Abstract
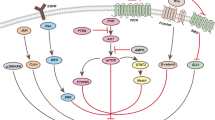
G-protein-coupled receptor (GPCR) 48, also known as leucine-rich repeat-containing G-protein-coupled receptor (LGR) 4, is an orphan receptor belonging to the GPCR superfamily, which plays an important role in the development of various organs and multiple cancers. However, the function of GPCR48/LGR4 in prostate cancer has not been fully investigated. Herein, GPCR48/LGR4 was overexpressed and silenced in prostate cancer cells via plasmid and shRNA transfection, respectively. The expression of GPCR48/LGR4 in mRNA and protein levels was analyzed using RT-qPCR and Western blotting, respectively. Subsequently, we demonstrated the effects of GPCR48/LGR4 on the migration, invasion, proliferation and apoptosis of prostate cancer cells, including Du145 and PC-3 cells. Next, we investigated the relationship between GPCR48/LGR4 and phosphatidylinositol-4,5-bisphosphate 3-kinase (PI3K)/Akt signaling pathway. The results showed that the overexpression of GPCR48/LGR4 was associated with the up-regulation of Akt, a key effector of PI3K/Akt signaling pathway, which meantime up-regulated the expression of mammalian target of rapamycin (mTOR) and glycogen synthase kinase 3β (GSK-3β), while down-regulated forkhead box, class O (FOXO), all of whom are the downstream targets of PI3K/Akt signaling pathway. Hence, the results suggested that GPCR48/LGR4 may regulate prostate cancer cells and tumor growth via the PI3K/Akt signaling pathway and could provide a better therapeutic target for the diagnosis and treatment of prostate cancer.







Similar content being viewed by others
References
Tenesa A, Dunlop MG. New insights into the aetiology of colorectal cancer from genome-wide association studies. Nat Rev Genet. 2009;10:353–8.
Narita S, Tsuchiya N, Habuchi T. Treatment for high-risk localized prostate cancer. Nihon Rinsho. 2014;72:2212–6.
Ghosh D, Yu H, Tan XF, Lim TK, Zubaidah RM, Tan HT, et al. Identification of key players for colorectal cancer metastasis by iTRAQ quantitative proteomics profiling of isogenic SW480 and SW620 cell lines. J Proteome Res. 2011;10:4373–87.
O’Neill PR, Giri L, Karunarathne WK, Patel AK, Venkatesh KV, Gautam N. The structure of dynamic GPCR signaling networks. Wiley Interdiscip Rev Syst Biol Med. 2014;6:115–23.
Zhu YB, Xu L, Chen M, Ma HN, Lou F. GPR48 promotes multiple cancer cell proliferation via activation of Wnt signaling. Asian Pac J Cancer Prev. 2013;14:4775–8.
Luo W, Rodriguez M, Valdez JM, Zhu X, Tan K, Li D, et al. Lgr4 is a key regulator of prostate development and prostate stem cell differentiation. Stem Cells. 2013;31:2492–505.
Luo J, Zhou W, Zhou X, Li D, Weng J, Yi Z, et al. Regulation of bone formation and remodeling by G-protein-coupled receptor 48. Development. 2009;136:2747–56.
Kudo M, Chen T, Nakabayashi K, Hsu SY, Hsueh AJ. The nematode leucine-rich repeat-containing, G protein-coupled receptor (LGR) protein homologous to vertebrate gonadotropin and thyrotropin receptors is constitutively active in mammalian cells. Mol Endocrinol. 2000;14:272–84.
Mendive F, Laurent P, Van Schoore G, Skarnes W, Pochet R, Vassart G. Defective postnatal development of the male reproductive tract in LGR4 knockout mice. Dev Biol. 2006;290:421–34.
Yamashita R, Takegawa Y, Sakumoto M, Nakahara M, Kawazu H, Hoshii T, et al. Defective development of the gall bladder and cystic duct in Lgr4-hypomorphic mice. Dev Dyn. 2009;238:993–1000.
Weng J, Luo J, Cheng X, Jin C, Zhou X, Qu J, et al. Deletion of G protein-coupled receptor 48 leads to ocular anterior segment dysgenesis (ASD) through down-regulation of Pitx2. Proc Natl Acad Sci USA. 2008;105:6081–6.
Daaka Y. G proteins in cancer: the prostate cancer paradigm. Sci STKE. 2004;2004:re2.
Dorsam RT, Gutkind JS. G-protein-coupled receptors and cancer. Nat Rev Cancer. 2007;7:79–94.
Rosenberg J, Small EJ. Prostate cancer update. Curr Opin Oncol. 2003;15:217–21.
Harada M, Noguchi M, Itoh K. Target molecules in specific immunotherapy against prostate cancer. Int J Clin Oncol. 2003;8:193–9.
Wu J, Xie N, Xie K, Zeng J, Cheng L, Lei Y, et al. GPR48, a poor prognostic factor, promotes tumor metastasis and activates β-catenin/TCF signaling in colorectal cancer. Carcinogenesis. 2013;34:2861–9.
Hsu SY, Liang SG, Hsueh AJ. Characterization of two LGR genes homologous to gonadotropin and thyrotropin receptors with extracellular leucine-rich repeats and a G protein-coupled, seven-transmembrane region. Mol Endocrinol. 1998;12:1830–45.
Fresno Vara JA, Casado E, de Castro J, Cejas P, Belda-Iniesta C, Gonzalez-Baron M. PI3K/Akt signalling pathway and cancer. Cancer Treat Rev. 2004;30:193–204.
Kasbohm EA, Guo R, Yowell CW, Bagchi G, Kelly P, Arora P, et al. Androgen receptor activation by G(s) signaling in prostate cancer cells. J Biol Chem. 2005;280:11583–9.
Dai J, Shen R, Sumitomo M, Stahl R, Navarro D, Gershengorn MC, et al. Synergistic activation of the androgen receptor by bombesin and low-dose androgen. Clin Cancer Res. 2002;8:2399–405.
Lee LF, Guan J, Qiu Y, Kung HJ. Neuropeptide-induced androgen independence in prostate cancer cells: roles of nonreceptor tyrosine kinases Etk/Bmx, Src, and focal adhesion kinase. Mol Cell Biol. 2001;21:8385–97.
Xie Y, Wolff DW, Lin MF, Tu Y. Vasoactive intestinal peptide transactivates the androgen receptor through a protein kinase A-dependent extracellular signal-regulated kinase pathway in prostate cancer LNCaP cells. Mol Pharmacol. 2007;72:73–85.
Nelson JB. Endothelin inhibition: novel therapy for prostate cancer. J Urol. 2003;170:S65–7 discussion S7–8.
Growcott JW. Preclinical anticancer activity of the specific endothelin A receptor antagonist ZD4054. Anticancer Drugs. 2009;20:83–8.
Takahashi H, Ishii H, Nishida N, Takemasa I, Mizushima T, Ikeda M, et al. Significance of Lgr5(+ve) cancer stem cells in the colon and rectum. Ann Surg Oncol. 2011;18:1166–74.
Fan XS, Wu HY, Yu HP, Zhou Q, Zhang YF, Huang Q. Expression of Lgr5 in human colorectal carcinogenesis and its potential correlation with β-catenin. Int J Colorectal Dis. 2010;25:583–90.
Steffen JS, Simon E, Warneke V, Balschun K, Ebert M, Rocken C. LGR4 and LGR6 are differentially expressed and of putative tumor biological significance in gastric carcinoma. Virchows Arch. 2012;461:355–65.
Gao Y, Kitagawa K, Hiramatsu Y, Kikuchi H, Isobe T, Shimada M, et al. Up-regulation of GPR48 induced by down-regulation of p27Kip1 enhances carcinoma cell invasiveness and metastasis. Cancer Res. 2006;66:11623–31.
Polivka J Jr, Janku F. Molecular targets for cancer therapy in the PI3K/AKT/mTOR pathway. Pharmacol Ther. 2014;142:164–75.
Sabbah DA, Brattain MG, Zhong H. Dual inhibitors of PI3K/mTOR or mTOR-selective inhibitors: which way shall we go? Curr Med Chem. 2011;18:5528–44.
Jiang K, Zhong B, Ritchey C, Gilvary DL, Hong-Geller E, Wei S, et al. Regulation of Akt-dependent cell survival by Syk and Rac. Blood. 2003;101:236–44.
Laplante M, Sabatini DM. mTOR signaling in growth control and disease. Cell. 2012;149:274–93.
Gupta C, Kaur J, Tikoo K. Regulation of MDA-MB-231 cell proliferation by GSK-3β involves epigenetic modifications under high glucose conditions. Exp Cell Res. 2014;324:75–83.
Acknowledgments
None.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Fang Liang and Junmin Yue have contributed equally to the work.
Rights and permissions
About this article
Cite this article
Liang, F., Yue, J., Wang, J. et al. GPCR48/LGR4 promotes tumorigenesis of prostate cancer via PI3K/Akt signaling pathway. Med Oncol 32, 49 (2015). https://doi.org/10.1007/s12032-015-0486-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12032-015-0486-1