Abstract
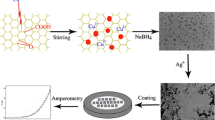
Employing a graphene-modified glassy carbon electrode, a sensitive and convenient electrochemical method for the determination of phoxim by linear sweep voltammetry was developed. The electrochemical behavior of phoxim at the modified electrode was studied by cyclic voltammetry. In citric acid–phosphate buffers, the modified electrode exhibited excellent electrocatalytical effect on the reduction of phoxim and this was further used for the determination of phoxim. Under optimized analytical conditions, the reduction peak current showed a linear relationship with the concentration of phoxim in a range of 5.97 to 5,966 μg L−1, with a correlation coefficient of 0.9993 and a detection limit of 2.39 μg L−1. The proposed method shows excellent sensitivity, selectivity, and linearity and has been successfully applied for the determination of phoxim in a variety of food samples with satisfactory results.






Similar content being viewed by others
References
Bard AJ, Faulkner LR (2001) Electrochemical Methods, 2nd edn. Wiley, New York
Chao M, Ma X, Li X (2012) Int J Electrochem Sci 7:2201–2213
European Commission (2010) Commission Regulation (EU) No 37/2010 of 22 December 2009 on pharmacologically active substances and their classification regarding maximum residue limits in foodstuffs of animal origin. Off J Eur Union L15:1–72
Geim AK, Novoselov KS (2007) Nat Mater 6:183–191
Gu C, Xiang B, Xu J (2012) Spectrochim Acta Part A 97:594–599
Guo L, Liang P, Zhang TZ, Liu Y, Liu S (2005) Chromatographia 61:523–526
Hamscher G, Priess B, Nau H (2007) Anal Chim Acta 586:330–335
Huang J, Zhang X, Lin Q, He X, Xing X, Huai H, Lian W, Zhu H (2011) Food Control 22:786–791
Huanshun Y, Shiyun A, Jing X, Weijie S, Lusheng Z (2009) J Electroanal Chem 637:21–27
Jin W, Yang G, Wu L, Wang Q, Shao H, Qin A, Yu B, Li D, Cai B (2011) Food Control 22:1609–1616
Keawkim K, Chuanuwatanakul S, Chailapakul O, Motomizu S (2013) Food Control 31:14–21
Laviron E (1979) J Electroanal Chem Interfacial Electrochem 101:19–28
Lee JH, Park S, Jeong WY, Park HJ, Kim HG, Lee SJ, Shim JH, Kim ST, El-Aty AMA, Im MH, Choi OJ, Shin SC (2010) Anal Chim Acta 674:64–70
Liang P, Guo L, Liu Y, Liu S, Zhang TZ (2005) Microchem J 80:19–23
Liang P, Xu J, Guo L, Song F (2006) J Sep Sci 29:366–370
Liu Q, Liu X, Qiu C, Wang X, Ren H (2009) Chin J Chromatogr 27:476–479
Lu X, Bai H, Ruan Q, Yang M, Yang G, Tan L, Yang Y (2008) Int J Environ Anal Chem 88:813–824
Lv Z, Gao L, Gao H, Hou Z, Zhang B (2009) J Food Sci 74:T37–T41
Ma X, Chao M, Wang Z (2012) Anal Methods 4:1687–1692
Ma X, Chao M, Chen M (2014) Russ J Electrochem 50:154–161
Ni Y, Cao D, Kokot S (2007) Anal Chim Acta 588:131–139
Qu G, Yang J, Li J, Xie L (1997) Chin J Chromatogr 15:252–253
Shen F, Yan Z, Ye Z, Ying Y (2009) Spectroscopy and Spectral Analysis 29:2421–2424
Wang L, Qi W, Su R, He Z (2014) Food Anal Methods 7:109–115
Zhang C, Zhang J, Wang K, Dai Z (2012) Acta Chim Sinica 70:1008–1012
Acknowledgements
This work was financially supported by a project of Shandong Province Higher Educational Science and Technology Program (J12LD53) and Heze University Scientific Research Fund (XY12BS07).
Conflict of Interest
Mingyong Chao declares that he has no conflict of interest. Meifeng Chen declares that she has no conflict of interest. This article does not contain any studies with human or animal subjects.
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
ESM 1
(DOC 0 kb)
Rights and permissions
About this article
Cite this article
Chao, M., Chen, M. Electrochemical Determination of Phoxim in Food Samples Employing a Graphene-Modified Glassy Carbon Electrode. Food Anal. Methods 7, 1729–1736 (2014). https://doi.org/10.1007/s12161-014-9813-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12161-014-9813-y