Abstract
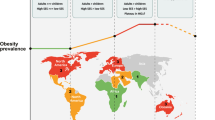
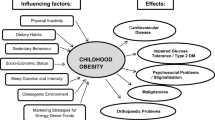
The idea that nutrition in early life (such as before conception, during pregnancy and in infancy) can influence, or programme, long-term health, known as the ‘Developmental Origins of Health and Disease Hypothesis’, has generated great scientific interest. This concept is particularly relevant for the development of obesity and its complications, arguably the most important public health issue of the twenty-first century worldwide. The concept is strongly supported by evidence from animal studies, both observational and experimental (randomised) studies in humans, and is highly relevant for population health in both low-income and high-incomes countries. For instance, optimising nutrition in pregnancy (both in terms of under-nutrition and over-nutrition) and preventing too fast infant weight gain have been shown to reduce the risk of future obesity. Proposed mechanisms have included effects of early nutrition on the epigenome, hormones such as insulin, and regulation of appetite, that effect long-term risk of obesity. Although further data from experimental studies is required to support a causal link between early nutrition and future adiposity, the developmental origins hypothesis is already changing health policy and practice globally. The present review considers the evidence for the developmental origins of obesity, the mechanisms involved, and the implications for public health.
Similar content being viewed by others
References
Heymsfield SB, Wadden TA. Mechanisms, pathophysiology, and management of obesity. N Engl J Med. 2017;376:254–66.
Hanson M, Mullins E, Modi N. Time for the UK to commit to tackling child obesity. BMJ. 2017;356:j762.
Baird J, Jacob C, Barker M, et al. Developmental origins of health and disease: a lifecourse approach to the prevention of non-communicable diseases. Healthcare (Basel). 2017;5.pii:E14.
Lucas A. Programming by early nutrition in man. In: Bock GR, Whelan J, editors. The Childhood Environment and Adult Disease. (CIBA Foundation Symposium 156): Chichester, UK: Whiley; 1991. p. 38–55.
Singhal A. The role of infant nutrition in the global epidemic of non-communicable disease. Proc Nutr Soc. 2016;75:162–8.
Jain V, Singhal A. Catch up growth in low birth weight infants: striking a healthy balance. Rev Endocr Metab Disord. 2012;13:141–7.
Tomar AS, Tallapragada DS, Nongmaithem SS, et al. Intrauterine programming of diabetes and adiposity. Curr Obes Rep. 2015;4:418–28.
Crowther NJ. Early determinants of chronic disease in developing countries. Best Pract Res Clin Endocrinol Metab. 2012;26:655–65.
Reddy SP, Mbewu AD. The implications of the developmental origins of health and disease on public health policy and health promotion in South Africa. Healthcare (Basel). 2016;4.pii:E83.
Barker DJ, Winter PD, Osmond C, Margetts B, Simmonds SJ. Weight in infancy and death from ischaemic heart disease. Lancet. 1989;2:577–80.
Barker DJP. Fetal origins of coronary heart disease. BMJ. 1995;311:171–4.
Oken E, Gillman MW. Fetal origins of obesity. Obes Res. 2003;11:496–506.
Bateson P, Barker D, Clutton-Brock T, et al. Developmental plasticity and human health. Nature. 2004;430:419–21.
Hanson MA, Gluckman PD. Developmental processes and the induction of cardiovascular function: conceptual aspects. J Physiol. 2005;565:27–34.
Hales CN, Barker DJ. Type 2 (non-insulin-dependent) diabetes mellitus: the thrifty phenotype hypothesis. Diabetologia. 1992;35:595–601.
Hattersley AT, Beards F, Ballantyne E, et al. Mutations in the glucokinase gene of the fetus result in reduced birth weight. Nat Genet. 1998;19:268–70.
Horikoshi M, Beaumont RN, Day FR, et al. Genome-wide associations for birth weight and correlations with adult disease. Nature. 2016;538:248–52.
Singhal A, Lucas A. Early origins of cardiovascular disease; is there a unifying hypothesis? Lancet. 2004;363:1642–5.
Ozanne SE, Hales N. Lifespan: catch-up growth and obesity in male mice. Nature. 2004;427:411–2.
Metcalfe NB, Monaghan P. Compensation for a bad start: grow now, pay later? Trends Ecol Evol. 2001;16:254–60.
Whincup PH, Kaye SJ, Owen CG, et al. Birth weight and risk of type 2 diabetes: a systematic review. JAMA. 2008;300:2886–97.
Fall CH. Fetal malnutrition and long-term outcomes. Nestle Nutr Inst Workshop Ser. 2013;74:11–25.
Haider BA, Bhutta ZA. Multiple-micronutrient supplementation for women during pregnancy. Cochrane Database Syst Rev. 2015;11:CD004905.
Ota E, Hori H, Mori R, Tobe-Gai R, Farrar D. Antenatal dietary education and supplementation to increase energy and protein intake. Cochrane Database Syst Rev. 2015;6:CD000032.
Devakumar D, Fall CH, Sachdev HS, et al. Maternal antenatal multiple micronutrient supplementation for long-term health benefits in children: a systematic review and meta-analysis. BMC Med. 2016;14:90.
Potdar RD, Sahariah SA, Gandhi M, et al. Improving women's diet quality preconceptionally and during gestation: effects on birth weight and prevalence of low birth weight--a randomized controlled efficacy trial in India (Mumbai maternal nutrition project). Am J Clin Nutr. 2014;100:1257–68.
Hawkesworth S. Conference on "multidisciplinary approaches to nutritional problems". Postgraduate symposium. Exploiting dietary supplementation trials to assess the impact of the prenatal environment on CVD risk. Proc Nutr Soc. 2009;68:78–88.
Godfrey KM, Reynolds RM, Prescott SL, et al. Influence of maternal obesity on the long-term health of offspring. Lancet Diabetes Endocrinol. 2017;5:53–64.
Woo Baidal JA, Locks LM, Cheng ER, et al. Risk factors for childhood obesity in the first 1,000 days: a systematic review. Am J Prev Med. 2016;50:761–79.
Smith J, Cianflone K, Biron S, et al. Effects of maternal surgical weight loss in mothers on intergenerational transmission of obesity. J Clin Endocrinol Metab. 2009;94:4275–83.
Patro B, Liber A, Zalewski B, et al. Maternal and paternal body mass index and offspring obesity: a systematic review. Ann Nutr Metab. 2013;63:32–41.
Dodd JM, Grivell RM, Louise J, et al. The effects of dietary and lifestyle interventions among pregnant women who are overweight or obese on longer-term maternal and early childhood outcomes: protocol for an individual participant data (IPD) meta-analysis. Syst Rev. 2017;6:51.
Patel N, Godfrey KM, Pasupathy D, et al. Infant adiposity following a randomised controlled trial of a behavioural intervention in obese pregnancy. Int J Obes. 2017;41:1018–26.
Patro-Gołąb B, Zalewski BM, Kołodziej M, et al. Nutritional interventions or exposures in infants and children aged up to 3 years and their effects on subsequent risk of overweight, obesity and body fat: a systematic review of systematic reviews. Obes Rev. 2016;17:1245–57.
Singhal A, Lanigan J. Breastfeeding, early growth and later obesity. Obes Rev. 2007;8:51–4.
Weber M, Grote V, Closa-Monasterolo R, et al. Lower protein content in infant formula reduces BMI and obesity risk at school age: follow-up of a randomized trial. Am J Clin Nutr. 2014;99:1041–51.
Gruszfeld D, Weber M, Gradowska K, et al; European Childhood Obesity Study Group. Association of early protein intake and pre-peritoneal fat at five years of age: follow-up of a randomized clinical trial. Nutr Metab Cardiovasc Dis. 2016;26:824–32.
Savage JS, Birch LL, Marini M, Anzman-Frasca S, Paul IM. Effect of the INSIGHT responsive parenting intervention on rapid infant weight gain and overweight status at age 1 year: a randomized clinical trial. JAMA Pediatr. 2016;170:742–9.
Fewtrell M, Bronsky J, Campoy C, et al. Complementary feeding: a position paper by the European Society for Paediatric Gastroenterology, hepatology, and nutrition (ESPGHAN) committee on nutrition. J Pediatr Gastroenterol Nutr. 2017;64:119–32.
Rolland-Cachera MF, Akrout M, Péneau S. Nutrient intakes in early life and risk of obesity. Int J Environ Res Public Health. 2016;13. Pii:E564.
Cole TJ. Children grow and horses race: is the adiposity rebound a critical period for later obesity? BMC Pediatr. 2004;4:6.
Michaelsen KF, Greer FR. Protein needs early in life and long-term health. Am J Clin Nutr. 2014;99:718S–22S.
Elks CE, Heude B, de Zegher F, et al. Associations between genetic obesity susceptibility and early postnatal fat and lean mass: an individual participant meta-analysis. JAMA Pediatr. 2014;168:1122–30.
Ramamoorthy TG, Begum G, Harno E, White A. Developmental programming of hypothalamic neuronal circuits: impact on energy balance control. Front Neurosci. 2015;9:126.
Alexander DD, Yan J, Bylsma LC, et al. Growth of infants consuming whey-predominant term infant formulas with a protein content of 1.8 g/100 kcal: a multicenter pooled analysis of individual participant data. Am J Clin Nutr. 2016;104:1083–92.
Acknowledgements
A. Singhal is supported by Great Ormond Street Hospital Children’s Charity and is a Stellenbosch Institute for Advanced Study (STIAS) Fellow, Wallenberg Research Centre at Stellenbosch University, Stellenbosch 7600, South Africa.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
None.
Source of Funding
Atul Singhal is supported by Great Ormond Street Hospital Children’s Charity.
Rights and permissions
About this article
Cite this article
Singhal, A. Early Life Origins of Obesity and Related Complications. Indian J Pediatr 85, 472–477 (2018). https://doi.org/10.1007/s12098-017-2554-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12098-017-2554-3