Abstract
Three new reference materials: MODAS-3 Herring Tissue (M-3 HerTis), MODAS-4 Cormorant Tissue (M-4 CormTis), and MODAS-5 Cod Tissue (M-5 CodTis) were prepared and certified on the basis of results of a worldwide intercomparison exercise. Independently of our proven method of establishing the certified and information values, the content of several essential and toxic elements was additionally determined by the use of ratio primary reference measurement procedures (definitive methods) based on radiochemical neutron activation analysis (RNAA) in the case of As, Cd, Co, Cr, Fe, Mo, Se, and U and isotope dilution mass spectrometry (IDMS) in the case of Hg, respectively. Good agreement of the established certified values and the results obtained by ratio primary reference measurement procedures confirmed the validity of the certification procedure. The total number of elements which could be certified amounted to 30, 21, 18 in M-3 HerTis, M-4 CormTis, and M-5 CodTis, respectively. The relative frequency of use of individual analytical techniques in this intercomparison campaign was calculated and discussed. Inductively coupled plasma mass spectrometry (ICP-MS) is now a dominant technique, followed by atomic absorption spectroscopy (AAS), NAA, and emission spectroscopy (ES). The decreasing share of NAA as compared to several earlier intercomparison exercises should be noticed. NAA is the only method in the array of highly sensitive methods of inorganic trace analysis, which is essentially free from blank. The lack of this method in the foreseeable future may be an obstacle in the prospective certification campaigns and may endanger the implementation of quality assurance in trace analysis.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Ecosphere is subject to constant evolution not only due to natural processes, local catastrophes as e.g. forest fire, tsunami, or eruption of volcanos but also due to the human influence like mining, transport, and industrial activity, waste disposal, etc. Human activity may cause substantial changes in the composition of water, air, soil, plants, and animal tissues what in consequence may lead to pollution of food products of vegetable and animal origin (Kabata-Pendias and Mukherjee 2007). Several elements e.g. Mn, Fe, Co, Cu, Zn, Mo, I, Ni, Se, and Sn have already been identified as essential to humans or toxic (Pb, Hg, As, Cd). The essentiality or toxicity of others e.g. rare earth elements (REE) was not unequivocally established although some researchers suggested that REE may play some biological role in the life systems (Ozaki et al. 1997; Tyler 2004). Chinese scientists maintained that REE accumulated in plants may enter via the food chain the bodies of animals and humans and affect regulating some biological functions (Qiu et al. 2005a, b). In this context, it is perhaps worth quoting the opinion of a medieval scientist and physician Paracelsus, reminded in the book describing the path of trace elements from soil to human (Kabata-Pendias and Mukherjee 2007), who wrote: “All substances are poisonous, there are none which is not a poison; the right dose is what differentiates a poison from a remedy”. The growing concern about environmental pollution created the situation when it became necessary to monitor several components of the environment for the content of essential and toxic elements. We are living in an era when many important decisions (administrative, forensic, technological, ecological, biomedical, etc.) may depend upon the results of chemical analysis including also trace analysis (Dybczyński and Polkowska-Motrenko 2015; Dybczyński 2019). It is obvious that the correct decisions can be taken only when the analytical results on which they are based are reliable. The reliability of determination of several trace elements in various natural materials often leaves much to be desired. As an example, it can serve the data on the concentration levels of 18 trace elements in the blood plasma of apparently healthy individuals published in the literature over the time span of almost a quarter of century as compiled by Versieck and Cornelis (Versieck and Cornelis 1980). The dispersion of results for some essential elements like Ni, Mo, Mn, Co, and Cr reached a few orders of magnitude. It was expected that normally the concentrations of these elements should be kept within narrow limits because they are homeostatically controlled. Commenting on the observed disparities between the values reported by different investigators the authors of the compilation ascribe the probable reasons for inadequate sampling and sample handling or to defective analysis. Similar pessimistic conclusions can be drawn from the results of worldwide intercomparisons observed over a period of 40 years. The spread of results for “difficult” trace elements like As, Cd, Co, Hg, Pb, and many others exceeded, in the case of some of the intercomparison exercises, two, three or even five orders of magnitude (Dybczyński et.al 1979, Dybczyński 1980a, Dybczyński et.al. 1980b, Dybczyński 1981, Dybczyński et.al. 1989a, Dybczyński at.al. 1991, Dybczyński 1997, Dybczyński and Polkowska-Motrenko 2015, Dybczyński 2019). This indicates inadequate quality assurance in several laboratories. On the other hand, one should remember that trace analysis is a difficult task, often consisting of several steps, and there are many occasions to commit grave errors (Dybczyński 2002a).
Quality assurance (QA) of analytical procedures and quality control (QC) are essential for the quantitative measurements of elements in complex matrices. The correct practice to implement effectively those principles is the use of appropriate certified reference materials (CRMs) (Dybczyński and Polkowska-Motrenko 2015; Emons et al. 2006; Quevauviller and Griepink 1996). It is also necessary to use and incorporate CRMs in analytical protocols for the development and validation of new measurement methods and for the assessment of the reliability and comparability of the obtained results.
Good CRM should resemble as much as possible the real samples that are routinely analyzed. The feasible combinations of the type of matrix–concentration level(s) of the analyte(s) are practically unlimited. Therefore, the new CRMs should always be welcome. The availability of the CRMs certified for the content of trace and minor elements was summarized in several textbooks and review papers (Quevauviller and Griepink 1996, Dybczyński 2002b, Phillips et.al. 2007, Dybczyński and Polkowska-Motrenko 2015). Problems associated with the novel CRMs were recently discussed by Wise 2018. It should be noticed that currently on the market there is a very limited number of CRMs of fish origin for multi-elemental analysis. In fact, there are only 10 such materials in stock and available: 2 CRMs produced by the Joint Research Centre Geel (JRC Geel, Belgium), 2 CRMs produced by the International Atomic Energy Agency (IAEA, Monaco), 2 CRMs produced by the National Institute of Standards and Technology (NIST, USA), 2 CRMs produced by the National Research Council Canada ( NRCC, Canada), and 2 CRMs produced by the National Metrology Institute in Japan (NMI, Japan) ( cf. Table 1).
In the case of a cormorant matrix-matching sample, there is no certified material of such origin. It is well known that effective bioindicators of contamination in the environment are fishes and piscivorous birds (e.g. cormorants, which occur at the top of aquatic food chain) as many elements (especially heavy metals) can be stored at higher concentrations in bodies of predators (Aazami and KianiMehr 2018; Houserova et al., 2007; Skoric et al., 2012). Matrix-matching fish and cormorant materials can be widely used for QA/QC of analysis result of metals pollution of food products of aquatic environment and thus to the risk assessment of human health. Moreover, reference materials available on the market (e.g. animal tissue) do not fully meet the needs of analytical laboratories, due to different compositions of the matrix, source, and geochemical characteristics, other natural and anthropogenic contaminants compared to real analyzed samples. In this paper, the preparation and certification of the three new CRMs: MODAS-3 Herring Tissue (M-3 HerTis), MODAS-4 Cormorant Tissue (M-4 CormTis), and MODAS-5 Cod Tissue (M-5 CodTis) intended for food, environmental, and biomedical trace analysis is reported. In the case of a few selected trace elements, the validity of the certified values, established in the course of elaboration of results of the worldwide interlaboratory comparison, was additionally confirmed by the results of “definitive methods” (Uriano and Gravatt 1977), which now, according to VIM 3, are called “primary reference measurement procedures” (ISO/IEC Guide 99:2007 2007).
The work was performed within the MODAS consortium, which was formed at the initiative of the late professor J. Namieśnik of the Gdańsk University of Technology. Other works executed within the frame of the consortium activity were already published (Baranowska et al. 2015, Baranowska et al. 2017, Rutkowska et al. 2018).
Methods and Materials
Strategy of Certification
Preparation and certification of reference materials is a complex task consisting of several stages which must be executed with utmost care to achieve the final success. The general approach used in our laboratory has been described in several earlier publications (Dybczyński et.al.1989a, Dybczyński et al. 1991, Dybczyński et al. 1993, Dybczyński 2002b, Polkowska-Motrenko et al.2010, Samczyński et al. 2012, Dybczyński and Polkowska-Motrenko 2015). All steps in the preparation and certification process are important but evaluation of the intercomparison results and assigning of certified (“recommended “) values has been considered to be the most critical one. In the early times of CRM production, practically every team of researchers employed his own method of elaboration of results. Some of these approaches were a combination of statistics and arbitrary decisions. Moreover, established “recommended” values were usually quoted without uncertainty limits. The review of these early times was published by S. Abbey (1977). Abbey himself was the supporter of some arbitrary decisions writing: “My philosophy is simple. The reliability of a result depends more on who produced it than on how it was done “ (Abbey 1981). The program which we are using for the establishing of certified values on the basis of intercomparison results was originally devised for the Analytical Quality Control Service of the International Atomic Energy Agency (IAEA) (Dybczyński et al. 1978, Dybczyński 1980a). The aim was to create a fully objective method of data evaluation with the simultaneous setting free the general population from outlying results. This purpose was achieved by the concurrent use of four different statistical tests to minimize the so-called masking effects. The method was thoroughly tested and proved to yield very reliable results when applied to data sets from several intercomparison exercises, organized by IAEA on the determination of trace elements or low levels of radionuclides in which the “true values” were known (Dybczyński et al. 1978; Dybczyński, 1980a). Not every overall mean with its associated confidence limits (after rejection of outlying results) is automatically given the status of “certified value.” To do so, several criteria must be simultaneously fulfilled. These criteria were formulated at the beginning of using this program (Dybczyński et al. 1979, Dybczyński 1980a, Dybczyński et al. 1980b), and with slight refinements (Dybczyński et al. 1989a, 1991; Samczyński et al. 2012; Dybczyński and Polkowska-Motrenko 2015) have been used also in this study. The advantages of our method were discussed in the literature (Abbey and Rousseau 1985). Several groups of researchers used this method for the certification of their reference materials (e.g., Hołyńska et al 1987, Kocman and Foley 1987).
The general strategy of preparation and certification of reference materials has been previously presented in detail in the literature. It was also used in the case of all CRMs issued by the Institute of Nuclear Chemistry and Technology (INCT) (Polkowska-Motrenko et al. 2010). These CRMs included materials with mineral matrix i.e. Apatite concentrate (CTA-AC-1) and Fine fly ash (CTA-FFA-1) (Dybczyński et al. 1989a, 1991) and a big group of materials with biological matrix, viz., Oriental tobacco leaves (CTA-OTL-1) (Dybczyński et al. 1993); Virginia tobacco leaves (CTA-VTL-2) (Dybczyński et al. 1998); Tea leaves (INCT-TL-1), and Mixed Polish herbs (INCT-MPH-2) (Dybczyński et al. 2004a, 2004b); Corn flour (INCT-CF-3) and Soya bean flour (INCT-SBF-4) (Polkowska-Motrenko et al. 2007); Oriental Basma tobacco leaves (INCT-OBTL-5), and Polish Virginia tobacco leaves (INCT-PVTL-6) (Samczyński et al. 2012).
Collection and Preparation of Materials
MODAS–3 Herring Tissue
Herring tissue, originating from fishes living in the North Sea, was prepared by the Gdańsk University of Technology. All operations were performed taking care to minimize any possibility of the contamination of future CRM. After freeze-drying and grinding the material was sieved through the 200-μm sieve. Homogenization was performed in the Institute of Nuclear Chemistry and Technology, (INCT). The whole lot of the material (ca. 70 kg) was placed in a 110 dm3 polyethylene (PE) drum and mixed by rotating in three directions for 16 h. The homogenized material was distributed in 50 g portions to 100 cm3 amber glass bottles with screw cap (future MODAS–3 Herring Tissue (M–3 HerTis) certified reference material) and in 10 g portions to 60 cm3 (samples to be sent to the laboratories participating in the interlaboratory comparison), respectively. In order to ensure long-term stability of the new CRM, all containers with M–3 HerTis were sterilized by electron beam radiation (energy 13 MeV) from a linear accelerator LAEA-13. The dose amounted to approximately 27 kGy.
MODAS–4 Cormorant Tissue
Cormorant tissue, originating from the Czech Republic, was prepared by the Gdańsk University of Technology. After freeze-drying, grinding and sieving through the 200-μm sieve ca. 45 kg of the material was obtained. Homogenization, distribution, and sterilization were performed in the Institute of Nuclear Chemistry and Technology as described above.
MODAS–5 Cod Tissue
Cod tissue, originating from fishes living in the Baltic Sea, was prepared by the Gdańsk University of Technology. After freeze-drying, grinding and sieving through the 200-μm sieve ca. 70 kg of the material was obtained. Homogenization, distribution, and sterilization were performed in the Institute of Nuclear Chemistry and Technology as described above.
Testing of the Materials
Determination of Moisture Content
The moisture content (“dry state”) was established individually for MODAS–3 Herring Tissue (M–3 HerTis), MODAS–4 Cormorant Tissue (M–4 CormTis), and MODAS–5 Cod Tissue (M–5 CodTis) materials on the basis of the water desorption curves recorded at several selected temperatures. For all the above materials, the adopted moisture determination procedure involved drying a 5 g subsample for 48 h at 85 °C.
Homogeneity Studies
Homogeneity testing of M–3 HerTis, M–4 CormTis, and M–5 CodTis was performed for the sample size of 100 mg. Two subsamples were taken from different eleven containers, chosen at random from the whole population of units of the given material. They were analyzed by inductively coupled plasma mass spectrometry (ICP-MS), whereas statistical evaluation of results was carried out applying the analysis of variance (ANOVA) approach (ISO Guide 35:2017 2017, Linsinger et al. 2000, 2001a; Van der Veen and Pauwels, 2000; Van der Veen et al. 2001a). The following analytical procedure was used. Microwave-assisted digestion of samples was applied by means of a multiwave 3000 high-pressure microwave system (Anton Paar GmbH). Known amounts (ca. 100 mg) of the materials were weighed into special Teflon vessels and at first 6 mL of conc. HNO3 and 2 mL of 40% HF were added. When decomposition was completed, 6 mL portions of 4% H3BO3 solution were introduced to the samples and the vessels were re-sealed. This method ensures decomposition of silica and further removal of the excess of fluoride ions by complexation with boric acid. Solutions obtained as a result of microwave digestion were suitably diluted with 2% HNO3 + 5 ng mL−1 In-115 (internal standard) and then the mass fraction of selected elements was determined by ICP-MS. On the basis of obtained analytical results, the so-called mean squares among containers (MSa) as well as within containers (MSw) were calculated.
The standard uncertainty resulting from the inhomogeneity of M–3 HerTis, M–4 CormTis, and M–5 CodTis were evaluated using the ANOVA method (ISO Guide 35:2017 2017, Van der Veen et al. 2001a, b). Its estimation (so-called between bottle variance) was calculated from the MSa and MSw values (ISO Guide 35:2017 2017, Van der Veen et al. 2001a, b).
Stability Studies
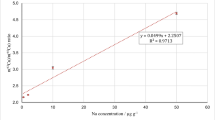
Long-term stability studies of newly issued CRMs are mandatory for their manufacturers (Ellison et al. 2001, ISO 17,034 2016, ISO Guide 35:2017 2017, Linsinger et al. 2000, Linsinger et al. 2001a, Linsinger et al. 2001b, Pauwels et al. 1998, Van der Veen et al. 2001b). In the case of M–3 HerTis, M–4 CormTis, and M–5 CodTis, it was realized by comparing the results obtained for samples stored under controlled conditions i.e. in the air-conditioned room at 20 °C (normal storage). Samples of the given material were taken from the randomly chosen container after 0, 2, 4, 6, 10, 12, and 15 months of storage. Mass fractions of selected elements were determined by ICP-MS in order to establish the relationship between the content of a given element (C) and time (x). Assuming a linear regression model of degradation, an estimate was fitted using the least square method (Linsinger et. al. 2001a, Linsinger et al. 2001b, Pauwels et al. 1998, Van der Veen et al. 2001b):
where b is the slope of the line (degradation rate). Long-term stability of the material was assessed by comparing |b|/ub to the value of the t-test (α = 0.05 and n – 2 degrees of freedom) where ub is the standard deviation of the slope of the fitted regression line (Linsinger et al. 2001a, b; Van der Veen et al. 2001b, ISO Guide 35:2017 2017).
The standard uncertainties associated with the long-term stability of M–3 HerTis, M–4 CormTis, and M–5 CodTis were determined on the basis of the obtained regression lines, calculating the standard deviation of their slope (ISO Guide 35:2017 2017, Linsinger et al. 2001a, Linsinger et al. 2001b).
The stability of reference materials in transport conditions (short-term stability) was also investigated. The materials were stored in CO2 incubator at 37 °C, 100% humidity, and 5% CO2 for 2 mounts and the changes in the concentration of selected elements were examined.
Element Content Characterization of the Materials
Interlaboratory Comparison
In order to certify the trace elements content in M–3 HerTis, M–4 CormTis, and M–5 CodTis candidate reference materials, a worldwide interlaboratory comparison was organized. The number of participating laboratories from eleven countries was 47, 46, and 50, respectively, providing the data for over 60 elements.
Samples of the mass 10 g of the candidate reference materials (so-called intercomparison samples) together with the information sheets were sent to the laboratories, which had previously confirmed participation in this study. It was requested to make at least three, but preferably six separate determinations for as many elements as they were able to, using a method of choice. The results should have been provided electronically in the specially designed MS Excel file (sent by e-mail). The participants were requested to report the determined concentrations (mass fractions) of elements as net values i.e. after correcting for blank (if any), moisture content, etc. The laboratories were also asked to provide for each element the following information: the average mass of the analyzed sample, detection limit of the method used, and the combined standard uncertainty together with the short description of the whole analytical procedure. This information was expected to contain the sample pretreatment method, preconcentration and separation procedure (if any), the technique of quantitative determination, and certified reference material(s) used for the quality control. In order to secure anonymity, the laboratories were coded and the code number has been known exclusively to the participant himself and to the organizers.
The components of the whole analytical procedure were also coded as shown in Table 2 for presumptive further more detailed analysis of the data. The excerpt of the so constructed database is shown in Fig. 1. In order to avoid any possible errors, the results of each laboratory were extracted individually from the database and sent back to the participant for checking. In the case of indicating any discrepancies, all necessary corrections were immediately made.
Data Evaluation
Results delivered by the laboratories participating in the interlaboratory comparison were organized in the form of a large database. It had a special structure, enabling work with the AQCS-1 package—the software employed for the data processing (Szopa et al. 1996). Analogously like in the case of our former certification campaigns, the database was subjected to statistical evaluation. The essential part of this process is the outlier’s rejection procedure. The population of laboratory averages for a given element is examined applying four statistical tests, namely those of Dixon (D), Grubbs (G), Skewness (S), and Kurtosis (K) at the significance level of 0.05. When a given result is classified as an outlier by any test, it is rejected from the population until no outlying result is detected. The final value of the overall mean is calculated from laboratory averages remaining after outliers’ rejection (c.f. Figure 2).
Results and Discussion
Materials Homogeneity and Stability Study
The candidate reference materials were prepared in such a way that they met the requirements of ISO Guide 35 (ISO 35:2017 2017) and ISO 17,034 (ISO 17034:20162016). Their homogeneity and stability were tested.
To assess the homogeneity, the minimum sample mass for which a given material can be considered homogeneous were specified, as well as the uncertainty associated with heterogeneity.
Natural matrix CRMs are always inhomogeneous on a microscopical scale. Apparent homogeneity is achieved by grinding, sieving, and mixing the material. When a great number of individual particles are present in the subsample of a given mass, then it can be assumed that all such subsamples have the same average elemental composition. Using a very simplified model (Harris and Kratochvil 1973, Dybczyński et.al. 2000, Dybczyński et.al. 2001), it was possible to derive a formula for the relative standard deviation due to inhomogeneity (sampling error), Rs:
where Ks is Ingamells sampling constant (Ingamells and Switzer 1973), msample is the sample mass (g), mparticle is the average mass of a single particle (g), and CA is the concentration (mass fraction) of an element A (g g−1). So, the sampling error is directly proportional to the square root of the mass of the single particle and inversely proportional to the square root of the mass fraction of an analyte (at constant sample mass). The sampling error increases strongly with the increase of dimension of the single particle (mparticle is roughly proportional to the third power of particle’s dimension) (Dybczyński et al. 2000, 2001; Dybczyński 2015).
As far as our previous CRMs are concerned, the minimum sample mass for which homogeneity was proved, usually amounted to 100 mg (Dybczyński et al. 1991, 1993; Dybczyński et al. 2004a, b; Polkowska-Motrenko et al. 2007; Samczyński et al. 2012). However, in one case (Virginia tobacco leaves CTA-VTL-2), i.e., for the material with a specially fine particle size (15–35 µm), thorough investigations revealed that the material was sufficiently homogenous even for the sample sizes of single milligrams (Dybczyński et al. 1998). Because of the specific properties of the materials being subject of this work, it was difficult to obtain a significant amount of particles much smaller than 200 μm. Basing on our previous experience, it was decided that the rational sample mass for which the homogeneity could be guaranteed should be around 100 mg. On the basis of obtained results (c.f. Table 3), it was stated that the materials are homogenous for the sample size of 100 mg or greater. The evaluated standard uncertainty due to inhomogeneity amounted to 2.00%, 1.90%, and 1.80% for the M-3 HerTis, M-4 CormTis, and M-5 CodTis, respectively.
Long-term stability studies of newly issued CRMs were performed in accordance with the recommendation for the manufacturers (Ellison et al. 2001, ISO 17,034 2016, ISO Guide 35:2017 2017, Linsinger et al. 2000, Linsinger et al. 2001a, b, Pauwels et al. 1998, Van der Veen et al. 2001b). On the basis of a long-term stability study, it was found that the presented CRMs should be stored at room temperature, protected from light, in a tightly closed container. In such conditions, it remains stable at least 5 years since the year of production. The values of standard uncertainty resulting from long-term storage of the materials were estimated to be 1.10%, 1.13%, and 1.07%, respectively. The uncertainty related to the short-term stability of the materials was found to be not significant and can be ignored. Also the dry states of the new CRMs were defined on the basis of a plateau of the water desorption curves. The reliable and reproducible procedure for the determination of moisture content was developed for all the materials to enable the analytical results to be related to the same state of the examined material.
Assigning of the Certified and Information Values
As the candidate reference materials met the criteria, it was possible to conduct an interlaboratory comparison and use the obtained data to assign the certified values for concentration (mass fraction) of elements. Those values were assigned when the overall mean calculated from laboratory averages remaining after outliers’ rejection met the quality criteria for the analytical uncertainty, the number of results, and number of analytical methods (Polkowska-Motrenko et al. 2010).
Each certified value has been assigned its combined uncertainty. In accordance with current ISO standards (ISO Guide 35:2017 2017, Linsinger et al. 2000), the combined standard uncertainty of the certified value uc, is calculated taking into account the following four components:
where uinterlab is standard analytical uncertainty estimated as the standard deviation of the overall mean, ulstab the standard uncertainty estimated from the long-term stability studies, uinhom the standard uncertainty estimated from the homogeneity studies, and um the standard uncertainty due to moisture determination. Afterwards, the expanded uncertainty (U), which corresponds to 95% confidence level, is calculated as the product of uc and the coverage factor k=t0.05 (Student’s t-test parameter for α=0.05 and n-1 degrees of freedom, where n is the number of laboratory averages). Certified values must always be quoted together with their uncertainties: \(\stackrel{-}{X\pm U}\). Traceability to the International System of Units (SI) was maintained, because participating laboratories along with the analysis of the candidate reference materials analyzed also CRMs of a similar matrix. Additionally, the traceability was established by use, when available, ratio primary reference measurement procedures (definitive methods) based on radiochemical neutron activation analysis (RNAA) and isotope dilution mass spectrometry (IDMS) as well as neutron activation analysis (NAA) procedures calibrated against pure metals or oxides with full uncertainty budget. Certified values with their uncertainties are quoted in Tables 4 and 5.
Several elements in M–3 HerTis, M–4 CormTis, and M–5 CodTis did not meet the criteria for the certified values however fulfilled the criterion for assigning the information values. The information values (cf. Table 5) are quoted as numbers only. Information value provides some idea about the mass fraction of a given element in the material, but should never be made equal with the certified value.
Finally, it was possible to assign the certified concentration (mass fraction) value to 30 elements in MODAS–3 Herring Tissue, 21 in MODAS–4 Cormorant Tissue, and 18 in MODAS–5 Cod Tissue, respectively. The information values were assigned for 3, 4, and 8 elements in the case of M-3 HerTis, M-4 CormTis, and M-5 CodTis, respectively.
Primary Reference Measurement Procedures (Definitive Methods) as an Auxiliary Tool in the Process of Certification of the Candidate Reference Materials.
Definitive methods (Uriano and Gravatt 1977) or primary reference measurement procedures (ISO/IEC Guide 99:2007 2007) are the methods of the highest metrological quality, which are used to check the accuracy of other routine methods employed in the chemical analysis of elements. Isotope dilution mass spectrometry (IDMS) is a well-established technique used for the very accurate determination of trace elements (De Bievre 1993; Vogl 2007). The other methods of similar metrological quality which can be used in the field of trace analysis are ratio primary reference measurement procedures (RPRMP) or definitive methods based on radiochemical neutron activation analysis (RNAA) (Dybczyński et al. 1989b, 2007, 2014; Dybczyński 1997, 2015). The essence of these methods is the combination of neutron activation with the post-irradiation selective and quantitative isolation of the desired radionuclide by column chromatography followed by γ-ray spectrometric measurement.
These methods have the advantage over the IDMS that they can be devised and used also in the case of monoisotopic elements. Both varieties of RPRMP (definitive methods) were used in this study for the determination of selected trace elements, employing the procedures previously devised for arsenic (Chajduk and Dybczyński 2010), cadmium (Samczyński and Dybczyński 1996), cobalt (Polkowska-Motrenko et al. 2004; Dybczyński and Danko 1994), chromium (Zuba and Polkowska-Motrenko 2019), iron (Dybczyński et al. 2012), mercury (Bulska et al. 2017), molybdenum (Danko and Dybczyński 1997), selenium (Chajduk et al. 2008), and uranium (Danko and Dybczyński 1997). Results by the definitive methods were not included in the population of intercomparison results but served for independent validation of the process of certification (cf. Table 6). As can be inferred from Table 6 in all cases, in which we managed to perform the measurements, there is a good agreement between the certified value and the result obtained by RPRMP (the confidence interval of the result by definitive method at 95% confidence level overlaps the uncertainty range of the certified value). The graphical presentation of selected examples together with the original range of results sent in by participants of the intercomparison is shown in Fig. 3.
a Comparison of the certified value and its confidence limits for Cd in M–4 CormTis with the results obtained using the definitive method (ratio primary reference measurement procedure) on the background of the original range of results submitted by participating laboratories. The “range of accepted laboratory averages” refers to the data after rejection of outlying results. b Comparison of the certified value and its confidence limits for As in M–5 CodTis with the results obtained using the definitive method (ratio primary reference measurement procedure) on the background of the original range of results submitted by participating laboratories. The “range of accepted laboratory averages” refers to the data after rejection of outlying results
The Share of Analytical Techniques in the Certification Process Now, in the Past, Its Evolution, and Consequences
Each certification campaign provides valuable information concerning analytical techniques employed when determining the individual elemental content in the candidate reference materials. Using the descriptions of the analytical procedure as supplied by the participating laboratories it was possible to calculate the relative frequency of the use of particular analytical techniques employed in this study. Graphical presentation of the share of the methods applied when analyzing M–4 CormTis is shown in Fig. 4. In the case of M–3 HerTis and M–5 Cod-Tis, the picture is roughly the same. As is evident, four methods occupy a predominant position in the certification, namely, inductive coupled plasma mass spectrometry (ICP-MS), atomic absorption spectrometry (AAS), neutron activation analysis (NAA), and emission spectroscopy (ES). Among them, ICP-MS distinctly prevails, what is the effect of its constant and dynamic development observed since the new millennium. The contribution of individual analytical techniques to the certification process and its evolution as a function of time was discussed in the literature (Dybczyński et al. 2003) and deserves a profound reflection. As follows from a recent review encompassing the time span of 40 years, in the years 1975–1980, the NAA was a dominating technique and its share was in the range of 40–66% (Dybczyński 2019). AAS occupied the second place followed by X-ray fluorescence (XRF), and ES. The contribution of the mass spectrometry at that time was minor or negligible. This trend was continued in the nineties although the prevalence of NAA became smaller (30–43%). At the end of the century, AAS became the technique most frequently used, followed by NAA and ICP-MS already showed its presence. In the first decade of the twenty-first century, four methods i.e. ICP-MS, NAA, ES, and AAS had almost equal share (23–25%) with marginal contribution from other methods (Samczyński et.al. 2012). Less than a decade later, the prevalence of ICP-MS is already clearly visible, similarly, as the further decline of the use of NAA (cf. Figure 4). If this trend will continue, it may lead to monopoly of ICP-MS and unavailability of NAA in the near future. The ICP-MS is an excellent method but as in the case of any other method, the systematic errors may occur for certain element–matrix combinations. The examples are e.g. the determination of scandium in INCT-TL-1 CRM (Tea leaves) (Dybczyński et al. 2017) and determination of iron in INCT-OBTL-5 CRM (Oriental Basma tobacco leaves) (Dybczyński et al., 2012). On the other hand, NAA is a nuclear method, less sensitive to matrix effects than the other methods of inorganic trace analysis, and at the same time, NAA is the only method that is essentially free from blank problems. Definitive methods by RNAA can be an addendum or alternative to IDMS as the methods of the highest metrological quality in the inorganic trace analysis. ICP-MS and NAA complement each other. It would be a pity if NAA would disappear or be unavailable in future certification campaigns. So, as was recently stated:”It should be in the interest of the whole scientific community, not only radioanalytical chemists, to keep this method alive still for many years to come” (Dybczyński 2019).
Conclusions
The new materials described in this paper extend the number of available CRMs of biological origin by two new species of sea fish tissue and one of bird tissue, respectively. The general rule for the use of CRMs is that the reference material should possibly closely mimic the samples being analyzed routinely, both with respect to matrix composition and to the mass fraction level(s) of the analyte(s). So, the new CRMs should be of interest to numerous researchers engaged in the study on the content of trace elements in food, environmental, and biomedical samples. The important scientific novelty of this work is the simultaneous use of a large number of ratio primary reference measurement procedures RPRMP (definitive methods) for independent confirmation and validation of certified values for nine essential or toxic trace elements. Very good agreement between the certified values assigned in this study and the results obtained by definitive methods for these elements, indirectly confirms also the validity and traceability of other certified values established by our standard certification procedure.
References
Abbey S (1977) Standard samples: how standard are they? Geostandards Newsletter 1:39–35
Abbey S (1981) Reliability in the analysis of rocks and minerals. Analytical Chemistry 53:528-534A
Abbey S, Rousseau RM (1985) Pragmatism vs. rigour: a debate on the resolution of disparate analytical data on four Canadian iron-formation reference samples. Geostandards Newsletter 9:1–16
Aazami J, KianiMehr N (2018) Survey of heavy metals in internal tissues of great cormorant collected from southern wetlands of Caspian Sea. Iranian Environment Monitoring and Assessment 190:52. https://doi.org/10.1007/s10661-017-6433-1
Baranowska I, Kowalski B, Polkowska-Motrenko H, Samczyński Z (2015) Trace metal determinations using voltamperometric (DPV-HMDE) and atomic absorption spectrometry (F-AAS and ET-AAS) in bottom sediment, cod, herring and cormorant tissue samples – candidates for certified reference materials. Polish Journal of Environmental Studies 204:1911–1017
Baranowska I, Buszewski B, Namieśnik J, Konieczka P, Magiera S, Polkowska-Motrenko H, Kościelniak P, Gadzała-Kopciuch R, Woźniakiewicz A, Samczyński Z, Kochańska K, Rutkowska M (2017) Development of potential candidate reference materials for drugs in bottom sediment, cod and herring tissues. Chemosphere 169:181–187
De Bievre P (1993) SAC 92. Isotope dilution mass spectrometry as a primary method of analysis. Analytical Proceedings 30:328–333. https://doi.org/10.1039/AP9933000328
Bulska E, Krata AA, Kałabun M, Wojciechowski M (2017) On the use of certified reference materials for assuring the quality of results for the determination of mercury in environmental samples. Environmental Science and Pollution Research 24:7889–7897. https://doi.org/10.1007/s11356-016-7262-4
Chajduk E, Polkowska-Motrenko H, Dybczyński RS (2008) A definitive RNAA method for selenium determination in biological samples. Uncertainty evaluation and assessment of degree of accuracy. Accreditation and Quality Assurance 13:443–451
Chajduk E, Dybczyński RS (2010) Highly accurate radiochemical neutron activation analysis of arsenic in biological materials involving selective isolation of arsenic by hybrid and conventional ion exchange. Microchimica Acta 168:37–44
Clement RE, Keith LH, Siu KWM (1997) 1997. Reference materials for environmental analysis. CRC Lewis Publishers, Boca Raton
Danko B, Dybczyński R (1997) Determination of molybdenum and uranium in biological materials by radiochemical neutron activation analysis. Journal of Radioanalytical and Nuclear Chemistry 216:51–57
Dybczyński R, Tugsavul A, Suschny O (1978) Problems of accuracy and precision of trace element determination in water as shown by recent IAEA intercomparison tests. Analyst 103:733–744
Dybczyński R, Tugsavul A, Suschny O (1979) Soil-5, a new IAEA certified reference material for trace element determinations. Geostandards Newsletter 3:61–87
Dybczyński R (1980) Comparison of effectiveness of various procedures for the rejection of outlying results and assigning consensus values in interlaboratory programs involving determination of trace elements or radionuclides. Analytica Chimica Acta 117:53–70
Dybczyński, R., Veglia, A., Suschny, O. (1980b). Milk powder (A–11) – a new IAEA reference material for trace and other element analysis. In: P. Bratter, P. Schramel (Eds.), Trace Element Analytical Chemistry in Medicine and Biology pp.657–674. New York: W.de Gruyter.
Dybczyński R (1981) Letter to the Editor, Rubeŝka-Dybczyński. Geostandards Newsletter 5:213–215
Dybczyński, R., Polkowska-Motrenko, H., Szopa, Z., Samczyński, Z. (1989a). A strategy of certification of a new CRM: Apatite Concentrate (CTA-AC-1) for multielement trace analysis and observations on performance of various analytical techniques, in: Proceedings of ISCRM'89, Certified Reference Materials, pp. 312–321, International Academic Publishers-Pergamon Press, Beijing, China, May 15–18, 1989.
Dybczyński R, Wasek M, Maleszewska H (1989b) A definitive method for the determination of small amounts of copper in biological materials by neutron activation analysis. Journal of Radioanalytical and Nuclear Chemistry 130:365–388
Dybczyński R, Polkowska-Motrenko H, Samczyński Z, Szopa Z (1991) Two new Polish geological-environmental reference materials: Apatite Concentrate (CTA-AC-1) and Fine Fly Ash (CTA-FFA-1). Geostandards Newsletter 15:163–185
Dybczyński R, Polkowska-Motrenko H, Samczyński Z, Szopa Z (1993) New Polish certified reference materials for multielement inorganic trace analysis. Fresenius’ Journal of Analytical Chemistry 345:99–103
Dybczyński R, Danko B (1994) Accurate determination of cobalt traces in several biological reference materials. Biological Trace Elements Research 43(45):615–625
Dybczyński, R. (1997) . The significance of very accurate RNAA methods RNAA methods for quality assurance and certification of candidate reference materials. Proc.Intern.Symp. on harmonization of health related environmental measurements using nuclear and isotopic techniques, Hyderabad, India, 4–7 November 1996. IAEA, Vienna, pp. 199–211.
Dybczyński R, Polkowska-Motrenko H, Samczyński Z, Szopa Z (1998) Virginia Tobacco Leaves (CTA-VTL-2) – new Polish CRM for inorganic trace analysis including microanalysis. Fresenius’ Journal of Analytical Chemistry 360:384–387
Dybczyński R, Danko B, Polkowska-Motrenko H (2000) NAA study on homogeneity of reference materials and their suitability for microanalytical techniques. Journal of Radioanalytical and Nuclear Chemistry 245:97–104
Dybczyński R, Danko B, Polkowska-Motrenko H (2001) Some difficult problems still existing in the preparation and certification of CRMs. Fresenius’ Journal of Analytical Chemistry 370:126–130
Dybczyński R (2002a) Considerations on the accuracy of determination of some essential and/or toxic elements in biological materials. Chemia Analityczna (warsaw) 47:325–334
Dybczyński R (2002b) ). Preparation and use of reference materials for quality assurance in inorganic trace analysis. Food Additives and Contaminants 19:928–938
Dybczyński R, Danko B, Kulisa K, Maleszewska E, Polkowska-Motrenko H, Samczyński Z, Szopa Z (2003) Performance and frequency of use of NAA and other techniques during the certification of two new Polish CRMs prepared by INCT. Czechoslovak Journal of Physics 53:A171–A179
Dybczyński R, Danko B, Kulisa K, Maleszewska E, Polkowska-Motrenko H, Samczyński Z, Szopa Z (2004a) Preparation and preliminary certification of two new Polish CRMs for inorganic trace analysis. Journal of Radioanalytical and Nuclear Chemistry 259:409–413
Dybczyński R, Danko B, Kulisa K, Maleszewska E, Polkowska-Motrenko H, Samczyński Z, Szopa Z (2004b) Final certification of two new reference materials for inorganic trace analysis. Chemia Analityczna (Warsaw) 49:143–158
Dybczyński RS, Danko B, Polkowska-Motrenko H, Samczyński Z (2007) RNAA in metrology: a highly accurate (definitive) method. Talanta 71:529–536
Dybczyński RS, Danko B, Pyszynska M, Polkowska-Motrenko H (2012) Ratio primary reference measurement procedure (RPRMP) for the determination of iron in biological materials by RNAA. Radiochimica Acta 100:409–416
Dybczyński RS, Polkowska-Motrenko H, Chajduk E, Danko B, Pyszynska M (2014) Recent advances in ratio primary reference measurement procedures (definitive methods) and their use in certification of reference materials and controlling assigned values in proficiency testing. Journal of Radioanalytical and Nuclear Chemistry 302:1295–1302. https://doi.org/10.1007/s10967-014-3607-y
Dybczyński RS, Polkowska-Motrenko H (2015) Certified reference materials for inorganic trace analysis. In: Baranowska I (ed) Handbook of Trace Analysis. Springer, Heidelberg, pp 49–73
Dybczyński RS (2015) 50 Years of adventures with neutron activation analysis with the special emphasis on radiochemical separations. Journal of Radioanalytical and Nuclear Chemistry 303:1067–1090. https://doi.org/10.1007/s10967-014-3822-6
Dybczyński RS, Samczyński Z, Bartosiewicz I, Kulisa K, Polkowska-Motrenko H, Pyszynska M, Zuba I (2017) Two new separation schemes for group isolation of rare earth elements (REE) from biological and other matrices and their determination by ICP-MS. NAA and Chromatographic Methods Nukleonika 62(3):199–211. https://doi.org/10.1515/nuka-2017-0030
Dybczyński RS (2019) The role of NAA in securing the accuracy of analytical results in the inorganic trace analysis. Journal of Radioanalytical and Nuclear Chemistry 322:1505–1515. https://doi.org/10.1007/s10967-019-06675-7
Hołyńska, B., Jasion, J., Lankosz, M., Ostrowski, A. (1987). Cabbage leaves CL–1 certified reference material for the trace analysis of plant materials.·Fresenius’ Journal of Analytical Chemistry, 328(7), 588–591.
Houserova P, Kuban V, Kracmar S, Sitko J (2007) Total mercury and mercury species in birds and fish in an aquatic ecosystem in the Czech Republic. Environmental Pollution 145:185–194
Ellison SLR, Burke S, Walker RF, Heydorn K, Månsson M, Pauwels J, Wegscheider W, Te Nijenhuis B (2001) Uncertainty for reference materials certified by interlaboratory study: recommendations of an international study group. Accreditation and Quality Assurance 6:274–277
Emons H, Held A, Ulberth F (2006) Reference materials as crucial tools for quality assurance and control in food analysis. Pure and Applied Chemistry 78:135–143
Harris WE, Kratochvil B (1973) Sampling variance in analysis for trace components in solids. Analytical Chemistry 46:313–315
Ingamells CO, Switzer P (1973) A proposed sampling constant for use in geochemical analysis. Talanta 20(6):547–568
ISO Guide 35 (2017). Certification of reference materials – general and statistical principles. Geneva: ISO.
ISO 17034:2016 (2016). General requirements for the competence of reference materials producers. Geneva: ISO.
ISO/IEC Guide 99:2007 (2007). International vocabulary of metrology – basic and general concepts and associated terms (VIM3). Geneva: ISO.
Kabata-Pendias A, Mukherjee AB (2007) Trace elements from soil to human. Springer, Berlin
Kocman, V., Foley L. M. Certification of four North-American gypsum rock samples type: calcium sulphate dehydrate, CaSO4 ·2H2O, GYP-A, GYP-B, GYP-C and GYP-D. Geostandards Newsletter 11, 87–102.
Linsinger TPJ, Pauwels J, Schimmel H, Lamberty A, Van der Veen AMH, Schumann G, Siekmann L (2000) Estimation of the uncertainty of CRMs in accordance with GUM: application to the certification of four enzyme CRMs. Fresenius’ Journal of Analytical Chemistry 368:589–594
Linsinger TPJ, Pauwels J, Van der Veen AMH, Schimmel H, Lamberty A (2001a) Homogeneity and stability of reference materials. Accreditation and Quality Assurance 6:20–25
Linsinger TPJ, Pauwels J, Lamberty A, Schimmel HG, Van der Veen AMH, Siekmann L (2001b) Estimating the uncertainty of stability for matrix CRMs. Fresenius’ Journal of Analytical Chemistry 370:183–188
Ozaki T, Enomoto S, Minai Y, Ambe S, Tominaga T (1997) Determination of lanthanides and other trace elements in ferns by instrumental neutron activation analysis. Journal of Radioanalytical and Nuclear Chemistry 217:117–124
Pauwels J, Lamberty A, Schimmel H (1998) Quantification of the expected shelf-life of certified reference materials. Fresenius’ Journal of Analytical Chemistry 361:395–399
Phillips KM, Wolf WR, Patterson KY, Sharpless KA, Amanna KR, Holden JM (2007) Summary of reference materials for the determination of the nutrient composition of foods. Accreditation and Quality Assurance 12:126–133
Polkowska-Motrenko H, Danko B, Dybczyński R (2004) Metrological assessment of the high accuracy RNAA method of Co determination in biological materials. Analytical and Bioanalytical Chemistry 379:221–226
Polkowska-Motrenko H, Dybczyński R, Chajduk E, Danko B, Kulisa K, Samczyński Z, Sypuła M, Szopa Z (2007) New Polish certified reference materials for inorganic trace analysis: corn flour (INCT-CF-3) and Soya Bean Flour (INCT-SBF-4). Chemia Analityczna (Warsaw) 52:361–376
Polkowska-Motrenko H, Dybczyński RS, Chajduk E (2010) Certification of reference materials for inorganic trace analysis: the INCT approach. Accreditation and Quality Assurance 15:245–250
Quevauviller PH, Griepink B (1996) Reference materials for quality assurance. In: Günzler H (ed) Accreditation and quality assurance in analytical chemistry. Springer, Berlin, pp 195–217
Qiu G, Li W, Li X, Zhou W (2005) Biological function of REE in plants and microbes. Journal of Rare Earths 23:645–652
Qiu G, Li W, Li X, Zhou W, Yang C (2005) Biological intelligence of rare earth elements in animal cells. Journal of Rare Earths 23:554–573
Rutkowska M, Kochańska K, Kandel L, Bajger-Nowak G, Słomińska M, Marć M, Chojnacka K, Polkowska-Motrenko H, Zabiegała B, Namieśnik J, Konieczka P (2018) Homogeneity study of candidate reference material (contaminated soil) based on determination of selected metals, PCBs and PAHs. Measurement 128:1–12
Samczyński Z, Dybczyński R (1996) Ion exchange behaviour of cadmium on amphoteric ion exchange resin Retardion 11A8 and its application for the determination of cadmium in biological materials by neutron activation analysis. Chemia Analityczna (Warsaw) 41:873–890
Samczyński, Z., Dybczyński, R. S., Polkowska-Motrenko, H., Chajduk, E., Pyszynska, M., Danko, B., Czerska, E., Kulisa, K., Doner, K., Kalbarczyk, P. (2012). Two new reference materials based on tobacco leaves: certification for over a dozen of toxic and essential elements. The Scientific World Journal Volume 2012, Article ID 216380, 16 pages. 10. 1100/2012/216380.
Skoric S, Visnjić-Jeftic Z, Jaric I, Djikanovic V, Mickovic B, Nikcevic M, Lenhardt M (2012) Accumulation of 20 elements in great cormorant (Phalacrocorax carbo) and its main prey, common carp (Cyprinus carpio) and Prussian carp (Carassius gibelio). Ecotoxicology and Environmental Safety 80:244–251
Szopa Z, Jaszczuk J, Dybczyński R (1996) A new multifunctional PC program for evaluation of interlaboratory comparison results. Nukleonika 41:117–127
Tyler G (2004) Rare earth elements in soil and plant systems – a review. Plant and Soil 267:191–206
Uriano GA, Gravatt CC (1977) The role of reference materials and reference methods in chemical analysis. Critical Reviews in Analytical Chemistry 6:361–411
Van der Veen AMH, Pauwels J (2000) Uncertainty calculations in the certification of reference materials. 1. Principles of analysis of variance. Accreditation and Quality Assurance 5:464–469
Van der Veen AMH, Linsinger T, Pauwels J (2001) Uncertainty calculations in the certification of reference materials. 2. Homogeneity Study Accreditation and Quality Assurance 6:26–30
Van der Veen AMH, Linsinger T, Lamberty A, Pauwels J (2001) Uncertainty calculations in the certification of reference materials 3. Stability Study Accreditation and Quality Assurance 6:257–263
Versieck J, Cornelis R (1980) Normal levels of trace elements in human blood or plasma. Analytica Chimica Acta 116(2):217–254
Vogl J (2007) Characterization of reference materials by isotope dilution mass spectrometry. Journal of Analytical Atomic Spectrometry 22:475–492. https://doi.org/10.1039/b614612k
Wise SA (2018) What is novel about certified reference materials ? Analytical and Bioanalytical Chemistry 410:2045–2049
Zuba I, Polkowska-Motrenko H (2019) Ratio primary reference measurement procedure (RPRMP) for the certification of chromium content in biological materials. Radiochimica Acta 107(2):141–147
Acknowledgements
The authors would like to thank all laboratories which participated in the laboratory intercomparison and provided the results of the analysis.
Funding
This work was supported by funds from the National Centre for Research and Development in the frame of the project MODAS No. INNOTECH-K1/IN1/43/138947/NCBR/12 for the period 2012–2015, Warsaw, Poland.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed Consent
Informed consent not applicable.
Conflict of Interest
Halina Polkowska-Motrenko declares that she has no conflict of interest. Zbigniew Samczyński declares that she has no conflict of interest. Rajmund S. Dybczyński declares that he has no conflict of interest. Ewelina Chajduk declares that she has no conflict of interest. Bożena Danko declares that she has no conflict of interest. Paweł Kalbarczyk declares that he has no conflict of interest. Agnieszka A. Krata declares that she has no conflict of interest. Marta Pyszynska declares that she has no conflict of interest. Iga Zuba declares that she has no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Polkowska-Motrenko, H., Samczyński, Z., Dybczyński, R.S. et al. Preparation of Three New Certified Reference Materials for Food and Environmental Analysis and Certification Using Laboratory Intercomparison as well as Primary Reference Measurement Procedures. Food Anal. Methods 15, 377–390 (2022). https://doi.org/10.1007/s12161-021-02081-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12161-021-02081-6