ubx2 as a critical regulator of lipid saturation and lipid bilayer stress. Mol Cell 51:519–530. https://doi.org/10.1016/j.molcel.2013.06.014
Temple MD, Perrone GG, Dawes IW (2005) Complex cellular responses to reactive oxygen species. Trends Cell Biol 15:319–32610. https://doi.org/10.1016/j.tcb.2005.04.003
Thevenod F (2009) Cadmium and cellular signaling cascades: to be or not to be? Toxicol Appl Pharmacol 238:221–239
Thibault G, Ng DTW (2012) The endoplasmic reticulum-associated degradation pathways of budding yeast. Cold Spring Harb Perspect Biol. https://doi.org/10.1101/cshperspect.a013193
Thibault G, Shui G, Kim W, McAlister GC, Ismail N, Gygi SP, Wenk MR, Ng DTW (2012) The membrane stress response buffers lethal effects of lipid disequilibrium by reprogramming the protein homeostasis network. Mol Cell 48:16–27. https://doi.org/10.1016/j.molcel.2012.08.016
Thompson JE, Froese CD, Madey E, Smith MD, Hong Y (1998) Lipid metabolism during plant senescence. Prog Lipid Res 37:119–141
Thorburn A (2018) Autophagy and disease. J Biol Chem 293(15):5425–5430. https://doi.org/10.1074/jbc.R117.810739
Tong AHY, Lesage G, Bader GD, Ding H, Xu H, Xin X, Young J, Berriz GF, Brost RL, Chang M, Chen Y, Cheng X, Chua G, Friesen H, Goldberg DS, Haynes J, Humphries C, He G, Hussein S, Ke L, Krogan N, Li Z, Levinson JN, Lu H, Ménard P, Munyana C, Parsons AB, Ryan O, Tonikian R, Roberts T, Sdicu A-M, Shapiro J, Sheikh B, Suter B, Wong SL, Zhang LV, Zhu H, Burd CG, Munro S, Sander C, Rine J, Greenblatt J, Peter M, Bretscher A, Bell G, Roth FP, Brown GW, Andrews B, Bussey H, Boone C (2004) Global mapping of the yeast genetic interaction network. Science 303:808–813. https://doi.org/10.1126/science.1091317
Traaseth NJ, Ha KN, Verardi R, Shi L, Buffy JJ, Masterson LR, Veglia G (2008) Structural and dynamic basis of phospholamban and sarcolipin inhibition of Ca (2+)-ATPase. Biochemistry 47:3–13. https://doi.org/10.1021/bi701668v
Tsaluchidu S, Cocchi M, Tonello L, Puri BK (2008) Fatty acids and oxidative stress in psychiatric disorders. BMC Psychiatry 8:S5. https://doi.org/10.1186/1471-244X-8-S1-S5
Upchurch RG (2008) Fatty acid unsaturation, mobilization, and regulation in the response of plants to stress. Biotechnol Lett 30:967–977. https://doi.org/10.1007/s10529-008-9639-z
Verkhratsky A (2005) Physiology and pathophysiology of the calcium store in the endoplasmic reticulum of neurons. Physiol Rev 85:201–279. https://doi.org/10.1152/physrev.00004.2004
Vido K, Spector D, Lagniel G, Lopez S, Toledano MB, Labarre J (2001) A proteome analysis of the cadmium response in Saccharomyces cerevisiae. J Biol Chem 276:8469–8474. https://doi.org/10.1074/jbc.M008708200
Visram M, Radulovic M, Steiner S, Malanovic N, Eichmann TO, Wolinski H, Rechberger GN, Tehlivets O (2018) Homocysteine regulates fatty acid and lipid metabolism in yeast. J Biol Chem 293:5544–5555. https://doi.org/10.1074/jbc.M117.809236
Walther TC, Farese RV Jr (2012) Lipid droplets and cellular lipid metabolism. Annu Rev Biochem 81:687–714. https://doi.org/10.1146/annurev-biochem-061009-102430
Wang C-W (2015) Lipid droplet dynamics in budding yeast. Cell Mol Life Sci 72:2677–2695. https://doi.org/10.1007/s00018-015-1903-5
Wang XH, Liu H, Yi HL (2014) Involvement of ROS and calcium in cadmium-induced yeast cell death. Acta Sci Circumst 34:1869–1873
Wei CC, Luo Z, Hogstrand C, Xu YH, Wu LX, Chen GH, Pan YX, Song YF (2018) Zinc reduces hepatic lipid deposition and activates lipophagy via Zn2+/MTF-1/PPARα and Ca2+/CaMKKβ/AMPK pathways. FASEB J 32:6666–6680. https://doi.org/10.1096/fj.201800463
Weisshaar N, Welsch H, Guerra-Moreno A, Hanna J (2017) Phospholipase Lpl1 links lipid droplet function with quality control protein degradation. Mol Biol Cell 28:716–725. https://doi.org/10.1091/mbc.E16-10-0717
Wemmie JA, Szczypka MS, Thiele DJ, Moye-Rowley WS (1994) Cadmium tolerance mediated by the yeast AP-1 protein requires the presence of an ATP-binding cassette transporter-encoding gene, YCF1. J Biol Chem 269:32592–32597
Wenk MR (2005) The emerging field of lipidomics. Nat Rev Drug Discov 4:594–610. https://doi.org/10.1038/nrd1776
Wu C-Y, Roje S, Sandoval FJ, Bird AJ, Winge DR, Eide DJ (2009) Repression of sulfate assimilation is an adaptive response of yeast to the oxidative stress of zinc deficiency. J Biol Chem 284:27544–27556. https://doi.org/10.1074/jbc.M109.042036
Wysocki R, Tamás MJ (2010) How Saccharomyces cerevisiae copes with toxic metals and metalloids. FEMS Microbiol Rev 34:925–951. https://doi.org/10.1111/j.1574-6976.2010.00217.x
Xie Z, Shao M, Lai L, Liu Y, Fan J (2016) Inhibition of autophagy contributes to the toxicity of cadmium telluride quantum dots in Saccharomyces cerevisiae. Int J Nanomedicine 11:3371–3383. https://doi.org/10.2147/IJN.S108636
Xiong B, Zhang L, Xu H, Yang Y, Jiang L (2015) Cadmium induces the activation of cell wall integrity pathway in budding yeast. Chem Biol Interact 240:316–323
Zhang GH, Yamaguchi M, Kimura S, Higham S, Kraus-Friedmann N (1990) Effects of heavy metal on rat liver microsomal Ca (2+)-ATPase and Ca2+ sequestering. Relation to SH groups. J Biol Chem 265:2184–2189
Zhang Y, Qi H, Taylor R, Xu W, Liu LF, Jin S (2007) The role of autophagy in mitochondria maintenance: characterization of mitochondrial functions in autophagy-deficient S. cerevisiae strains. Autophagy 3:337–346. https://doi.org/10.4161/auto.4127
Zhao H, Eide D (1996a) The ZRT2 gene encodes the low affinity zinc transporter in Saccharomyces cerevisiae. J Biol Chem 271:23203–23210
Zhao H, Eide D (1996b) The yeast ZRT1 gene encodes the zinc transporter protein of a high-affinity uptake system induced by zinc limitation. Proc Natl Acad Sci U S A 93:2454–2458
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Rajakumar, S., Abhishek, A., Selvam, G.S. et al. Effect of cadmium on essential metals and their impact on lipid metabolism in Saccharomyces cerevisiae. Cell Stress and Chaperones 25, 19–33 (2020). https://doi.org/10.1007/s12192-019-01058-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12192-019-01058-z