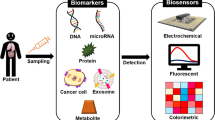
Abstract
Cancer stands as the reigning champion of life-threatening diseases, casting a shadow with the highest global mortality rate. Unleashing the power of early cancer treatment is a vital weapon in the battle for efficient and positive outcomes. Yet, conventional screening procedures wield limitations of exorbitant costs, time-consuming endeavors, and impracticality for repeated testing. Enter bio-marker-based cancer diagnostics, which emerge as a formidable force in the realm of early detection, disease progression assessment, and ultimate cancer therapy. These remarkable devices boast a reputation for their exceptional sensitivity, streamlined setup requirements, and lightning fast response times. In this study, we embark on a captivating exploration of the most recent advancements and enhancements in the field of electrochemical marvels, targeting the detection of numerous cancer biomarkers. With each breakthrough, we inch closer to a future where cancer’s grip on humanity weakens, guided by the promise of personalized treatment and improved patient outcomes. Together, we unravel the mysteries that cancer conceals and illuminate a path toward triumph against this daunting adversary. This study celebrates the relentless pursuit of progress, where electrochemical innovations take center stage in the quest for a world free from the clutches of carcinoma.



© 2018 Elsevier B.V.)

© 2020 Elsevier B.V.)

(Adapted from Zouari et al. 2018 [150] under an ACS AuthorChoice License. © 2018 American Chemical Society)
Similar content being viewed by others
Data availability
All data is available in the script.
Abbreviations
- AFP::
-
alpha-fetoprotein
- CA125::
-
cancer antigen 125
- CA15-3::
-
cancer antigen 15–3
- CEA::
-
carcinoembryonic antigen
- PSA::
-
prostate-specific antigen
- WHO::
-
world health organization
- EBs::
-
electrochemical biosensors
- PCR::
-
polymerase chain reaction
- RIA::
-
radioimmunoassay
- ELISA::
-
enzyme-linked immunosorbent assay
- HPLC::
-
high-performance liquid chromatography
- PSA::
-
prostate-specific antigen
- RF::
-
radio frequency
- MW::
-
microwave
- mmW::
-
millimeter wave
- THz::
-
tetrahertz
- EIS::
-
electrochemical impedance spectroscopy
- DDPpy::
-
DNA dendrimers and polypyrrole
- ITO::
-
indium-tin oxide
- DNA::
-
deoxyribonucleic acid
- CV::
-
cyclic voltammogram
- EIS::
-
electrochemical impedance spectroscopy
- GLC::
-
gas liquid chromatography VI
- ROS::
-
reactive oxygen species
- LPO::
-
lipid per oxidation
- H2O2::
-
hydrogen peroxide
- dsDNA::
-
double-strand DNA
- ssDNA::
-
single-strand DNA
- SPGEs::
-
screen-printed gold electrodes
- BESs::
-
bio-electrochemical systems
- HPLC::
-
high-performance liquid chromatography
- GC–MS::
-
gas chromatography–mass spectrometry
- LC–MS::
-
liquid chromatography–mass spectrometry
- AFM::
-
atomic force microscopy
- SEM::
-
scanning electron microscopy
- TEM::
-
transmission electron microscopy
- US EPA::
-
U.S. environmental protection agency
- IARC::
-
international agency for research on cancer
- SAW::
-
surface acoustic wave
References
GBD. 2015 Mortality and causes of death collaborators. Global, regional, and national life expectancy, all-cause mortality, and cause-specific mortality for 249 causes of death, 1980–2015: a systematic analysis for the Global burden of disease study 2015. Lancet. 2016;388:1459–544.
Do N, Grossman R, Feldman T, Fillmore N, Elbers D, Tuck D, Dhond R, et al. "The Veterans Precision Oncology Data Commons: transforming VA data into a national resource for research in precision oncology. In Sem Oncol. 2019;46(4–5):314–20.
Horton S, Gauvreau CL. Cancer in Low- and Middle Income Countries: an Economic Overview. In: Gelband H, Jha P, Sankaranarayanan R, et al. editors. SourceCancer: Disease Control Priorities, Third Edition (Volume 3). Washington (DC): The International Bank for Reconstruction and Development / The World Bank; 2015 Nov. Chapter 16
Nagai H, Kim YH. Cancer prevention from the perspective of global cancer burden patterns. J Thorac Dis. 2017;9(3):448.
Li J, Li S, Yang CF. Electrochemical biosensors for cancer biomarker detection. Electroanalysis. 2012;24(12):2213–29.
Goossens N, Nakagawa S, Sun X, Hoshida Y. Cancer biomarker discovery and validation. Transl Cancer Res. 2015;4(3):256–69.
Khoury JD, Wang WL, Prieto VG, Medeiros LJ, Kalhor N, Hameed M, Broaddus R, Hamilton SR. Validation of immunohistochemical assays for integral biomarkers in the NCI-MATCH EAY131 clinical trial validation of IHC integral marker assays. Clin Cancer Res. 2018;24(3):521–31.
Ausch C, Kim YH, Tsuchiya KD, Dzieciatkowski S, Washington MK, Paraskeva C, Radich J, Grady WM. Comparative analysis of PCR-based biomarker assay methods for colorectal polyp detection from fecal DNA. Clin Chem. 2009;55(8):1559–63.
Geisler C, Gaisa NT, Pfister D, Fuessel S, Kristiansen G, Braunschweig T, Gostek S, Beine B, Diehl HC, Jackson AM, Borchers CH. Identification and validation of potential new biomarkers for prostate cancer diagnosis and prognosis using 2D-DIGE and MS. BioMed Res Int. 2015.
Shlyapnikov YM, Malakhova EA, Vinarov AZ, Zamyatnin AA Jr, Shlyapnikova EA. Can new immunoassay techniques improve bladder cancer diagnostics with protein biomarkers? Front Mol Biosci. 2021;7:620687.
Al-Wajeeh AS, Salhimi SM, Al-Mansoub MA, Khalid IA, Harvey TM, Latiff A, Ismail MN. Comparative proteomic analysis of different stages of breast cancer tissues using ultra high performance liquid chromatography tandem mass spectrometer. PloS One. 2020;15(1):e0227404.
Hu R, Sou K, Takeoka S. A rapid and highly sensitive biomarker detection platform based on a temperature-responsive liposome-linked immunosorbent assay. Sci Rep. 2020;10(1):1–11.
Pulumati A, Pulumati A, Dwarakanath BS, Verma A, Papineni RVL. Technological advancements in cancer diagnostics: improvements and limitations. Cancer Rep (Hoboken). 2023;6(2):e1764.
Stobiecka M, Ratajczak K, Jakiela S. Toward early cancer detection: focus on biosensing systems and biosensors for an anti-apoptotic protein survivin and survivin mRNA. Biosens Bioelectron. 2019;137:58–71.
Bohunicky B, Mousa SA. Biosensors: the new wave in cancer diagnosis. Nanotechnol Sci Appl. 2010;30(4):1–10.
Arya S. K. and Estrela P., 2018 Recent advances in enhancement strategies for electrochemical ELISA-based immunoassays for cancer biomarker detection. Sensors (Switzerland), 18
Ranjan R, Esimbekova EN, Kratasyuk VA. Rapid biosensing tools for cancer biomarkers. Biosens Bioelectron. 2017;87:918.
Mahato K, Kumar A, Maurya PK, Chandra P. Shifting paradigm of cancer diagnoses in clinically relevant samples based on miniaturized electrochemical nanobiosensors and microfluidic devices. Biosens Bioelectron. 2018;100:411.
Freitas M, Nouws HPA, Delerue-Matos C. Electrochemical biosensing in cancer diagnostics and follow-up. Electroanalysis. 2018;30:1576.
Lassere MN. The biomarker-surrogacy evaluation schema: a review of the biomarker-surrogate literature and a proposal for a criterion-based, quantitative, multidimensional hierarchical levels of evidence schema for evaluating the status of biomarkers as surrogate endpoints. Stat Meth Med Res. 2008;17(3):303–40.
WHO. 2001 Biomarkers in risk assessment: Validity and validation. WHO
Biomarkers Definitions Working Group. Biomarkers and surrogate endpoints: preferred definitions and conceptual framework. Clin Pharmacol Ther. 2001;69(3):89–95.
Topkaya SN, Azimzadeh M, Ozsoz M. Electrochemical biosensors for cancer biomarkers detection: recent advances and challenges. Electroanalysis. 2016;28(7):1402–19.
Cui F, Zhou Z, Zhou HS. Measurement and analysis of cancer biomarkers based on electrochemical biosensors. J Electrochem Soc. 2019;167(3):037525.
Varol T. Ö. 2020 Electrochemical Sensors and Biosensors for the Detection of Cancer Biomarkers and Drugs. In: H. S. Tuli (eds) Drug Targets in Cellular Processes of Cancer: From Nonclinical to Preclinical Models. https://doi.org/10.1007/978-981-15-7586-0_2
Sarhadi VK, Armengol G. Molecular biomarkers in cancer. Biomolecules. 2022;12(8):1021.
Liu H, Xu Y, Xiang J, Long L, Green S, Yang Z, Zimdahl B, Lu J, Cheng N, Horan LH, Liu B. Targeting alpha-fetoprotein (AFP)–MHC complex with CAR T-cell therapy for liver cancer CAR T therapy against AFP for the treatment of liver cancer. Clin Cancer Res. 2017;23(2):478–88.
Qin YY, Wu YY, Xian XY, Qin JQ, Lai ZF, Liao L, Lin FQ. Single and combined use of red cell distribution width, mean platelet volume, and cancer antigen 125 for differential diagnosis of ovarian cancer and benign ovarian tumors. J Ovarian Res. 2018;11(1):1–6.
Li X, Dai D, Chen B, Tang H, Xie X, Wei W. Clinicopathological and prognostic significance of cancer antigen 15–3 and carcinoembryonic antigen in breast cancer: a meta-analysis including 12,993 patients. Dis Mark. 2018.
Sekiguchi M, Matsuda T. Limited usefulness of serum carcinoembryonic antigen and carbohydrate antigen 19–9 levels for gastrointestinal and whole-body cancer screening. Sci Rep. 2020;10(1):1–10.
Kelleher M, Singh R, O’Driscoll CM, Melgar S. Carcinoembryonic antigen (CEACAM) family members and inflammatory bowel disease. Cytokine Growth Factor Rev. 2019;47:21–31.
Betz, D. and Fane, K., 2018. Human chorionic gonadotropin (HCG). 2023 Aug 14. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2023
Nordström T, Akre O, Aly M, Grönberg H, Eklund M. Prostate-specific antigen (PSA) density in the diagnostic algorithm of prostate cancer. Prostate Cancer Prostat Diseas. 2018;21(1):57–63.
Chinen AB, Guan CM, Ferrer JR, Barnaby SN, Merkel TJ, Mirkin CA. Nanoparticle probes for the detection of cancer biomarkers, cells, and tissues by fluorescence. Chem Rev. 2015;115(19):10530–74.
Nikhil B, Pawan J, Nello F, Pedro E. Introduction to biosensors. Essays Biochem. 2016;60(1):1–8.
Heineman WR, Jensen WB. Leland C. Clark Jr. (1918–2005). Biosens Bioelectron. 2006;8(21):1403–4.
Cremer, M., 1906. Über die Ursache der elektromotorischen Eigenschaften der Gewebe, zugleich ein Beitrag zur Lehre von den polyphasischen Elektrolytketten. R. Oldenbourg.
Sörensen, S.P.L., 1909. Uber die Messung und die Bedeutung der Wasserstoffionenkonzentration bei enzymatischen Prozessen.
Griffin EG, Nelson JM. The influence of certain substances on the activity of invertase. J Am Chem Soc. 1916;38(3):722–30.
Nelson JM, Griffin EG. Adsorption of invertase. J American Chem Soc. 1916;38(5):1109–15.
Hughes WS. The potential difference between glass and electrolytes in contact with the glass. J American Chem Soc. 1922;44(12):2860–7.
Karunakaran, C., Rajkumar, R. and Bhargava, K., 2015. Introduction to biosensors. In Biosensors and bioelectronics (pp. 1–68). Elsevier.
Heineman WR, Jensen WB. Leland c clark jr (1918–2005). Biosens Bioelectron. 2006;8(21):1403–4.
Clark LC Jr, Lyons C. Electrode systems for continuous monitoring in cardiovascular surgery. Ann New York Acad Sci. 1962;102(1):29–45.
Updike SJ, Hicks GP. The enzyme electrode. Nature. 1967;214:986–8.
Guilbault GG, Montalvo JG Jr. Urea-specific enzyme electrode. J American Chem Soc. 1969;91(8):2164–5.
Bergveld P. Development of an ion-sensitive solid-state device for neurophysiological measurements. IEEE Trans Biomed Eng. 1970;1:70–1.
Guilbault GG, Lubrano GJ. An enzyme electrode for the amperometric determination of glucose. Analytica chimica acta. 1973;64(3):439–55.
Mosbach K, Danielsson B. An enzyme thermistor. Biochimica et Biophysica Acta (BBA)-Enzymology. 1974;364(1):140–5.
Lübbers DW, Opitz N. The pCO2-/pO2-optode: a new probe for measurement of pCO2 or pO in fluids and gases (authors transl). Zeitschrift fur Naturforschung, Biosci. 1975;30(4):532–3.
Newman JD, Turner AP. Home blood glucose biosensors: a commercial perspective. Biosens Bioelectron. 2005;20(12):2435–53.
D’Orazio P. Biosensors in clinical chemistry. Clinica chimica acta. 2003;334(1–2):41–69.
Suzuki S, Takahashi F, Satoh I, Sonobe N. Ethanol and lactic acid sensors using electrodes coated with dehydrogenase—Collagen membranes. Bullet Chem Soc Japan. 1975;48(11):3246–9.
Clemens, A.H., Chang, P.H. and Myers, R.W., 1976. Le développement d’un système automatique d’infusion d’insuline controle par la glycemie, son système de dosage du glucose et ses algorithmes de controle. Journ. Annu. Diabétol. Hotel Dieu, pp.269–278.
Yoo EH, Lee SY. Glucose biosensors: an overview of use in clinical practice. Sensors. 2010;10(5):4558–76.
Schultz, J.S. 1982 Optical Sensor of Plasma Constituents. U.S. Patent No. 4,344,438A
Liedberg B, Nylander C, Lunström I. Surface plasmon resonance for gas detection and biosensing. Sens Actuat. 1983;4:299–304.
Roederer JE, Bastiaans GJ. Microgravimetric immunoassay with piezoelectric crystals. Analytic Chem. 1983;55(14):2333–6.
Cass AE, Davis G, Francis GD, Hill HAO, Aston WJ, Higgins IJ, Plotkin EV, Scott LD, Turner AP. Ferrocene-mediated enzyme electrode for amperometric determination of glucose. Analyt Chem. 1984;56(4):667–71.
Mun’delanji, C.V.; Tamiya, E. Nanobiosensors and nanobioanalyses: A Review. In Nanobiosensors and Nanobioanalyses, 1st ed.; Mun’delanji, C.V., Kerman, K., Hsing, I.M., Tamiya, E., Eds.; Springer: Tokyo, Japan, 2015; pp. 3–20.
Poncharal P, Wang ZL, Ugarte D, De Heer WA. Electrostatic deflections and electromechanical resonances of carbon nanotubes. Science. 1999;283(5407):1513–6.
Serra, P.A. 2011. Biosensors for health, environment and biosecurity. BoD–Books on Demand. https://doi.org/10.5772/928
Nayak V, Singh KR, Verma R, Pandey MD, Singh J, Singh RP. Recent advancements of biogenic iron nanoparticles in cancer theranostics. Mater Lett. 2022;313:131769.
Chaturvedi VK, Sharma B, Tripathi AD, Yadav DP, Singh KR, Singh J, Singh RP. Biosynthesized nanoparticles: a novel approach for cancer therapeutics. Front Med Technol. 2023.
Augustine S, Singh J, Srivastava M, Sharma M, Das A, Malhotra BD. Recent advances in carbon based nanosystems for cancer theranostics. Biomater Sci. 2017;5(5):901–52.
Nisar S, Chansi Mathur A, Basu T, Singh KR, Singh J. Template free anisotropically grown gold nanocluster based electrochemical immunosensor for ultralow detection of cardiac troponin I. Biosensors. 2022;12(12):1144.
Pundir CS, Chauhan N. Acetylcholinesterase inhibition-based biosensors for pesticide determination: a review. Analyt Biochem. 2012;429(1):19–31.
Ogi H. Wireless-electrodeless quartz-crystal-microbalance biosensors for studying interactions among biomolecules: a review. Proc Jan Acad Ser B. 2013;89(9):401–17.
Ko PJ, Ishikawa R, Sohn H, Sandhu A. Porous silicon platform for optical detection of functionalized magnetic particles biosensing. J Nanosci Nanotechnol. 2013;13(4):2451–60.
Vigneshvar S, Senthilkumaran B. Current technological trends in biosensors, nanoparticle devices and biolabels: Hi-tech network sensing applications. Med Dev Sens. 2018;1(2):e10011.
Schneider E, Clark DS. Cytochrome P450 (CYP) enzymes and the development of CYP biosensors. Biosens Bioelectron. 2013;39(1):1–13.
Khimji I, Kelly EY, Helwa Y, Hoang M, Liu J. Visual optical biosensors based on DNA-functionalized polyacrylamide hydrogels. Methods. 2013;64(3):292–8.
Peng F, Su Y, Zhong Y, Fan C, Lee ST, He Y. Silicon nanomaterials platform for bioimaging, biosensing, and cancer therapy. Accounts Chem Res. 2014;47(2):612–23.
Wang B, Anzai JI. Recent progress in electrochemical HbA1c sensors: a review. Materials. 2015;8(3):1187–203.
Randriamampita C, Lellouch AC. Imaging early signaling events in T lymphocytes with fluorescent biosensors. Biotechnol J. 2014;9(2):203–12.
Oldach L, Zhang J. Genetically encoded fluorescent biosensors for live-cell visualization of protein phosphorylation. Chem Biol. 2014;21(2):186–97.
Kunzelmann, S., Solscheid, C. and Webb, M.R., 2014. Fluorescent biosensors: design and application to motor proteins. Fluorescent Methods for Molecular Motors. Springer Basel. Basel. pp. 25–47
Wang S, Poon GM, Wilson WD. Quantitative investigation of protein-nucleic acid interactions by biosensor surface plasmon resonance. Methods Mol Biol. 2015;1334:313–32.
Garcia-Gomez NA, Balderas-Renteria I, Garcia-Gutierrez DI, Mosqueda HA, Sánchez EM. Development of mats composed by TiO2 and carbon dual electrospun nanofibers: a possible anode material in microbial fuel cells. Mater Sci Eng: B. 2015;193:130–6.
Gribi S, Bois Du, de Dunilac S, Ghezzi D, Lacour SP. A microfabricated nerve-on-a-chip platform for rapid assessment of neural conduction in explanted peripheral nerve fibers. Nat Communicat. 2018;9(1):1–10.
Kim J, Campbell AS, de Ávila BEF, Wang J. Wearable biosensors for healthcare monitoring. Nat Biotechnol. 2019;37(4):389–406.
Saylan Y, Erdem Ö, Ünal S, Denizli A. An alternative medical diagnosis method: biosensors for virus detection. Biosensors. 2019;9(2):65.
Jiang Z, Feng B, Xu J, Qing T, Zhang P, Qing Z. Graphene biosensors for bacterial and viral pathogens. Biosens Bioelectron. 2020;166:112471.
Inda ME, Lu TK. Microbes as biosensors. Annual Rev Microbiol. 2020;74:337–59.
Jain S, Nehra M, Kumar R, Dilbaghi N, Hu T, Kumar S, Kaushik A, Li CZ. Internet of medical things (IoMT)-integrated biosensors for point-of-care testing of infectious diseases. Biosens Bioelectron. 2021;179:113074.
Karimi-Maleh H, Orooji Y, Karimi F, Alizadeh M, Baghayeri M, Rouhi J, Tajik S, Beitollahi H, Agarwal S, Gupta VK, Rajendran S. A critical review on the use of potentiometric based biosensors for biomarkers detection. Biosens Bioelectron. 2021;184:113252.
Alwarappan S, Nesakumar N, Sun D, Hu TY, Li CZ. 2D metal carbides and nitrides (MXenes) for sensors and biosensors. Biosens Bioelectron. 2022.
Kaur B, Kumar S, Kaushik BK. Recent advancements in optical biosensors for cancer detection. Biosens Bioelectron. 2022;197:113805.
Fu X, Ding B, D’Alessandro D. Fabrication strategies for metal-organic framework electrochemical biosensors and their applications. Coordinat Chem Rev. 2023;475:214814.
Galyamin D, Liébana S, Esquivel JP, Sabaté N. Immuno-battery: a single use self-powered immunosensor for reassured diagnostics. Biosens Bioelectron. 2023;220:114868.
Naresh V, Lee N. A review on biosensors and recent development of nanostructured materials-enabled biosensors. Sensors. 2021;21(4):1109.
Coccia M, Roshani S, Mosleh M. Scientific developments and new technological trajectories in sensor research. Sensors. 2021;21(23):7803.
Khan MA, Mujahid M. Recent advances in electrochemical and optical biosensors designed for detection of Interleukin 6. Sensors. 2020;20(3):646.
Damborský P, Švitel J, Katrlík J. Optical biosensors. Essays Biochem. 2016;60(1):91–100.
Kurbanoglu S, et al. Chemical nanosensors in pharmaceutical analysis. In: New developments in nanosensors for pharmaceutical analysis. Amsterdam: Elsevier; 2019. p. 141–70.
Qian L, Li Q, Baryeh K, Qiu W, Li K, Zhang J, Yu Q, Xu D, Liu W, Brand RE, Zhang X. Biosensors for early diagnosis of pancreatic cancer: a review. Trans Res. 2019;213:67–89.
Chang J, Wang X, Wang J, Li H, Li F. Nucleic acid-functionalized metal-organic framework-based homogeneous electrochemical biosensor for simultaneous detection of multiple tumor biomarkers. Anal Chem. 2019;91:3604–10.
Zhang X, Xie G, Gou D, Luo P, Yao Y, Chen H. A novel enzyme-free electrochemical biosensor for rapid detection of Pseudomonas aeruginosa based on high catalytic Cu-ZrMOF and conductive Super P. Biosens Bioelectron. 2019;142:111486.
Zhang X, Yu Y, Shen J, Qi W, Wang H. Design of organic/inorganic nanocomposites for ultrasensitive electrochemical detection of a cancer biomarker protein. Talanta. 2020;212:120794.
Khanmohammadi A, Aghaie A, Vahedi E, Qazvini A, Ghanei M, Afkhami A, Hajian A, Bagheri H. Electrochemical biosensors for the detection of lung cancer biomarkers: a review. Talanta. 2020;206:120251.
Vijayan VM, Jothi L, Arunagirinathan RS, Nageswaran G. Recent advances in the electrochemical sensing of lung cancer biomarkers. Biosens Bioelectron: X. 2022;12:100235.
da Silva ET, Souto DE, Barragan JT, De Giarola J, De Moraes AC, Kubota LT. Electrochemical biosensors in point-of-care devices: recent advances and future trends. Chemelectrochem. 2017;4(4):778–94.
Sandhyarani N. Surface modification methods for electrochemical biosensors. In: Electrochemical biosensors. Amsterdam: Elsevier; 2019. p. 45–75.
Scholz F. Voltammetric techniques of analysis: the essentials. ChemTexts. 2015;1(4):17.
Anik Ü. Electrochemical medical biosensors for POC applications. In: Medical biosensors for point of care (POC) applications. Amsterdam: Elsevier; 2017. p. 275–92.
Luo Y, Wang Y, Yan H, Wu Y, Zhu C, Du D, Lin Y. SWCNTs@GQDs composites as nanocarriers for enzyme-free dual-signal amplification electrochemical immunoassay of cancer biomarker. Anal Chim Acta. 2018;1042:44–51.
Paimard G, et al. An Impedimetric Immunosensor modified with electrospun core-shell nanofibers for determination of the carcinoma embryonic antigen. Sens Actuat B Chem. 2020;311:127928.
Butmee P, et al. An ultrasensitive immunosensor based on manganese dioxide-graphene nanoplatelets and core shell Fe3O4@ Au nanoparticles for label-free detection of carcinoembryonic antigen. Bioelectrochemistry. 2020;132:107452.
Ehzari H, Amiri M, Safari M. Enzyme-free sandwich-type electrochemical immunosensor for highly sensitive prostate specific antigen based on conjugation of quantum dots and antibody on surface of modified glassy carbon electrode with core–shell magnetic metal-organic frameworks. Talanta. 2020;210:120641.
Heidari R, Rashidiani J, Abkar M, Taheri RA, Moghaddam MM, Mirhosseini SA, Seidmoradi R, Nourani MR, Mahboobi M, Keihan AH, Kooshki H. CdS nanocrystals/graphene oxide-AuNPs based electrochemiluminescence immunosensor in sensitive quantification of a cancer biomarker: p53. Biosens Bioelectron. 2019;126:7–14.
Jeong S, Barman SC, Yoon H, Park JY. A Prostate cancer detection immunosensor based on nafion/reduced graphene oxide/aldehyde functionalized methyl pyridine composite electrode. J Electrochem Soc. 2019;166:B920.
Ibau C, Arshad MK, Gopinath SCB, Nuzaihan Fathil MMF, Estrela P. Gold interdigitated triple-microelectrodes for label-free prognosticative aptasensing of prostate cancer biomarker in serum. Biosens Bioelectron. 2019;136:118.
Sharifuzzaman M, Barman SC, Rahman MT, Zahed MA, Xuan X, Park JY. Green synthesis and layer-by-layer assembly of amino-functionalized graphene oxide/carboxylic surface modified trimetallic nanoparticles nanocomposite for label-free electrochemical biosensing. J Electrochem Soc. 2019;166:B983.
Jafari-Kashi A, Rafiee-Pour HA, Shabani-Nooshabadi M. A new strategy to design label-free electrochemical biosensor for ultrasensitive diagnosis of CYFRA 21–1 as a biomarker for detection of non-small cell lung cancer. Chemosphere. 2022;301:134636.
Chen M, Wang Y, Su H, Mao L, Jiang X, Zhang T, Dai X. Three-dimensional electrochemical DNA biosensor based on 3D graphene-Ag nanoparticles for sensitive detection of CYFRA21-1 in non-small cell lung cancer. Sens Actuat B: Chem. 2018;255:2910–8.
Zeng Y, Bao J, Zhao Y, Huo D, Chen M, Yang M, Fa H, Hou C. A sensitive label-free electrochemical immunosensor for detection of cytokeratin 19 fragment antigen 21–1 based on 3D graphene with gold nanoparticle modified electrode. Talanta. 2018;178:122–8.
Shi P, et al. Non-covalent modification of glassy carbon electrode with isoorientin and application to alpha-fetoprotein detection by fabricating an immunosensor. Sens Actuat B Chem. 2020;305:127494.
Zhang X, Li Y, Lv H, Gao Z, Zhang C, Zhang S, Wang Y, Xu Z, Zhao Z. Electrochemical immunosensor with enhanced stability for sensitive detection of α-fetoprotein based on Pd@Ag@CeO2 as signal amplification label. J Electrochem Soc. 2018;165:B931.
Zhang X, et al. Sandwich-type electrochemical immunosensor based on Au@ Ag supported on functionalized phenolic resin microporous carbon spheres for ultrasensitive analysis of α-fetoprotein. Biosens Bioelectron. 2018;106:142–8.
Ma E, et al. Electrochemical immunosensors for sensitive detection of neuron-specific enolase based on small size trimetallic Au@ Pd^Pt nanocubes functionalized on ultrathin MnO2 nanosheets as signal labels. ACS Biomater Sci Eng. 2020;6(3):1418–27.
Zhao L, Kong D, Wu Z, Liu G, Gao Y, Yan X, Liu F, Liu X, Wang C, Cui J, Lu G. Interface interaction of MoS2 nanosheets with DNA based aptameric biosensor for carbohydrate antigen 15–3 detection. Microchem J. 2020;155:104675.
Khoshroo A, Mazloum-Ardakani M, Forat-Yazdi M. Enhanced performance of labelfree electrochemical immunosensor for carbohydrate antigen 15–3 based on catalytic activity of cobalt sulfide/graphene nanocomposite. Sens Actuat B Chem. 2018;255:580–7.
Senel M, Dervisevic M, Kokkokoğlu F. Electrochemical DNA biosensors for label-free breast cancer gene marker detection. Analyt Bioanalytic Chem. 2019;411(13):2925–35.
Saadati A, et al. A novel biosensor for the monitoring of ovarian cancer tumor protein CA 125 in untreated human plasma samples using a novel nano-ink: a new platform for efficient diagnosis of cancer using paper based microfluidic technology. Anal Methods. 2020;12(12):1639–49.
Chauhan D, Nohwal B, Pundir C. An electrochemical CD59 targeted noninvasive immunosensor based on graphene oxide nanoparticles embodied pencil graphite for detection of lung cancer. Microchem J. 2020;156:104957.
Jalil O, Pandey CM, Kumar D. Electrochemical biosensor for the epithelial cancer biomarker EpCAM based on reduced graphene oxide modified with nanostructured titanium dioxide. Microchim Acta. 2020;187:1–9.
Li X, Peng G, Cui F, Qiu Q, Chen X, Huang H. Double determination of long noncoding RNAs from lung cancer via multi-amplified electrochemical genosensor at sub-femtomole level. Biosens Bioelectron. 2018;113:116–23.
Jayanthi VSPKSA, Das AB, Saxena U. Fabrication of an immunosensor for quantitative detection of breast cancer biomarker UBE2C. RSC Adv. 2019;9:16738.
Xu W, et al. A signal-decreased electrochemical immunosensor for the sensitive detection of LAG-3 protein based on a hollow nanobox-MOFs/AuPt alloy. Biosens Bioelectron. 2018;113:148–56.
Kuntamung K, Jakmunee J, Ounnunkad K. A label-free multiplex electrochemical biosensor for the detection of three breast cancer biomarker proteins employing dye/metal ion-loaded and antibody-conjugated polyethyleneimine-gold nanoparticles. J Mater Chem B. 2021;9(33):6576–85.
Shoja Y, Kermanpur A, Karimzadeh F. Diagnosis of EGFR exon21 L858R point mutation as lung cancer biomarker by electrochemical DNA biosensor based on reduced graphene oxide/functionalized ordered mesoporous carbon/Ni-oxytetracycline metallopolymer nanoparticles modified pencil graphite electrode. Biosens Bioelectron. 2018;113:108–15.
Amani J, et al. An electrochemical immunosensor based on poly p-phenylenediamine and graphene nanocomposite for detection of neuron-specific enolase via electrochemically amplified detection. Anal Biochem. 2018;548:53–9.
Khodadoust A, Nasirizadeh N, Seyfati SM, Taheri RA, Ghanei M, Bagheri H. High-performance strategy for the construction of electrochemical biosensor for simultaneous detection of miRNA-141 and miRNA-21 as lung cancer biomarkers. Talanta. 2023;252:123863.
Sabahi A, Salahandish R, Ghaffarinejad A, Omidinia E. Electrochemical nano-genosensor for highly sensitive detection of miR-21 biomarker based on SWCNT-grafted dendritic Au nanostructure for early detection of prostate cancer. Talanta. 2020;209:120595.
Hasanzadeh M, Shadjou N, de la Guardia M. Early-stage screening of breast cancer using electrochemical biomarker detection. TrAC - Trends Anal Chem. 2017;91:67.
Raji MA, Amoabediny G, Tajik P, Hosseini M, Ghafar-Zadeh E. An aptabiosensor for colon cancer diagnostics. Sensors (Switzerland). 2015;15:22291.
Zeng Y, Bao J, Zhao Y, Huo D, Chen M, Qi Y, Yang M, Fa H, Hou C. A sandwich-type electrochemical immunoassay for ultrasensitive detection of non-small cell lung cancer biomarker CYFRA21-1. Bioelectrochemistry. 2018;120:183–9.
Freitas M, Nouws HPA, Keating E, Delerue-Matos C. High-performance electrochemical immunomagnetic assay for breast cancer analysis. Sens Actuat B Chem. 2020;308:127667.
Boriachek K, Umer M, Islam MN, Gopalan V, Lam AK, Nguyen NT, Shiddiky MJA. An amplification-free electrochemical detection of exosomal miRNA-21 in serum samples. Analyst. 2018;143:1662–9.
Pacheco JG, Rebelo P, Freitas M, Nouws HPA, Delerue-Matos C. Breast cancer biomarker (HER2-ECD) detection using a molecularly imprinted electrochemical sensor. Sens Actuat B Chem. 2018;273:1008–14.
Pacheco JG, Silva MSV, Freitas M, Nouws HPA, Delerue-Matos C. Molecularly imprinted electrochemical sensor for the point-of-care detection of a breast cancer biomarker (CA 15–3). Sensor Actuator B Chem. 2018;256:905–12.
An L, Wang G, Han Y, Li T, Jin P, Liu S. Electrochemical biosensor for cancer cell detection based on a surface 3D micro-array. Lab Chip. 2018;18:335.
Zandi A, Gilani A, Abbasvandi F, Katebi P, Tafti SR, Assadi S, Moghtaderi H, Parizi MS, Saghafi M, Khayamian MA, Davarish Z, Hoseinpour P, Gity M, Sanati H, Abdolahad M. Carbon nanotube based dielectric spectroscopy of tumor secretion; electrochemical lipidomics for cancer diagnosis. Biosens Bioelectron. 2019;142:111566.
Dowling CM, Herranz Ors C, Kiely PA. Using real-time impedance-based assays to monitor the effects of fibroblast-derived media on the adhesion, proliferation, migration and invasion of colon cancer cells. Biosci Rep. 2014;34:415.
Parekh A, Das D, Das S, Dhara S, Biswas K, Mandal M, Das S. Bioimpedimetric analysis in conjunction with growth dynamics to differentiate aggressiveness of cancer cells. Sci Rep. 2018;8:1.
Xiao F, Wang L, Duan H. Nanomaterial based electrochemical sensors for in vitro detection of small molecule metabolites. Biotechnol Adv. 2016;34(3):234–49.
Cardoso AR, et al. Novel and simple electrochemical biosensor monitoring attomolar levels of miRNA-155 in breast cancer. Biosens Bioelectron. 2016;80:621–30.
Nayak V, Patra S, Singh KR, Ganguly B, Kumar DN, Panda D, Maurya GK, Singh J, Majhi S, Sharma R, Pandey SS. Advancement in precision diagnosis and therapeutic for triple-negative breast cancer: Harnessing diagnostic potential of CRISPR-cas & engineered CAR T-cells mediated therapeutics. Environ Res. 2023;235:116573.
Luo L, Wang L, Zeng L, Wang Y, Weng Y, Liao Y, Chen T, Xia Y, Zhang J, Chen J. A ratiometric electrochemical DNA biosensor for detection of exosomal MicroRNA. Talanta. 2020;207:120298.
Zouari M, Campuzano S, Pingarrón JM, Raouafi N. Amperometric biosensing of miRNA-21 in serum and cancer cells at nanostructured platforms using anti-DNA-RNA hybrid antibodies. ACS Omega. 2018;3(8):8923–31.
Deswal R, Narwal V, Kumar P, Verma V, Dang AS, Pundir CS. An improved amperometric sarcosine biosensor based on graphene nanoribbon/chitosan nanocomposite for detection of prostate cancer. Sensors Int. 2022;3:100174.
Acknowledgements
The authors would like to thank the Department of Chemistry, Faculty of Science, University of Tabuk, Saudi Arabia, for their ardent encouragement of their current project.
Funding
This research received no external funding (self-funded).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declares no conflict of interest in this study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mustafa, S.K., Khan, M.F., Sagheer, M. et al. Advancements in biosensors for cancer detection: revolutionizing diagnostics. Med Oncol 41, 73 (2024). https://doi.org/10.1007/s12032-023-02297-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12032-023-02297-y