Abstract
Purpose
Calcitonin gene-related peptide (CGRP) is a neuropeptide widely distributed in the central and peripheral nervous systems, which is known as a potent vasodilator. Postmenopausal women who experience hot flushes have high levels of plasma CGRP, suggesting its involvement in menopausal vasomotor symptoms.
Methods
In this review, we describe the biochemical aspects of CGRP and its effects associated with deficiencies of sexual hormones on skin temperature, vasodilatation, and sweating as well as the possible peripheral and central mechanisms involved in these events.
Results
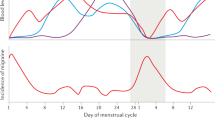
Several studies have shown that the effects of CGRP on increasing skin temperature and inducing vasodilatation are potentiated by a deficiency of sex hormones, a common condition of postmenopausal women. Additionally, the medial preoptic area of the hypothalamus, involved in thermoregulation, contains over 25-fold more CGRP-immunoreactive cells in female rodents compared with male rodents, reinforcing the role of female sex hormones on the action of CGRP. Some studies suggest that ovarian hormone deficiency decreases circulating endogenous CGRP, inducing an upregulation of CGRP receptors. Consequently, the high CGRP receptor density, especially in blood vessels, amplifies the stimulatory effects of this neuropeptide to raise skin temperature in postmenopausal women during hot flushes.
Conclusions
The duration of the perception of each hot flush in a woman is brief, while local reddening after intradermal administration of α-CGRP persists for 1 to 6 h. This contrast remains unclear.

Similar content being viewed by others
Abbreviations
- AM:
-
adrenomedullin;
- AMY:
-
amylin;
- CGRP:
-
calcitonin gene-related peptide;
- CLR:
-
calcitonin receptor-like receptor;
- CREB:
-
cAMP response element-binding;
- CTR:
-
Calcitonin receptor;
- GnRH:
-
gonadotropin-releasing hormone;
- MeSH:
-
medical subject heading;
- POA:
-
preoptic area;
- PVN:
-
hypothalamic paraventricular nucleus;
- RAMP:
-
receptor activity-modifying proteins;
References
S. Iyengar, M.H. Ossipov, K.W. Johnson, The role of calcitonin gene-related peptide in peripheral and central pain mechanisms including migraine. Pain (2017). https://doi.org/10.1097/j.pain.0000000000000831
F.A. Russell, R. King, S.J. Smillie, X. Kodji, S.D. Brain, Calcitonin gene-related peptide: physiology and pathophysiology. Physiol. Rev. (2014). https://doi.org/10.1152/physrev.00034.2013
A.J. Sorby-Adams, A.M. Marcoionni, E.R. Dempsey, J.A. Woenig, R.J. Turner, The role of neurogenic inflammation in blood-brain barrier disruption and development of cerebral oedema following acute central nervous system (CNS) injury. Int. J. Mol. Sci. (2017). https://doi.org/10.3390/ijms18081788
W.G. Lima, G.H. Marques-Oliveira, T.M. da Silva, V.E. Chaves, Role of calcitonin gene-related peptide in energy metabolism. Endocrine (2017). https://doi.org/10.1007/s12020-017-1404-4
D.L. Hay, D.R. Poyner, Calcitonin gene-related peptide, adrenomedullin and flushing. Maturitas (2009). https://doi.org/10.1016/j.maturitas.2009.08.011
S. Sharma, A. Mahajan, V.R. Tandon, Calcitonin gene-related peptide and menopause. J. Midlife. Health (2010). https://doi.org/10.4103/0976-7800.66985
D.W. Sturdee, M.S. Hunter, P.M. Maki, P. Gupta, J. Sassarini, J.C. Stevenson, M.A. Lumsden, The menopausal hot flush: a review. Climacteric (2017). https://doi.org/10.1080/13697137.2017.1306507
H.R. Morris, M. Panico, T. Etienne, J. Tippins, S.I. Girgis, I. MacIntyre, Isolation and characterization of human calcitonin gene-related peptide. Nature 308, 746–748 (1984)
P.H. Steenbergh, J.W.M. Höppener, J. Zandberg, C.J.M. Lips, H.S. Jansz, A second human calcitonin/CGRP gene. FEBS Lett. (1985). https://doi.org/10.1016/0014-5793(85)80820-6
J.W. Hoppener, P.H. Steenbergh, J. Zandberg, A.H. Geurts van Kessel, S.B. Baylin, B.D. Nelkin, H.S. Jansz, C.J. Lips, The second human calcitonin/CGRP gene is located on chromosome 11. Hum. Genet. 70, 259–263 (1985)
J.W. Hoppener, P.H. Steenbergh, J. Zandberg, E. Bakker, P.L. Pearson, A.H. Geurts van Kessel, H.S. Jansz, C.J. Lips, Localization of the polymorphic human calcitonin gene on chromosome 11. Hum. Genet. 66, 309–312 (1984)
D. Van Rossum, U.K. Hanisch, R. Quirion, Neuroanatomical localization, pharmacological characterization and functions of CGRP, related peptides and their receptors. Neurosci. Biobehav. Rev. 21, 649–678 (1997)
D.R. Poyner, P.M. Sexton, I. Marshall, D.M. Smith, R. Quirion,W. Born, R. Muff, J.A. Fischer, S.M. Foord, International Union of Pharmacology. XXXII. The mammalian calcitonin gene-related peptides, adrenomedullin, amylin, and calcitonin receptors. Pharmacol. Rev. (2002). https://doi.org/10.1124/pr.54.2.233
L.M. McLatchie, N.J. Fraser, M.J. Main, A. Wise, J. Brown, N. Thompson, R. Solari, M.G. Lee, S.M. Foord, RAMPs regulate the transport and ligand specificity of the calcitonin-receptor-like receptor. Nature 93, 333–339 (1998)
S.P. Alexander, A.P. Davenport, E. Kelly, N. Marrion, J.A. Peters, H.E. Benson, E. Faccenda, A.J. Pawson, J.L. Sharman, C. Southan, J.A. Davies, The Concise Guide to PHARMACOLOGY 2015/16: G protein-coupled receptors. Br. J. Pharmacol. (2015). https://doi.org/10.1111/bph.13348
C. Juaneda, Y. Dumont, R. Quirion, The molecular pharmacology of CGRP and related peptide receptor subtypes. Trends Pharmacol. Sci. (2000). https://doi.org/10.1016/S0165-6147(00)01555-8
Amgen Inc. Erenumab (Aimovig™): US prescribing information. https://www.accessdata.fda.gov/drugsatfdadocs/label/2018/761077s000lbl.pdf. (2018). Accessed 11 June 2018
A. Markham, Erenumab: first global approval. Drugs (2018). https://doi.org/10.1007/s40265-018-0944-0
ClinicalTrials.gov., Telcagepant for prevention of menstrually related migraine in female participants with episodic migraine (MK-0974-065). https://clinicaltrials.gov/ct2/show/NCT01125774?term=telcagepant&rank=1. (2018). Accessed 11 June 2018
M. Deen, E. Correnti, K. Kamm, T. Kelderman, L. Papetti, E. Rubio-Beltran, S. Vigneri, L. Edvinsson, A. Maassen Van Den Brink, Blocking CGRP in migraine patients - a review of pros and cons. J. Headache Pain (2017). https://doi.org/10.1186/s10194-017-0807-1
T.W. Ho, A.P. Ho, Y.J. Ge, C. Assaid, R. Gottwald, E.A. MacGregor, L.K. Mannix, W.P.J. van Oosterhout, J. Koppenhaver, C. Lines, M.D. Ferrari, D. Michelson, Randomized controlled trial of the CGRP receptor antagonist telcagepant for prevention of headache in women with perimenstrual migraine. Cephalalgia (2016). https://doi.org/10.1177/0333102415584308
D.J. Hewitt, S.K. Aurora, D.W. Dodick, P.J. Goadsby, Y.J. Ge, R. Bachman, D. Taraborelli, X. Fan, C. Assaid, C. Lines, T.W. Ho, Randomized controlled trial of the CGRP receptor antagonist MK-3207 in the acute treatment of migraine. Cephalalgia (2011). https://doi.org/10.1177/0333102411398399
H.-C. Diener, P. Barbanti, C. Dahlof, U. Reuter, J. Habeck, J. Podhorna, BI44370 TA, an oral CGRP antagonist for the treatment of acute migraine attacks: results from a phase II study. Cephalalgia (2011). https://doi.org/10.1177/0333102410388435
L. Kruger, P.W. Mantyh, C. Sternini, N.C. Brecha, C.R. Mantyh, Calcitonin gene-related peptide (CGRP) in the rat central nervous system: patterns of immunoreactivity and receptor binding sites. Brain Res. (1988). https://doi.org/10.1016/0006-8993(88)90395-2
P.M. Sexton, J.S. McKenzie, R.T. Mason, J.M. Moseley, T.J. Martin, F.A. Mendelsohn, Localization of binding sites for calcitonin gene-related peptide in rat brain by in vitro autoradiography. Neuroscience 19, 1235–1245 (1986)
L. Edvinsson, E. Nilsson, I. Jansen-Olesen, Inhibitory effect of BIBN4096BS, CGRP(8-37), a CGRP antibody and an RNA-Spiegelmer on CGRP induced vasodilatation in the perfused and non-perfused rat middle cerebral artery. Br. J. Pharmacol. (2007). https://doi.org/10.1038/sj.bjp.0707134
D.L. Hay, M.L. Garelja, D.R. Poyner, C.S. Walker, Update on the pharmacology of calcitonin/CGRP family of peptides: IUPHAR Review 25. Br. J. Pharmacol. (2018). https://doi.org/10.1111/bph.14075
H. Yoshizaki, M. Takamiya, T. Okada, Characterization of picomolar affinity binding sites for [125I]-human calcitonin gene-related peptide in rat brain and heart. Biochem. Biophys. Res. Commun. 146, 443–451 (1987)
T. Dennis, A. Fournier, A. Cadieux, F. Pomerleau, F.B. Jolicoeur, S. St Pierre, R. Quirion, hCGRP8-37, a calcitonin gene-related peptide antagonist revealing calcitonin gene-related peptide receptor heterogeneity in brain and periphery. J. Pharmacol. Exp. Ther. 254, 123–128 (1990)
H. Nakamuta, Y. Fukuda, M. Koida, N. Fujii, A. Otaka, S. Funakoskhi, H. Yajima, N. Mitsuyasu, R.C. Orlowshi, Binding sites of calcitonin gene-related peptide (CGRP): abundant occurrence in visceral organs. Jpn. J. Pharmacol. 42, 175–180 (1986)
M. Roa, J.P. Changeux, Characterization and developmental evolution of a high-affinity binding site for calcitonin gene-related peptide on chick skeletal muscle membrane. Neuroscience (1991). https://doi.org/10.1016/0306-4522(91)90349-S
C.E. Hill, D.J. Gould, J. Strigas, E. Burcher, M. Vidovic, Sensory nerves play an efferent role in the function of the arterioles, but not the dilator muscle, of the rat iris. J. Auton. Nerv. Syst. 58, 89–100 (1996)
J.C.W. Mak, P.J. Barnes, Autoradiographic localization of calcitonin gene-related peptide (CGRP) binding sites in human and guinea pig lung. Peptides 9, 957–963 (1988)
J.P. McGillis, S. Humphreys, V. Rangnekar, J. Ciallella, Modulation of B lymphocyte differentiation by calcitonin gene-related peptide (CGRP). I. Characterization of high-affinity CGRP receptors on murine 70Z/3 cells. Cell. Immunol. 150, 391–404 (1993)
A.M. Salmon, M.I. Damaj, L.M. Marubio, M.P. Epping-Jordan, E. Merlo-Pich, J.P. Changeux, Altered neuroadaptation in opiate dependence and neurogenic inflammatory nociception in alpha CGRP-deficient mice. Nat. Neurosci. (2001). https://doi.org/10.1038/86001
K. Takami, Y. Kawai, S. Uchida, M. Tohyama, Y. Shiotani, H. Yoshida, P.C. Emson, S. Girgis, C.J. Hillyard, I. MacIntyre, Effect of calcitonin gene-related peptide on contraction of striated muscle in the mouse. Neurosci. Lett. 60, 227–230 (1985)
S. Uchida, H. Yamamoto, S. Lio, N. Matsumoto, X.B. Wang, N. Yonehara, Y. Imai, R. Inoki, H. Yoshida, Release of calcitonin gene-related peptide-like immunoreactive substance from neuromuscular junction by nerve excitation and its action on striated muscle. J. Neurochem. 54, 1000–1003 (1990)
C. Gennari, J.A. Fischer, Cardiovascular action of calcitonin gene-related peptide in humans. Calcif. Tissue Int. (1985). https://doi.org/10.1007/BF02554909
M.D. Harzenetter, A.R. Novotny, P. Gais, C.A. Molina, F. Altmayr, B. Holzmann, Negative regulation of TLR responses by the neuropeptide CGRP is mediated by the transcriptional repressor ICER. J. Immunol. (2007). https://doi.org/10.4049/jimmunol.179.1.607
E. Tanaka, S. Uchiyama, S. Nakano, Effects of calcitonin gene-related peptide and vasoactive intestinal peptide on nicotine-induced sweating in man. J. Auton. Nerv. Syst. 30, 265–268 (1990)
J. Machado, L.H. Manfredi, W.A. Silveira, D.A.P. Goncalves, D. Lustrino, N.M. Zanon, I.C. Kettelhut, L.C. Navegantes, Calcitonin gene-related peptide inhibits autophagic-lysosomal proteolysis through cAMP/PKA signaling in rat skeletal muscles. Int. J. Biochem. Cell Biol. (2016). https://doi.org/10.1016/j.biocel.2015.12.011
C.S. Walker, D.L. Hay, S.M. Fitzpatrick, G.J.S. Cooper, K.M. Loomes, alpha-Calcitonin gene related peptide (alpha-CGRP) mediated lipid mobilization in 3T3-L1 adipocytes. Peptides. (2014). https://doi.org/10.1016/j.peptides.2014.05.011
T. Schinke, S. Liese, M. Priemel, M. Haberland, A.F. Schilling, P. Catala-Lehnen, D. Blicharski, J.M. Rueger, R.F. Gagel, R.B. Emeson, M. Amling, Decreased bone formation and osteopenia in mice lacking alpha-calcitonin gene-related peptide. J. Bone Miner. Res. (2004). https://doi.org/10.1359/JBMR.040915
N. Takahashi, Y. Matsuda, K. Sato, P.R. de Jong, S. Bertin, K. Tabeta, K. Yamazaki, Neuronal TRPV1 activation regulates alveolar bone resorption by suppressing osteoclastogenesis via CGRP. Sci. Rep. (2016). https://doi.org/10.1038/srep29294
S.D. Brain, T.J. Williams, J.R. Tippins, H.R. Morris, I. MacIntyre, Calcitonin gene-related peptide is a potent vasodilator. Nature (1985). https://doi.org/10.1038/313054a0
S.D. Brain, Vascular actions of calcitonin gene-related peptide and adrenomedullin. Physiol. Rev. (2004). https://doi.org/10.1152/physrev.00037.2003
R. Uddman, L. Edvinsson, E. Ekblad, R. Hakanson, F. Sundler, Calcitonin gene-related peptide (CGRP): perivascular distribution and vasodilatory effects. Regul. Pept. 15, 1–23 (1986)
K. Kumazawa, G. Sobue, T. Mitsuma, T. Ogawa, Modulatory effects of calcitonin gene-related peptide and substance P on human cholinergic sweat secretion. Clin. Auton. Res. (1994). https://doi.org/10.1007/BF01821532
T. Schlereth, J.O. Dittmar, B. Seewald, F. Birklein, Peripheral amplification of sweating – a role for calcitonin gene-related peptide. J. Physiol. (2006). https://doi.org/10.1113/jphysiol.2006.116111
T. Hasegawa, K. Yokotani, Y. Okuma, M. Manabe, M. Hirakawa, Y. Osumi, Microinjection of ƒα-calcitonin gene-related peptide into the hypothalamus activates sympathetic outflow in rats. J. Pharmacol. 61, 325–332 (1993)
Research on the menopause in the 1990s. Report of a WHO Scientific Group (WHO, Switzerland, 1996)
Y. Handelsman, J.I. Mechanick, S. Dagogo-Jack, J.A. Davidson, AACE Guidelines. Endocr. Pract. (2011). https://doi.org/10.4158/EP.17.S2.1
P.K. Dalal, M. Agarwal, Postmenopausal syndrome. Indian J. Psychiatry. (2015). https://doi.org/10.4103/0019-5545.161483
J.T. Bromberger, K.A. Matthews, L.H. Kuller, R.R. Wing, E.N. Meilahn, P. Plantinga, Prospective study of the determinants of age at menopause. Am. J. Epidemiol. (1997). https://doi.org/10.1093/oxfordjournals.aje.a009083
T. Okeke, U. Anyaehie, C. Ezenyeaku, Premature menopause. Ann. Med. Health Sci. Res. (2013). https://doi.org/10.4103/2141-9248.109458
B.K. Jacobsen, I. Heuch, G. Kvale, Age at natural menopause and all-cause mortality: a 37-year follow-up of 19,731 Norwegian women. Am. J. Epidemiol. (2003). https://doi.org/10.1093/aje/kwg066
R. Cui, H. Iso, H. Toyoshima, C. Date, A. Yamamoto, S. Kikuchi, T. Kondo, Y. Watanabe, A. Koizumi, Y. Inaba, A. Tamakoshi, Relationships of age at menarche and menopause, and reproductive year with mortality from cardiovascular disease in Japanese postmenopausal women: the JACC study. J. Epidemiol. 16, 177–184 (2006)
J. Kozakowski, M. Gietka-Czernel, D. Leszczynska, A. Majos, Obesity in menopause - our negligence or an unfortunate inevitability?. Menopause Rev. (2017). https://doi.org/10.5114/pm.2017.68594
R. Bijelic, S. Milicevic, J. Balaban, Risk factors for osteoporosis in postmenopausal women. Med. Arch. (2017). https://doi.org/10.5455/medarh.2017.71.25-28
B.E. Bernstein, T.S. Mikkelsen, X. Xie, M. Kamal, D.J. Huebert, J. Cuff, B. Fry, A. Meissner, M. Wernig, M., K. Plath, R. Jaenisch, A. Wagschal, R. Feil, S.L. Schreiber, E.S. Lander, A bivalent chromatin structure marks key developmental genes in embryonic stem cells. Cell (2006). https://doi.org/10.1016/j.cell.2006.02.041
D. Kritz-Silverstein, E. Barrett-Connor, Early menopause, number of reproductive years, and bone mineral density in postmenopausal women. Am. J. Public Health 83, 983–988 (1993)
ACOG Practice Bulletin No. 141: management of menopausal symptoms. Obstet. Gynecol. (2014). https://doi.org/10.1097/01.AOG.0000441353.20693.78
I. Goranitis, L. Bellanca, A.J. Daley, A. Thomas, H. Stokes-Lampard, A.K. Roalfe, S. Jowett, Aerobic exercise for vasomotor menopausal symptoms: a cost-utility analysis based on the Active Women trial. PLoS One (2017). https://doi.org/10.1371/journal.pone.0184328
R.W. Rebar, I.B. Spitzer, The physiology and measurement of hot flushes. Am. J. Obstet. Gynecol. (1987). https://doi.org/10.1016/0002-9378(87)90165-7
C.M. Estrada, V. Ghisays, E.T. Nguyen, J.L. Caldwell, J. Streicher, M.B. Solomon, Estrogen signaling in the medial amygdala decreases emotional stress responses and obesity in ovariectomized rats. Horm. Behav. (2018). https://doi.org/10.1016/j.yhbeh.2017.12.002
R.R. Freedman, Imaging and menopausal hot flashes. Temperature (Austin.) (2016). https://doi.org/10.1080/23328940.2016.1208318
P.K.H. Morrow, D.N. Mattair, G.N. Hortobagyi, Hot flashes: a review of pathophysiology and treatment modalities. Oncologist (2011). https://doi.org/10.1634/theoncologist.2011-0174
A.A. Krull, S.A. Larsen, D.K. Clifton, G. Neal-Perry, R.A. Steiner, A. comprehensive method to quantify adaptations by male and female mice with hot flashes induced by the neurokinin B receptor agonist senktide. Endocrinology (2017). https://doi.org/10.1210/en.2017-00142
G.W. Molnar, Body temperatures during menopausal hot flashes. J. Appl. Physiol. (1975). https://doi.org/10.1152/jappl.1975.38.3.499
S. Valdemarsson, L. Edvinsson, P. Hedner, R. Ekman, Hormonal influence on calcitonin gene-related peptide in man: effects of sex difference and contraceptive pills. Scand. J. Clin. Lab. Invest. (1990). https://doi.org/10.3109/00365519009091595
A. Valentini, F. Petraglia, D. De Vita, C. Nappi, A. Margutti, E.C. Degli Uberti, A.R. Genazzani, Changes of plasma calcitonin gene-related peptide levels in postmenopausal women. Am. J. Obstet. Gynecol. (1996). https://doi.org/10.1053/ob.1996.v175.a74287
Y.A. Wyon, A.C. Spetz, G.E. Theodorsson, M.L. Hammar, Concentrations of calcitonin gene-related peptide and neuropeptide Y in plasma increase during flushes in postmenopausal women. Menopause (2000). https://doi.org/10.1097/00042192-200007010-00005
J.T. Chen, M. Shiraki, Menopausal hot flash and calcitonin gene-related peptide; effect of Keishi-bukuryo-gan, a kampo medicine, related to plasma calciotonin gene-related peptide level. Maturitas (2003). https://doi.org/10.1016/S0378-5122(03)00128-2
P. Gupta, A. Harte, D.W. Sturdee, A. Sharma, A.H. Barnett, S. Kumar, P.G. McTernan, Effects of menopausal status on circulating calcitonin gene-related peptide and adipokines: implications for insulin resistance and cardiovascular risks. Climacteric (2008). https://doi.org/10.1080/13697130802378493
Y. Wyon, J. Frisk, T. Lundeberg, E. Theodorsson, M. Hammar, Postmenopausal women with vasomotor symptoms have increased urinary excretion of calcitonin gene-related peptide. Maturitas 30, 289–294 (1998)
A.C. Spetz, B. Pettersson, E. Varenhorst, E. Theodorsson, L.H. Thorell, M Hammar, Momentary increase in plasma calcitonin gene-related peptide is involved in hot flashes in men treated with castration for carcinoma of the prostate. J Urol. (2001). http://www.ncbi.nlm.nih.gov/pubmed/11586209
A.C. Holm, L.H. Thorell, E. Theodorsson, M. Hammar, Hot flushes in healthy aging men differ from those in men with prostate cancer and in menopausal women. Gynecol. Endocrinol. (2012). https://doi.org/10.3109/09513590.2011.588744
P.R.R. Gangula, M. Chauhan, L. Reed, C. Yallampalli, Age-related changes in dorsal root ganglia, circulating and vascular calcitonin gene-related peptide (CGRP) concentrations in female rats: effect of female sex steroid hormones, Neurosci Lett. (2009). https://doi.org/10.1016/j.neulet.2009.02.068
M. Noguchi, Y. Ikarashi, M. Yuzurihara, K. Mizoguchi, K. Kurauchi, J. Chen, A. Ishige, Up-regulation of calcitonin gene-related peptide receptors underlying elevation of skin temperature in ovariectomized rats. J. Endocrinol. 175, 177–183 (2002)
A. Spinetti, A. Margutti, S. Bertolini, F. Bernardi, G. BiFulco, E.C. Degli Uberti, F. Petraglia, A.R. Genazzani, Hormonal replacement therapy affects calcitonin gene-related peptide and atrial natriuretic peptide secretion in postmenopausal women. Eur. J. Endocrinol. 137, 664–669 (1997)
P.R.R. Gangula, S.J. Wimalawansa, C. Yallampalli, Pregnancy and sex steroid hormones enhance circulating calcitonin gene-related peptide concentrations in rats. Hum. Reprod. (2000). http://www.ncbi.nlm.nih.gov/pubmed/10739848
M. Noguchi, Y. Ikarashi, M. Yuzurihara, Y. Kase, S. Takeda, M. Aburada, Effects of 17 beta-estradiol and the Japanese herbal medicine Keishi-bukuryo-gan on the release and synthesis of calcitonin gene-related peptide in ovariectomized rats. J. Pharmacol. Sci. (2003). https://doi.org/10.1254/jphs.93.80
C.N. Mowa, S. Usip, J. Collins, M. Storey-Workley, K.M. Hargreaves, R.E. Papka, The effects of pregnancy and estrogen on the expression of calcitonin gene-related peptide (CGRP) in the uterine cervix, dorsal root ganglia and spinal cord. Peptides (2003). https://doi.org/10.1016/j.peptides.2003.07.009
G. Gon, A. Giaid, J.H. Steel, D.J.O. Halloran, S.V.A.N. Noorden, M.A. Ghatei, P.M. Jones, G. Susan, S.R. Bloom, J.M. Polak, Localization of immunoreactivity for calcitonin gene-related peptide in the rat anterior pituitary during ontogeny and gonadal steroid manipulations and detection of its messenger ribonucleid acid. Endocrinology 127, 2618–2629 (1990)
Y. Lamari, M. Ghorbel, J.M. Garel, Effects of 17 beta-estradiol on calcitonin-gene-related peptide secretions and contents in a murine medullary thyroid carcinoma C-cell line (CA-77). Reprod. Nutr. Dev. 35, 655–661 (1995)
V. Pota, V. Quagliariello, E. Armenia, C. Aurilio, M.B. Passavanti, P. Sansone, M. Iannotti, M. Catauro, S. Coaccioli, M. Barbarisi, M.C. Pace, CGRP and visceral pain: the role of sex hormones in in vitro experiment. J. Cell. Biochem. (2017). https://doi.org/10.1002/jcb.25680
M. Aggarwal, V. Puri, S. Puri, Effects of estrogen on the serotonergic system and calcitonin generelated peptide in trigeminal ganglia of rats. Ann. Neurosci. (2012). https://doi.org/10.5214/ans.0972.7531.190403
R.T. Dos Santos Pereira, C.S. Porto, F.M.F. Abdalla, Ovariectomy and 17beta-estradiol replacement play a role on the expression of Endonuclease-G and phosphorylated cyclic AMP response element-binding (CREB) protein in hippocampus. Mol. Cell. Endocrinol. (2014). https://doi.org/10.1016/j.mce.2013.09.037
L. Yang, Q.-G. Zhang, C. Zhou, F. Yang, Y. Zhang, R. Wang, D.W. Brann, Extranuclear estrogen receptors mediate the neuroprotective effects of estrogen in the rat hippocampus. PLoS One (2010). https://doi.org/10.1371/journal.pone.0009851
S.M. Aronica, W.L. Kraus, B.S. Katzenellenbogen, Estrogen action via the cAMP signaling pathway: stimulation of adenylate cyclase and cAMP-regulated gene transcription. Proc. Natl. Acad. Sci. USA 91, 8517–8521 (1994)
T.D. Purves-Tyson, J.R. Keast, Rapid actions of estradiol on cyclic amp response-element binding protein phosphorylation in dorsal root ganglion neurons. Neuroscience (2004). https://doi.org/10.1016/j.neuroscience.2004.08.019
M. Genua, G. Pandini, D. Sisci, G. Castoria, M. Maggiolini, R. Vigneri, A. Belfiore, Role of cyclic AMP response element-binding protein in insulin-like growth factor-i receptor up-regulation by sex steroids in prostate cancer cells. Cancer Res. (2009). https://doi.org/10.1158/0008-5472.CAN-09-0088
K. Freeland, Y.Z. Liu, D.S. Latchman, Distinct signalling pathways mediate the cAMP response element (CRE)-dependent activation of the calcitonin gene-related peptide gene promoter by cAMP and nerve growth factor. Biochem. J. 345(Pt 2), 233–238 (2000)
C.S. Walker, X. Li, L. Whiting, S. Glyn-Jones, S. Zhang, A.J. Hickey, M.A. Sewell, K. Ruggiero, A.R.J.Phillips, E.W. Kraegen, D.L. Hay, G.J.S.., Cooper, K.M. Loomes, Mice lacking the neuropeptide alpha-calcitonin gene-related peptide are protected against diet-induced obesity. Endocrinology. (2010). https://doi.org/10.1210/en.2010-0284
T. Kobayashi, O. Ushijima, J.T. Chen, M. Shiraki, T. Ohta, M. Kiyoki, Basal tail skin temperature elevation and augmented response to calcitonin gene-related peptide in ovariectomized rats. J. Endocrinol. (1995). https://doi.org/10.1677/joe.0.1460431
M. Noguchi, Y. Ikarashi, M. Yuzurihara, Y. Kase, S. Takeda, M. Aburada, Significance of measured elevation of skin temperature induced by calcitonin gene-related peptide in anaesthetized rats. J. Pharm. Pharmacol. (2003). https://doi.org/10.1211/0022357022142
M. Noguchi, M. Yuzurihara, Y. Ikarashi, Effects of the vasoactive neuropeptides calcitonin gene-related peptide, substance P and vasoactive intestinal polypeptide on skin temperature in ovariectomized rats. Neuropeptides (2002). https://doi.org/10.1016/S0143-4179(02)00090-2
M. Noguchi, Y. Ikarashi, M. Yuzurihara, Y. Kase, K. Watanabe, G.A. Plotnikoff, S. Takeda, M. Aburada, Skin temperature rise induced by calcitonin gene-related peptide in gonadotropin-releasing hormone analogue-treated female rats and alleviation by Keishi-bukuryo-gan, a Japanese herbal medicine. Life Sci. (2005). https://doi.org/10.1016/j.lfs.2004.09.032
M. Yuzurihara, Y. Ikarashi, M. Noguchi, Y. Kase, S. Takeda, M. Aburada, Prevention by 17β-estradiol and progesterone of calcitonin gene-related peptide-induced elevation of skin temperature in castrated male rats. Urology (2004). https://doi.org/10.1016/j.urology.2004.06.013
P. Holzer, Peptidergic sensory neurons in the control of vascular functions: mechanisms and significance in the cutaneous and splanchnic vascular beds, Rev. Physiol. Biochem. Pharmacol. (1992). https://doi.org/10.1007/BFb0033194
D. Roosterman, T. Goerge, S.W. Schneider, N.W. Bunnett, M. Steinhoff, Neuronal control of skin function: the skin as a neuroimmunoendocrine organ. Physiol. Rev. (2006). https://doi.org/10.1152/physrev.00026.2005
S.R. Hughes, S.D. Brain, A calcitonin gene-related peptide (CGRP) antagonist (CGRP8-37) inhibits microvascular responses induced by CGRP and capsaicin in skin. Br. J. Pharmacol. 104, 738–742 (1991)
K.J. Escott, S.D. Brain, Effect of a calcitonin gene-related peptide antagonist (CGRP8-37) on skin vasodilatation and oedema induced by stimulation of the rat saphenous nerve. Br. J. Pharmacol. 110, 772–776 (1993)
K.J. Escott, D.T. Beattie, H.E. Connor, S.D. Brain, Trigeminal ganglion stimulation increases facial skin blood flow in the rat: a major role for calcitonin gene-related peptide. Brain Res. 669, 93–99 (1995)
C.A. Salvatore, E.L. Moore, A. Calamari, J.J. Cook, M.S. Michener, S. O’Malley, P.J. Miller, C. Sur, D.L.J. Williams, Z. Zeng, A. Danziger, J.J. Lynch, C.P. Regan, J.F. Fay, Y.S. Tang, C.-C. Li, N.T. Pudvah, R.B. White, I.M. Bell, S.N. Gallicchio, S. L. Graham, H.G. Selnick, J.P. Vacca, S.A. Kane, Pharmacological properties of MK-3207, a potent and orally active calcitonin gene-related peptide receptor antagonist. J. Pharmacol. Exp. Ther. (2010). https://doi.org/10.1124/jpet.109.163816
S.R. Sinclair, S. A. Kane, B.J. Van der Schueren, A. Xiao, K.J. Willson, J. Boyle, I. de Lepeleire, Y. Xu, L. Hickey, W.S. Denney, C.-C. Li, J. Palcza, F.H.M. Vanmolkot, M. Depre, A. Van Hecken, M.G. Murphy, T.W. Ho, J.N. de Hoon, Inhibition of capsaicin-induced increase in dermal blood flow by the oral CGRP receptor antagonist, telcagepant (MK-0974). Br. J. Clin. Pharmacol. (2010). https://doi.org/10.1111/j.1365-2125.2009.03543.x
C.-C. Li, S. Vermeersch, W.S. Denney, W. P. Kennedy, J. Palcza, A. Gipson, T.H. Han, R. Blanchard, I. De Lepeleire, M. Depre, M.G. Murphy, K. Van Dyck, J.N. de Hoon, JCharacterizing the PK/PD relationship for inhibition of capsaicin-induced dermal vasodilatation by MK-3207, an oral calcitonin gene related peptide receptor antagonist. Br. J. Clin. Pharmacol. (2015). https://doi.org/10.1111/bcp.12547
T. Vu, P. Ma, J.S. Chen, J. de Hoon, A. Van Hecken, L. Yan, L.S. Wu, L. Hamilton, G. Vargas, Pharmacokinetic-pharmacodynamic relationship of erenumab (AMG 334) and capsaicin-induced dermal blood flow in healthy and migraine subjects. Pharm. Res. (2017). https://doi.org/10.1007/s11095-017-2183-6
B.J. Van der Schueren, A. Rogiers, F.H. Vanmolkot, A. Van Hecken, M. Depre, S.A. Kane, I. De Lepeleire, S.R. Sinclair, J.N. de Hoon, Calcitonin gene-related peptide8-37 antagonizes capsaicin-induced vasodilation in the skin: evaluation of a human in vivo pharmacodynamic model. J. Pharmacol. Exp. Ther. (2008). https://doi.org/10.1124/jpet.107.133868
H. Minato, A. Ikeno, N. Watanabe, J.Tsuji, Effect of OS-0689, a novel SERM, on periarterial nerve function in tail arteries of ovariectomized rats. Maturitas (2005). https://doi.org/10.1016/j.maturitas.2004.11.003
S.P. Han, L. Naes, T.C. Westfall, Inhibition of periarterial nerve stimulation-induced vasodilation of the mesenteric arterial bed by CGRP (8-37) and CGRP receptor desensitization. Biochem. Biophys. Res. Commun. 168, 786–791 (1990)
Y. Oh-hashi, T. Shindo, Y. Kurihara, T. Imai, Y. Wang, H. Morita, Y. Imai, Y. Kayaba, H. Nishimatsu, Y. Suematsu, Y. Hirata, Y. Yazaki, R. Nagai, T. Kuwaki, H. Kurihara, Elevated sympathetic nervous activity in mice deficient in alphaCGRP. Circ. Res. (2001). https://doi.org/10.1161/hh2301.100812
P.R. Gangula, H. Zhao, S.C. Supowit, S.J. Wimalawansa, D.J. Dipette, K.N. Westlund, R.F. Gagel, C. Yallampalli, Increased blood pressure in alpha-calcitonin gene-related peptide/calcitonin gene knockout mice.Hypertension 35, 470–475 (2000).
J. Jernbeck, M. Edner, C.J. Dalsgaard, B. Pernow, The effect of calcitonin gene-related peptide (CGRP) on human forearm blood flow. Clin. Physiol. 10, 335–343 (1990)
K. Jager, R. Muench, H. Seifert, C. Beglinger, A. Bollinger, J.A. Fischer, Calcitonin gene-related peptide (CGRP) causes redistribution of blood flow in humans. Eur. J. Clin. Pharmacol. 39, 491–494 (1990)
S.C. Landis, J.R. Fredieu, Coexistence of calcitonin gene-related peptide and vasoactive intestinal peptide in cholinergic sympathetic innervation of rat sweat glands. Brain Res. 377, 177–181 (1986)
N. Charkoudian, E.C.J. Hart, J.N. Barnes, M.J. Joyner, Autonomic control of body temperature and blood pressure: influences of female sex hormones. Clin. Auton. Res. (2017). https://doi.org/10.1007/s10286-017-0420-z
C.J. Smith, J.M. Johnson, Responses to hyperthermia. Optimizing heat dissipation by convection and evaporation: neural control of skin blood flow and sweating in humans. Auton. Neurosci. (2016). https://doi.org/10.1016/j.autneu.2016.01.002
R.R. Freedman, W. Krell, Reduced thermoregulatory null zone in postmenopausal women with hot flashes. Am. J. Obstet. Gynecol. (1999). https://doi.org/10.1016/S0002-9378(99)70437-0
A.E. Herbison, Identification of a sexually dimorphic neural population immunoreactive for calcitonin gene-related peptide (CGRP) in the rat medial preoptic area. Brain Res. 591, 289–295 (1992)
A.E. Herbison, S. Dye, Perinatal and adult factors responsible for the sexually dimorphic calcitonin gene-related peptide-containing cell population in the rat preoptic area. Neuroscience (1993). https://doi.org/10.1016/0306-4522(93)90590-C
D.C. Braasch, E.M. Deegan, E.R. Grimm, J.D. Griffin, Calcitonin gene-related peptide alters the firing rates of hypothalamic temperature sensitive and insensitive neurons. BMC Neurosci. (2008). https://doi.org/10.1186/1471-2202-9-64
A. Kobayashi, T. Osaka, Y. Namba, S. Inoue, S. Kimura, CGRP microinjection into the ventromedial or dorsomedial hypothalamic nucleus activates heat production. Brain Res. (1999). https://doi.org/10.1016/S0006-8993(99)01333-5
R.R. Freedman, Menopausal hot flashes: mechanisms, endocrinology, treatment. J. Steroid Biochem. Mol. Biol. (2014). https://doi.org/10.1016/j.jsbmb.2013.08.010
C.U. Pae, M.H. Park, D.M. Marks, C. Han, A.A. Patkar, P.S. Masand, Desvenlafaxine, a serotonin-norepinephrine uptake inhibitor for major depressive disorder, neuropathic pain and the vasomotor symptoms associated with menopause. Curr. Opin. Investig. Drugs 10, 75–90 (2009)
S.F. Morrison, Central control of body temperature. F1000Res (2016). https://doi.org/10.12688/f1000research.7958.1
Y. Hu, C. Converse, M.C. Lyons, W.H. Hsu, Neural control of sweat secretion: a review. Br. J. Dermatol. (2017). https://doi.org/10.1111/bjd.15808
K.F. Malik, H.H. Feder, J.I. Morrell, Estrogen receptor immunostaining in the preoptic area and medial basal hypothalamus of estradiol benzoate- and prazosin-treated female guinea-pigs. J. Neuroendocrinol. 5, 297–306 (1993)
J.A. Butler, I. Kallo, M. Sjoberg, C.W. Coen, Evidence for extensive distribution of oestrogen receptor alpha-immunoreactivity in the cerebral cortex of adult rats. J. Neuroendocrinol. 11, 325–329 (1999)
R.R. Freedman, M.D. Benton, R.J. Genik 2nd, F.X. Graydon, Cortical activation during menopausal hot flashes. Fertil. Steril. (2006). https://doi.org/10.1016/j.fertnstert.2005.08.026
D.F. Archer, D.W. Sturdee, R. Baber, T.J. de Villiers, A. Pines, R.R. Freedman, A. Gompel, M. Hickey, M.S. Hunter, R.A. Lobo, M.A. Lumsden, A.H. MacLennan, P. Maki, S. Palacios, D. Shah, P. Villaseca, M. Warren, Menopausal hot flushes and night sweats: where are we now?. Climacteric (2011). https://doi.org/10.3109/13697137.2011.608596
O.H. Franco, T. Muka, V. Colpani, S. Kunutsor, S. Chowdhury, R. Chowdhury, M. Kavousi, Vasomotor symptoms in women and cardiovascular risk markers: systematic review and meta-analysis. Maturitas (2015). https://doi.org/10.1016/j.maturitas.2015.04.016
X. Wu, J.-T.Zhang, J. Liu, S.Yang, T. Chen, J.-G. Chen, F. Wang, Calcitonin gene-related peptide erases the fear memory and facilitates long-term potentiation in the central nucleus of the amygdala in rats. J. Neurochem. (2015). https://doi.org/10.1111/jnc.13246
J.S. Han, H. Adwanikar, Z. Li, G. Ji, V. Neugebauer, Facilitation of synaptic transmission and pain responses by CGRP in the amygdala of normal rats. Mol. Pain. (2010). https://doi.org/10.1186/1744-8069-6-10
A.Y. Deutch, R.H. Roth, Calcitonin gene-related peptide in the ventral tegmental area: selective modulation of prefrontal cortical dopamine metabolism. Neurosci. Lett. 74, 169–174 (1987)
A.A. Mathe, P. Hertel, G.G. Nomikos, S. Gruber, J.M. Mathe, T.H. Svensson, The psychotomimetic drugs D-amphetamine and phencyclidine release calcitonin gene-related peptide in the limbic forebrain of the rat. J. Neurosci. Res. 46, 316–323 (1996)
Acknowledgements
The authors would like to thank Renato Helios Migliorini (in memoriam) for being an exemplary scientist and professor.
Funding
This work was supported by the Federal University of São João del-Rei. W.G.L. received a fellowship from the Fundação de Amparo à Pesquisa do Estado de Minas Gerais (FAPEMIG).
Author contributions
All authors contributed to the development, analysis, and drafting of this article.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Oliveira, M.A., Lima, W.G., Schettini, D.A. et al. Is calcitonin gene-related peptide a modulator of menopausal vasomotor symptoms?. Endocrine 63, 193–203 (2019). https://doi.org/10.1007/s12020-018-1777-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-018-1777-z