Abstract
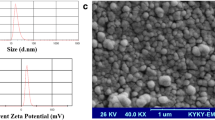
Induction of immunogenic cell death (ICD) is a promising strategy for cancer immunotherapy. Chrysin, which has potential anticancer effects, faces limitations in clinical applications due to its poor water solubility. This study aimed to formulate chrysin with PEG-poly(α-benzylcarboxylate-ε-caprolactone) (PBCL) nanoparticles (NPs) and assess their anticancer and ICD-inducing potency in melanoma cells, comparing with free chrysin. The co-solvent evaporation method was employed to develop chrysin-loaded NPs. UV spectroscopy, dynamic light scattering, and the dialysis bag method were used to evaluate the encapsulation efficiency (EE), particle size, polydispersity index (PDI), and drug release profile, respectively. The anticancer effects of the drugs were assessed using the MTT and trypan blue exclusion assays. Flow cytometry was employed to evaluate apoptosis and calreticulin (CRT) expression. ELISA and western blotting were used to detect heat shock protein 90 (HSP90), Annexin A1, GRP78 (Glucose-related protein78), and activated protein kinase R-like endoplasmic reticulum kinase (p-PERK). Chrysin-loaded PEG-PBCL NPs (chrysin-PEG-PBCL) showed an EE of 97 ± 1%. Chrysin-PEG-PBCL was 38.18 ± 3.96 nm in size, with a PDI being 0.62 ± 0.23. Chrysin-PEG-PBCL showed an initial burst release, followed by sustained release over 24 h. Chrysin-PEG-PBCL exhibited a significantly stronger anticancer effect in B16 cells. Chrysin-PEG-PBCL was found to be more potent in inducing apoptosis. Both free chrysin and chrysin NPs induced ICD as indicated by an increase in the levels of ICD biomarkers. Interestingly, chrysin NPs were found to be more potent inducers of ICD than the free drug. These findings demonstrate that chrysin and chrysin-PEG-PBCL NPs can induce ICD in B16 cells. PEG-PBCL NPs significantly enhanced the potency of chrysin in inducing ICD compared to its free form.






Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author upon reasonable request.
References
Senga SS, Grose RP. Hallmarks of cancer—the new testament. Open Biol. 2021;11(1):200358.
Jafari S, Heydarian S, Lai R, Aghdam EM, Molavi O. Silibinin induces immunogenic cell death in cancer cells and enhances the induced immunogenicity by chemotherapy. Bioimpacts. 2023;13(1):51.
Krysko O, Løve Aaes T, Bachert C, Vandenabeele P, Krysko DV. Many faces of DAMPs in cancer therapy. Cell Death Dis. 2013;4(5):e631. https://doi.org/10.1038/cddis.2013.156.
Pitt JM, Kroemer G, Zitvogel L. Immunogenic and non-immunogenic cell death in the tumor microenvironment. In: Kalinski P, editor. Tumor immune microenvironment in cancer progression and cancer therapy. Cham: Springer; 2017. p. 65–79.
Kroemer G, Galluzzi L, Kepp O, Zitvogel L. Immunogenic cell death in cancer therapy. Annu Rev Immunol. 2013;31:51–72. https://doi.org/10.1146/annurev-immunol-032712-100008.
Yatim N, Cullen S, Albert ML. Dying cells actively regulate adaptive immune responses. Nat Rev Immunol. 2017;17(4):262–75. https://doi.org/10.1038/nri.2017.9.
Casares N, Pequignot MO, Tesniere A, Ghiringhelli F, Roux S, Chaput N, et al. Caspase-dependent immunogenicity of doxorubicin-induced tumor cell death. J Exp Med. 2005;202(12):1691–701. https://doi.org/10.1084/jem.20050915.
Menger L, Vacchelli E, Adjemian S, Martins I, Ma Y, Shen S, et al. Cardiac glycosides exert anticancer effects by inducing immunogenic cell death. Sci Transl Med. 2012;4(143):143ra99. https://doi.org/10.1126/scitranslmed.3003807.
Golden EB, Frances D, Pellicciotta I, Demaria S, Helen Barcellos-Hoff M, Formenti SC. Radiation fosters dose-dependent and chemotherapy-induced immunogenic cell death. Oncoimmunology. 2014;3:e28518. https://doi.org/10.4161/onci.28518.
Kepp O, Menger L, Vacchelli E, Locher C, Adjemian S, Yamazaki T, et al. Crosstalk between ER stress and immunogenic cell death. Cytokine Growth Factor Rev. 2013;24(4):311–8.
Yang Y, Li X-J, Chen Z, Zhu X-X, Wang J, Zhang L, et al. Wogonin induced calreticulin/annexin A1 exposure dictates the immunogenicity of cancer cells in a PERK/AKT dependent manner. PLoS ONE. 2012;7(12):e50811. https://doi.org/10.1371/journal.pone.0050811.
Jafari S, Dabiri S, Mehdizadeh Aghdam E, Fathi E, Saeedi N, Montazersaheb S, et al. Synergistic effect of chrysin and radiotherapy against triple-negative breast cancer (TNBC) cell lines. Clin Transl Oncol. 2023;25:2599–2568.
Naz S, Imran M, Rauf A, Orhan IE, Shariati MA, Shahbaz M, et al. Chrysin: Pharmacological and therapeutic properties. Life Sci. 2019;235:116797.
Khaledi S, Jafari S, Hamidi S, Molavi O, Davaran S. Preparation and characterization of PLGA-PEG-PLGA polymeric nanoparticles for co-delivery of 5-Fluorouracil and Chrysin. J Biomater Sci Polym Ed. 2020;31(9):1107–26.
Saadat M, Jafari S, Zakeri-Milani P, Shahbazi-Mojarrad J, Valizadeh H. Stearoylcholine and oleoylcholine: synthesis, physico-chemical characterization, nanoparticle formation, and toxicity studies. J Drug Deliv Sci Technol. 2020;59:101872.
Jafari S, Bakhshaei A, Eskandani M, Molavi O. Silibinin-loaded nanostructured lipid carriers for growth inhibition of cisplatin-resistant ovarian cancer cells. Assay Drug Dev Technol. 2022;20(8):339–48.
Banstola A, Poudel K, Kim JO, Jeong J-H, Yook S. Recent progress in stimuli-responsive nanosystems for inducing immunogenic cell death. J Control Release. 2021;337:505–20.
Masoumzadeh H, Hoseinzad N, Jafari S, Shayanfar A, Vaez H, Molavi O. Development of polymeric micelles loaded with STAT3 inhibitory, stattic, for cancer treatment. Med J Tabriz Univ Med Sci. 2021;43(4):335–45.
Zheng H, Li S, Pu Y, Lai Y, He B, Gu Z. Nanoparticles generated by PEG-Chrysin conjugates for efficient anticancer drug delivery. Eur J Pharm Biopharm. 2014;87(3):454–60. https://doi.org/10.1016/j.ejpb.2014.03.011.
Sulaiman GM, Jabir MS, Hameed AH. Nanoscale modification of chrysin for improved of therapeutic efficiency and cytotoxicity. Artif Cells Nanomed Biotechnol. 2018;46(sup1):708–20. https://doi.org/10.1080/21691401.2018.1434661.
Ragab EM, El Gamal DM, Mohamed TM, Khamis AA. Therapeutic potential of chrysin nanoparticle-mediation inhibition of succinate dehydrogenase and ubiquinone oxidoreductase in pancreatic and lung adenocarcinoma. Eur J Med Res. 2022;27(1):172. https://doi.org/10.1186/s40001-022-00803-y.
Soleimani AH, Garg SM, Paiva IM, Vakili MR, Alshareef A, Huang Y-H, et al. Micellar nano-carriers for the delivery of STAT3 dimerization inhibitors to melanoma. Drug Deliv Transl Res. 2017;7:571–81.
Farjami A, Siahi-Shadbad M, Akbarzadehlaleh P, Roshanzamir K, Molavi O. Evaluation of the physicochemical and biological stability of cetuximab under various stress condition. J Pharm Pharm Sci. 2019;22:171–90.
Farjami A, Akbarzadehlaleh P, Molavi O, Siahi-Shadbad M. Stability-indicating size exclusion chromatography method for the analysis of IgG mAb-cetuximab. Chromatographia. 2019;82:767–76.
Molavi O, Torkzaban F, Jafari S, Asnaashari S, Asgharian P. Chemical compositions and anti-proliferative activity of the aerial parts and rhizomes of squirting cucumber, Cucurbitaceae. Jundishapur J Nat Pharm Prod. 2020. https://doi.org/10.5812/jjnpp.82990.
Montazersaheb S, Kabiri F, Saliani N, Nourazarian A, Avci ÇB, Rahbarghazi R, et al. Prolonged incubation with Metformin decreased angiogenic potential in human bone marrow mesenchymal stem cells. Biomed Pharmacother. 2018;108:1328–37.
Valipour B, Abedelahi A, Naderali E, Velaei K, Movassaghpour A, Talebi M, et al. Cord blood stem cell derived CD16+ NK cells eradicated acute lymphoblastic leukemia cells using with anti-CD47 antibody. Life Sci. 2020;242:117223.
Montazersaheb S, Avci ÇB, Bagca BG, Ay NPO, Tarhriz V, Nielsen PE, et al. Targeting TdT gene expression in Molt-4 cells by PNA-octaarginine conjugates. Int J Biol Macromol. 2020;164:4583–90.
Rahimi M, Bagheri A, Bagheri Y, Fathi E, Bagheri S, Nia A, et al. Renoprotective effects of prazosin on ischemia-reperfusion injury in rats. Hum Exp Toxicol. 2021;40(8):1263–73.
Adangale SC, Wairkar S. Potential therapeutic activities and novel delivery systems of chrysin-a nature’s boon. Food Biosci. 2022;45:101316.
Fang D-L, Chen Y, Xu B, Ren K, He Z-Y, He L-L, et al. Development of lipid-shell and polymer core nanoparticles with water-soluble salidroside for anti-cancer therapy. Int J Mol Sci. 2014;15(3):3373–88.
Muzzio M, Li J, Yin Z, Delahunty IM, Xie J, Sun S. Monodisperse nanoparticles for catalysis and nanomedicine. Nanoscale. 2019;11(41):18946–67.
Firouzi-Amandi A, Dadashpour M, Nouri M, Zarghami N, Serati-Nouri H, Jafari-Gharabaghlou D, et al. Chrysin-nanoencapsulated PLGA-PEG for macrophage repolarization: possible application in tissue regeneration. Biomed Pharmacother. 2018;105:773–80.
El-Hussien D, El-Zaafarany GM, Nasr M, Sammour O. Chrysin nanocapsules with dual anti-glycemic and anti-hyperlipidemic effects: chemometric optimization, physicochemical characterization and pharmacodynamic assessment. Int J Pharm. 2021;592:120044.
Yoo J-W, Chambers E, Mitragotri S. Factors that control the circulation time of nanoparticles in blood: challenges, solutions and future prospects. Curr Pharm Des. 2010;16(21):2298–307.
Rajani C, Borisa P, Karanwad T, Borade Y, Patel V, Rajpoot K et al. Cancer-targeted chemotherapy: emerging role of the folate anchored dendrimer as drug delivery nanocarrier. In: Chauhan A, Kulhari H, editors. Pharmaceutical applications of dendrimers. Amsterdam: Elsevier; 2020. p. 151–98.
Lungare S, Hallam K, Badhan RK. Phytochemical-loaded mesoporous silica nanoparticles for nose-to-brain olfactory drug delivery. Int J Pharm. 2016;513(1–2):280–93.
Mehnath S, Arjama M, Rajan M, Annamalai G, Jeyaraj M. Co-encapsulation of dual drug loaded in MLNPs: implication on sustained drug release and effectively inducing apoptosis in oral carcinoma cells. Biomed Pharmacother. 2018;104:661–71.
Rad AH, Asiaee F, Jafari S, Shayanfar A, Lavasanifar A, Molavi O. Poly(ethylene glycol)-poly(ε-caprolactone)-based micelles for solubilization and tumor-targeted delivery of silibinin. Bioimpacts. 2020;10(2):87.
Mohammadinejad S, Akbarzadeh A, Rahmati-Yamchi M, Hatam S, Kachalaki S, Zohreh S, Zarghami N. Preparation and evaluation of chrysin encapsulated in PLGA-PEG nanoparticles in the T47-D breast cancer cell line. Asian Pac. J. Cancer Prev. 2015;16(9):3753–8.
Zhang Y, Zhao J, Afzal O, Kazmi I, Al-Abbasi FA, Altamimi AS, et al. Neuroprotective role of chrysin-loaded poly (lactic-co-glycolic acid) nanoparticle against kindling-induced epilepsy through Nrf2/ARE/HO-1 pathway. J Biochem Mol Toxicol. 2021;35(2):e22634.
Siddhardha B, Pandey U, Kaviyarasu K, Pala R, Syed A, Bahkali AH, et al. Chrysin-loaded chitosan nanoparticles potentiates antibiofilm activity against Staphylococcus aureus. Pathogens. 2020;9(2):115.
Khoo BY, Chua SL, Balaram P. Apoptotic effects of chrysin in human cancer cell lines. Int J Mol Sci. 2010;11(5):2188–99.
Pichichero E, Cicconi R, Mattei M, Canini A. Chrysin-induced apoptosis is mediated through p38 and Bax activation in B16–F1 and A375 melanoma cells. Int J Oncol. 2011;38(2):473–83.
Jafari S, Lavasanifar A, Hejazi MS, Maleki-Dizaji N, Mesgari M, Molavi O. STAT3 inhibitory stattic enhances immunogenic cell death induced by chemotherapy in cancer cells. DARU J Pharm Sci. 2020;28:159–69.
Panaretakis T, Kepp O, Brockmeier U, Tesniere A, Bjorklund AC, Chapman DC, et al. Mechanisms of pre-apoptotic calreticulin exposure in immunogenic cell death. EMBO J. 2009;28(5):578–90.
Ryu S, Lim W, Bazer FW, Song G. Chrysin induces death of prostate cancer cells by inducing ROS and ER stress. J Cell Physiol. 2017;232(12):3786–97. https://doi.org/10.1002/jcp.25861.
Xu Y, Tong Y, Ying J, Lei Z, Wan L, Zhu X, et al. Chrysin induces cell growth arrest, apoptosis, and ER stress and inhibits the activation of STAT3 through the generation of ROS in bladder cancer cells. Oncol Lett. 2018;15(6):9117–25.
Shimizu A, Kaira K, Yasuda M, Asao T, Ishikawa O. Clinical and pathological significance of ER stress marker (BiP/GRP78 and PERK) expression in malignant melanoma. Pathol Oncol Res. 2017;23(1):111–6. https://doi.org/10.1007/s12253-016-0099-9.
Kepp O, Bezu L, Kroemer G. The endoplasmic reticulum chaperone BiP: a target for immunogenic cell death inducers? OncoImmunology. 2022;11:2092328.
Martin S, Hill DS, Paton JC, Paton AW, Birch-Machin MA, Lovat PE, et al. Targeting GRP78 to enhance melanoma cell death. Pigment Cell Melanoma Res. 2010;23(5):675–82.
Acknowledgements
Not applicable.
Funding
This work was financially supported by the faculty of pharmacy, Tabriz University of Medical Sciences, Tabriz, Iran (Pazhoohan ID: 69585).
Author information
Authors and Affiliations
Contributions
YO and SHD performed the experiments and wrote the first draft of the manuscript text. OM designed the experiment and wrote the manuscript. MS.H and S.D edited the manuscript. SM and SJ designed the experiments and cooperated in data analysis, and wrote the manuscript. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
We declare that there are no conflicts of interest associated with this study.
Ethical approval
Ethical consent was approved by an ethics committee at Tabriz University of Medical Sciences, Tabriz, Iran. Ethic Code No: IR.TBZMED.VCR.REC.1401.054.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Oliyapour, Y., Dabiri, S., Molavi, O. et al. Chrysin and chrysin-loaded nanocarriers induced immunogenic cell death on B16 melanoma cells. Med Oncol 40, 278 (2023). https://doi.org/10.1007/s12032-023-02145-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12032-023-02145-z