Abstract
Purpose
DGAT2 is the critical catalyzing enzyme for triglyceride biosynthesis, and excess triglyceride accumulation in fat tissues is a fundamental process for obesity. Mutations in DGAT2 or other genes interacting with DGAT2 associated with adiposity have not been reported in human to date.
Methods
DGAT2 mutation was identified based on our in-home database-exome sequencing 227 young obese subjects (body-mass index (BMI), 35.1–61.7 kg/m2) and 219 lean controls (BMI, 17.5–23.0 kg/m2), further validated in 1190 lean subjects and the pedigree of the proband. The trios of the proband were further subjected to whole-exome sequencing to explore the candidate genes for obesity. The mutations in DGAT2 and FAAH were functionally evaluated in vitro.
Results
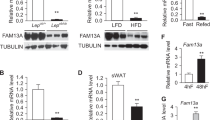
We detected two rare variants in DGAT2 with no significant difference between obese and lean individuals. One novel heterozygous nonsense variant c.382C > T (p.R128*) was identified in one obese subject but not in 219 lean subjects and another 1190 lean subjects. Notably, in vitro study showed that R128* mutation severely damaged the TG-biosynthesis ability of DGAT2, and all other R128* carriers in the pedigree were lean. Thus, we further identified a loss-of-function variant c. 944G > T (p.R315I) in FAAH in the proband inheriting from his obese father. Importantly, FAAH overexpression inhibited DGAT2 expression and TG synthesis, while R315I mutant largely eliminated this inhibitory effect.
Summary
We first report loss-of-function mutations in DGAT2 and FAAH in one obese subject, which may interact with each other to affect the adiposity penetrance, providing a model of genetic interaction associated with human obesity.





Similar content being viewed by others
References
F. Picard, P. dos Santos, B. Catargi, [Diabetes, obesity and heart complications]. Rev. Prat. 63(6), 759–764 (2013)
A.G. Comuzzie, The search for human obesity genes. Science 280(5368), 1374–1377 (1998). doi:10.1126/science.280.5368.1374
J.M. Friedman, A war on obesity, not the obese. Science 299(5608), 856–858 (2003). doi:10.1126/science.1079856
S. Ramachandrappa, I.S. Farooqi, Genetic approaches to understanding human obesity. J. Clin. Invest. 121(6), 2080–2086 (2011). doi:10.1172/JCI46044
A.E. Locke, B. Kahali, S.I. Berndt et al., Genetic studies of body mass index yield new insights for obesity biology. Nature 518(7538), 197–206 (2015). doi:10.1038/nature14177
T.A. Manolio, F.S. Collins, N.J. Cox et al., Finding the missing heritability of complex diseases. Nature 461(7265), 747–753 (2009). doi:10.1038/nature08494
P. Xu, B.A. Grueter, J.K. Britt et al., Double deletion of melanocortin 4 receptors and SAPAP3 corrects compulsive behavior and obesity in mice. Proc. Natl. Acad. Sci. USA 110(26), 10759–10764 (2013). doi:10.1073/pnas.1308195110
J. Wang, R. Liu, F. Wang et al., Ablation of LGR4 promotes energy expenditure by driving white-to-brown fat switch. Nat. Cell. Biol. 15(12), 1455–1463 (2013). doi:10.1038/ncb2867
H. Jiao, A. Kulyte, E. Naslund, A. Thorell, P.Gerdhem, J. Kere, P. Arner, I. Dahlman, Whole exome sequencing suggests LAMB3 as a susceptibility gene for morbid obesity. Diabetes (2016). doi:10.2337/db16-0522
T. Wang, M. Li, B. Chen et al., Urinary bisphenol A (BPA) concentration associates with obesity and insulin resistance. J. Clin. Endocrinol. Metab. 97(2), E223–E227 (2012). doi:10.1210/jc.2011-1989
C.A. Harris, J.T. Haas, R.S. Streeper et al., DGAT enzymes are required for triacylglycerol synthesis and lipid droplets in adipocytes. J. Lipid Res. 52(4), 657–667 (2011). doi:10.1194/jlr.M013003
S.J. Smith, S. Cases, D.R. Jensen et al., Obesity resistance and multiple mechanisms of triglyceride synthesis in mice lacking Dgat. Nat. Genet. 25(1), 87–90 (2000). doi:10.1038/75651
S.J. Stone, Lipopenia and skin barrier abnormalities in DGAT2-deficient mice. J. Biol. Chem. 279(12), 11767–11776 (2004). doi:10.1074/jbc.M311000200
K. Saar, F. Geller, F. Ruschendorf et al., Genome scan for childhood and adolescent obesity in German families. Pediatrics 111(2), 321–327 (2003)
S. Friedel, K. Reichwald, A. Scherag et al., Mutation screen and association studies in the diacylglycerol O-acyltransferase homolog 2 gene (DGAT2), a positional candidate gene for early onset obesity on chromosome 11q13. BMC Genet. 8(1), 17 (2007). doi:10.1186/1471-2156-8-17
N. Ahituv, N. Kavaslar, W. Schackwitz et al., Medical sequencing at the extremes of human body mass. Am. J. Hum. Genet. 80(4), 779–791 (2007). doi:10.1086/513471
D. Piomelli, The molecular logic of endocannabinoid signalling. Nat. Rev. Neurosci. 4(11), 873–884 (2003). doi:10.1038/nrn1247
C. Touriño, F. Oveisi, J. Lockney, D. Piomelli, R. Maldonado, FAAH deficiency promotes energy storage and enhances the motivation for food. Int. J. Obes. 34(3), 557–568 (2009). doi:10.1038/ijo.2009.262
J.C. Cable, G.D. Tan, S.P.H. Alexander, S.E. O Sullivan, The effects of obesity, diabetes and metabolic syndrome on the hydrolytic enzymes of the endocannabinoid system in animal and human adipocytes. Lipids Health Dis. 13(1), 43 (2014). doi:10.1186/1476-511x-13-43
N. Jamshidi, D.A. Taylor, Anandamide administration into the ventromedial hypothalamus stimulates appetite in rats. Br. J. Pharmacol. 134(6), 1151–1154 (2001). doi:10.1038/sj.bjp.0704379
D. Osei-Hyiaman, M. DePetrillo, P. Pacher, J. Liu, S. Radaeva, S. Batkai, J. Harvey-White, K. Mackie, L. Offertaler, L. Wang, G. Kunos, Endocannabinoid activation at hepatic CB1 receptors stimulates fatty acid synthesis and contributes to diet-induced obesity. J. Clin. Invest. 115(5), 1298–1305 (2005). doi:10.1172/JCI23057
D. Cota, G. Marsicano, M. Tschop et al., The endogenous cannabinoid system affects energy balance via central orexigenic drive and peripheral lipogenesis. J. Clin. Invest. 112(3), 423–431 (2003). doi:10.1172/JCI17725
S. Karaliota, A. Siafaka-Kapadai, C. Gontinou, K. Psarra, M. Mavri-Vavayanni, Anandamide increases the differentiation of rat adipocytes and causes PPARγ and CB1 receptor upregulation. Obesity 17(10), 1830–1838 (2009). doi:10.1038/oby.2009.177
R.S. Padwal, S.R. Majumdar, Drug treatments for obesity: orlistat, sibutramine, and rimonabant. Lancet 369(9555), 71–77 (2007). doi:10.1016/s0140-6736(07)60033-6
Q. Wang, X.D. Perrard, J.L. Perrard, A. Mansoori, C.W. Smith, C.M. Ballantyne, H. Wu, Effect of the cannabinoid receptor-1 antagonist rimonabant on inflammation in mice with diet-induced obesity. Obesity (Silver Spring) 19(3), 505–513 (2011). doi:10.1038/oby.2010.213
J.C. Sipe, J. Waalen, A. Gerber, E. Beutler, Overweight and obesity associated with a missense polymorphism in fatty acid amide hydrolase (FAAH). Int. J. Obes. 29(7), 755–759 (2005). doi:10.1038/sj.ijo.0802954
P. Monteleone, A. Tortorella, V. Martiadis, C. Di Filippo, B. Canestrelli, M. Maj, The cDNA 385C to A missense polymorphism of the endocannabinoid degrading enzyme fatty acid amide hydrolase (FAAH) is associated with overweight/obesity but not with binge eating disorder in overweight/obese women. Psychoneuroendocrinology 33(4), 546–550 (2008). doi:10.1016/j.psyneuen.2008.01.004
M. Xu, X. Lv, L. Xie et al., Discrete associations of the GCKR variant with metabolic risk in a Chinese population: longitudinal change analysis. Diabetologia 59(2), 307–315 (2016). doi:10.1007/s00125-015-3788-0
J. Liu, S. Batkai, P. Pacher, J. Harvey-White, J.A. Wagner, B.F. Cravatt, B. Gao, G. Kunos, Lipopolysaccharide induces anandamide synthesis in macrophages via CD14/MAPK/phosphoinositide 3-kinase/NF- B independently of platelet-activating factor. J. Biol. Chem. 278(45), 45034–45039 (2003). doi:10.1074/jbc.M306062200
B.F. Zhou, Cooperative Meta-Analysis Group of the Working Group on Obesity in, C., Predictive values of body mass index and waist circumference for risk factors of certain related diseases in Chinese adults--study on optimal cut-off points of body mass index and waist circumference in Chinese adults. Biomed. Environ. Sci. 15(1), 83–96 (2002)
G. Arreaza, D.G. Deutsch, Deletion of a proline-rich region and a transmembrane domain in fatty acid amide hydrolase. FEBS Lett. 454(1–2), 57–60 (1999)
M. Bouaboula, S. Hilairet, J. Marchand, L. Fajas, G.L. Fur, P. Casellas, Anandamide induced PPARγ transcriptional activation and 3T3-L1 preadipocyte differentiation. Eur. J. Pharmacol. 517(3), 174–181 (2005). doi:10.1016/j.ejphar.2005.05.032
S. Engeli, J. Jordan, The endocannabinoid system: body weight and metabolic regulation. Clin. Cornerstone 8(Suppl 4), S24–S35 (2006)
S.P. Bapat, J. Myoung Suh, S. Fang, S. Liu et al., Depletion of fat-resident Treg cells prevents age-associated insulin resistance. Nature 528(7580), 137–141 (2015). doi:10.1038/nature16151
H.R. Wurie, L. Buckett, V.A. Zammit, Diacylglycerol acyltransferase 2 acts upstream of diacylglycerol acyltransferase 1 and utilizes nascent diglycerides andde novosynthesized fatty acids in HepG2 cells. FEBS J. 279(17), 3033–3047 (2012). doi:10.1111/j.1742-4658.2012.08684.x
R. Christensen, P.K. Kristensen, E.M. Bartels, H. Bliddal, A. Astrup, Efficacy and safety of the weight-loss drug rimonabant: a meta-analysis of randomised trials. Lancet 370(9600), 1706–1713 (2007). doi:10.1016/s0140-6736(07)61721-8
S.A. Tucci, E.K. Rogers, M. Korbonits, T.C. Kirkham, The cannabinoid CB1 receptor antagonist SR141716 blocks the orexigenic effects of intrahypothalamic ghrelin. Br. J. Pharmacol. 143(5), 520–523 (2004). doi:10.1038/sj.bjp.0705968
A.R. Alvheim, B.E. Torstensen, Y.H. Lin, H.H. Lillefosse, E.-J. Lock, L. Madsen, L. Frøyland, J.R. Hibbeln, M.K. Malde, Dietary linoleic acid elevates the endocannabinoids 2-AG and anandamide and promotes weight gain in mice fed a low fat diet. Lipids 49(1), 59–69 (2013). doi:10.1007/s11745-013-3842-y
B.F. Cravatt, K. Demarest, M.P. Patricelli, M.H. Bracey, D.K. Giang, B.R. Martin, A.H. Lichtman, Supersensitivity to anandamide and enhanced endogenous cannabinoid signaling in mice lacking fatty acid amide hydrolase. Proc. Natl. Acad. Sci. 98(16), 9371–9376 (2001). doi:10.1073/pnas.161191698
D. Rohrborn, N. Wronkowitz, J. Eckel, DPP4 in diabetes. Front. Immunol. 6, 386 (2015). doi:10.3389/fimmu.2015.00386
Acknowledgements
We thank all the staff and participants for their contributions. This study was funded by grants from the National Natural Science Foundation of China (81522011, 81570757, 81370963, 81370949, 81500651), National Basic Research Program of China (2015CB553601), National International Science Cooperation Foundation (2015DFA30560).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. And all applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Ning, T., Zou, Y., Yang, M. et al. Genetic interaction of DGAT2 and FAAH in the development of human obesity. Endocrine 56, 366–378 (2017). https://doi.org/10.1007/s12020-017-1261-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-017-1261-1