Abstract
Analyses of transient dynamics are critical to understanding infectious disease transmission and persistence. Identifying and predicting transients across scales, from within-host to community-level patterns, plays an important role in combating ongoing epidemics and mitigating the risk of future outbreaks. Moreover, greater emphases on non-asymptotic processes will enable timely evaluations of wildlife and human diseases and lead to improved surveillance efforts, preventive responses, and intervention strategies. Here, we explore the contributions of transient analyses in recent models spanning the fields of epidemiology, movement ecology, and parasitology. In addition to their roles in predicting epidemic patterns and endemic outbreaks, we explore transients in the contexts of pathogen transmission, resistance, and avoidance at various scales of the ecological hierarchy. Examples illustrate how (i) transient movement dynamics at the individual host level can modify opportunities for transmission events over time; (ii) within-host energetic processes often lead to transient dynamics in immunity, pathogen load, and transmission potential; (iii) transient connectivity between discrete populations in response to environmental factors and outbreak dynamics can affect disease spread across spatial networks; and (iv) increasing species richness in a community can provide transient protection to individuals against infection. Ultimately, we suggest that transient analyses offer deeper insights and raise new, interdisciplinary questions for disease research, consequently broadening the applications of dynamical models for outbreak preparedness and management.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Since the pioneering work of Kermack and McKendrick (1927), a large proportion of epidemiological modeling has been based on analyses of deterministic, nonlinear dynamical systems (Guckenheimer and Holmes 1983; Strogatz 2018). The primary focus of such studies has been to identify the stability of attractors, the special solutions to which all trajectories eventually converge, e.g., equilibria, limit cycles, or chaotic attractors (Schwartz and Smith 1983; Diekmann and Heesterbeek 2000; Brauer et al. 2019). However, in the broader context of ecological systems, there has been a growing recognition that asymptotic dynamics of models give only a partial and potentially misleading impression of what we should expect to observe in the real world (Hastings et al. 2018). The dynamical characteristics of transients, trajectories that have not reached an attractor, may tell us more about real systems than can be gleaned from attractors. If transients play an important dynamical role in ecological systems, then it stands to reason that they deserve attention when assessing epidemics across human, wildlife, and agricultural systems and designing management strategies to combat them.
As an example, the general concept of transient disease dynamics has recently entered public consciousness in the ongoing COVID-19 pandemic. As of May 2021, more than three million deaths have been reported worldwide, accompanied by major disruptions to the global economy (McKibbin and Fernando 2020), transportation networks (e.g., travel bans, control of mass population movement; Chen et al. 2020), and social norms (e.g., physical distancing, household quarantine, closures of schools and university campuses; Wilder-Smith and Freedman 2020). In response, there have been numerous media headlines early on urging the public to help “flatten the curve”, a message promoting the idea that preventive actions can help reduce transmission and avoid overloading healthcare systems. In a world that continues to grapple with this crisis, transient analyses (e.g., Arthur et al. 2020; Ferguson et al. 2020) have assisted in forward projections of outbreak severity, estimations of resurgence risk, and policy recommendations ahead of worst-case scenarios.
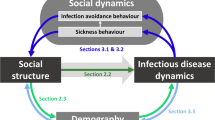
As COVID-19 information accumulates, so too has evidence of transient dynamics related to its pathogenesis, transmission window, and management efforts. Symptoms of some infected individuals under clinical care have been shown to subside temporarily before the sudden onset of severe illness (Zhou et al. 2020). SARS-CoV-2 emerged in the human population following a rare cross-species transmission event from a wildlife reservoir (Andersen et al. 2020; Wu et al. 2020). Cases were initially localized to the region where the virus spilled over into humans, individual movement and metapopulation connectivity drove subsequent transmissions and spatial spread across the globe (Wu and McGoogan 2020). As the outbreak spread, connections into and out of affected populations were transiently strengthened, suppressed, or redirected as a result of mass exodus, government-imposed lockdown, and self-quarantines (Jia et al. 2020). These short-lived patterns at the within-host, individual host, and host metapopulation levels show that, to fully understand the disease and control the pandemic effectively, it is not sufficient to only predict the system’s transient dynamics at the population level, e.g., measuring disease incidence. Instead, we must also account for transient structural changes that are reflected in lower- and higher-level processes (Fig. 1).
Examples of transient disease dynamics organized along an ecological hierarchy from within-host processes to host communities. Section III: Within an infected host, illness-induced anorexia can strongly influence infection dynamics prior to host recovery. Section II: Between hosts, transitional space use patterns before a home range or territory is stabilized can provide rare opportunities for infective contacts. Section I: At the host population level, the critical epidemiological questions address the spread of a transmissible pathogen through the population over time. Section IV: Within a host metapopulation, landscape disturbances (e.g., flooding, forest clearcutting, road construction, wildfires) may lead to transient windows of connectivity between habitat patches that reappear at various frequencies (solid lines: low; dashed lines: intermediate; dotted lines: high). Section V: At the host community level, interspecific variation in reservoir competence—shown here using examples for Lyme disease (see Levi et al. 2016)—can reduce the risk of infection to a particular host species, a phenomenon referred to as the dilution effect, or increase it as per the amplification effect. Roman numerals next to the ecological scales indicate corresponding sections in the text. Illustration credit: Life Science Studios (https://lifesciencestudios.com)
In part one of this paper, we examine the prevalence and properties of transients in observed time-series of infectious disease incidence and the analytical methods used to detect them. In subsequent parts, we draw from multiple disciplines transecting disease ecology, underlining timely case studies and theories that demonstrate different forms of structural transients.
We use case studies of both human and animal diseases to specifically show that at the individual host level, transient movement dynamics can modify opportunities for transmission events over time. We also show that within-host energetic processes can lead to transient dynamics in individual immunity, pathogen load, and transmission potential. In metapopulations, transient connectivity between discrete populations in response to environmental factors and outbreak dynamics can determine outbreak trajectory and pathogen spread across spatial networks. Finally, we show that increasing species richness in a community can provide transient protection to individuals against infection. Our survey of transient disease dynamics across ecological scales reinforces the idea that hierarchical factors affecting pathogen exposure and susceptibility to infection must be aligned in space and time to convert transmission risk to reality (Plowright et al. 2017).
I. Transient behaviors in population models
The term “population” can be broadly applied to any collection of conspecifics that are not explicitly connected through a spatial network (for that description, see metapopulation level transients in Section IV). The transient nature of outbreak dynamics in a population can be observed across a variety of temporal scales determined by the duration of pathogen persistence.
Common scales: regional to national, single epidemic duration
When a pathogen emerges or re-emerges, such as COVID-19 or Ebola, respectively, asymptotic dynamics are not the primary concern for disease management efforts. Instead, attention is focused on characterizing the initial pattern of spread, forecasting cases and deaths in the absence of interventions, and investigating (usually through simulations) whether and how the outbreak can be contained (Ferguson et al. 2016). Typically, there is a flurry of activity to estimate the basic reproduction number (Anderson et al. 1992; van den Driessche and Watmough 2002; Wallinga and Teunis 2004; Pourbohloul et al. 2009; Park et al. 2020), initial growth rate (Wallinga and Lipsitch 2007; Ma et al 2014; Champredon and Earn 2016; Chowell 2017; Earn et al. 2020), expected final epidemic size (Kermack and McKendrick 1927; Ma and Earn 2006), etc., and how the estimated values of these quantities should influence control strategies (Bauch et al. 2003; Earn et al. 2012; Molina and Earn 2015). In most models, the dynamics of interest are transient. Thus, in the context of forecasting and controlling outbreaks, whether for SARS (Lipsitch et al. 2003), pandemic influenza (Yang et al. 2009), Ebola (Bellan et al. 2014), Zika (Gao et al. 2016), COVID-19 (Anderson et al. 2020), or other diseases, there has always been an appreciation for, and emphasis on, transient dynamics.
The transients that span only a single epidemic are short-lived relative to any kind of asymptotic behavior. The timing, intensity, and targets of response during the transient phase can have drastic consequences for intervention success (e.g., Ferguson et al. 2001; Haydon et al. 2006; Tao et al. 2021). Recently, there has been an increased interest in dynamic control strategies that can adapt to the transient states of the epidemic (e.g., Handel et al. 2007; Probert et al. 2019) and resolve the epistemic uncertainties about transmission and treatment via real-time surveillance (Shea et al. 2014; Bradbury et al. 2017). These temporally nuanced approaches have typically led to significant improvement in management outcomes. As public health faces a growing threat from emerging infectious diseases (Jones et al. 2008; Heymann et al. 2015), new theories of dynamic control strategies that can provide timely, context-dependent solutions (e.g., when to reprioritize control objectives) may be better positioned to facilitate discourses between modelers, stakeholders, and policymakers.
In the age of social media, fears regarding the safety of recommended practices (e.g., vaccination, use of face masks) are prone to amplify general resistance to control strategies intended to lower susceptible recruitment and transmission opportunities. Recent game-theoretical models (e.g., Bauch and Earn 2004; Bauch and Bhattacharyya 2012) have explored individual vaccination behavior during bouts of vaccine scares. Additional theories that anticipate transients in the dynamics of both disease incidence and public trust could help manage policy expectations according to social currents and identify effective alternative strategies.
Common scales: regional, decadal
In contrast to novel epidemics, for endemic diseases, the system’s dynamical behavior away from the attractor is generally maintained much longer. Transients could play a dominant role since temporal forcing and perturbations might keep orbits away from an attractor (Schwartz and Smith 1983; Keeling et al. 2001; Bharti et al. 2011; Becket et al. 2019). These types of dynamics are notably associated with oscillatory incidence time-series found in endemic childhood diseases (Fig. 2), in particular, diseases such as measles, rubella, and whooping cough that are strongly affected by contact differences between school holidays and school terms (London and Yorke 1973; Earn et al. 2000; Keeling et al. 2001; Bauch and Earn 2003). In early studies, identifying the periods of the system attractors alone correctly predicted the annual or biennial spectral peaks in the incidence time-series (Earn et al. 2000). However, the same approach does not capture the “non-resonant peaks” that occur at arbitrary frequencies and are sometimes found to be the most significant for the long-term dynamics for these diseases. Bauch and Earn (2003) identified noise-sustained transients to be the source of the non-resonant peaks. Their method of linear perturbation analysis involves converting a continuous-time model into a Poincaré map and then computing the eigenvalues of the linearization about its attractors. The results show that the greater presence of demographic noise associated with a smaller population can slow or prevent convergence to the attractor. Reduction of susceptible recruitment level over time (i.e., fewer births or more vaccination) can also promote transient effects for (at least) two reasons: lower effective reproduction number yields (i) more prominent non-resonant peaks associated with non-convergence to attractors (e.g., Fig. 2c of Bauch and Earn 2003) and (ii) more likely stochastic switching between basins of co-existing attractors (Earn et al. 2000; Hempel and Earn 2015).
Comparison of predicted vs. observed measles dynamics in New York City (NYC), 1891–1984. Reprinted with permission from Hempel and Earn (2015). Top panel: Square root of measles case reports, normalized by total concurrent population. Middle panel: Approximate volumes of basins of attraction near the observed NYC measles incidence for each year from 1891 to 1984. For each year, we display the proportion of 10,000 simulations that reach each period. See Hempel and Earn (2015) for details. Bottom panel: Color depth plot of a continuous wavelet transform of the square root of normalized observed NYC measles cases (color warmth scales with spectral power and 95% significance contours are shown in black). The predicted attractor and transient periods are overlaid (only the periods of annual and biennial attractors are short enough to appear on this graph and to be observable). Unfilled black circles identify predicted attractor periods that are consistent with the observed data. White vertical bars show the possible ranges of the transient period associated with these attractors; the filled white dot on each bar marks the median transient period. There is a good qualitative agreement between the predicted transient periods and the observed power near periods 2 and 3 during 1891–1909, 1917–1945, and 1964–1973
Although non-resonant spectral peaks in incidence time-series have been identified largely in the context of human diseases, similar dynamical patterns can also be expected to unfold in temporally forced zoonotic diseases. For instance, widespread outbreaks of Rift Valley fever, an endemic, arthropod-borne disease that affects both human and ruminant populations, typically occur in cycles varying between 7 and 11 years (Manore and Beechler 2015). How the pathogen is able to persist during the inter-epidemic periods remains unclear. Recent model explanations have been limited to vertical transmission, seasonality of vector abundance, and environmental stochasticity (e.g., Cavalerie et al. 2015; Manore and Beechler 2015). Extending perturbation analyses to such systems might offer a new, mechanistically simpler explanation: much like the rubella dynamics in the pre-vaccine era (cf. Bauch and Earn 2003, Figs. 1c,g and 3), these infrequent large outbreaks, with frequent small outbreaks or no detectable outbreaks in between, could be no more than a manifestation of intrinsically driven non-resonant spectral peaks. This line of investigation might therefore help to better inform the surveillance programs for zoonotic pathogens that have potential for long-term, silent circulation.
Contrasting model descriptions of a traveling host (or host group behaving as a unit) coming into contact with vector clusters in a constant environment. A ruminant herd is depicted to encounter mosquitoes when moving near watering holes, a key transmission pathway of Rift Valley fever (Manore and Beechler 2015). To account for transient space use dynamics (a), the movement of the host was modeled as a utilization distribution (UD) converging toward an equilibrium (yellow circles of increasing opaqueness) centered around a spatial point attractor (triangle). Here, time evolution of the UD is determined by a Fokker–Planck equation with constant coefficients in the advection and diffusion terms. Infective contact occurs at a rate proportional to the instantaneous overlap between the UD and the vector distribution (blue circles). See Fieberg and Kochanny (2005) for candidate indices of overlap, e.g., integral of the joint distribution. The resultant time-series of contact rate is characterized by irregular peaks. In b, the transient UDs are aggregated to produce a time-independent probability density surface of the host’s location, yielding a constant contact rate. The model in c incorporates only the asymptotic UD. This reduces the contact process to a constant, which could drastically underestimate the animal’s risk of infection over the course of its resettlement
Transient structural dynamics in disease systems at sub-population levels
Despite the extensive use of population models, there remain epidemiological challenges where the needs to reduce uncertainties are better addressed by anticipating the potential experience of individuals at various stages of infection than by predicting the population dynamics. For instance, targeted conservation may require close surveillance of individual host movement near a small number of colonies. Clinical outcomes could also be improved by personalizing treatment based on the infection dynamics within each patient. In models suited for these purposes, the spatial scale of transient disease dynamics ranges from the host body size to the parts of an environment in which a host (or a group of hosts behaving as a single unit) may traverse; the relevant time scales tend to be short compared to those in population models.
II. Effects of transient host movement dynamics
Common scales: individuals to collective groups, short-term encounters to long-term range shifts
Within a population, the risk of pathogen transmission to a recipient host is largely driven by host movement (Fig. 1, Section II). Direct transmission occurs through contact between a susceptible and an infected host while indirect transmission can occur when an infected and susceptible host spatially overlap with some temporal lag (Dougherty et al. 2018). Advanced knowledge of host movement contributes to our understanding and management of emerging zoonotic diseases. Numerous studies have highlighted the downstream effects of myriad movement behaviors including bat foraging activities during periods of agricultural and urban intensification (e.g., Nipah virus: Pulliam et al. 2012, Hendra virus: Plowright et al. 2011), long-distance dispersals of pre-breeding ruminants (e.g., bovine tuberculosis, Rift Valley fever: Caron et al. 2016), and worker resettlements and air travel (e.g., Ebola: Alexander et al. 2015, Zika virus: Ali et al. 2017). Despite the importance of host movement as a predictor of outbreaks, many compartmental models assume populations are well-mixed and the per-capita contact rate is time-invariant in the absence of temporal forcing. These simplifying conditions imply homogeneity in population contact structures and that each host has a stationary range distribution unless changes happen in the environment (e.g., seasonal transitions). By not recognizing contact rate as a host-specific variable that is dependent on both space and time, important transmission opportunities could be missed.
Mechanistic space use models (see a historical overview in Potts and Lewis 2014) have laid the foundation for predicting movement dynamics of individual hosts. The analyses predominantly focus on the utilization distribution (UD), a probability density surface that describes the range and uncertainties of an individual’s possible locations at any point in time. UDs can be solved numerically when they are modeled using partial differential equations or estimated from outputs of agent-based simulations (see Potts and Lewis 2014 and references therein). A host’s UD may equilibrate slowly, if at all (Fryxell et al. 2008; Potts and Lewis 2016). It may also be sensitive to local conditions, such as terrain (Moorcroft et al. 2006), resource availability (Moorcroft et al. 2006; Bateman et al. 2015), and the presence of conspecifics or predators (Lewis and Moorcroft 2001; Bateman et al. 2015; Tao et al. 2016; Potts et al. 2018). The transitional UDs may be highly variable and create short-term opportunities for host-pathogen contacts that impact population, metapopulation, and community level transmissions (Plowright et al. 2011; Ramsey et al. 2014; White et al. 2020). Conversely, an individual’s exposure to pathogens could temporarily change where it may go (Zidon et al. 2017). Thus, explicit inclusion of transient movement dynamics in disease models could have far-reaching consequences for the implementation of biosurveillance systems.
In basic compartmental models, transmission is commonly treated as a single process: a product of infective contact rate, averaged over both time and individuals, and the probability of transmission during contact. We propose separating these two components and describing the encounter process as a function \({f}_{c}\) of the transient space use pattern at the host level, such that, without vital dynamics,
where \({u}_{i}(\mathbf{x},t)\) and \({v}_{j}(\mathbf{x},t)\) denote the UD of each susceptible host and the distributional pattern of each pathogen source, respectively. \(S\) is the number of susceptible hosts. \(\widehat{I}\) may refer to the number of (a) infected hosts, (b) vector-infested sites, or (c) patches containing pathogen. Upon contact, transmission happens with probability \(\kappa\). Epidemic predictions that account for transient space use dynamics can differ considerably from those based on asymptotic, mean field approaches (Tao et al. 2018). We conjecture that, by tracking, at minimum, \({u}_{i}\left(\mathbf{x},t\right)\) and the consequent rate of contact over time, this framework will help bring into focus the period and spatial region of host movement most critical for disease transmission (Fig. 3).
Mass vaccination campaigns in regions with underperforming health services often require mobilization (e.g., house-to-house visits) of healthcare workers for vaccine delivery (polio: Curry et al. 2014; measles: Mbabazi et al. 2015), reaching inhabitants in a limited geographical region at a time. In such scenarios, the deployment of response personnel into a target population can also be modeled as a UD evolving over time. Evaluating the impact of this strategy on management success allows us to address operational questions, namely, how fast or from where response effort should be distributed, thus extending control recommendations beyond the equilibrium criterion (e.g., threshold herd immunity). Tao et al. (2018) did so by combining a diffusion model of probabilistic vaccine delivery with an agent-based simulation of disease transmission. This produces site-specific, time-dependent rates of vaccine uptake. The race between local vaccinations and disease spread determines the optimal deployment strategy to minimize overall outbreak severity. We expect to see further development of models centered on transient phases of human movement that can be used to optimize resource allocation and minimize delays during an emergency response.
III. Effects of transient within-host dynamics
Common scales: pathogen population inside a host, time to recovery or death
Host level movement processes that drive pathogen transmission are often tightly coupled to environments within hosts: a host’s immunity against invading pathogens weaken following energetically costly migration (Owen and Moore 2008); individuals carrying high pathogen loads may also become sedentary and shed pathogens in concentrated areas that are frequently visited by conspecifics (Patterson and Ruckstuhl 2013). Modeling infection dynamics of individual hosts could therefore help us understand the epidemiological costs of certain host behaviors (Lunn et al. 2019) and identify short-term ecological contexts that might promote the existence of superspreaders (Hawley and Altizer 2011).
Infection dynamics arise from within-host interactions between host immunity and pathogens, which behave, respectively, as predators and prey that also compete over limited resources (Smith and Holt 1996; Cressler et al. 2014; Greenspoon et al. 2018). The onset and duration of specific immune phases in an infected host, and the associated shifts in its metabolic processes, can regulate infection dynamics, affecting the time until pathogen clearance, i.e., host recovery from infection. Dynamic models have been recently developed to track energetic flows within hosts. The included mechanisms may describe energy (or resources) being assimilated after feeding, stolen by pathogens, and allocated toward host immunity, maintenance, reproduction, and growth (see Hall et al. 2009; Hite and Cressler 2018; Civitello et al. 2018; Van Leeuwen et al. 2019). The model framework has helped explain divergences in disease chronicity, i.e., why some hosts clear a pathogen rapidly (acute infection) while others develop persistent, sometimes life-long, infection (chronic infection). For instance, Van Leeuwen et al. (2019) demonstrate how a high pathogen dose can strongly modulate a host’s metabolism and lead to long-term pathogen persistence, as opposed to a low dose that could be quickly cleared. Transient analysis may thus resolve well-documented uncertainties in host pathologies (see Wilber et al. 2017) and project individual risks of onward transmission.
Although immune responses are often energetically expensive, many infected individuals have been found to voluntarily suppress their feeding behavior in order to “starve out” the pathogen (Fig. 1, Section III). Illness-mediated anorexia (reviewed in Hite et al. 2020), a temporary but substantial suppression of appetite during key phases of infection, can function as a first line of defense by limiting pathogen growth and replication (“antigrowth resistance”) or infection-induced pathology (“tolerance”). This caloric restriction begins after seconds to days of exposure and appears strategically tuned to specific stages of pathogen life cycles. To further complicate matters, empirical evidence indicates that anorexia can be manipulated, directly or indirectly, by pathogens to increase their rate of shedding or vector transmission (Adamo et al. 2007; Rogers and Bates 2007; Rao et al. 2017). There is currently only a small number of studies that model the interactions between infection dynamics and anorexia (see Hite and Cressler 2019; Hite et al. 2020). Nevertheless, subverting the loss of appetite has been a part of many standard treatments for humans and livestock (Fox et al. 2002; Schütz et al. 2014; Rodrigues et al. 2015; Hite et al. 2020). Future theoretical advances in this area could help evaluate their efficacies across different individuals and optimize treatment schedules to minimize infection duration.
Models of within-host dynamics have mostly been used to predict population-level consequences such as infection prevalence and evolution of virulence. Typically, the pathogen is assumed to reach carrying capacity immediately after successful host invasion and colonization, irrespective of local metabolic condition (Mideo et al. 2008). Resource availability outside the host is sometimes incorporated as a slowly changing external factor. This time-scale separation has aided mathematical tractability and provided general insights into pathogenesis, transmission fitness, and unwelcome repercussions of vector-reduction programs (Mideo et al. 2008; Handel and Rohani 2015; Civitello et al. 2018). However, an alternative framework could integrate multiple intrinsic time scales: i.e., slow environmental resource dynamics interacting with fast infection dynamics. Models that “internalize” the slowly changing variables might reveal long, relatively disease-free periods in individuals interrupted by infection during population-wide epidemics (see Hastings et al. 2018 on how slow-fast systems promote long-lived, quasi-stable transients).
Transient structural dynamics in disease systems above the host population level
In metapopulations and communities, inter-population and interspecific interactions drive transient disease dynamics (Fig. 1, Sections IV and V). At this upper echelon of the ecological hierarchy, transient behaviors typically operate over an area sufficiently large to accommodate substantial heterogeneity in contact structure, disease susceptibility, and pathogen transmissibility. Unlike population models, the time scale can be a fraction of an epidemic cycle (or a natural disturbance cycle); alternatively, it can persist for many host generations until significant species turnover has been achieved.
IV. Effects of transient network dynamics in host metapopulation
Common scales: multiple populations, phase of an outbreak
Since Hanski’s classic works on the ecological importance of metapopulation structure (e.g., Hanski 1998), the notion of connectivity between geographic areas (e.g., habitat patches) has been the cornerstone of network-related models, including the many theoretical frameworks currently available for predicting disease spread. In metapopulation models, transient dynamics related to local and global population densities have long been a subject of interest (Saravia et al. 2000; Ovaskainen and Hanski 2002; Labra et al. 2003); nevertheless, connectivity itself, i.e., the spatial distribution of population flow rates, is typically assumed to exist in an asymptotic state (but see Perry and Lee 2019; Karnatak and Wollrab 2020). Given their enormous implications for transmission dynamics (Hess 1996; Grenfell and Harwood 1997; Hanski 1998; Keeling et al. 2004), models that can explicitly account for the transient dynamics of connectivity can help quickly predict and prevent the spread of infectious diseases. Below, we outline some of the most common types of transient dynamics of connectivity pertinent to disease modeling. These are classified as network advection, suppression, and diffusion (Supplementary Table).
Network advection (Fig. 4a) denotes transient immigration to a patch. This can describe safety or health-seeking behavior in humans, such as when patients travel to areas with more stability or higher quality, more accessible health resources (Bharti et al. 2015). We observed this trend during the 2013 Ebola outbreak in West Africa (Bogoch et al. 2015; Fallah et al. 2018), which was facilitated by improvements in transportation infrastructure relative to previous sites of Ebola spillover (Chowell et al. 2004; Malvy et al. 2019). Advection may also describe species range shifts. Marsh fritillary butterflies may relocate en masse following land cover changes (e.g., forest clearcutting) that result in the conversion of an inhospitable matrix habitat into a transient movement corridor (see Wahlberg et al. 2002; Driscoll et al. 2013). In both scenarios, the short-term establishment of directional bias in connectivity, especially early in an outbreak, may allow pathogen dispersal into and colonization of new, susceptible host populations (Helble 2011; Alegana et al. 2012).
Examples of outbreak-induced transients in human network connectivity: a network advection: health-seeking behavior; b network suppression: disease avoidance behavior; c network diffusion: disease escape and avoidance. Top row: Metapopulation level incidence time-series with (black solid) and without (black dashed) the transient population flows pictured in the bottom schematic. Middle row: Metapopulation level connectivity time-series between an infected patch (red circle below) and its neighboring uninfected patches (blue circles). Red lines show network connectivity in the presence (solid) and absence (dashed) of the illustrated transient population flows out of the focal infected patch; blue lines show it in the presence (solid) and absence (dashed) of transient flows into the focal infected patch. Bottom row: Transient population flows between patches (circles). Patch size is proportional to local population size. Straight lines show unchanged connectivity dynamics; triangles show increased flow from point to wide end; bar lines show no immigration. Brackets above the schematic mark the time window during an outbreak when these transient dynamics might occur
Under network advection, the potential impact of an outbreak would be strongly dependent on travel time relative to the mean infectious period. Direct and rapid travel toward health care while infectious is essential for timely treatment and recovery of an individual; yet, it also enables onward transmission at the destination patch. Conversely, circuitous routing that delays travel time past the infectious stage may increase the chance of pathogen introduction at stopover sites but prevent introduction at the destination. Therefore, differences in transportation infrastructures and habitat matrix conditions (terrain, elevation, favorability, and other attributes affecting crossings) might have long-term, opposing effects on disease dynamics that should be explicitly considered in multi-patch models.
When new infections are concentrated in one or a few patches, severing connections between infected and uninfected or low-prevalence populations, i.e., network suppression (Fig. 4b), becomes critical for containment (Chinazzi et al. 2020; Wells et al. 2020). Suppression may arise intrinsically through disease avoidance behavior (Widmar et al. 2017), or be a result of top-down quarantine measures (SARS: Riley et al. 2003, COVID-19: Ainslie et al. 2020; Chinazzi et al. 2020). In wildlife systems, transient habitat fragmentation caused by extreme weather events can produce an analogous effect. For example, after a riverine floodplain has experienced unusually high precipitation, terrestrial species may be temporarily unable to move between habitat patches due to localized flooding (e.g., Angelone et al. 2011; Erös and Grant 2015).
There are significant geographic differences in compliance with prevention guidelines across socio-economic and urban-rural divides (see Painter and Qiu 2020; Wright et al. 2020), particularly if polarization in attitude towards public health measures grows. Future multi-patch epidemic models might need to investigate the correlation between local compliance and the duration and spatial scale of network suppression. On the other hand, to study the effects of suppression on epizootics, metapopulation models may consider incorporating sudden appearances of impermeable matrices and their restorations to suitable habitats over time (see the concept of transient connectivity window in Ziegler and Fagan 2014).
As incidence within a patch surges, resident individuals might attempt to escape toward disease-free patches until the outbreak is brought under control. Meanwhile, the immigration rate to infected patches should decline due to disease avoidance or restrictions on patch entries (Médecins Sans Frontières 2009; Bengtsson et al. 2015). Co-occurrence of these two transient responses, termed network diffusion (Fig. 4c), has been observed in management scenarios characterized by “leaky lockdowns” (Ebola: Dellicour et al. 2018; Mukandavire et al. 2011, cholera: Bengtsson et al. 2015). Diffusion can also stem from sporadic, patchy disturbances. Low-intensity wildfires, for instance, can induce local wildlife to search for suitable habitats beyond the fire boundary (Nimmo et al. 2019). This temporary change in connectivity, which could disseminate pathogens via infected individuals into neighboring populations within a short time interval, promotes the emergence of an outbreak cluster at the metapopulation level.
Network diffusion aptly describes the Syrian refugee crisis, where large cohorts with low levels of immunity against vaccine-preventable diseases (e.g., polio, measles, rubella) entered new environments, increasing the transmission risk in refugee camps as well as in the imperfectly immunized communities to which they were displaced (Eichner and Brockmann 2013). We postulate that synchronous pathogen invasions into multitudes of new susceptible pools, in responses to humanitarian crises or stochastic habitat losses, are widespread across human and wildlife systems. Models that can capture the effects of such events could be critical for predicting epidemiological repercussions of socio-political unrest and human-accelerated environmental changes.
V. Transient effects of host community dynamics
Common scales: ecosystem, multiple host generations
The idea that increased species richness reduces disease transmission, referred to as the “dilution effect” (Keesing et al. 2010), is popular yet controversial. This hypothesis posits that adding infection-incompetent host species, or species that are incapable of efficient onward pathogen transmission, to a community will increase the density of pathogen sinks and thus reduce the spread of a host-specific pathogen (Fig. 1, Section V). Therefore, in a diverse community composed of relatively abundant incompetent host species, a susceptible, competent host individual would have a lower risk of infection by benefiting from “safety in numbers” (Johnson et al. 2013).
Negative correlations between host diversity and infection rate have been broadly found in recent studies, leading to suggestions of its generality (Keesing et al. 2010; Civitello et al. 2015). However, large-scale studies (e.g., Dunn et al. 2010; Randolph and Dobson 2012; Lafferty and Wood 2013; Wood et al. 2017) tend to indicate an opposite trend, with evidence of biodiversity increasing disease transmission, presumably due to an amplification effect characterized by a disproportionate abundance of competent host species for a generalist pathogen (Keesing et al. 2010). These contradictory patterns may be explained by the dilution effect being a scale-dependent phenomenon that is observed either in small-scale experiments or under short durations (see Cohen et al. 2016; Halliday and Rohr 2019; Rohr et al. 2019). The latter explanation considers that added hosts, irrespective of their competence, might buffer the original hosts against infections early on by intercepting free-living stages of the pathogen (e.g., parasite eggs, cysts). However, if the added host is competent, the initially observed dilution effect will weaken over time and the original hosts could experience a net increase of infection risk after the pathogen spreads through the newcomers. In other words, amplification can transiently appear as dilution in the short term. The same principle may apply to human communities. For example, immigration of many vaccine-immunized hosts into a susceptible population may provide it with transient herd immunity via a temporary increase in immune diversity. However, the risk of infection will be amplified in the long run (due to high population density) if immunity fades or if new offspring are not sufficiently vaccinated.
We present a heuristic model to demonstrate how infection risk changes over time within a diversifying community. It assumes that a new host species is seeded randomly in a well-mixed system that previously contains a single competent host species. The pathogen’s questing, attacking, and consuming stages are combined using a separation of time scales, giving a saturating transmission function that is necessary for dilution to occur (after Lafferty et al. 2015). We further assume the transition from infection to pathogen production is fast relative to host generation time. In this system,
where \(I\) is the density of infected individuals of any host species and \({X}_{1}\) is the density of all individuals of target host species 1. Free-living pathogen stages are produced at rate \(b\) per infected host and removed at rate \(d\). Contact with any host (susceptible, infected, or non-competent) occurs at rate \(\beta\) and leads to the loss of the pathogen stage. A new infection is generated if the host is uninfected and competent. To maintain constant host density, infected hosts are replaced with susceptibles at rate \(\gamma\) upon death or recovery. When species 1 is the only host present, \({X}_{1}-I\) is the density of susceptible hosts, and this model would reduce to the familiar \(SI\) model if one assumes that the pathogen stage has such a high background morality rate that loss due to contact is trivial.
Adding a competent host species \({X}_{2}\) at the system equilibrium modifies the time derivative to
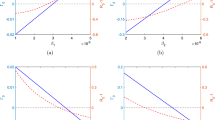
where \(K=d/\beta\) is the familiar Michaelis-Menton half-saturation constant from enzyme kinetics (Lafferty et al. 2015). A simple derivation shows that, initially, the per-capita infection rate for host species 1\(,\)
decreases with the addition of another competent host species, much in resemblance to the dilution effect (Fig. 5). However, as \(t\to \infty\), the rate
Per-capita infection rate of a target host species over time under three biodiversity scenarios. The addition of a non-competent host in a single-species community leads to a dilution effect (white space), characterized by the target host species being infected at a permanently reduced rate (cf. blue and black lines). In contrast, adding a competent host results in only a temporary rate reduction (dashed line), reflecting a transient dilution effect. As the system approaches its new attractor, the target host species becomes infected at a higher rate than it would without the increase in species richness (cf. red and black lines), in accordance with an amplification effect (gray space). Model parameters \({X}_{1}\), \({X}_{2}\), and \({X}_{3}=1\), \(K=1.2\), \(\gamma =0.1\), \(b=5\)
exceeds the single-species threshold. In contrast, adding a non-competent host species \({X}_{3}\) always reduces the per-capita infection rate for the target host,
even as the system heads to a new equilibrium (Fig. 5).
In summary, at the initiation of a study where a second host species is introduced, both diluting and amplifying hosts transiently appear to dilute transmission. Over time, however, their variable levels of competence lead to divergent epidemic potentials. In practice, an amplification effect could be very difficult to detect from field and experimental data collected over short time spans. We therefore caution that the support for a dilution effect by these studies should not be extrapolated to predict long-term system behavior, or be generalized. Moreover, the existence of transients in our model results suggests that disease-control strategies with immediate benefits may have future negative consequences that should be carefully considered.
Concluding remarks
Analyses of transients in dynamical systems are of vital importance to disease research. Although the scientific interest in transients was first motivated by rigorous mathematics, the results of many recent models carry implications for actionable measures that are far from theoretical abstractions. Predictions of transient dynamics, with a prevailing emphasis on an “ecologically relevant time scale” (Hastings 2004), increasingly demonstrate their wide-ranging utilities to problems of applied epidemiology. Here, using a sample of models across ecological scales, we highlight the importance of understanding transient dynamics at different stages of disease transmission and outbreak response. This approach can improve standard protocols of drug delivery, timeliness of outbreak surveillance and intervention, accuracy of epidemic forecast, and effectiveness of large-scale disease management policies.
We observed that transient patterns at a population level may erupt early and rapidly, e.g., during an outbreak, or may reoccur periodically as in the case of many endemic childhood diseases. Individually, spatial interactions between a host and its environment through movement determine infective contact opportunities and the window of transmission; transient immune and physiological responses could mean the difference between recovery and chronic illness. At the metapopulation level, transient increase or loss of connectivity between patches may significantly reshape an epidemic curve. Within a host community, observations of a dilution effect can mislead conclusions regarding a species’ long-term infection risk.
We discussed a wide range of disease models to show transient behaviors may be critical for a variety of reasons. The importance of studying transient behavior depends on how long the system remains away from the attractors and how information collected during those periods might be interpreted and applied in an epidemiological context. For instance, the transients discussed in population and within-host models (Sections I and III) share the common feature that the asymptotic state can be disease-free. The adverse effects on individuals or populations occur during the transient phase in such cases; an ability to predict transients is advantageous for clinical care and outbreak management no matter how short the transient phase. This stands in contrast to the transient dynamics that are slow to equilibrate, which may describe endemic incidence, animal space use patterns, as well as conspecific rates of infection in a diversified community (Sections I, II, and V). The dynamics likely persist over the time scales that are most relevant for addressing questions in epidemiology. In the special case where the transient regimes are quasi-stable for a long time (see Hastings et al. 2018; Morozov et al. 2020), their short-term dynamics could be mistaken for an asymptotic state, which would undermine long-term forecasting (e.g., mislead conclusions on the generality of the dilution effect) and scenario planning. We also featured systems in which opposing inferences are drawn from the transient and the asymptotic behaviors (e.g., dilution versus amplification effects; Section V), and systems that contain multiple attractors with divergent outcomes (e.g., host recovery versus chronic infection; Section III). Here, the need for understanding transient dynamics is tied to the importance of early interventions that aim to maintain a system in, or force it towards, a more desirable state. Finally, in models of individual host movement and host metapopulations (Sections II and IV), the transient behaviors, which can be fleeting, reflect changes in the probabilities of rare, brief, or localized events (e.g., spillover infections, emergency travel restrictions) that might have long-term, large-scale implications for disease spread. Transient analyses of these systems could therefore be critical to developing early detection methods that are able to forestall low-risk but high-impact transmissions.
Recognizing the impacts of transient disease dynamics can help modelers decide which processes should be included in a system. At sub-population levels (Sections II and III), scheduled pharmaceutical interventions, territorial conflicts, and other events that delay equilibration tend to occur on a time scale shorter than the transient’s lifetime in the absence of these perturbations; as a result, the predicted transient phase could be substantially shortened by excluding them. By contrast, if the transient behavior were to dampen quickly such that the system’s controlling parameters stay near-constant during the transient’s lifetime, incorporating external processes might still be important in long-term studies since it may identify repeated transitions between the different transient and asymptotic states. In population and metapopulation models where changes in management and environmental conditions are often infrequent (Sections I and IV), predicting the timing of their recurrences could help evaluate multi-generational trends and sudden shifts in large-scale dynamics, thereby providing a stronger test of the system’s epidemiological resilience. Finally, in the two systems driven by spatial behaviors (Sections II and IV), the transients (e.g., individual migrations away from pathogen hotspots, changes in population flow patterns) are direct responses to the disease itself. A model that couples movement and disease dynamics may then contribute to the development of control strategies that are robust to behavioral changes by anticipating how hosts might balance the trade-offs between infection risk and travel cost as an outbreak continues.
Transients do not always lend themselves easily to examinations by conventional mathematical methods. Nevertheless, ongoing advances in dynamical systems analysis, numerical simulations, and network analysis show that transient dynamics can be modeled explicitly and combined with asymptotic analysis to enhance responses to infections, contagion, and outbreaks. The recent proposal of applying adaptive management approaches to guide public health decision-making (Shea et al. 2014) further suggests that our description of a disease system and the recommended management actions are transient in themselves due to inherent model uncertainty, which can be resolved over time through continuous surveillance. Consequently, a greater acknowledgment of transients will not only influence our model conclusions, but will also provide an objective basis for real-time model improvement.
Availability of data and material
Epidemiological data used in the present manuscript are available at http://iidda.mcmaster.ca.
Code availability
Software and custom code used in the present manuscript are available upon request.
References
Adamo SA, Fidler TL, Forestell CA (2007) Illness-induced anorexia and its possible function in the caterpillar, Manduca sexta. Brain Behav Immun 21:292–300
Ainslie KEC, Walters C, Fu H et al (2020) Report 11: Evidence of initial success for China exiting COVID-19 social distancing policy after achieving containment. http://imperial.ac.uk/media/imperial-college/medicine/sph/ide/gida-fellowships/Imperial-College-COVID19-Exiting-Social-Distancing2020.pdf. Accessed 24 March 2020
Alegana VA, Wright JA, Pentrina U et al (2012) Spatial modelling of healthcare utilisation for treatment of fever in Namibia. Int J Health Geogr 11:1–13.
Alexander KA, Sanderson CE, Marathe M et al (2015) What factors might have led to the emergence of Ebola in West Africa? PLoS Negl Trop Dis 9:e0003652
Ali S, Gugliemini O, Harber S et al (2017) Environmental and social change drive the explosive emergence of Zika virus in the Americas. PLoS Negl Trop Dis 11:e0005135
Andersen KG, Rambaut A, Lipkin WI et al (2020) The proximal origin of SARS-CoV-2. Nat Med 26:450–452
Anderson RM, May RM (1992) Infectious diseases of humans: dynamics and control. Oxford University Press, Oxford
Anderson RM, Heesterbeek H, Klinkenberg D, Hollingsworth TD (2020) How will country-based mitigation measures influence the course of the COVID-19 epidemic? Lancet 395:931–934
Angelone S, Kienast F, Holderegger R (2011) Where movement happens: scale-dependent landscape effects on genetic differentiation in the European tree frog. Ecography 34:714–722
Arthur RF, Jones JH, Bonds MH et al (2021) Adaptive social contact rates induce complex dynamics during epidemics. PLoS Comput Biol 17:e1008639
Bateman AW, Lewis MA, Gall G et al (2015) Territoriality and home-range dynamics in meerkats, Suricata suricatta: a mechanistic modelling approach. J Anim Ecol 84:260–271
Bauch CT, Earn DJD (2003) Transients and attractors in epidemics. Proc Biol Sci 270:1573–1578
Bauch CT, Galvani AP, Earn DJD (2003) Group interest versus self-interest in smallpox vaccination policy. Proc Natl Acad Sci 100:10564–10567
Bauch CT, Earn DJD (2004) Vaccination and the theory of games. Proc Natl Acad Sci 101:13391–13394
Bauch CT, Bhattacharyya S (2012) Evolutionary game theory and social learning can determine how vaccine scares unfold. PLoS Comput Biol 8:e1002452
Bellan SE, Pulliam JRC, Dushoff J, Meyers LA (2014) Ebola control: effect of asymptomatic infection and acquired immunity. Lancet 384:1499–1500
Bengtsson L, Gaudart J, Lu X et al (2015) Using mobile phone data to predict the spatial spread of cholera. Sci Rep 5:8923
Bharti N, Lu X, Bengtsson L et al (2015) Remotely measuring populations during a crisis by overlaying two data sources. Int Health 7:90–98
Bharti N, Tatem AJ, Ferrari MJ et al (2011) Explaining seasonal fluctuations of measles in Niger using nighttime lights imagery. Science 334:1424–1427
Bogoch II, Creatore MI, Cetron MS et al (2015) Assessment of the potential for international dissemination of Ebola virus via commercial air travel during the 2014 west African outbreak. Lancet 385:29–35
Bradbury NV, Probert WJM, Shea K et al (2017) Quantifying the value of perfect information in emergency vaccination campaigns. PLoS Comput Biol 13:e1005318
Brauer F, Castillo-Chavez C, Feng Z (2019) Mathematical models in epidemiology. Springer, New York
Caron A, Cornelis D, Foggin C et al (2016) African buffalo movement and zoonotic disease risk across transfrontier conservation areas. Southern Africa Emerg Infect Dis 22:277
Cavalerie L, Charron MVP, Ezanno P et al (2015) A stochastic model to study Rift Valley fever persistence with different seasonal patterns of vector abundance: new insights on the endemicity in the tropical island of Mayotte. PLoS One 10:e0130838
Champredon D, Earn DJD (2016) Understanding apparently non-exponential outbreaks. Comment on “Mathematical models to characterize early epidemic growth: A review” by Gerardo Chowell et al. Phys Life Rev 18:105–108
Chen S, Yang J, Yang W et al (2020) COVID-19 control in China during mass population movements at New Year. Lancet 395:764–766
Chinazzi M, Davis JT, Ajelli M et al (2020). The effect of travel restrictions on the spread of the 2019 novel coronavirus (COVID-19) outbreak. Science 368:395–400
Chowell G (2017) Fitting dynamic models to epidemic outbreaks with quantified uncertainty: a primer for parameter uncertainty, identifiability, and forecasts. Infect Dis Model 2:379–398
Chowell G, Hengartner NW, Castillo-Chavez C et al (2004) The basic reproductive number of Ebola and the effects of public health measures: the cases of Congo and Uganda. J Theor Biol 229:119–126
Civitello DJ, Cohen J, Fatima H et al (2015) Biodiversity inhibits parasites: broad evidence for the dilution effect. Proc Natl Acad Sci 112:8667–8671
Civitello DJ, Fatima H, Johnson LR et al (2018) Bioenergetic theory predicts infection dynamics of human schistosomes in intermediate host snails across ecological gradients. Ecol Lett 21:692–701
Cohen JM, Civitello DJ, Brace AJ et al (2016) Spatial scale modulates the strength of ecological processes driving disease distributions. Proc Natl Acad Sci 113:E3359–E3364
Cressler CE, Nelson WA, Day T, McCauley E (2014) Disentangling the interaction among host resources, the immune system and pathogens. Ecol Lett 17:284–293
Curry DW, Perry HB, Tirmizi SN et al (2014) Assessing the effectiveness of house-to-house visits on routine oral polio immunization completion and tracking of defaulters. J Health Popul Nutr 32:356–366
Dellicour S, Baele G, Dudas G et al (2018) Phylodynamic assessment of intervention strategies for the West African Ebola virus outbreak. Nat Commun 9:222
Diekmann O, Heesterbeek JAP (2000) Mathematical epidemiology of infectious diseases: model building, analysis and interpretation. John Wiley & Sons
Dougherty ER, Seidel DP, Carlson CJ et al (2018) Going through the motions: incorporating movement analyses into disease research. Ecol Lett 21:588–604
Driscoll DA, Banks SC, Barton PS et al (2013) Conceptual domain of the matrix in fragmented landscapes. Trends Ecol Evol 28:605–613
Dunn RR, Jonathan DT, Harris NC, Gavin MC (2010) Global drivers of human pathogen richness and prevalence. Proc R Soc B Biol Sci 277:2587–2595
Earn DJD, Rohani P, Bolker BM, Grenfell BT (2000) A simple model for complex dynamical transitions in epidemics. Science 287:667–670
Earn DJD, He D, Loeb, MB et al (2012) Effects of school closure on incidence of pandemic influenza in Alberta, Canada. Ann Intern Med 156:173–181
Earn DJD, Ma J, Poinar H et al (2020) Acceleration of plague outbreaks in the second pandemic. Proc Natl Acad Sci 117:27703–27711
Eichner M, Brockmann SO (2013) Polio emergence in Syria and Israel endangers Europe. Lancet 382:1777
Erős T, Campbell Grant EH (2015) Unifying research on the fragmentation of terrestrial and aquatic habitats: patches, connectivity and the matrix in riverscapes. Freshwater Biol 60:1487–1501
Fallah MP, Skrip LA, Enders J (2018) Preventing rural to urban spread of Ebola: lessons from Liberia. Lancet 392:279–280
Ferguson NM, Donnelly CA, Anderson RM (2001) The foot-and-mouth epidemic in Great Britain: pattern of spread and impact of interventions. Science 292:1155–1160
Ferguson NM, Cucunuba ZM, Dorigatti I et al (2016) Countering the Zika epidemic in Latin America. Science 353:353–354
Ferguson NM, Laydon D, Nedjati-Gilani G et al (2020) Impact of non-pharmaceutical interventions (NPIs) to reduce COVID-19 mortality and healthcare demand. http://imperial.ac.uk/media/imperial-college/medicine/sph/ide/gida-fellowships/Imperial-College-COVID19-NPI-modelling-16-03-2020.pdf. Accessed 20 March 2020
Fieberg J, Kochanny CO (2005) Quantifying home-range overlap: the importance of the utilization distribution. J Wildl Manage 69:1346–1359
Fox MT, Uche UE, Vaillant C et al (2002) Effects of Ostertagia ostertagi and omeprazole treatment on feed intake and gastrin-related responses in the calf. Vet Parasitol 105:285–301
Fryxell JM, Hazell M, Börger L et al (2008) Multiple movement modes by large herbivores at multiple spatiotemporal scales. Proc Natl Acad Sci 105:19114–19119
Gao D, Lou Y, He D et al (2016) Prevention and control of Zika as a mosquito-borne and sexually transmitted disease: a mathematical modeling analysis. Sci Rep 6:1–10
Greenspoon PB, Banton S, Mideo N (2018) Immune system handling time may alter the outcome of competition between pathogens and the immune system. J Theor Biol 447:25–31
Grenfell B, Harwood J (1997) (Meta)population dynamics of infectious diseases. Trends Ecol Evol 12:395–399
Guckenheimer J, Holmes P (1983) Nonlinear oscillations, dynamical systems, and bifurcations of vector fields. Springer, New York
Hall SR, Simonis JL, Nisbet RM et al (2009) Resource ecology of virulence in a planktonic host-parasite system: an explanation using dynamic energy budgets. Am Nat 174:149–162
Halliday FW, Rohr JR (2019) Measuring the shape of the biodiversity-disease relationship across systems reveals new findings and key gaps. Nat Commun 10:5032
Handel A, Longini IM Jr, Antia R (2007) What is the best control strategy for multiple infectious disease outbreaks? Proc Biol Sci 274:833–837
Handel A, Rohani P (2015) Crossing the scale from within-host infection dynamics to between-host transmission fitness: a discussion of current assumptions and knowledge. Philos Trans R Soc Lond B Biol Sci 370:20140302
Hanski I (1998) Metapopulation dynamics. Nature 396:41–49
Hastings A (2004) Transients: the key to long-term ecological understanding? Trends Ecol Evol 19:39–45
Hastings A, Abbott KC, Cuddington K et al (2018) Transient phenomena in ecology. Science 361:1–9
Hawley DM, Altizer SM (2011) Disease ecology meets ecological immunology: understanding the links between organismal immunity and infection dynamics in natural populations. Funct Ecol 25:48–60
Haydon DT, Randall DA, Matthews L et al (2006) Low-coverage vaccination strategies for the conservation of endangered species. Nature 443:692–695
Helble M (2011) The movement of patients across borders: challenges and opportunities for public health. Bull World Health Organ 89:68–72
Hempel K, Earn DJD (2015) A century of transitions in New York City’s measles dynamics. J R Soc Interface 12:20150024
Hess G (1996) Disease in metapopulation models: implications for conservation. Ecology 77:1617–1632
Heymann DL, Chen L, Takemi K et al (2015) Global health security: the wider lessons from the west African Ebola virus disease epidemic. The Lancet 385:1884–1901
Hite JL, Cressler CE (2018) Resource-driven changes to host population stability alter the evolution of virulence and transmission. Philos Trans R Soc Lond B Biol Sci 373:20170087
Hite JL, Cressler CE, (2019) Parasite-mediated anorexia and nutrition modulate virulence evolution. Integr Comp Biol 59:1264–1274
Hite JL, Pfenning AC, Cressler CE (2020) Starving the enemy? Feeding behavior shapes host-parasite interactions. Trends Ecol Evol 35:68–80
Jia JS, Lu X, Yuan Y et al (2020) Population flow drives spatio-temporal distribution of COVID-19 in China. Nature 582:389–394
Johnson PTJ, Preston DL, Hoverman JT, Richgels KLD (2013) Biodiversity decreases disease through predictable changes in host community competence. Nature 494:230–233
Jones KE, Patel NG, Levy MA et al (2008) Global trends in emerging infectious diseases. Nature 451:990–993
Karnatak R, Wollrab S (2020) A probabilistic approach to dispersal in spatially explicit meta-populations. Sci Rep 10:22234
Keeling MJ, Rohani P, Grenfell BT (2001) Seasonally forced disease dynamics explored as switching between attractors. Physica D 148:317–335
Keeling M, Bjornstad ON, Grenfell BT (2004) Metapopulation dynamics of infectious diseases. In Hanski I, Gaggiotti O (eds) Ecology, genetics, and evolution of metapopulations. Elsevier Academic Press. 415–445
Keesing F, Belden LK, Daszak P et al (2010) Impacts of biodiversity on the emergence and transmission of infectious diseases. Nature 468:647–652
Kermack WO, McKendrick AG (1927) A contribution to the mathematical theory of epidemics. Proc R Soc Lond A 115:700–721
Labra FA, Lagos NA, Marquet PA (2003) Dispersal and transient dynamics in metapopulations. Ecol Lett 6:197–204
Lafferty KD, DeLeo G, Briggs CJ et al (2015) A general consumer-resource population model. Science 349:854–857
Lafferty KD, Wood CL (2013) It’s a myth that protection against disease is a strong and general service of biodiversity conservation: response to Ostfeld and Keesing. Trends Ecol Evol 28:503–504
Levi T, Keesing F, Holt RD et al (2016) Quantifying dilution and amplification in a community of hosts for tick-borne pathogens. Ecol Appl 26:484–498
Lewis MA, Moorcroft P (2001) ESS analysis of mechanistic models for territoriality: the value of scent marks in spatial resource partitioning. J Theor Biol 210:449–461
Lipsitch M, Cohen T, Cooper B et al (2003) Transmission dynamics and control of severe acute respiratory syndrome. Science 300:1966–1970
London WP, Yorke JA (1973) Recurrent outbreaks of measles, chickenpox and mumps: I. seasonal variation in contact rates. Am J Epidemiol 98:453–468
Lunn TJ, Restif O, Peel AJ et al (2019) Dose–response and transmission: the nexus between reservoir hosts, environment and recipient hosts. Philos Trans R Soc Lond B Biol Sci 374:20190016
Ma J, Earn DJD (2006) Generality of the final size formula for an epidemic of a newly invading infectious disease. Bull Math Biol 68:679–702
Ma J, Dushoff J, Bolker BM, Earn DJD (2014) Estimating initial epidemic growth rates. Bull Math Biol 76:245–260
Malvy D, McElroy AK, de Clerck H et al (2019) Ebola virus disease. The Lancet 393:936–948
Manore CA, Beechler BR (2015) Inter-epidemic and between-season persistence of Rift Valley Fever: vertical transmission or cryptic cycling? Transbound Emerg Dis 62:13–23
Mbabazi WB, Tabu CW, Chemirmir C et al (2015) Innovations in communication technologies for measles supplemental immunization activities: lessons from Kenya measles vaccination campaign, November 2012. Health Policy Plan 30:638–644
McKibbin WJ, Fernando R (2020) The global macroeconomic impacts of COVID-19: Seven Scenarios. http://brookings.edu/research/the-global-macroeconomic-impacts-of-covid-19-seven-scenarios/. Accessed 24 March 2020
Médecins Sans Frontières (2009) Raging cholera just tip of Zimbabwe’s humanitarian crisis. https://msf.org/raging-cholera-just-tip-zimbabwes-humanitarian-crisis. Accessed 24 March 2020
Mideo N, Alizon S, Day T (2008) Linking within- and between-host dynamics in the evolutionary epidemiology of infectious diseases. Trends Ecol Evol 23:511–517
Molina C, Earn DJD (2015) Game theory of pre-emptive vaccination before bioterrorism or accidental release of smallpox. J R Soc Interface 12:20141387
Moorcroft PR, Lewis MA, Crabtree RL (2006) Mechanistic home range models capture spatial patterns and dynamics of coyote territories in Yellowstone. Proc Biol Sci 273:1651–1659
Morozov A, Abbott K, Cuddington K et al (2020) Long transients in ecology: theory and applications. Phys Life Rev 32:1–40
Mukandavire Z, Liao S, Wang J (2011) Estimating the reproductive numbers for the 2008–2009 cholera outbreaks in Zimbabwe. Proc Natl Acad Sci U S A 108:8767–8772
Nimmo DG, Avitabile S, Banks SC et al (2019) Animal movements in fire-prone landscapes. Biol Rev 94:981–998
Ovaskainen O, Hanski I (2002) Transient dynamics in metapopulation response to perturbation. Theor Popul Biol 61:285–295
Owen JC, Moore FR (2008) Relationship between energetic condition and indicators of immune function in thrushes during spring migration. Can J Zool 86:638–647
Painter M, Qiu T (2020) Political beliefs affect compliance with COVID-19 social distancing orders. https://papers.ssrn.com/sol3/papers.cfm?abstract_id=3569098. Accessed 24 March 2020
Park SW, Bolker BM, Champredon D et al (2020) Reconciling early-outbreak estimates of the basic reproductive number and its uncertainty: framework and applications to the novel coronavirus (SARS-CoV-2) outbreak. J R Soc Interface 17:20200144
Patterson JEH, Ruckstuhl KE (2013) Parasite infection and host group size: a meta-analytical review. Parasitology 140:803–813
Perry GLW, Lee F (2019) How does temporal variation in habitat connectivity influence metapopulation dynamics? Oikos 128:1277–1286
Plowright RK, Foley P, Field HE et al (2011) Urban habituation, ecological connectivity and epidemic dampening: the emergence of Hendra virus from flying foxes (Pteropus spp.). Proc Biol Sci 278:3703–3712
Plowright RK, Parrish CR, McCallum H et al (2017) Pathways to zoonotic spillover. Nat Rev Microbiol 15:502–510
Potts JR, Lewis MA (2014) How do animal territories form and change? Lessons from 20 years of mechanistic modelling. Proc Biol Sci 281:20140231
Potts JR, Lewis MA (2016) How memory of direct animal interactions can lead to territorial pattern formation. J R Soc Interface 13:20160059
Potts JR, Fagan WF, Mourão G (2018) Deciding when to intrude on a neighbour: quantifying behavioural mechanisms for temporary territory expansion. Theor Ecol 12:307–318
Pourbohloul B, Ahued A, Davoudi B et al (2009) Initial human transmission dynamics of the pandemic (H1N1) 2009 virus in North America. Influenza Other Resp 3:215–222
Probert WJM, Lakkur S, Fonnesbeck CJ et al (2019) Context matters: using reinforcement learning to develop human-readable, state-dependent outbreak response policies. Philos Trans R Soc Lond B Biol Sci 374:20180277
Pulliam JRC, Epstein JH, Dushoff J et al (2012) Agricultural intensification, priming for persistence and the emergence of Nipah virus: a lethal bat-borne zoonosis. J R Soc Interface 9:89–101
Ramsey DSL, O’brien DJ, Cosgrove MK et al (2014) Forecasting eradication of bovine tuberculosis in Michigan white‐tailed deer. J Wildl Manage 78:240–254
Randolph SE, Dobson ADM (2012) Pangloss revisited: a critique of the dilution effect and the biodiversity-buffers-disease paradigm. Parasitology 139:847–863
Rao S, Schieber AMP, O’Connor CP et al (2017) Pathogen-mediated inhibition of anorexia promotes host survival and transmission. Cell 168:503–516
Riley S, Fraser C, Donnelly CA et al (2003) Transmission dynamics of the etiological agent of SARS in Hong Kong: impact of public health interventions. Science 300:1961–1966
Rodrigues MC, Cooke RF, Marques RS et al (2015) Effects of vaccination against respiratory pathogens on feed intake, metabolic, and inflammatory responses in beef heifers. J Anim Sci 93:4443–4452
Rogers ME, Bates PA (2007) Leishmania manipulation of sand fly feeding behavior results in enhanced transmission. PLoS Pathog 3:e91
Rohr JR, Civitello DJ, Halliday FW et al (2019) Towards common ground in the biodiversity–disease debate. Nat Ecol Evol 4:24–33
Saravia LA, Ruxton GD, Coviella CE (2000) The importance of transient’s dynamics in spatially extended populations. Proc R Soc Lond B 267:1781–1785
Schütz P, Bally M, Stanga Z, Keller U (2014) Loss of appetite in acutely ill medical inpatients: physiological response or therapeutic target? Swiss Med Wkly 144:w13957
Schwartz IB, Smith HL (1983) Infinite subharmonic bifurcation in an SEIR epidemic model. J Math Biol 18:233–253
Shea K, Tildesley MJ, Runge MC et al (2014) Adaptive management and the value of information: learning via intervention in epidemiology. PLoS Biol 12:e1001970
Smith VH, Holt RD (1996) Resource competition and within-host disease dynamics. Trends Ecol Evol 11:386–389
Strogatz SH (2018) Nonlinear dynamics and chaos: with applications to physics, biology, chemistry, and engineering. CRC Press
Tao Y, Börger L, Hastings A (2016) Dynamic range size analysis of territorial animals: an optimality approach. Am Nat 188:460–474
Tao Y, Shea K, Ferrari M (2018) Logistical constraints lead to an intermediate optimum in outbreak response vaccination. PLoS Comput Biol 14:e1006161
Tao Y, Probert WJM, Shea K et al (2021) Causes of delayed outbreak responses and their impacts on epidemic spread. J R Soc Interface 18:20200933
van den Driessche P, Watmough J (2002) Reproduction numbers and sub-threshold endemic equilibria for compartmental models of disease transmission. Math Biosci 180:29–48
Van Leeuwen A, Budischak SA, Graham AL, Cressler CE (2019) Parasite resource manipulation drives bimodal variation in infection duration. Proc R Soc B 286:20190456
Wahlberg N, Klemetti T, Hanski I (2002) Dynamic populations in a dynamic landscape: the metapopulation structure of the marsh fritillary butterfly. Ecography 25:224–232
Wallinga J, Teunis P (2004) Different epidemic curves for severe acute respiratory syndrome reveal similar impacts of control measures. Am J Epidemiol 160:509–516
Wallinga J, Lipsitch M (2007) How generation intervals shape the relationship between growth rates and reproductive numbers. Proc R Soc B Biol Sci 274:599–604
Wells CR, Sah P, Moghadas SM et al (2020) Impact of international travel and border control measures on the global spread of the novel 2019 coronavirus outbreak. Proc Natl Acad Sci 117:7504–7509
White LA, VandeWoude S, Craft ME (2020) A mechanistic, stigmergy model of territory formation in solitary animals: territorial behavior can dampen disease prevalence but increase persistence. PLoS Comput Biol 16:e1007457
Wilber MQ, Johnson PTJ, Briggs CJ (2017) When can we infer mechanism from parasite aggregation? A constraint-based approach to disease ecology. Ecology 98:688–702
Widmar NJO, Dominick SR, Ruple A, Tyner WE (2017) The influence of health concern on travel plans with focus on the Zika virus in 2016. Prev Med Rep 6:162–170
Wilder-Smith A, Freedman DO (2020) Isolation, quarantine, social distancing and community containment: pivotal role for old-style public health measures in the novel coronavirus (2019-nCoV) outbreak. J Travel Med 27:1–4
Wood CL, McInturff A, Young HS et al (2017) Human infectious disease burdens decrease with urbanization but not with biodiversity. Philos Trans R Soc Lond B Biol Sci 372:20160122
Wright AL, Sonin K, Driscoll J, Wilson J (2020) Poverty and Economic Dislocation Reduce Compliance with COVID-19 Shelter-in-Place Protocols. J Econ Behav Organ 180:544–554
Wu F, Zhao S, Yu B et al (2020) A new coronavirus associated with human respiratory disease in China. Nature 579:265–269
Wu Z, McGoogan JM (2020) Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: summary of a report of 72314 cases from the Chinese Center for Disease Control and Prevention. http://jamanetwork.com/journals/jama/fullarticle/2762130. Accessed 24 March 2020
Yang Y, Sugimoto JD, Halloran ME et al (2009) The Transmissibility and control of pandemic influenza A (H1N1) virus. Science 326:729–733
Zeigler SL, Fagan WF (2014) Transient windows for connectivity in a changing world. Mov Ecol 2:1–10
Zhou F, Yu T, Du R et al (2020) Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet 395:1054–1062
Zidon R, Garti S, Getz WM, Saltz D (2017) Zebra migration strategies and anthrax in Etosha National Park. Namibia Ecosphere 8:e01925
Acknowledgements
This project began at the NIMBioS Investigative Workshop on Transients in Biological Systems on 30 May 2019. The authors thank the organizers of the workshop, as well as Holly Bik and Noah Legall for help and comments.
Funding
YT was supported by an appointment to the Intelligence Community Postdoctoral Research Fellowship Program at UC Santa Barbara, administered by Oak Ridge Institute for Science and Education through an interagency agreement between the U.S. Department of Energy and the Office of the Director of National Intelligence. JLH was supported by the National Institute of General Medical Sciences of the National Institutes of Health under Award Number F32GM128246. NB was supported by the Huck Institutes of the Life Sciences, the National Science Foundation CNH:DEB-1716698 and the DARPA PREEMPT program Cooperative Agreement # D18AC00031. The contents of the information do not necessarily reflect the position or the policy of the U.S. government, and no official endorsement should be inferred. KDL was supported by the Ecosystem Mission Area of the U.S. Geological Survey. This article has been peer reviewed and approved for publication consistent with USGS Fundamental Science Practices (https://pubs.usgs.gov/circ/1367/). Any use of trade, firm, or product names is for descriptive purposes only and does not imply endorsement by the U.S. Government. DJDE was supported by the Natural Sciences and Engineering Research Council of Canada (NSERC). SHARCnet (www.sharcnet.ca) provided computational resources.
Author information
Authors and Affiliations
Contributions
All authors conceived, discussed, and contributed to figure generations and writing of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Tao, Y., Hite, J.L., Lafferty, K.D. et al. Transient disease dynamics across ecological scales. Theor Ecol 14, 625–640 (2021). https://doi.org/10.1007/s12080-021-00514-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12080-021-00514-w