Abstract
In 2011, the Spanish Society of Medical Oncology (SEOM) first published a clinical guideline of venous thromboembolism (VTE) and cancer. This guideline was updated in 2014, and since then, multiple studies and clinical trials have changed the landscape of the treatment and prophylaxis of VTE in cancer patients. To incorporate the most recent evidence, including data from direct oral anticoagulants (DOACs) randomized clinical trials, SEOM presents a new update of the guideline.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cancer and venous thromboembolism (VTE) are two events that walk together [1]. Cancer patients have a four-to-sevenfold increased risk of VTE, being the second cause of preventable death in cancer patients. Therefore, the risk of suffering a severe hemorrhage is doubled when patients are on anticoagulation [2]. There are multiple factors contributing to increase that risk related to specific characteristics of the patient, tumor, and treatments. Moreover, VTE survival in cancer patients is shortened when we compared with similar non-oncologic patients. In recent years, new data have appeared on the pathophysiological relationship between cancer and thrombosis and the number of studies targeting specifically this population has increased. Although to date, the standard treatment for VTE in cancer is low-molecular-weight heparins (LMWH), studies with new anticoagulants open new horizons of research and could increase the therapeutic armamentarium for cancer patients with VTE. In this update of the SEOM guidelines to thrombosis and cancer, these data and their respective levels of evidence are reviewed as a useful tool for clinicians.
Methodology
SEOM guidelines have been developed with the consensus of ten oncologists from the Spanish Society of Medical Oncology (SEOM) and Cancer and Thrombosis Section. To assess the level and quality of evidence and to establish a grade of recommendation of the different statements in this guideline, we based ourselves on The Infectious Diseases Society of America-US Public Health Service Grading System (Table 1). The final text was reviewed and approved by all the authors. The goal of this document consists of providing clear practical recommendations about the management of VTE.
Prophylaxis
Prophylaxis of VTE in hospitalized medical cancer patients
Hospitalization is an important VTE-risk factor. Three large randomized phase III trials [3,4,5] reported a significant reduction in VTE following treatment with LMWH or fondaparinux compared with placebo, but none of them specifically in hospitalized cancer patients. The only evidence available is the subgroup analysis of the aforementioned trials and no major bleeding rates were reported (Table 2). Carrier et al. recently published a meta-analysis of the cancer subgroup of the three randomized clinical trials with contradictory results, showing no significant reduction in the incidence of VTE in cancer patients treated with prophylaxis [relative risk (RR) 0.91; 95% confidence interval (CI) 95% 0.21 to 4.0] [6]. This result was mainly driven by the ARTEMIS clinical trial, with an incidence rate of 17.0% of VTE with fondaparinux versus 3.0% with placebo. This higher incidence of VTE in the prophylaxis arm has not been well understood. Other reasons that could explain this result are the low number of patients included in the meta-analysis (overall 307 patients) and different VTE-risk cancer populations. These contradictory results highlight the lack of conclusive studies. Thus, randomized studies should be designed in this setting with a higher number of patients, and stratifications according to VTE risk and LMWH dose adjustment to obtain definitive conclusions.
Regarding thromboprophylaxis in surgical cancer patients, to extend thromboprophylaxis beyond hospitalization should be considered a matter of importance; however, there is no evidence yet to make this recommendation.
Finally, no specific trials have been performed with direct oral anticoagulants (DOACS) in this setting.
Recommendations
Although data from hospital populations in general cannot be directly extrapolated to cancer patients, in the absence of specific randomized studies for this population, anticoagulation should be considered in the case of hospitalized cancer patients with acute medical illness in the absence of contraindications. The preferred agents are LMWH (level of evidence: grade 1B).
Prophylaxis of VTE in surgical cancer patients
VTE is a common complication in cancer patients undergoing surgery, with a twofold or greater increased risk of DVT and four times more risk of a fatal postoperative PE compared to non-cancer population. VTE becomes the cause of a 10% postoperative early mortality rate.
The risk of VTE depends on specific factors of the patient, the tumor and the surgical procedure, the type and duration of the anesthesia, the advanced age of the subject, the residual disease after surgery, obesity, advanced stages of disease, prolonged immobility (more than 3 days), and the most important: background thromboembolism [7, 8].
Several randomized studies and meta-analysis have demonstrated the benefit of pharmacologic prophylaxis with LMWH and UFH over no prophylaxis or placebo unless there is a contraindication [9].
Pharmacological prophylaxis should be started preoperatively, and mechanical methods can be added to increase efficacy [10]. Mechanical methods should not be used as the sole treatment strategy unless there are contraindications for pharmacological prophylaxis (active bleeding or high-risk bleeding).
Classically in clinical practice, prophylaxis is continued for at least 7–10 days. Considering that 40% of VTE events may occur later than 21 days from the surgical intervention [11] and more than 50% after hospital discharge [12], multiple randomized clinical trials and meta-analysis have assessed the benefit of extended thromboprophylaxis in high-risk patients [13, 14]. Bergqvist et al. [15] reported the results of a double-blind trial that enrolled patients undergoing curative surgery for abdominal or pelvic cancer. Patients received 40 mg of enoxaparin daily for 6–10 days, randomized to receive either enoxaparin or placebo for another 21 days. The primary endpoint was the incidence of VTE between days 25 and 31. The incidence of VTE was 4.8% among patients treated with enoxaparin compared with 12.0% treated with placebo (p = 0.02). This difference was maintained after 3 months with a similar rate of bleeding between groups (5.5% vs. 13.8%). In 2016, Fagarasanu et al. [14] published a systematic review and meta-analysis that included seven randomized and prospective studies comprising 4807 patients. Extended thromboprophylaxis decreased the incidence of all VTE (RR 0.44, 95% CI 0.28–0.70) without significant difference in the incidence of major bleeding (RR 1.19, 95% CI 0.47–2.97). The update of the Cochrane review [13] concludes that prolonged thromboprophylaxis with LMWH significantly reduces the risk of VTE compared to thromboprophylaxis during hospital admittance without increasing bleeding complications or mortality.
The only trial that has assessed the benefit of extended thromboprophylaxis in laparoscopic surgery was published by Vedovati et al. [16]. Two hundred and twenty-five patients that underwent laparoscopic surgery for colorectal cancer were randomized to 1 week or 4 weeks of thromboprophylaxis with LMWH. VTE incidence rate at 4 weeks after surgery was 9.7% in the 1-week arm compared to 0% in the extended treatment arm (p = 0.001), with a similar incidence of bleeding and mortality rates. Similar efficacy results were observed at 3 months (VTE incidence 9.7% vs. 0.9%; p = 0.005).
Recommendations
In the absence of contraindications, all patients undergoing major surgical intervention should receive pharmacologic thromboprophylaxis (level of evidence: grade 1A). The preferred agents are LMWH and prophylaxis should be started before surgery or as soon as possible in the postoperative period. Mechanical methods may be added to pharmacologic prophylaxis in high-risk patients, but should not be used as monotherapy, unless pharmacologic prophylaxis is contraindicated (level of evidence: grade 2C). Patients should receive at least 7–10 days of prophylaxis and patients undergoing major abdominal or pelvic cancer surgery with high-risk features should be considered for extended thromboprophylaxis for 4 weeks (level of evidence: grade 1A). We suggest the same recommendations for laparoscopic surgery; risk factors and the duration and type of the procedure must be assessed (level of evidence: grade 2C).
Prophylaxis of VTE in ambulatory cancer patients during systemic therapy
Chemotherapy, hormonotherapy, and different biological and supportive care drugs have been identified as an independent risk factor for VTE. Incidence of thrombosis in these patients varies widely, so it is important to identify patients at higher risk of developing VTE whom prophylaxis may be beneficial. Recently, VTE incidence over 30% have been described in pancreatic cancer [17, 18] and specific molecular subtypes of non-small cell lung cancer (NSCLC) with ROS-1 [19] and ALK rearrangement [20, 21].
Cancer patients are usually unaware of VTE risk [22]. Self-consciousness and education can contribute to identify high-risk patients. In this sense, risk models may be useful to provide them with educational information and warnings.
A recent experience by The Ottawa Regional Cancer Center has used electronic medical records to calculate real-time risks of thrombosis [23]. Out of 580 patients included, 25% were identified as VTE high-risk patients, 11% of which developed VTE. Since many health centers use electronic medical records, the implementation of VTE-risk scores into clinical practice could be a feasible and meaningful measure. Although there is no clear evidence on ratios between them, based on available clinical data, it seems that performing a dynamic assessment and educating cancer patients on VTE risk would improve patients’ performance and outcomes.
Khorana score was the first validated risk assessment model (RAM) for identifying VTE high-risk patients receiving chemotherapy. After this publication, different RAM have been published and some of them have been validated (Table 3) [24,25,26,27,28,29,30,31,32]. Other RAM addressed to specific tumors such as THROLY [33] or testicular germ cell tumors [34].
Different randomized clinical trials and meta-analyses comparing anticoagulant prophylaxis with no intervention or placebo have evaluated the role of primary thromboprophylaxis with LMWH in ambulatory patients receiving anticancer therapy (Table 4) [35,36,37,38,39,40,41]. Overall, a significant reduction of 50% in VTE incidence rate was reported across such meta-analyses with LMWH compared with no intervention or placebo [42, 43]. This reduction was higher in patients with pancreatic cancer (82–74%), with an estimated number of patients needed to treat of 11 subjects to prevent one symptomatic VTE event [44], and lung cancer (58%) [34, 45]. Risk of minor bleeding is increased in the LMWH prophylaxis group without significant increase in major bleeding. No significant difference in mortality was reported by any of these studies.
Nowadays, DOACs have broken into clinical practice in cancer patients. Considering VTE prophylaxis, two trials have been recently published in moderate–high-risk ambulatory patients (Table 5). In the AVERT [46], trial apixaban was associated with a significantly lower incidence of VTE in the modified intention-to-treat population but also with a higher incidence of major bleeding episodes. Overall survival was similar in both arms. In contrast, in the CASSINI [47] trial, the incidence of VTE was not lower with rivaroxaban than placebo in the primary intention-to-treat analysis [6% rivaroxaban vs. 8.8% placebo, hazard ratio (HR) 0.66, IC 95% 0.40–1.09; p = 0.1]. However, in the per-protocol analysis, the VTE incidence was significantly lower with rivaroxaban than placebo (2.6% vs. 6.4%, HR 0.40, IC 95% 0.20–0.80). No differences in major bleeding or death were observed. Despite both studies used Khorana score ≥ 2 as inclusion criteria, the VTE incidence in the placebo arms was low (10.2–8.8%). Considered together, these trials showed a significant benefit of the oral anticoagulants for the prevention of VTE with an acceptable incidence of major bleeding. Recently, at least two meta-analyses have assessed the role of DOACs in this setting [36, 48]. Beccatini et al. [36] included three randomized clinical trials and describe a significant reduction of VTE with DOACs [odds ratio (OR) 0.49; 95% CI 0.33–0.74]. A similar VTE reduction was also observed in this study with parenteral thromboprophylaxis (OR 0.43; 95% CI 0.33–0.56). The second meta-analysis by Ll et al. showed an RR with DOACs for overall and symptomatic VTE incidence by 6 months of 0.56 (0.35–0.89) and 0.58 (0.29–1.13), respectively. No difference in major bleeding (RR 1.96, 95% CI 0.80–4.82) and clinically relevant non-major bleeding (RR 1.28, 95% CI 0.74–2.20) while on-treatment were observed. When pool together studies of thromboprophylaxis with primary endpoint VTE and survival, VTE was reduced by 50% (OR 0.49, 9 5% CI 0.43–0.61) [36]. There is a lack of data coming from these trials regarding drug interactions; therefore, DOACs are not recommended to be used concomitantly with potent inhibitors or inducers of P-glycoprotein or cytochrome P450 3A4.
Recommendations
Assessment of VTE risk in cancer patients in the outpatient setting is recommended at initiation of systemic therapy and during evolution of treatment and disease. It is recommended to use a validated RAM to assess VTE risk (level of evidence: grade 2C).
Routine thromboprophylaxis is not recommended in ambulatory patients with cancer (level of evidence: grade 1B).
Pharmacological thromboprophylaxis with LMWH or DOACs may be considered in high-risk ambulatory cancer patients, as advanced pancreatic cancer, NSCLC with ROS-1 or ALK rearrangement, patients with a Khorana score ≥ 2 or considered high-risk based on a validated RAM, starting of receiving systemic therapy and no contraindications to anticoagulation and low risk of bleeding. There is no consensus about the dose and duration of the thromboprophylaxis; it is suggested at least 12 weeks after the initiation a new systemic therapy. If the choice is thromboprophylaxis with DOACs, a specific drug–drug interaction assessment must be done. It is recommended to discuss with the patient the indication of pharmacological thromboprophylaxis and the potential risk and benefits. Patients who are receiving primary thromboprophylaxis should be closely monitored (level of evidence: grade 1B).
It is recommended to educate patient regarding VTE specifically, including risk factor and early symptoms, at the time of cancer diagnosis and during cancer evolution (level of evidence: 2A).
Prophylaxis of VTE in cancer patients with central venous catheters
Long-term central venous catheters (CVCs) are commonly used in patients with cancer. The placement of a CVC is associated with an increased risk of thrombotic events in upper limbs and pulmonary embolisms (PE). The reported incidence of CVC-associated thrombosis varies widely between studies (0.3–28% symptomatic events and 27–66% asymptomatic events detected by venography) [49]. Different factors may increase the risk of CVC-associated thrombosis, including material, placement technique, location and type of the catheter, tumor characteristics, treatment (chemotherapy, antiangiogenic therapy, hormone therapy, parenteral nutrition, and radiotherapy), and patient factors. Several randomized trials and meta-analyses evaluated the efficacy and safety of (vitamin K antagonists) VKA, UFH, LMWH, and thrombolytics in the prevention of CVC-associated thrombosis. These studies do not support the use of routine thromboprophylaxis for CVC in cancer patients. Several studies [50] including one meta-analysis [51] have suggested that CVC should be placed on the right side, in the jugular vein, and the catheter tip should be positioned at the right atrium/superior vena cava junction.
Recommendations
Routine thromboprophylaxis in cancer patients with CVCs is not recommended (level of evidence: grade 1A).
Catheters should be placed on the right side, in the jugular vein, and the distal tip should be located at the junction of the superior vena cava and the right atrium (level of evidence: grade 1B).
Treatment
The goals of anticoagulant therapy in cancer patients with cancer-associated thrombosis (CAT) are to improve symptoms, reduce risk of recurrent VTE, and decrease the risk of post-thrombotic syndrome (PTS). Anticoagulation is the cornerstone of treatment. Cancer patients present a higher risk of recurrent VTE and anticoagulant treatment-related bleeding compared to those without malignancy during anticoagulation therapy.
Initial treatment of VTE in cancer patients (5–10 days)
Since our previous recommendation of 2014 two Cochrane reviews have been published [52, 53]. Hakoum et al. [52] showed a trend to a decrease in mortality at 3 months (RR 0.66, 95% CI 0.40–1.10) and VTE recurrence (RR 0.69, 95% CI 0.27–1.76) with LMWH compared to unfractionated heparin (UFH). Compared to LMWH or UFH, fondaparinux was not statistically different in all endpoints including mortality, recurrence VTE, and bleeding. In the second meta-analysis [53], a subgroup analysis of cancer patients reported a significant reduction in mortality with LMWH compared to UFH (OR 0.53, 95% CI 0.33–0.85; p = 0.009). In the SELECT-D trial, upfront treatment with rivaroxaban showed similar efficacy compared to LMWH [54].
Recommendations
LMWH at a body weight-adjusted dose is the drug of choice for the initial treatment of CAT (level of evidence: grade 1B).
Rivaroxaban can be used as initial treatment of CAT if low risk of bleeding and in the absence of significant drug interactions (level of evidence: grade 1B).
UFH and fondaparinux can be considered alternative agents to LMWH or DOACs (level of evidence: grade 1B).
Long-term treatment of VTE in cancer patients
Since the previous update of the SEOM guideline in 2014, four randomized clinical trials have been presented, three with DOACs [54,55,56] and one with LMWH [57] (Table 6).
Multiple meta-analyses have shown that LMWH is more effective than VKAs at reducing the risk of recurrent VTE in patients with cancer [58]. The CATCH trial randomized 900 patients with active cancer and compared tinzaparin 175 IU/kg once daily versus warfarin for 6 months. At 6 months, it showed a non-significantly lower incidence of recurrent VTE with tinzaparin (7.2 vs. 10.5%, HR 0.65, 95% CI 0.41–1.03; p = 0.07). No differences in major bleeding (HR 0.89, 95% CI 0.40–1.99; p = 0.77) and mortality (HR 1.08, 95% CI 0.85–1.36; p = 0.54) were observed, though a significant reduction in clinically relevant non-major bleeding (CRNMB) was described in the tinzaparin arm (10.9 vs. 15.3%, HR 0.58, 95% CI 0.40–0.84; p = 0.004).
HOKUSAI Cancer VTE is the largest trial published to date regarding VTE treatment in cancer patient. It randomized 1050 patients to edoxaban after an initial course of at least 5 days of LMWH or dalteparin based on the CLOT regimen. It was designed as a non-inferiority trial. Edoxaban was administered at a fixed dose of 60 mg daily except in patients with creatinine clearance of 30–50 ml per minute, body weight of 60 kg or less, or in those receiving concomitant treatment with potent P-glycoprotein inhibitors that all received a reduced dose of 30 mg daily. The duration of the study was at least 6 months and up to 12 months. The primary endpoint was a composite of recurrent VTE or major bleeding up to 12 months after randomization. The median drug exposure was higher with edoxaban than dalteparin, 211 vs. 84 days. It should be noted that inconvenience dosing (patient decision) was the reason for permanent study drug discontinuation in 1 in 7 patients in the dalteparin arm compared to 1 in 25 patients in the edoxaban arm. Around one-fourth of the study population met the criteria to received edoxaban 30 mg. All types of cancer were represented in the trial including metastatic or primary brain tumors, and all systemic anticancer therapies were allowed. The primary endpoint was achieved; edoxaban was not inferior to dalteparin for the composite of recurrent VTE and major bleeding (edoxaban 12.8% vs. dalteparin 13.5%, HR 0.97, 95% CI 0.70–1.36; p = 0.006 for non-inferiority). In the secondary endpoints, a trend to a reduced recurrent VTE was observed for edoxaban (7.9% vs. 11.3%, HR 0.71, 95% CI 0.48–1.06; p = 0.09) with a significant increase in major bleeding (6.9% vs. 4.0%, HR 1.77, 95% CI 1.03–3.04; p = 0.04). The most common bleeding location with edoxaban was the gastrointestinal (GI) tract, in particular in the upper GI tract. In addition to genitourinary (GU) bleedings were more frequent with the oral anticoagulation. In a post hoc analysis, Kraaijpoel et al. [59] published later showed that tumors associated with major bleeding were predominantly GI cancers (major bleeding in GI cancers treated with edoxaban 12.5% vs. dalteparin 3.5%, HR 4.0, 95% CI 1.5–10.6, p = 0.005). The event-free survival and mortality rate were similar in the two arms, and must be pointed out that the main cause of death was cancer related (34.7% in the experimental arm and 32.8% with dalteparin) and only a minority of deaths were VTE related (1.1% with edoxaban and 0.8 in the LMWH arm).
SELECT-D trial is a pilot study that randomized 406 patients to rivaroxaban versus dalteparin (CLOT regimen). Rivaroxaban was administered orally 15 mg twice daily for 3 weeks, then 20 mg once daily up to 6 months. The primary endpoint was VTE recurrence, though no formal hypothesis was established. It was planned a second randomization after 6 months to continue rivaroxaban versus placebo, but it was closed due to poor recruitment based on data and safety monitoring committee recommendation and also the sample size was reduced to 406 patients. After an interim analysis, esophageal and gastric cancer were excluded because of a higher incidence of major bleeding with rivaroxaban compared to LMWH (36% vs. 11%). The VTE recurrence rate at 6 months was significantly lower with rivaroxaban compared to LMWH (4 vs. 11%, HR 0.43 95% CI 0.19–0.99). Again, as observed in the HOKUSAI trial, more major bleeding was described with DOACs compared to subcutaneous treatment (6-month cumulative rate of major bleeding for rivaroxaban 6% vs. 4% for dalteparin, HR 1.83, 95% CI 0.68–4.96). Also, a significant increase in the rate of CRNMB was associated to rivaroxaban (13 vs. 4%, HR 3.76, 95% CI 1.63–8.69). Most major bleeding and CRNMB occurred in the GI and GU tract.
In a recent meta-analysis [60] of these two trials, a non-significant lower incidence of 6-month recurrent VTE was observed (RR 0.65, 95% CI 0.42–1.01) together with a significant increase in the risk of major bleeding (RR 1.74, 95% CI 1.05–2.88) for DOACs in comparison with LMWH. Mortality was comparable in both arms (RR 1.03, 95% CI 0.85–1.26).
The ADAM VTE trial was presented in the 60th American Society of Hematology (ASH) meeting. It randomized 300 patients to apixaban 10 mg twice daily for 7 days followed by 5 mg twice daily or dalteparin (CLOT trial regimen) for 6 months. The primary endpoint was major bleeding. Major bleeding rate was similar in both arms (apixaban 0.0 vs. 2.1% dalteparin; p = 0.99) and a significant reduction in VTE recurrent rate was described with apixaban (3.4 vs. 14.1%, HR 0.26, 95% CI 0.09–0.80; p = 0.018). CRNMB was higher with apixaban than LMWH (6.2 vs. 4.2%). The secondary safety composite endpoint, major bleeding plus CRNMB, was comparable in both arms (apixaban 6.2 vs. dalteparin 6.3%, HR 0.9, 95% CI 0.41–1.94; p = 0.88). The quality of life showed globally better results for apixaban including concern for excess bruising, stress, irritation, burden of delivery, and overall satisfaction with anticoagulant therapy. The mortality rate at 6 months was similar comparing apixaban with dalteparin (15.9 vs. 10.6%, HR 1.36, 95% CI 0.79–2.35). These results must be taken with caution and must await the final publication and the outcomes of Caravaggio trial [61] (NCT03045406), that will randomize 1168 patients to the same regimens of apixaban and dalteparin used in ADAM trial for 6 months. The primary outcome of the study is objectively confirmed recurrent VTE and this trial will be the largest trial ever performed in CAT treatment setting.
Recommendations
LMWH at a body weight-adjusted dose and DOACs for 6 months are the drugs of choice for long-term treatment of VTE in cancer patients. DOACs must be used in low-risk bleeding patients (increased risk of GI and probably GU bleeding) and with no significant drug–drug interactions (level of evidence: grade 1A).
Extended duration of anticoagulation therapy after 6 months should be considered for high-risk patients such as those with active cancer and those receiving systemic therapy. Beyond 6 months, patients should be re-evaluated frequently to assess the risk–benefit ratio of continuing anticoagulant therapy (level of evidence: grade 2C).
Treatment of CVC-associated thrombosis (CVCAT)
The scientific evidence is scarce. It comes from some retrospective studies [62] and a small prospective study [63] but especially information from lower limb DVT is applied. LMWH is the treatment of choice during 3–6 months and considered indefinite treatment if the catheter is not removed and the cancer is present. The catheter should not be removed [64] unless it is no longer needed, it is infected, there is any contraindication to anticoagulation treatment, or there is no response to it. Removal should be done after 5–7 days of anticoagulant treatment. Related to DOACs, experience in CVCAT is limited [65] and comparison with LMWH is not available, but could be considered with the same nuances than in the rest of VTE.
Recommendations
It is recommended treatment with LMWH during 3–6 months or consider indefinite treatment if CVC is not removed (level of evidence: 2B). DOACs could be considered an option of CVCAT (level of evidence: 2C). We recommend not to remove the catheter unless it is not necessary, infected, anticoagulation treatment is contraindicated, or anticoagulation failure of appropriate therapy (level of evidence 2B).
Treatment of incidental thromboembolic events
Incidental VTE (iVTE) is a growing problem in cancer patients and accounts up to 50% of all VTE events in some retrospective studies, and it is likely to increase further with the improvements of imaging techniques. In the recent randomized clinical trials of DOACs vs. LMWH, incidental VTE events were included [54, 55]. Similar recurrent VTE, bleeding complications, and mortality rates, comparing incidental PE and deep vein thrombosis (DVT) with symptomatic events has been described [66, 67]. However, there is limited evidence about the optimal management of isolated, incidental subsegmental PE and incidental visceral vein thrombosis (iVVT), in particular splanchnic vein thrombosis (SVT) [68]. Some observational retrospective and prospective studies have suggested a similar outcome of incidental subsegmental PE compared to patients with more proximal clots or symptomatic events [67, 69], but the precise role of anticoagulation remains unknown in isolated cases. The available data in SVT or iVVT are even more scarce. Two international registries [70, 71] that included cancer patients (35–45% of the overall population) analyzed the outcomes and prognosis of incidental SVT. The prognosis of incidental SVT was comparable to the symptomatic SVT, and in the multivariable analysis, anticoagulation seemed to reduce the incidence of thrombotic events (HR 0.85, 95% CI 0.76–0.96) without impact in major bleeding risk.
Recommendations
Incidental VTE (PE and DVT) should be treated as symptomatic VTE and anticoagulation therapy with LMWH or DOACs is considered the standard treatment (level of evidence: 1B).
Treatment of isolated, incidental subsegmental PE, or incidental SVT should be individualized in every patient. Despite the low evidence available, it is suggested to consider anticoagulation therapy (level of evidence: 2C).
Treatment of recurrent VTE during anticoagulation therapy
The recurrent rate of VTE on anticoagulation therapy in the most recent randomized trials ranges between 4 and 11%. Type of tumor, stage, active cancer, cancer progression, and prior history of VTE have been found to be risk factors for VTE recurrence. There is a lack of randomize clinical to guide the management of recurrent VTE in cancer patients. An empirical approach for the management of recurrent VTE is proposed. First of all, in the setting of a recurrent event, doses and treatment compliance should be checked and also the absence of heparin induced thrombocytopenia (HIT) ruled out. Patient receiving VKA while the recurrence is diagnosed should switch to LWMH or DOACs. However, currently, the majority of cancer patients with VTE are treated with LWMH. If recurrent VTE occurs while infra-therapeutic LMWH, it is recommended to increase to therapeutic dose or switch to DOACs. If therapeutic dose of LMWH is being used, two retrospective cohort studies [72, 73] support the use of 25% dose escalation of LWMH. Even a small number of patients with another recurrent event despite dose escalation were managed successfully using further dose escalation. Monitoring anti-Xa levels to guide dose escalation could be considered, but the association between clinical efficacy of LMWH and anti-Xa levels is not completely probed. Alternatively, a switch to DOACs could be proposed based on the lower recurrent rate compared to LMWH described in the randomized clinical trials HOKUSAI, SELECT-D, and ADAM. Probably, the number of patients treated with DOACs will increase in the near future, and if recurrence VTE occurs, it is recommended to switch to LMWH based on expert opinions. Vena cava filters have been associated to an increased long-term risk of VTE and no impact in short- or long-term survival has been proved with its used [74,75,76]. Use of vena cava filter only could be justified if anticoagulant therapy is contraindicated or further recurrences occur despite proper anticoagulation is administered. Due to the uncertain benefit of vena cava filter insertion a retrievable filter is preferred and anticoagulant therapy should be resumed as soon as possible.
Recommendations
It is suggested that if recurrence occurs on VKA or prophylactic or intermediate doses of LMWH, therapeutic doses of LMWH or switch to DOACs should be initiated (level of evidence: grade 2B).
If recurrence occurs on therapeutic doses of LMWH, a 25% dose escalation should be considered or a switch to DOACs (level of evidence: grade 2B).
If recurrence occurs while treatment with DOACs switch to LMWH should be considered or alternatively DOACs dose escalation if infra-therapeutic dose was used (level of evidence: grade 3C).
Vena cava filter insertion may be considered if anticoagulation is contraindicated or further recurrent events occur despite appropriate anticoagulant therapy (failure of anticoagulation). Insertion of vena cava filter is associated with a lack of survival benefit and increased risk of long-term VTE. It is preferred a retrievable filter and anticoagulation should be resumed as soon as possible (level of evidence: grade 2B).
VTE treatment of central nervous system (CNS) primary tumors and metastasis
Since there are not specific trials for brain tumors, general rules for anticoagulation in CAT should be applied to these cases. However, some clinical data are available.
In a retrospective study with 182 patients with primary or metastatic brain tumors and 182 with other tumors with CAT, median duration of anticoagulation was 6.7 months [77]. No differences in the incidence of recurrence (11.0 vs. 13.5 cases per 100 patients-year, p = 0.26) or major bleeding (8.9 vs. 6.0 cases per 100 patients-year, p = 0.80) were observed.
At a second retrospective study with 293 patients with brain metastases, 104 patients with VTE were treated with enoxaparin [78]. No differences in intracranial bleeding were described for treated or not treated groups (44 vs. 37%, p = 0.13). Nevertheless, the incidence of hemorrhage was significantly higher for patients with melanoma or renal cancer, although not related to heparin used.
A recent meta-analysis including 1480 patients with neoplastic cerebral involvement studied the incidence of intracranial bleeding between anticoagulated and not patients [79]. The OR obtained were: 2.13 (95% CI 1.0–4.56) for all patients; 1.07 (95% CI, 0.61–1.88) for metastatic patients; and 3.75 (95% CI 1.42–9.95) for patients with glioma. Interestingly, the incidence of hemorrhage was not due to use of low-molecular weight heparin (OR 0.75, 95% CI 0.24–2.33).
A retrospective cohort study showed that DOACs are not associated with an increased incidence of intracranial bleeding compared to LMWH in patients with brain metastases or primary brain tumors.
Recommendations
In absence of contraindications, same recommendations for management of CAT as in other solid tumors should be applied for patients with primary or metastatic CNS tumors (level of evidence: grade 2B).
For patients with brain metastases from melanoma or kidney cancer, due to a greater risk of bleeding, a 25–50% reduction of LMWH dose may be considered (level of evidence: grade 2C).
For patients with brainstem glioma, due to potential severity of bleeding, a 25–50% reduction of LMWH is suggested (level of evidence: grade 2C).
Anticoagulation in the absence of VTE to improve survival in cancer patients
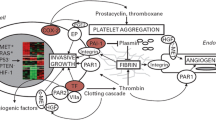
The existence of a relation between pathophysiological pathways of the hemostatic system and cancer development and progression has been confirmed by several preclinical research studies [80]. Hemostatic system activation would favor tumor transformation and progression, throughout angiogenesis, increasing local invasion and remote metastasis dissemination and growth. Quite relevant are: tissue factor, thrombin, and activated protease receptors. Tumor growth and metastatic process require a fibrin matrix as well as selectins, both implied in metastatic growth. Coagulation inhibition with anticlotting treatments might also play an antitumor role beyond antithrombotic effects. Preclinical studies have demonstrated that LMWH might inhibit or slow down tumor growth while binding to cell membrane selectins, inducing angiogenesis blockade, growth factors, and cell signaling inhibition which difficult extracellular tumor matrix formation [81].
Different clinical trials designed to study the prevention role of anticlotting treatments in the progression or relapse of VTE in cancer patients, which confirmed a potential survival benefit, larger than the one expected from VTE-risk reduction [82].
Several studies, comprising different patients and treatments, conducted to see the effect of LMWH (study primary end point) on global survival in the absence of VTE have shown contradictory results, in some cases statistically significant in subsets of patients with non-metastatic diseases, of a better prognosis, which triggered the hypothesis that they would benefit the most from adding LMWH to a standard treatment [83, 84].
In recent years, several studies have been published, focused on tumors with a high thrombotic potential, as lung cancer. These studies have shown no survival benefit from adding LMWH to ambulatory treatments or they have given uneven results. ABEL clinical trial evidenced a significant enhancement of the progression-free survival (HR 2.58) and overall survival (HR 2.96) by adding bemiparin to chemotherapy for treating small cell lung cancer patients with limited disease [85]. Other similar purpose studies conducted in lung cancer patients with different histological subtypes, including a bigger number of patients, have not confirmed any significant survival benefit from adding LMWH [40, 86].
Recent evidence analysis by Kahale et al. [87] could not confirm any survival benefit from treatments with oral anticoagulant agents, VKA, or DOACs in cancer patients with no previous VTE, but they did confirm an increase of bleeding risk.
Recently, it has been suggested that the determination of biomarkers related to the coagulation cascade activation and tumor progression would favor a correct selection of those patients that will benefit from anticoagulant treatments for enhancing survival [88].
Recommendation
Anticlotting treatment in cancer patients should not be prescribed with the purpose of enhancing survival, unless studied within the context of clinical trials (evidence level 1B).
References
Timp JF, Braekkan SK, Versteeg HH, Cannegieter SC. Epidemiology of cancer-associated venous thrombosis. Blood. 2013;122:1712–23.
Blon JW, Doggen CJ, Osanto S, Rosendaal FR. Malignancies, prothrombotic mutations, and the risk of venous thrombosis. JAMA. 2005;293:715–22.
Cohen AT, Davidson BL, Gallus AS, Lassen MR, Prins MH, Tomkowski W, et al. Efficacy and safety of fondaparinux for the prevention of venous thromboembolism in older acute medical patients: randomized placebo controlled trial. BMJ. 2006;332:325–9.
Samama MM, Cohen AT, Darmon JY, Desjardins L, Eldor A, Janbon C, et al. A comparison of enoxaparin with placebo for the prevention of venous thromboembolism in acutely ill medical patients. Prophylaxis in medical patients with enoxaparin study group. N Engl J Med. 1999;341:793–800.
Leizorovicz A, Cohen AT, Turpie AG, Olsson CG, Vaitkus PT, Goldhaber SZ, et al. Randomized, placebo-controlled trial of dalteparin for the prevention of venous thromboembolism in acutely ill medical patients. Circulation. 2004;110:874–9.
Carrier M, Khorana AA, Moretto P, Le Gal G, Karp R, Zwicker JI. Lack of evidence to support thromboprophylaxis in hospitalized medical patients with cancer. Am J Med. 2014;127:82–6.
Zurawska U, Parasuraman S, Goldhaber SZ. Prevention of pulmonary embolism in general surgery patients. Circulation. 2007;115(9):e302–7.
Agnelli G, Bolis G, Capussotti L, Scarpa RM, Tonelli F, Bonizzoni E, et al. A clinical outcome-based prospective study on venous thromboembolism after cancer surgery: the @RISTOS project. Ann Surg. 2006;243(1):89–95.
Matar CF, Kahale LA, Hakoum MB, Tsolakian IG, Etxeandia-Ikobaltzeta I, Yosuico VE, et al. Anticoagulation for perioperative thromboprophylaxis in people with cancer. Cochrane Database Syst Rev. 2018;7:CD009447.
Wille-Jørgensen P, Rasmussen MS, Andersen BR, Borly L. Heparins and mechanical methods for thromboprophylaxis in colorectal surgery. Cochrane Database Syst Rev. 2003;4:CD001217.
Agnelli G, Bolis G, Capussotti L, Scarpa RM, Tonelli F, Bonizzoni E, et al. A clinical outcome-based prospective study on venous thromboembolism after cancer surgery: the @RISTOS project. Ann Surg. 2006;243:89–95.
Bustos Merlo AB, Arcelus Martínez JI, Turiño Luque JD, Valero B, Villalobos A, Aibar MÁ, et al. Form of presentation, natural history and course of postoperative venous thromboembolism in patients operated on for pelvic and abdominal cancer. Analysis of the RIETE registry. Cir Esp. 2017;95:328–34.
Felder S, Rasmussen MS, King R, Sklow B, Kwaan M, Madoff R, et al. Prolonged thromboprophylaxis with low molecular weight heparin for abdominal or pelvic surgery. Cochrane Database Syst Rev. 2019;3:004318.
Fagarasanu A, Alotaibi GS, Hrimiuc R, Lee AY, Wu C, et al. Role of extended thromboprophylaxis after abdominal and pelvic surgery in cancer patients: a systematic review and meta-analysis. Ann Surg Oncol. 2016;23:1422–30.
Bergqvist D, Agnelli G, Cohen AT, Eldor A, Nilsson PE, Le Moigne-Amrani A, et al. Duration of prophylaxis against venous thromboembolism with enoxaparin after surgery for cancer. N Engl J Med. 2002;346:975–80.
Vedovati MC, Becattini C, Rondelli F, Boncompagni M, Camporese G, Balzarotti R, et al. A randomized study on 1-week versus 4-week prophylaxis for venous thromboembolism after laparoscopic surgery for colorectal cancer. Ann Surg. 2014;259(4):665–9.
Epstein AS, Soff GA, Capanu M, Crosbie C, Shah MA, Kelsen DP, et al. Analysis of incidence and clinical outcomes in patients with thromboembolic events and invasive exocrine pancreatic cancer. Cancer. 2012;118:3053–61.
Muñoz Martín AJ, García Alfonso P, Rupérez Blanco AB, Pérez Ramírez S, Blanco Codesido M, Martín Jiménez M. Incidence of venous thromboembolism (VTE) in ambulatory pancreatic cancer patients receiving chemotherapy and analysis of Khorana’s predictive model. Clin Transl Oncol. 2014;16:927–30.
Chiari R, Ricciuti B, Landi L, Morelli AM, et al. ROS1-rearranged non-small cell lung cancer is associated with high rate of venous thromboembolism: analysis from a phase II, prospective, multicentre, two arms trial (METROS). Clin Lung Cancer. 2019;pii: S1525-7304(19)30154-8. https://doi.org/10.1016/j.cllc.2019.06.012.
Zer A, Moskovitz M, Hwang DM, Hershko-Klement A, Fridel L, Korpanty GJ, et al. ALK-rearranged non-small-cell lung cancer is associated with a high rate of venous thromboembolism. Clin Lung Cancer. 2017;18:156–61.
Zugazagoitia J, Biosca M, Oliveira J, Olmedo ME, Dómine M, Nadal E, et al. Incidence, predictors and prognostic significance of thromboembolic disease in patients with advanced ALK-rearranged non-small cell lung cancer. Eur Respir J. 2018;51:1702431.
Aggarwal A, Fullam L, Brownstein AP, Maynard GA, Ansell J, Varga EA, et al. Deep vein thrombosis (DVT) and pulmonary embolism (PE): awareness and prophylaxis practices reported by patients with cancer. Cancer Invest. 2015;33:405–10.
Lustig DB, Rodriguez R, Wells PS. Implementation and validation of a risk stratification method at The Ottawa Hospital to guide thromboprophylaxis in ambulatory cancer patients at intermediate-high risk for venous thrombosis. Thromb Res. 2015;136:1099–102.
Khorana AA, Kuderer NM, Culakova E, Lyman GH, Francis CW. Development and validation of a predictive model for chemotherapy-associated thrombosis. Blood. 2008;111:4902–7.
Ay C, Dunkler D, Marosi C, Chiriac AL, Vormittag R, Simanek R, et al. Prediction of venous thromboembolism in cancer patients. Blood. 2010;116:5377–82.
Verso M, Agnelli G, Barni S, Gasparini G, LaBianca R. A modified Khorana risk assessment score for venous thromboembolism in cancer patients receiving chemotherapy: the Protecht score. Intern Emerg Med. 2012;7:291–2.
Pelzer U, Sinn M, Stieler J, Riess H. Primary pharmacological prevention of thromboembolic events in ambulatory patients with advanced pancreatic cancer treated with chemotherapy? Dtsch Med Wochenschr. 2013;138:2084–8.
Muñoz Martín AJ, Ortega I, Font C, Pachón V, Castellón V, Martínez-Marín V, et al. Multivariable clinical-genetic risk model for predicting venous thromboembolic events in patients with cancer. Br J Cancer. 2018;118:1056–61.
Muñoz A, Ay C, Gritz E, López S, Font C, Pachón V, et al. A clinical-genetic risk score to predict cancer-associated venous thromboembolism: a development and validation study in two independent prospective cohorts. Oral communication OC 15.5. In: ISTH 2019: the XXVII congress of the International Society On Thrombosis And Haemostasis, Melbourne, Australia. 2019.
Gerotziafas GT, Taher A, Abdel-Razeq H, AboElnazar E, Spyropoulos AC, El Shemmari S, et al. A predictive score for thrombosis associated with breast, colorectal, lung, or ovarian cancer: the prospective compass-cancer-associated thrombosis study. Oncologist. 2017;22(10):1222–31.
Cella CA, Di Minno G, Carlomagno C, Arcopinto M, Cerbone AM, Matano E, et al. Preventing venous thromboembolism in ambulatory cancer patients: the ONKOTEV study. Oncologist. 2017;22:601–8.
Pabinger I, van Es N, Heinze G, Posch F, Riedl J, Reitter EM, et al. A clinical prediction model for cancer-associated venous thromboembolism: a development and validation study in two independent prospective cohorts. Lancet Haematol. 2018;5:e289–98.
Antic D, Milic N, Nikolovski S, Todorovic M, Bila J, Djurdjevic P, et al. Development and validation of multivariable predictive model for thromboembolic events in lymphoma patients. Am J Hematol. 2016;91:1014–9.
Bezan A, Posch F, Ploner F, Bauernhofer T, Pichler M, Szkandera J, et al. Risk stratification for venous thromboembolism in patients with testicular germ cell tumors. PLoS ONE. 2017;12(4):e0176283.
Agnelli G, Gussoni G, Bianchini C, Verso M, Mandala M, Cavanna L, et al. Nadroparin for the prevention of thromboembolic events in ambulatory patients with metastatic or locally advanced solid cancer receiving chemotherapy: a randomised, placebo-contolled, double-blind study. Lancet Oncol. 2009;10:943–9.
Maraveyas A, Waters J, Roy R, Fyfe D, Propper D, Lofts F, et al. Gemcitabine versus gemcitabine plus dalteparin thromboprophylaxis in pancreatic cancer. Eur J Cancer. 2012;48:1283–92.
Pelzer U, Opitz B, Deutschinoff G, Pelzer U, Opitz B, Deutschinoff G, et al. Efficacy of prophylactic low-molecular weight heparin for ambulatory patients with advanced pancreatic cancer: outcomes from the CONKO-004 trial. J Clin Oncol. 2015;33:2028–34.
Agnelli G, George DJ, Kakkar AK, Fisher W, Lassen MR, Mismetti P, et al. Semuloparin for thromboprophylaxis in patients receiving chemotherapy for cancer. N Engl J Med. 2012;366:601–9.
Perry JR, Julian JA, Laperriere NJ, Geerts W, Agnelli G, Rogers LR, et al. PRODIGE: a randomized placebo-controlled trial of dalteparin low-molecular-weight heparin thromboprophylaxis in patients with newly diagnosed malignant glioma. J Thromb Haemost. 2010;8:1959–65.
Macbeth F, Noble S, Evans J, Ahmed S, Cohen D, Hood K, et al. Randomized phase III trial of standard therapy plus low molecular weight heparin in patients with lung cancer: FRAGMATIC trial. J Clin Oncol. 2016;34:488–94.
Khorana AA, Francis CW, Kuderer NM, Carrier M, Ortel TL, Wun T, et al. Dalteparin thromboprophylaxis in cancer patients at high risk for venous thromboembolism: a randomized trial. Thromb Res. 2017;151:89–95.
Di Nisio M, Porreca E, Candeloro M, De Tursi M, Russi I, Rutjes AW, et al. Primary prophylaxis for venous thromboembolism in ambulatory cancer patients receiving chemotherapy. Cochrane Database Syst Rev. 2016;12:CD008500.
Ben-Aharon I, Stemmer SM, Leibovici L, Shpilberg O, Sulkes A, Gafter-Gvili A. Low molecular weight heparin (LMWH) for primary thrombo-prophylaxis in patients with solid malignancies—systematic review and meta-analysis. Acta Oncol. 2014;53:1230–7.
Tun NM, Guevara E, Oo TH. Benefit and risk of primary thromboprophylaxis in ambulatory patients with advanced pancreatic cancer receiving chemotherapy: a systematic review and meta-analysis of randomized controlled trials. Blood Coagul Fibrinolysis. 2016;27:270–4.
Becattini C, Verso M, Muňoz A, Agnelli G. Updated meta-analysis on prevention of venous thromboembolism in ambulatory cancer patients. Haematologica. 2019;pii: haematol.2019.221424. https://doi.org/10.3324/haematol.2019.221424.
Carrier M, Abou-Nassar K, Mallick R, Tagalakis V, Shivakumar S, Schattner A, et al. Apixaban to prevent venous thromboembolism in patients with cancer. N Engl J Med. 2019;380:711–9.
Khorana AA, Soff GA, Kakkar AK, Vadhan-Raj S, Riess H, Wun T, et al. Rivaroxaban for thromboprophylaxis in high-risk ambulatory patients with cancer. N Engl J Med. 2019;380:720–8.
Li A, Kuderer NM, Garcia DA, Khorana AA, Wells PS, Carrier M, et al. Direct oral anticoagulant for the prevention of thrombosis in ambulatory patients with cancer: a systematic review and meta-analysis. J Thromb Haemost. 2019;17(12):2141–2151. https://doi.org/10.1111/jth.14613(Epub ahead of print).
Verso M, Agnelli G. Venous thromboembolism associated with long-term use of central venous catheters in cancer patients. J Clin Oncol. 2003;21:3665–75.
Lee AY, Levine MN, Butler G, Webb C, Constantitni L, Gu C, et al. Incidence, risk factors, and outcomes of catheter-related thrombosis in adult patients with cancer. J Clin Oncol. 2006;24:1404–8.
Saber W, Moua T, Williams EC, Verso M, Agnelli G, Couban S, et al. Risk factors of catheter-related thrombosis (CRT) in cancer patients: a patient-level data (IPD) meta-analysis of clinical trials and prospective studies. J Thromb Haemost. 2011;9:312–9.
Hakoum MB, Kahale LA, Tsolakian IG, Matar CF, Yosuico VE, Terrenato I, et al. Anticoagulation for the initial treatment of venous thromboembolism in people with cancer. Cochrane Database Syst Rev. 2018;1:CD006649.
Robertson L, Jones LE. Fixed dose subcutaneous low molecular weight heparins versus adjusted dose unfractionated heparin for the initial treatment of venous thromboembolism. Cochrane Database Syst Rev. 2017;2:CD001100.
Young AM, Marshall A, Thirlwall J, Chapman O, Lokare A, Hill C, et al. Comparison of an oral factor Xa inhibitor with low molecular weight heparin in patients with cancer with venous thromboembolism: results of a randomized trial (SELECT-D). J Clin Oncol. 2018;36:2017–23.
Raskob GE, van Es N, Verhamme P, Carrier M, Di Nisio M, Garcia D, et al. Edoxaban for the treatment of cancer-associated venous thromboembolism. N Engl J Med. 2018;378:615–24.
McBane RD 2nd, Wysokinski WE, Le-Rademacher JG, Zemla T, Ashrani A, Tafur A, et al. Apixaban and dalteparin in active malignancy-associated venous thromboembolism: the ADAM VTE trial. J Thromb Haemost. 2019. https://doi.org/10.1111/jth.14662.
Lee AYY, Kamphuisen PW, Meyer G, Bauersachs R, Janas MS, Jarner MF, et al. Tinzaparin vs warfarin for treatment of acute venous thromboembolism in patients with active cancer: a randomized clinical trial. JAMA. 2015;314(7):677–86.
Kahale LA, Hakoum MB, Tsolakian IG, Matar CF, Terrenato I, Sperati F, et al. Anticoagulation for the long-term treatment of venous thromboembolism in people with cancer. Cochrane Database Syst Rev. 2018;6:CD006650.
Kraaijpoel N, Di Nisio M, Mulder FI, van Es N, Beyer-Westendorf J, Carrier M, et al. Cinical impact of bleeding in cancer-associated venous thromboembolism: results from the Hokusai Vte cancer study. Thromb Haemost. 2018;118:1439–49.
Li A, Garcia DA, Lyman GH, Carrier M. Direct oral anticoagulant (DOAC) versus low-molecular-weight heparin (LMWH) for treatment of cancer associated thrombosis (CAT): a systematic review and meta-analysis. Thromb Res. 2019;173:158–63.
Agnelli G, Becattini C, Bauersachs R, Brenner B, Campanini M, Cohen A, et al. Apixaban versus dalteparin for the treatment of acute venous thromboembolism in patients with cancer: the caravaggio study. Thromb Haemost. 2018;118:1668–78.
Delluc A, Le Gal G, Scarvelis D, Carrier M. Outcome of central venous catheter associated upper extremity deep vein thrombosis in cancer patients. Thromb Res. 2015;135:298–302.
Savage KJ, Wells PS, Schulz V, Goudie D, Morrow B, Cruickshank M, et al. Outpatient use of low molecular weight heparin (Dalteparin) for the treatment of deep vein thrombosis of the upper extremity. Thromb Haemost. 1999;82:1008–10.
Kovacs MJ, Kahn SR, Rodger M, Anderson DR, Andreou R, Mangel JE, et al. A pilot study of central venous catheter survival in cancer patients using low-molecular-weight heparin (dalteparin) and warfarin without catheter removal for the treatment of upper extremity deep vein thrombosis (the Catheter study). J Thromb Haemost. 2007;5:1650–3.
Davies GA, Lazo-Langner A, Gandara E, Rodger M, Tagalakis V, Louzada M, et al. A prospective study of Rivaroxaban for central venous catheter associated upper extremity deep vein thrombosis in cancer patients (Catheter 2). Thromb Res. 2018;162:88–92.
den Exter PL, Hooijer J, Dekkers OM, Huisman MV. Risk of recurrent venous thromboembolism and mortality in patients with cancer incidentally diagnosed with pulmonary embolism: a comparison with symptomatic patients. J Clin Oncol. 2011;29:2405–9.
van der Hulle T, den Exter PL, Planquette B, Meyer G, Soler S, Monreal M, et al. Risk of recurrent venous thromboembolism and major hemorrhage in cancer-associated incidental pulmonary embolism among treated and untreated patients: a pooled analysis of 926 patients. J Thromb Haemost. 2016;14:105–13.
Kreuziger LB, Ageno W, Lee A. Management of incidental splanchnic vein thrombosis in cancer patients. Hematology Am Soc Hematol Educ Program. 2014;2014:318–20.
Kraaijpoel N, Bleker SM, Meyer G, Mahé I, Muñoz A, Bertoletti L, et al. Treatment and long-term clinical outcomes of incidental pulmonary embolism in patients with cancer: an international prospective cohort study. J Clin Oncol. 2019;37:1713–20.
Riva N, Ageno W, Schulman S, Beyer-Westendorf J, Duce R, Malato A, et al. Clinical history and antithrombotic treatment of incidentally detected splanchnic vein thrombosis: a multicentre, international prospective registry. Lancet Haematol. 2016;3:e267–75.
Tufano A, Ageno W, Di Micco P, Niglio A, Rosa V, Ballaz A, et al. Outcomes during anticoagulation in patients with symptomatic vs. incidental splanchnic vein thrombosis. Thromb Res. 2018;164:69–74.
Ihaddadene R, Le Gal G, Delluc A, Carrier M. Dose escalation of low molecular weight heparin in patients with recurrent cancer-associated thrombosis. Thromb Res. 2014;134:93–5.
Carrier M, Le Gal G, Cho R, Tierney S, Rodger M, Lee AY. Dose escalation of low molecular weight heparin to manage recurrent venous thromboembolic events despite systemic anticoagulation in cancer patients. J Thromb Haemost. 2009;7:760–5.
PREPIC Study Group. Eight-year follow-up of patients with permanent vena cava filters in the prevention of pulmonary embolism: the PREPIC (Prevention du Risque d’Embolie Pulmonaire par Interruption Cave) randomized study. Circulation. 2005;112:416–22.
Brunson A, Ho G, White R, Wun T. Inferior vena cava filters in patients with cancer and venous thromboembolism (VTE) does not improve clinical outcomes: a population-based study. Thromb Res. 2017;153:57–64.
Turner TE, Saeed MJ, Novak E, Brown DL. Association of inferior vena cava filter placement for venous thromboembolic disease and a contraindication to anticoagulation with 30-day mortality. JAMA Netw Open. 2018;1:e180452.
Chai-Adisaksopha C, Linkins LA, Alkindi SY, Cheah M, Crowther MA, Iorio A, et al. Outcomes of low-molecular-weight heparin treatment for venous thromboembolism in patients with primary and metastatic brain tumours. Thromb Haemost. 2017;117:589–94.
Donato J, Campigotto F, Uhlmann EJ, Coletti E, Neuberg D, Weber GM, et al. Intracranial hemorrhage in patients with brain metastases treated with therapeutic enoxaparin: a matched cohort study. Blood. 2015;126:494–9.
Zwicker JI, Leaf RK, Carrier M. A meta-analysis of intracranial hemorrhage in patients with brain tumors receiving therapeutic anticoagulation. J Thromb Haemost. 2016;14:1736–40.
Remiker AS, Palumbo JS. Mechanisms coupling thrombin to metastasis and tumorigenesis. Thromb Res. 2018;164(Suppl 1):S29–33.
Mousa SM, Peteresn LJ. Anti-cancer properties of low-molecular weight heparin: preclinical evidence. Thromb Haemost. 2009;102:258–67.
García-Escobar I, Beato-Zambrano C, Muñoz-Langa J, Brozos-Vázquez E, Obispo-Portero B, Gutiérrez Abad D, et al. Pleiotropic effects of heparins: does anticoagulant treatment increase survival in cancer patients? Clin Tansl Oncol. 2018;20:1097–108.
Klerk CP, Smorenburg SM, Otten HM, Lensing AW, Prins MH, Piovella F, et al. The effect of low molecular weight heparin on survival in patients with advanced malignancy. J Clin Oncol. 2005;23:2130–5.
Kakkar AK, Levine MN, Kadziola Z, Lemoine NR, Low V, Patel HK, et al. Low molecular weight heparin, therapy with dalteparin, and survival in advanced cancer: the Fragmin advanced malignancy outcome study (FAMOUS). J Clin Oncol. 2004;22:1944–8.
Lecumberri R, López-Vivanco G, Font A, Billalabeitia EG, Gurpide A, Codina JG, et al. Adjuvant therapy with bemiparin in patients with limited-stage small cell lung cancer: results from the ABEL study. Thromb Res. 2013;132:666–70.
Meyer G, Besse B, Doubre H, Charles-Nelson A, Aquilanti S, Izadifar A, et al. Anti-tumour effect of low molecular weight heparin in localised lung cancer: a phase III clinical trial. Eur Respir J. 2018;52:1801220.
Kahale LA, Hakoum MB, Tsolakian IG, Matar CF, Barba M, Yosuico VED, et al. Oral anticoagulation in people with cancer who have no therapeutic or prophylactic indication for anticoagulation. Cochrane Database Syst Rev. 2017;12:CD006466.
Spek CA, Versteeg HH, Borensztajn KS. Anticoagulant therapy of cancer patients: will patient selection increase overall survival? Thromb Haemost. 2015;114:530–6.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
AMM reports consulting or advisory role from Celgene, Sanofi, Pfizer, Bristol-Myers Squibb, LEO Pharma, Daiichi Sankyo, Bayer and Halozyme; Speakers’ Bureau from Rovi; Research Funding from Sanofi and LEO Pharma; Travel, Accommodations, Expenses from Celgene, Roche and Merck Serono. Patents, Royalties, Other Intellectual Property: Risk assessment model in venous thromboembolism in patients with cancer. EG reports from the work under consideration for publication Consultant or advisory role from Rovi, Daiichi Sankyo, Techdow, Sanofi, Pfizer and BMS. Speakers’ Bureau from Rovi, Daiichi Sankyo, Leo Pharma and Sanofi. Travel, Accommodations and Expenses from Rovi, Daiichi Sankyo and Leo Pharma. Relevant financial activities outside the submitted work: Consultant or Advisory Role from Bayer, Merck, Ipsen, Novartis, Eisai, EUSA Pharma, Roche, Janssen, Astellas and AstraZeneca. Speakers’ Bureau from Bayer, Pfizer, BMS, Ipsen, Novartis, Eisai, Roche, Astellas, Janssen, Menarini and MSD. Travel, Accommodations and Expenses from Pfizer, Bayer, Ipsen, Roche, Novartis, Eisai, BMS, Pierre Fabre, Astellas, Janssen and Sanofi. Grant support (personal/institutional) from Pfizer, Bayer, Ipsen, Roche, BMS, Astellas, Janssen, Sanofi, Ferrer. IGE reports from the work under consideration for publication, Consultant or Advisory Role from Rovi. Speakers’ Bureau from Rovi and Leo Pharma. Travel, Accommodations and Expenses from Rovi and Leo Pharma. Relevant financial activities outside the submitted work: Travel, Accommodations and Expenses from Roche and Sanofi. RMM has nothing to disclose. VM has nothing to disclose. VPO reports speaker honoraria from Rovi, Leo Pharma and Sanofi; Advisory from daiichi sankyo. PPS reports speaker honoraria from Leo pharma. TQV reports Consulting or Advisory Role from Novartis and Glaxo-Tesaro; Speakers’ Bureau from Rovi, Sanofi, Roche and Pfizer; Travel, Accommodations and Expenses from Roche. MS reports Consultant or Advisory Role from Rovi; Speakers’ Bureau from Rovi and Leo Pharma. Travel, Accommodations and Expenses from Rovi and Leo Pharma. Relevant financial activities outside the submitted work: Consultant or Advisory Role from Merck, Amgen and Celgene; Speakers’ Bureau from Celgene, Servier and Amgen. Travel, Accommodations and Expenses from Roche and Servier. Grant support (personal/institutional) from Mylan and Kern Pharma.
Ethical approval
The current study has been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments.
Informed consent
Inform consent is not needed.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Muñoz Martín, A.J., Gallardo Díaz, E., García Escobar, I. et al. SEOM clinical guideline of venous thromboembolism (VTE) and cancer (2019). Clin Transl Oncol 22, 171–186 (2020). https://doi.org/10.1007/s12094-019-02263-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-019-02263-z