Abstract
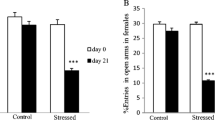
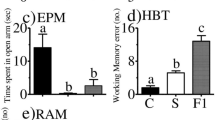
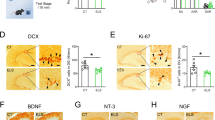
Studies show that gender plays an important role in stress-related disorders, and women are more vulnerable to its effect. The present study was undertaken to investigate differences in the change in expression of brain-derived neurotrophic factor (BDNF), and its tyrosine intracellular kinase-activating receptor (TrkB) genes in the male and female rats’ hippocampus (HPC) under chronic mild repeated stress (CMRS) conditions. In this experiment, male and female Wistar rats were randomly divided into two groups: the CMRS and the control group. To induce stress, a repeated forced swimming paradigm was employed daily for adult male and female rats for 21 days. At the end of the stress phase, elevated plus maze (EPM) was used for measuring the stress behavioral effects. Serum corticosterone level was measured by ELISA. BDNF and TrkB gene methylation and protein expression in the HPC were detected using real-time PCR and Western blotting. Chronic stress in the adolescence had more effects on anxiety-like behavior and serum corticosterone concentration in female rats than males. Furthermore, stressed female rats had higher methylation levels and following reduced protein expression of BDNF but not TrkB compared to stressed male rats. These findings suggest that in exposure to a stressor, sex differences in BDNF methylation may be root cause of decreased BDNF levels in females and may underlie susceptibility to pathology development.




Similar content being viewed by others
References
Lin Y, Westenbroek C, Bakker P, Termeer J, Liu A, Li X, Ter Horst GJ (2008) Effects of long-term stress and recovery on the prefrontal cortex and dentate gyrus in male and female rats. Cereb Cortex 18(12):2762–2774
Bale TL (2006) Stress sensitivity and the development of affective disorders. Horm Behav 50(4):529–533
Nan X, Ng H-H, Johnson CA, Laherty CD, Turner BM, Eisenman RN, Bird A (1998) Transcriptional repression by the methyl-CpG-binding protein MeCP2 involves a histone deacetylase complex. Nature 393(6683):386–389
Becker PB, Ruppert S, Schütz G (1987) Genomic footprinting reveals cell type-specific DNA binding of ubiquitous factors. Cell 51(3):435–443
Hill RA, van den Buuse M (2011) Sex-dependent and region-specific changes in TrkB signaling in BDNF heterozygous mice. Brain Res 1384:51–60
Hashimoto K, Shimizu E, Iyo M (2004) Critical role of brain-derived neurotrophic factor in mood disorders. Brain Res Rev 45(2):104–114
Buckley PF, Mahadik S, Pillai A, Terry A Jr (2007) Neurotrophins and schizophrenia. Schizophr Res 94(1):1–11
Schaaf M, De Kloet E, Vreugdenhil E (2000) Corticosterone effects on bdnf expression in the HPC implications for memory formation. Stress: Int J Biol Stress 3(3):201–208
Sirianni RW, Olausson P, Chiu AS, Taylor JR, Saltzman WM (2010) The behavioral and biochemical effects of BDNF containing polymers implanted in the HPC of rats. Brain Res 1321:40–50
Martinowich K, Manji H, Lu B (2007) New insights into BDNF function in depression and anxiety. Nat Neurosci 10(9):1089–1093
Dranovsky A, Hen R (2006) Hippocampal neurogenesis: regulation by stress and antidepressants. Biol Psychiatry 59(12):1136–1143
Gomez-Pinilla F, Vaynman S (2005) A “deficient environment” in prenatal life may compromise systems important for cognitive function by affecting BDNF in the HPC. Exp Neurol 192(2):235–243
Autry AE, Adachi M, Cheng P, Monteggia LM (2009) Gender-specific impact of brain-derived neurotrophic factor signaling on stress-induced depression-like behavior. Biol Psychiatry 66(1):84–90
Zuena AR, Mairesse J, Casolini P, Cinque C, Alemà GS, Morley-Fletcher S, Chiodi V, Spagnoli LG et al (2008) Prenatal restraint stress generates two distinct behavioral and neurochemical profiles in male and female rats. PLoS One 3(5), e2170
Bangasser DA, Valentino RJ (2012) Sex differences in molecular and cellular substrates of stress. Cell Mol Neurobiol 32(5):709–723
Altemus M (2006) Sex differences in depression and anxiety disorders: potential biological determinants. Horm Behav 50(4):534–538
Smith MA (1996) Hippocampal vulnerability to stress and aging: possible role of neurotrophic factors. Behav Brain Res 78(1):25–36
Li Y, Ji Y-J, Jiang H, Liu D-X, Zhang Q, Fan S-J, Pan F (2009) Effects of unpredictable chronic stress on behavior and brain-derived neurotrophic factor expression in CA3 subfield and dentate gyrus of the HPC in different aged rats. Chin Med J (Engl) 122(13):1564–1569
Badowska-Szalewska E, Spodnik E, Klejbor I, Morys J (2010) Effects of chronic forced swim stress on hippocampal brain-derived neutrophic factor (BDNF) and its receptor (TrkB) immunoreactive cells in juvenile and aged rats. Acta Neurobiol Exp 70:370–381
McEwen BS (2007) Physiology and neurobiology of stress and adaptation: central role of the brain. Physiol Rev 87(3):873–904
Shi S-S, S-h S, B-p Y, Pan F, Li Z-L (2010) Acute stress and chronic stress change brain-derived neurotrophic factor (BDNF) and tyrosine kinase-coupled receptor (TrkB) expression in both young and aged rat HPC. Yonsei Med J 51(5):661–671
Pingoud A, Alves J, Geiger R (1993) Restriction enzymes. In: Enzymes of molecular biology. Springer, pp 107-200
Yang C, Zhang M, Niu W, Yang R, Zhang Y, Qiu Z, Sun B, Zhao Z (2011) Analysis of DNA methylation in various swine tissues. PLoS One 6(1), e16229
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72(1):248–254
Boyce-Rustay JM, Cameron HA, Holmes A (2007) Chronic swim stress alters sensitivity to acute behavioral effects of ethanol in mice. Physiol Behav 91(1):77–86
Ulrich-Lai YM, Figueiredo HF, Ostrander MM, Choi DC, Engeland WC, Herman JP (2006) Chronic stress induces adrenal hyperplasia and hypertrophy in a subregion-specific manner. Am J Physiol-Endocrinol Metab 291(5):E965–E973
Avital A, Richter-Levin G, Leschiner S, Spanier I, Veenman L, Weizman A, Gavish M (2001) Acute and repeated swim stress effects on peripheral benzodiazepine receptors in the rat HPC, adrenal, and kidney. Neuropsychopharmacology 25(5):669–678
Luine V (2002) Sex differences in chronic stress effects on memory in rats. Stress: Int JBiol Stress 5(3):205–216
Viau V, Bingham B, Davis J, Lee P, Wong M (2005) Gender and puberty interact on the stress-induced activation of parvocellular neurosecretory neurons and corticotropin-releasing hormone messenger ribonucleic acid expression in the rat. Endocrinology 146(1):137–146
Iwasaki-Sekino A, Mano-Otagiri A, Ohata H, Yamauchi N, Shibasaki T (2009) Gender differences in corticotropin and corticosterone secretion and corticotropin-releasing factor mRNA expression in the paraventricular nucleus of the hypothalamus and the central nucleus of the amygdala in response to footshock stress or psychological stress in rats. Psychoneuroendocrinology 34(2):226
Livezey GT, Miller JM, Vogel WH (1985) Plasma norepinephrine, epinephrine and corticosterone stress responses to restraint in individual male and female rats, and their correlations. Neurosci Lett 62(1):51–56
Vamvakopoulos NC, Chrousos GP (1993) Evidence of direct estrogenic regulation of human corticotropin-releasing hormone gene expression. Potential implications for the sexual dimophism of the stress response and immune/inflammatory reaction. J Clin Investig 92(4):1896
Bao A, Fischer D, Wu Y, Hol E, Balesar R, Unmehopa U, Zhou J, Swaab D (2006) A direct androgenic involvement in the expression of human corticotropin-releasing hormone. Mol Psychiatry 11(6):567–576
Lund TD, Rovis T, Chung WC, Handa RJ (2005) Novel actions of estrogen receptor-β on anxiety-related behaviors. Endocrinology 146(2):797–807
Toufexis DJ, Myers KM, Bowser ME, Davis M (2007) Estrogen disrupts the inhibition of fear in female rats, possibly through the antagonistic effects of estrogen receptor α (ERα) and ERβ. J Neurosci 27(36):9729–9735
Handa RJ, Burgess LH, Kerr JE, O'Keefe JA (1994) Gonadal steroid hormone receptors and sex differences in the hypothalamo-pituitary-adrenal axis. Horm Behav 28(4):464–476
Seale J, Wood S, Atkinson H, Harbuz M, Lightman S (2004) Gonadal steroid replacement reverses gonadectomy‐induced changes in the corticosterone pulse profile and stress‐induced hypothalamic‐pituitary‐adrenal axis activity of male and female rats. J Neuroendocrinol 16(12):989–998
Calabrese F, Molteni R, Racagni G, Riva MA (2009) Neuronal plasticity: a link between stress and mood disorders. Psychoneuroendocrinology 34(Suppl 1):S208–S216
Sandanger I, Nygård JF, Sørensen T, Moum T (2004) Is women’s mental health more susceptible than men’s to the influence of surrounding stress? Soc Psychiatry Psychiatr Epidemiol 39(3):177–184
Weekes NY, Lewis RS, Goto SG, Garrison-Jakel J, Patel F, Lupien S (2008) The effect of an environmental stressor on gender differences on the awakening cortisol response. Psychoneuroendocrinology 33(6):766–772
Kessler RC (2003) Epidemiology of women and depression. J Affect Disord 74(1):5–13
Chertkow-Deutsher Y, Cohen H, Klein E, Ben-Shachar D (2010) DNA methylation in vulnerability to post-traumatic stress in rats: evidence for the role of the post-synaptic density protein Dlgap2. Int J Neuropsychopharmacol 13(3):347
Razin A (1998) CpG methylation, chromatin structure and gene silencing—a three-way connection. EMBO J 17(17):4905–4908
Martinowich K, Hattori D, Wu H, Fouse S, He F, Hu Y, Fan G, Sun YE (2003) DNA methylation-related chromatin remodeling in activity-dependent BDNF gene regulation. Science 302(5646):890–893
Martinowich K, Schloesser RJ, Jimenez DV, Weinberger DR, Lu B (2011) Activity-dependent brain-derived neurotrophic factor expression regulates cortistatin-interneurons and sleep behavior. Mol Brain 4(11)
Parrish RR, Buckingham SC, Mascia KL, Johnson JJ, Matyjasik MM, Lockhart RM, Lubin FD (2015) Methionine increases BDNF DNA methylation and improves memory in epilepsy. Ann Clin Transl Neurol 2(4):401–416
Roth TL, Zoladz PR, Sweatt JD, Diamond DM (2011) Epigenetic modification of hippocampal<i>Bdnf</i>DNA in adult rats in an animal model of post-traumatic stress disorder. J Psychiatr Res 45(7):919–926
McCormick CM, Mathews IZ, Thomas C, Waters P (2010) Investigations of HPA function and the enduring consequences of stressors in adolescence in animal models. Brain Cogn 72(1):73–85
Vollmayr B, Faust H, Lewicka S, Henn F (2001) Brain-derived-neurotrophic-factor (BDNF) stress response in rats bred for learned helplessness. Mol Psychiatry 6(4):471–474
Tsankova NM, Berton O, Renthal W, Kumar A, Neve RL, Nestler EJ (2006) Sustained hippocampal chromatin regulation in a mouse model of depression and antidepressant action. Nat Neurosci 9(4):519–525
Saarelainen T, Hendolin P, Lucas G, Koponen E, Sairanen M, MacDonald E, Agerman K, Haapasalo A et al (2003) Activation of the TrkB neurotrophin receptor is induced by antidepressant drugs and is required for antidepressant-induced behavioral effects. Jo Neurosci 23(1):349–357
Onishchenko N, Karpova N, Sabri F, Castrén E, Ceccatelli S (2008) Long‐lasting depression‐like behavior and epigenetic changes of BDNF gene expression induced by perinatal exposure to methylmercury. J Neurochem 106(3):1378–1387
Keller S, Sarchiapone M, Zarrilli F, Videtič A, Ferraro A, Carli V, Sacchetti S, Lembo F et al (2010) Increased BDNF promoter methylation in the Wernicke area of suicide subjects. Arch Gen Psychiatry 67(3):258–267
Keller S, Sarchiapone M, Zarrilli F, Tomaiuolo R, Carli V, Angrisano T, Videtic A, Amato F et al (2011) TrkB gene expression and DNA methylation state in Wernicke area does not associate with suicidal behavior. J Affect Disord 135(1):400–404
Ernst C, Deleva V, Deng X, Sequeira A, Pomarenski A, Klempan T, Ernst N, Quirion R et al (2009) Alternative splicing, methylation state, and expression profile of tropomyosin-related kinase B in the frontal cortex of suicide completers. Arch Gen Psychiatry 66(1):22
Koponen E, Rantamäki T, Voikar V, Saarelainen T, MacDonald E, Castrén E (2005) Enhanced BDNF signaling is associated with an antidepressant-like behavioral response and changes in brain monoamines. Cell Mol Neurobiol 25(6):973–980
Koponen E, WIKAR V, Riekki R, Saarelainen T, Rauramaa T, Rauvala H, Taira T, Castrén E (2004) Transgenic mice overexpressing the full-length neurotrophin receptor trkB exhibit increased activation of the trkB-PLCγ pathway, reduced anxiety, andfacilitated learning. Mol Cell Neurosci 26(1):166–181
Fumagalli F, Racagni G, Riva M (2005) The expanding role of BDNF: a therapeutic target for Alzheimer's disease? Pharmacogenomics J 6(1):8–15
Yu H, Z-y C (2010) The role of BDNF in depression on the basis of its location in the neural circuitry. Acta Pharmacol Sin 32(1):3–11
Sohrabji F, Lewis DK (2006) Estrogen–BDNF interactions: implications for neurodegenerative diseases. Front Neuroendocrinol 27(4):404–414
Sun M-K, Alkon DL (2006) Differential gender-related vulnerability to depression induction and converging antidepressant responses in rats. J Pharmacol Exp Ther 316(2):926–932
Sohrabji F, Miranda R, Toran-Allerand CD (1995) Identification of a putative estrogen response element in the gene encoding brain-derived neurotrophic factor. Proc Natl Acad Sci 92(24):11110–11114
Murphy DD, Cole NB, Segal M (1998) Brain-derived neurotrophic factor mediates estradiol-induced dendritic spine formation in hippocampal neurons. Proc Natl Acad Sci 95(19):11412–11417
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Niknazar, S., Nahavandi, A., Peyvandi, A.A. et al. Comparison of the Adulthood Chronic Stress Effect on Hippocampal BDNF Signaling in Male and Female Rats. Mol Neurobiol 53, 4026–4033 (2016). https://doi.org/10.1007/s12035-015-9345-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-015-9345-5