Abstract
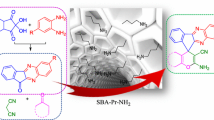
Incorporating sulfanilic acid as a hydrophobic Brønsted acid inside the nanospaces of SBA-15 led to a water-tolerant solid acid catalyst, SBA-15-PhSO3H, which showed excellent catalytic performance in synthesis of uracil-fused spirooxindoles in aqueous ethanol. The synthesized compounds were evaluated for their antioxidant activity by 1,1-diphenyl-2-picrylhydrazyl (DPPH) radical-scavenging assay.

Green synthesis of uracil-fused spirooxindoles using sulfanilic acidfunctionalized SBA-15 as a reusable heterogeneous acid catalyst and the antioxidant activity of the synthesized compounds are described.






Similar content being viewed by others
References
(a) Kefayati H, Narchin F and Rad-Moghadam K 2012 Tetrahedron Lett. 53 4573; (b) Kumar M, Sharma K and Arya A K 2012 Tetrahedron Lett. 53 4604; (c) Arya A K, Gupta S K and Kumar M 2012 Tetrahedron Lett. 53 6035; (d) Zhu J and Bienayme H 2005 In Multicomponent Reactions (Weinheim: Wiley-VCH)
Clark J H and Macquarrie D J 1996 Chem. Soc. Rev. 25 303
Adam F, Hello K M and Aisha M R B 2011 J. Taiwan Inst. Chem. E 42 843
(a) Martin-Aranda R M and Cejka J 2010 Top. Catal. 53 141; (b) Mondal J, Nandi M, Modak A and Bhaumik A 2012 J. Molecul. Catal. A 363–364 254
Karimi B and Zareyee D 2008 Org. Lett. 10 3989
Karimi B and Vafaeezadeh M 2012 Chem. Commun. 48 3327
Yuan C, Huang Z and Chen J 2012 Catal. Commun. 24 56
Parvin M N, Jin H, Ansari M B, Oh S M and Park S E 2012 Appl. Catal. A 413-414 205
Mohammadi Ziarania G, Mousavi S, Lashgari N and Badiei A 2013 J. Chem. Sci. 125 1359
Rostamnia S, Xin H, Liuc X and Lamei K 2013 J. Molecul. Catal. A 374–375 85
Zareyee D, Moosavi S M and Alaminezhad A 2013 J. Molecul. Catal. A 378 227
Rostamnia S and Hassankhani A 2014 Synlett 25 2753
Mohammadi Ziarania G, Lashgari N and Badiei A 2015 J. Molecul. Catal. A 397 166
Bahrami K, Khodaei M M and Fattahpour P 2015 J. Chem. Sci. 127 167
Abdel-Rahman A H, Keshk E M, Hanna M A and El-Bady S M 2004 Bioorg. Med. Chem. 12 2483
Kornet M J and Thio A P 1976 J. Med. Chem. 19 892
Okita T and Isobe M 1994 Tetrahedron 50 11143
Rosenmond P, Hosseini-Merescht M and Bub C 1994 Liebigs Ann. Chem. 2 151
Usui T, Kondoh M, Cui C B, Mayumi T and Osada H 1998 Biochem. J. 333 543
Kang T H, Matsumoto K, Murakami Y, Takayama H, Kitajima M, Aimi N and Watanabe H 2002 Eur. J. Pharmacol. 444 39
Litvinov Y M, Mortikov V Y and Shestopalov A M 2008 J. Comb. Chem. 10 741
Ghahremanzadeh R, Sayyafi M, Ahadi S and Bazgir A 2009 J. Comb. Chem. 11 393
Ghahremanzadeh R, Imani Shakibaei G, Ahadi S and Bazgir A 2010 J. Comb. Chem. 12 191
Balamurugan K, Perumal S and Menendez J C 2011 Tetrahedron 67 3201
Imani Shakibaei G, Feiz A, Khavasi H R, Abolhasani Soorki A and Bazgir A 2011 ACS Comb. Sci. 13 96
Jadidi K, Ghahremanzadeh R, Mirzaei P and Bazgir A 2011 J. Heterocycl. Chem. 48 1014
Arya A K and Kumar M 2011 Mol. Divers. 15 781
Kumar M, Sharma K and Arya A K 2012 Tetrahedron Lett. 53 4604
Rahmati A and Khalesi Z 2012 Tetrahedron 68 8472
Paul S and Das A R 2013 Tetrahedron Lett. 54 1149
Khalafi-Nezhad A and Mohammadi S 2013 ACS Comb. Sci. 15 512
Baharfar R and Azimi R 2014 Synth. Commun. 44 89
Baharfar R and Shariati N 2014 Aust. J. Chem. 67 1646
Azimi R and Baharfar R 2014 Can. J. Chem. 92 1163
Adam F, Hello K M and Ali T H 2011 Appl. Catal. A 399 42
Blois M S 1958 Nature 26 1199
Kumar M, Sharma K, Samarth R M and Kumar A 2010 Eur. J. Med. Chem. 45 4467
Kadhum A A H, Al-Amiery A A, Musa A Y and Mohamad A B 2011 Int. J. Mol. Sci. 12 5747
Acknowledgements
We are thankful to the Research Council of Mazandaran University for the financial support of this work.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information
All additional information relating to characterization of the catalyst using FT-IR, TGA, XRD and TEM techniques (figures S1–S4), 1H NMR and 13C NMR spectra for compounds 4a-j (figures S5-S24) are available in the supplementary information, which is available free of charge at www.ias.ac.in/chemsci.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
BAHARFAR, R., AZIMI, R. Sulfanilic acid functionalized mesoporous SBA-15: A water-tolerant solid acid catalyst for the synthesis of uracil fused spirooxindoles as antioxidant agents. J Chem Sci 127, 1389–1395 (2015). https://doi.org/10.1007/s12039-015-0910-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12039-015-0910-2