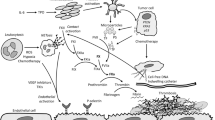
Opinion statement
Cancer patients are at high risk for venous thromboembolism (VTE) which is considered the second leading cause of death among this population. Both cancer and cancer treatment increase this risk. Since the risk of VTE is not the same in all cancer patients, it is important to understand what factors increase the risk of incident and the risk of recurrent VTE in this patient population. In an effort to combine multiple factors into a single risk stratification system, a scoring system for recurrent VTE risk in cancer patients has been developed and externally validated. While vitamin K antagonists (VKA) or the direct oral anticoagulants (DOACs) are first-line therapies for non-cancer-associated VTE treatment, low-molecular-weight heparin (LMWH) agents are the first-line anticoagulant for treatment of cancer-associated VTE. In this review, we discuss the epidemiology, pathophysiology, and risk stratification used in cancer-associated VTE. We also discuss the current therapies for cancer-associated VTE and the evidence supporting their use from the literature.
Similar content being viewed by others
References and Recommended Reading
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Deitcher SR. Cancer-related deep venous thrombosis: clinical importance, treatment challenges, and management strategies. Semin Thromb Hemost. 2003;29(3):247–58. doi:10.1055/s-2003-40963.
Blom JW, Doggen CM, Osanto S, Rosendaal FR. Malignancies, prothrombotic mutations, and the risk of venous thrombosis. JAMA. 2005;293(6):715–22. doi:10.1001/jama.293.6.715.
Lee AYY, Levine MN. Venous thromboembolism and cancer: risks and outcomes. Circulation. 2003;107(23 suppl 1):I-17-I-21. doi:10.1161/01.cir.0000078466.72504.ac.
Lip GYH, Chin BSP, Blann AD. Cancer and the prothrombotic state. Lancet Oncol. 2002;3(1):27–34. doi:10.1016/S1470-2045(01)00619-2.
Greene MT, Flanders SA, Woller SC, Bernstein SJ, Chopra V. The association between PICC use and venous thromboembolism in upper and lower extremities. Am J Med. 2015;128(9):986–993.e981. doi:10.1016/j.amjmed.2015.03.028.
Khorana AA, Francis CW, Culakova E, Kuderer NM, Lyman GH. Frequency, risk factors, and trends for venous thromboembolism among hospitalized cancer patients. Cancer. 2007;110(10):2339–46. doi:10.1002/cncr.23062.
Prandoni P, Lensing AWA, Piccioli A, Bernardi E, Simioni P, Girolami B, et al. Recurrent venous thromboembolism and bleeding complications during anticoagulant treatment in patients with cancer and venous thrombosis. Blood. 2002;100(10):3484–8. doi:10.1182/blood-2002-01-0108.
Monreal M, FalgÁ C, ValdÉS M, SuÁRez C, Gabriel F, Tolosa C, et al. Fatal pulmonary embolism and fatal bleeding in cancer patients with venous thromboembolism: findings from the RIETE registry. J Thromb Haemost. 2006;4(9):1950–6. doi:10.1111/j.1538-7836.2006.02082.x.
Trujillo-Santos J, Nieto JA, Tiberio G, Piccioli A, Micco PD, Prandoni P, et al. Predicting recurrences or major bleeding in cancer patients with venous thromboembolism—findings from the RIETE Registry. Thromb Haemost. 2008;100(9):435–9. doi:10.1160/th08-02-0125.
Louzada ML, Carrier M, Lazo-Langner A, Dao V, Kovacs MJ, Ramsay TO, et al. Development of a clinical prediction rule for risk stratification of recurrent venous thromboembolism in patients with cancer-associated venous thromboembolism. Circulation. 2012;126(4):448–54. doi:10.1161/circulationaha.111.051920. This study developed and validates a risk scoring system for recurrent VTE in cancer patients.
Douketis J, Tosetto A, Marcucci M, Baglin T, Cushman M, Eichinger S, et al. Patient-level meta-analysis: effect of measurement timing, threshold, and patient age on ability of d-Dimer testing to assess recurrence risk after unprovoked venous thromboembolism. Ann Intern Med. 2010;153(8):523–31. doi:10.7326/0003-4819-153-8-201010190-00009.
Janakiram M, Sullivan M, Shcherba M, Guo S, Billett HH. A systematic review of the utility of residual vein obstruction studies in primary and secondary venous thrombosis. Thrombosis. 2013;2013:9. doi:10.1155/2013/247913.
Linkins L-A, Stretton R, Probyn L, Kearon C. Interobserver agreement on ultrasound measurements of residual vein diameter, thrombus echogenicity and Doppler venous flow in patients with previous venous thrombosis. Thromb Res. 2006;117(3):241–7.
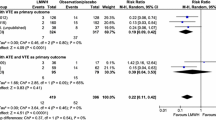
Lee AYY, Levine MN, Baker RI, Bowden C, Kakkar AK, Prins M, et al. Low-molecular-weight heparin versus a coumarin for the prevention of recurrent venous thromboembolism in patients with cancer. N Engl J Med. 2003;349(2):146–53. doi:10.1056/NEJMoa025313. This randomized trial demonstrates that LMWH is superior to warfarin for the prevention of recurrent VTE in cancer patients. The trial also demonstrates no increased risk of bleeding for the LMWH-treated group as compared to the warfarin-treated group.
Hull RD, Pineo GF, Brant RF, Mah AF, Burke N, Dear R, et al. Long-term low-molecular-weight heparin versus usual care in proximal-vein thrombosis patients with cancer. Am J Medicine. 2006;119(12):1062–72. doi:10.1016/j.amjmed.2006.02.022.
Lee AY, Kamphuisen PW, Meyer G, et al. Tinzaparin vs warfarin for treatment of acute venous thromboembolism in patients with active cancer: a randomized clinical trial. JAMA. 2015;314(7):677–86. doi:10.1001/jama.2015.9243. This randomized trial demonstrates that LMWH is at least as good as warfarin for the prevention of VTE-recurrence in cancer patients. As compared to the earlier CLOT trial, this trial included more diverse enrollment sites and found a lower rate of VTE recurrence in both treatment groups.
Meyer G, Marjanovic Z, Valcke J, et al. Comparison of low-molecular-weight heparin and warfarin for the secondary prevention of venous thromboembolism in patients with cancer: a randomized controlled study. Arch Intern Med. 2002;162(15):1729–35. doi:10.1001/archinte.162.15.1729.
Deitcher SR, Kessler CM, Merli G, Rigas JR, Lyons RM, Fareed J. Secondary prevention of venous thromboembolic events in patients with active cancer: enoxaparin alone versus initial enoxaparin followed by warfarin for a 180-day period. Clin Appl Thromb Hemost. 2006;12(4):389–96. doi:10.1177/1076029606293692.
Lee A, Bauersachs R, Janas M, Jarner M, Kamphuisen P, Meyer G, et al. CATCH: a randomised clinical trial comparing long-term tinzaparin versus warfarin for treatment of acute venous thromboembolism in cancer patients. BMC Cancer. 2013;13(1):284.
Carrier M, Cameron C, Delluc A, Castellucci L, Khorana AA, Lee AYY. Efficacy and safety of anticoagulant therapy for the treatment of acute cancer-associated thrombosis: a systematic review and meta-analysis. Thromb Res. 2014;134(6):1214–9. doi:10.1016/j.thromres.2014.09.039. This systematic review demonstrates the superiority of LMWH over warfarin for prevention of VTE recurrence in cancer patients. It also notes that DOAC therapy cannot be recommended over LMWH until head-to-head studies are performed.
Posch F, Königsbrügge O, Zielinski C, Pabinger I, Ay C. Treatment of venous thromboembolism in patients with cancer: a network meta-analysis comparing efficacy and safety of anticoagulants. Thromb Res. 2015;136(3):582–9. doi:10.1016/j.thromres.2015.07.011. This network meta-analysis suggests that LMWH and DOACs may have similar efficacy and safety for the treatment of cancer-associated VTE. However, only indirect comparisons were made in this analysis, limiting the confidence of its conclusions.
Lee AYY, Peterson EA. Treatment of cancer-associated thrombosis. Blood. 2013;122(14):2310–7. doi:10.1182/blood-2013-04-460162.
Hutten BA, Prins MH, Gent M, Ginsberg J, Tijssen JGP, Büller HR. Incidence of recurrent thromboembolic and bleeding complications among patients with Venous thromboembolism in relation to both malignancy and achieved international normalized ratio: a retrospective analysis. J Clin Oncol. 2000;18(17):3078–83.
Bauersachs R, Berkowitz SD, Brenner B, Buller HR, Decousus H, Gallus AS, et al. Oral rivaroxaban for symptomatic venous thromboembolism. N Engl J Med. 2010;363(26):2499–510. doi:10.1056/NEJMoa1007903.
Buller HR, Prins MH, Lensin AW, Decousus H, Jacobson BF, Minar E, et al. Oral rivaroxaban for the treatment of symptomatic pulmonary embolism. N Engl J Med. 2012;366(14):1287–97. doi:10.1056/NEJMoa1113572.
Buller HR, Decousus H, Grosso MA, Mercuri M, Middeldorp S, Prins MH, et al. Edoxaban versus warfarin for the treatment of symptomatic venous thromboembolism. N Engl J Med. 2013;369(15):1406–15. doi:10.1056/NEJMoa1306638.
Agnelli G, Buller HR, Cohen A, Curto M, Gallus AS, Johnson M, et al. Oral apixaban for the treatment of acute venous thromboembolism. N Engl J Med. 2013;369(9):799–808. doi:10.1056/NEJMoa1302507.
Schulman S, Kearon C, Kakkar AK, Mismetti P, Schellong S, Eriksson H, et al. Dabigatran versus warfarin in the treatment of acute venous thromboembolism. N Engl J Med. 2009;361(24):2342–52. doi:10.1056/NEJMoa0906598.
Vedovati MC, Germini F, Agnelli G, Becattini C. Direct oral anticoagulants in patients with VTE and cancer: a systematic review and meta-analysis. Chest. 2015;147(2):475–83. doi:10.1378/chest.14-0402. This meta-analysis of DOAC vs warfarin trials suggests that use of DOACs for VTE treatment in patients with cancer may be safe and effective. However, the cancer patients included in these trials may not be the same as the cancer-associated VTE patients from the CLOT and CATCH trials.
Wu C, Lee AYY. Novel or non-vitamin K antagonist oral anticoagulants and the treatment of cancer-associated thrombosis. Semin Thromb Hemost. 2015;41(2):237–43. doi:10.1055/s-0035-1544160.
Mandala M, Falanga A, Roila F, Group EGW. Management of venous thromboembolism (VTE) in cancer patients: ESMO Clinical Practice Guidelines. Ann Oncol. 2011;22 suppl 6:vi85–92.
Guyatt GH, Akl EA, Crowther M, Gutterman DD, Schuunemann HJ. Executive summary: antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest. 2012;141(2 Suppl):7S–47S. doi:10.1378/chest.1412S3. The American College of Chest Physicians guidelines on management of cancer-associated VTE. They favor the use of LMWH over wafarin without any recommendations on the use of DOACs in this clinical scenario.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Hazem Elewa and Riham Elrefai each declare no potential conflicts of interest.
Geoffrey D. Barnes reports grants from BMS/Pfizer, personal fees from Portola, during the conduct of the study, and grants from Blue Cross Blue Shield of Michigan.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical Collection on Vascular Disease
Rights and permissions
About this article
Cite this article
Elewa, H., Elrefai, R. & Barnes, G.D. Cancer-Associated Venous Thromboembolism. Curr Treat Options Cardio Med 18, 23 (2016). https://doi.org/10.1007/s11936-016-0445-y
Published:
DOI: https://doi.org/10.1007/s11936-016-0445-y