Abstract
The current SARS-CoV-2 outbreak, which causes COVID-19, is particularly devastating for individuals with chronic medical conditions, in particular those with Down Syndrome (DS) who often exhibit a higher prevalence of respiratory tract infections, immune dysregulation and potential complications. The incidence of Alzheimer’s disease (AD) is much higher in DS than in the general population, possibly increasing further the risk of COVID-19 infection and its complications. Here we provide a biological overview with regard to specific susceptibility of individuals with DS to SARS-CoV-2 infection as well as data from a recent survey on the prevalence of COVID-19 among them. We see an urgent need to protect people with DS, especially those with AD, from COVID-19 and future pandemics and focus on developing protective measures, which also include interventions by health systems worldwide for reducing the negative social effects of long-term isolation and increased periods of hospitalization.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Overview
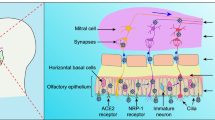
The current COVID-19 pandemic, caused by the SARS-CoV-2 virus, is of widespread concern for the elderly people and populations at risk due to co-existing medical conditions, including cardiovascular diseases, obesity, and diabetes. Individuals with Down syndrome (DS) appear to have a significantly higher risk of developing severe symptoms of infectious diseases, including those related to COVID-19, because of their complex trisomy and consequent numerous immune impairments, which render them susceptible to infections. Chromosome 21 (Chr21), triplicated in individuals with DS, includes genes directly involved in SARS-CoV-2 entry into cells, potentially augmenting COVID-19-specific susceptibility (Fig. 1). Clinically, these immune dysregulations often result in ineffective vaccination against infectious diseases. COVID-19-related risks, such as anatomical airway features, which enhance airway-related infections, congenital heart disease, hypertension, obesity, and diabetes, are prevalent in individuals with DS. The immune anomalies of individuals with DS suggest that any COVID-19-directed therapy developed for the general population requires careful testing prior to implementation in the clinic. Not least important is the additional aspect of social isolation experienced by numerous at-risk individuals in general and individuals with DS in particular (Fig. 1). Individuals with DS are often dependent on regular schedules, which, upon interruption, can impair their mental health. Measures to avert the implications of social isolation in this vulnerable population, therefore, need to be taken into consideration. This complex picture calls for an immediate reorganization towards better preparedness for future pandemics in vulnerable populations, including DS.
Down Syndrome–Prevalence and Lifespan
DS is the most common genetic disorder of cognition; essentially, all those with DS demonstrate intellectual disability (ID), though of variable degree. Chr21 contains approximately 233 protein-coding genes, some of which, like amyloid precursor protein (APP) and dual-specificity tyrosine phosphorylation-regulated kinase 1A (DYRK1A), have been linked to an Alzheimer’s disease (AD)-like phenotype. DS occurs in all populations with an average worldwide prevalence of 3.3–6.7 per 10,000 individuals that varies between countries (Antonarakis et al. 2020) (Fig. 2, Supplementary Data 1).
Worldwide prevalence of DS. Data collected from the Global Health Data Exchange (GHDx; http://ghdx.healthdata.org/) were used to calculate the total worldwide prevalence of DS according to sex in 194 countries
In the 1940s, the average life expectancy for people with DS was 12 years (Penrose, 1949). With advances and increased access to medical care and early surgical repair of congenital heart malformations, individuals with DS currently have an average life expectancy of 60 years (Bittles & Glasson, 2004). The risk for mortality in younger people with DS is increased mainly because of leukemia and respiratory infections. As a result of increased life expectancy, a greater number of individuals with DS now exhibit age-related diseases, such as AD—the principal cause of death in the elderly with DS, which may increase their risk for emerging pathogens such as SARS-CoV-2. A significant vulnerability to infections impacts individuals with DS after the age of 50 years, especially those with certain comorbidities, like seizures and age-related onset of dementia (Guffroy et al. 2019).
Comorbidities in DS as Risk Factors for Emerging Infectious Diseases
Obesity, increasing age, male sex, hypertension, and diabetes are risk factors associated with increased mortality from COVID-19 in the general population (Brenner et al. 2020; George et al. 2020; Palaiodimos et al. 2020) and are also present in DS, combining to increase susceptibility to infectious diseases including SARS-CoV-2. Certain anatomical features contribute a short stature, a flat and wide face with a flat nasal ridge, slanted eyes, a short neck, and a protruding tongue due to a small orofacial cavity and a relatively large tongue (Kanamori et al. 2000). These features impact the airway tract and may contribute to the increase in obstructive sleep apnea and upper airway obstruction found in 50% of those with DS (Horne et al. 2019; Marcus et al. 1991). Intervention with respiratory support may therefore prevent COVID-19-related consequences of sleep-disordered breathing (Trucco et al. 2018).
Pulmonary hypertension is diagnosed in 13% of those with DS (Berger et al. 2012). This condition and developmental lung anomalies (Cooney & Thurlbeck, 1982) may combine to increase the risk of COVID-19 in people with DS (Bush et al. 2020), due to reduced oxygen capacity and supply.
Obesity, due to lower thyroid and a lower metabolic rate, and ID co-occur significantly more often than in the general population. Obesity is associated with decreased expiratory reserve volume and functional capacity. Abdominal obesity compromises pulmonary function in immobilized patients by decreased diaphragmatic excursion and reducing ventilation efficacy. Obesity is also associated with higher levels of inflammatory cytokines, contributing to increased morbidity associated with obesity in COVID-19 infections (Dietz & Santos-Burgoa, 2020). Obesity is also associated with obstructive sleep apnea, another very common condition in individuals with DS, which, if left untreated, can lead to hypertension, heart failure and sudden death.
Additionally, the frequency of congenital heart disease varies from 40 to 54% in DS (Freeman et al. 1998). The cardiac defects are usually treated by pediatric cardiac surgery at an early age (Jensen et al. 2014). As for those with congenital heart diseases, individuals with DS are known to have a higher risk for complications with viral illnesses, including respiratory syncytial virus and influenza (Alsaied et al. 2020; Weijerman et al. 2010).
Prevalence of Infectious Diseases and Associated Hospitalizations in Down Syndrome
Bacteria such as Streptococcus pneumonia are the major cause of pneumonia in individuals with DS (Verstegen et al. 2014), while viruses such as influenza virus, respiratory syncytial virus (RSV), and parainfluenza virus account for most respiratory infections in DS (Chan et al. 2017). RSV, a double-stranded RNA (dsRNA) virus, is the primary cause of severe lower respiratory tract infections (LRI) in young children (< 5 years) (Beckhaus & Castro-Rodriguez, 2018; Perez-Padilla et al. 2010). Children with DS are at risk for RSV infections (Beckhaus & Castro-Rodriguez, 2018; Mitra et al. 2018), especially those who have chronic lung disease or congenital heart disease.
Following the outbreak of the H1NI 2009 pandemic in Mexico, a study found a 16-fold increase in hospitalization, an eightfold increase in endotracheal intubation, and a more than 300-fold increase in death in individuals with DS in Mexico (Perez-Padilla et al. 2010). Moreover, DS patients were significantly younger as compared with affected individuals of the general population. Thus, compared with the general population, DS is associated with increased susceptibility to bacterial and viral infections at a younger age, resulting in worse outcomes, higher hospitalization rates, longer hospital stays, and higher mortality rates.
These findings point to respiratory infections as a significant source of increased morbidity and mortality in children with DS and suggest that this will apply to infection with COVID-19 in both children and adults with DS.
A recent study, reported an increased severity of COVID-19 in hospitalized patients with DS (Malle et al. 2020). This dual-center study of 7246 patients hospitalized with COVID-19 in the city of New York, assessed the hospitalization rates, clinical characteristics, and outcomes. The researchers identified 12 patients with DS, and reported that their average age was ten years younger than patients without DS. The incident of severe disease was higher in those patients, specifically an increased incident of sepsis and mechanical ventilation. Moreover, a description of the clinical course in four COVID-19 patients with DS in Belgium, revealed severe illness in three of the four cases with fatal outcome in one patient (De Cauwer & Spaepen, 2020).
Immune Dysregulation and Aging in Down Syndrome vis-a-vis Vaccines
Individuals with DS have reduced lymphocyte numbers (Huggard et al. 2018; MacLean et al. 2018), thought to be a result of increased thymic dysfunction and apoptosis of immune cells (Schoch et al. 2017; Zampieri et al. 2014). The thymus is smaller in DS infants than in non-DS infants and exhibits structural abnormalities (Carsetti et al. 2015; Eijsvoogel et al. 2017), arguing that immune dysregulation and immunodeficiency are integral to the syndrome (Carsetti et al. 2015; Huggard et al. 2018). The selective cell-mediated immunodeficiency and impaired antibody response to pathogens in DS can be associated with the mortality associated with pneumonia and other respiratory diseases in children with DS (Chaushu et al. 2002; Costa-Carvalho et al. 2006).
Type I interferons (IFNs) are a group of proteins that help regulate the activity of the immune system. DS is characterized by constitutive IFN-I signaling; this may be a major cause of suboptimal vaccine responses in people with DS. This raises the question as to whether SARS-CoV-2 vaccines shown effective in otherwise healthy young hosts will prove effective in people with DS. It is premature to conclude that the DS population will fail to respond to SARS-CoV-2 vaccines that are effective in the general population. However, this should be proactively explored through studies in which strategies that block high levels of IFN-I signaling or upregulation of IFN receptors are tested. Alternatively, vaccine responses can be improved using immune modulators, such as interleukin (IL)-21, or activating downstream signaling effectors of IL21, such as the STAT3 pathway, known to contribute to long-lived antibody-secreting cells in humans (Avery et al. 2010).
Initial response to vaccines is generally adequate in individuals with DS but with lower mean titers and a need for more frequent booster immunizations compared to individuals without DS (Eijsvoogel et al. 2017). Many of these immunological alterations are age related and can be envisioned as within the spectrum of early senescence of the immune system, which is typically seen in the elderly (da Rosa Utiyama et al. 2008; Trotta et al. 2011). Indeed, premature aging in multiple body tissues is evident in DS (Zigman, 2013). Specifically, molecular aging in DS is more pronounced in individuals with DS that experience dementia compared with individuals with DS without apparent dementia. For example, telomeres are chromosome ends consisting of highly conserved TTAGGG repeats whose number progressively reduced with aging (Shay, 2018). Immune T cell lymphocyte cultures from people with DS and AD-related dementia have shorter telomeres than T-lymphocytes from age- and sex-matched people with DS with no dementia (Jenkins et al. 2008, 2010), a phenomenon that precedes the late stages of dementia in DS (Jenkins et al. 2012).
Specific Predisposition to Severe COVID-19 in DS
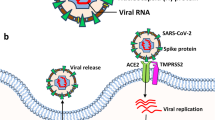
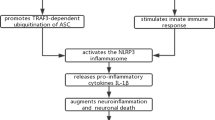
Cell attachment and entry is a critical step in the process of viral infection. Of particular relevance to DS is the TMPRSS2 gene, located within Chr21 (De Cauwer, 2020; Paoloni-Giacobino et al. 1997). TMPRSS2 is a protease involved in SARS-CoV-2 entry into cells via priming of S protein; TMPRSS2 overexpression might result in a higher number of infected cells that could potentially augment COVID-19 susceptibility in DS. It was recently reported that a TMPRSS2 inhibitor, approved for clinical use, blocked entry of SARS-2-S to lung cells (Hoffmann et al. 2020), therefore might constitute a treatment option for patients with DS. Furthermore, viruses, including SARS-CoV, enter cells by hijacking mechanisms, like phagocytosis via the endocytosis pathways (Cataldo et al. 2008; Inoue et al. 2007). Dysregulation of endocytosis in DS is likely a function of an increased dose of several endosomal pathway-related genes that map to Chr21, including APP, SYNJ1 (Synaptojanin-1), ITSN1 (Intersectin-1), and possibly RCAN1 (Regulator of calcineurin 1) (Botte & Potier, 2020; Patel et al. 2015). Studies focused on neurons showed that APP, known to mediate dementia in DS, is involved in altered trafficking of viral material and modified pH-dependent fusion of viruses with an endosomal membrane in DS (Botte & Potier, 2020; Jiang et al. 2016; Kim et al. 2016).
Adults with DS exhibit elevated levels of the proinflammatory cytokines IL-6 and TNF-α (Altable & de la Serna, 2021), along with hyperactivity of interferon signaling, possibly due to IFNAR1, IFNAR2, IFNGR2 and IL10RB overexpression (Espinosa, 2020). Hypothetically, these dysregulations support the notion that DS individuals are at increased risk of exacerbated cytokine storm following COVID-19 infection. However, this hypothesis is not yet backed with data from humans.
Children with ID have a higher prevalence of specific comorbidities associated with poorer COVID-19 outcomes (Turk et al. 2020). No specific data on COVID-19 prevalence in pediatric patients with DS is currently available. Nevertheless, several case studies were recently described in such patients (Alsahabi et al. 2021; Kantar et al. 2020; Newman et al. 2021; Pontes et al. 2020). Out of the total of 10 cases presented in these publications, 3 (33%) were admitted to the ICU, 2 (20%) required intubation and none had died.
Given this and related factors discussed above, individuals with DS may be at higher risk of SARS-CoV-2 infection and poorer clinical outcomes.
Vaccination Strategies
The majority of vaccination strategies in the race to control the COVID-19 aim to induce neutralizing antibodies to the SARS-CoV-2 by targeting its Spike (S) antigen and its binding to angiotensin converting enzyme 2 (ACE2), which serves as the primary mechanisms by which the virus enters cells. Elderly individuals, people with type-2-diabetes, morbidly obese individuals, and individuals with DS are in general poor responders to vaccines (Corretger, 2014). Two concerns attend considerations of vaccine development in the context of DS. COVID-19 vaccines may be challenged in their ability to elicit long-term protection in DS. Due to suboptimal primary and memory immune responses, vaccines often prove inefficient protection in DS (Costa-Carvalho et al. 2006). Moreover, even if a vaccine generates an adaptive immune response, it may induce persistent, even life-long, overactivation of innate immune cells in DS. Indeed, antibodies against SARS-CoV-2 antigens could exacerbate an already heightened immune response. Antibody-dependent enhancement of infection can also occur due to innate immune cell dysregulation, impaired destruction of phagocytosed immune complexes, or/and altered expression of Fc receptors, which recognize Fc fragment of antibodies and mediate downstream signaling. Achieving optimal responses in DS should be seen as an important research topic. One potential strategy would include the use of a conjugated pneumococcal vaccine, shown to induce antibodies responses in individuals with DS that are equally effective as in non-DS individuals (Kusters et al. 2013).
Data on the Susceptibility of Individuals with DS to COVID-19
For an initial assessment, data on the prevalence of DS per country, as well as death rates from different diseases in 61 countries, were collected from the Global Health Data Exchange (GHDx) (http://ghdx.healthdata.org/, Sep 2020). COVID-19-related data were obtained from the WORLDOMETER database (https://www.worldometers.info/, Sep 2020). A significant correlation was observed between COVID-19-related deaths (normalized to 1 million in every country) and the prevalence of DS per country (r = 0.39, p-value = 0.0022, Spearman Correlation Coefficient (SCC)). By means of SCC, we observed that SARS-CoV-2 infections were positively correlated with DS-related comorbidities such as AD and dementia (SCC = 0.73, p < 0.0001), cardiovascular diseases (SCC = 0.66, p < 0.0001), leukemia (SCC = 0.72, p < 0.0001), and thyroid cancer (SCC = 0.71, p < 0.001) (Fig. 3), suggesting that DS-related comorbidities are a risk factor for COVID-19.
Spearman correlation between COVID-19 and different diseases. Spearman correlation between COVID-19-related deaths (normalize to 1 million) and different pathologies. A positive significant Spearman correlation was found between COVID-19 and DS, AD, cardiovascular diseases, thyroid cancer and leukemia
‘Leumit’ Health Care Services (LHS) is a health maintenance organization that covers 712,773 residents in Israel, including 570 residents with DS (0.08%). The LHS study included 115,050 subjects, (from February 1st until December 31st, 2020) aged 1 month to 106 years old, who were tested for SARS-CoV-2. Among them, 10,469 (9.1%) had at least one positive result, and 104,581 (90.9%) were negative for SARS-CoV-2 (Table 1). COVID-19 positive individuals were of lower socioeconomic status (SES) and were mostly males (Table 1). The prevalence of DS in the general population in Israel is 0.11% (GHDx, Sep 2020). The prevalence of DS individuals that were tested positive to COVID-19 in the LHS database is higher than their proportion in the general population (0.19% of all COVID-19 positive cases). We then calculated the crude odds ratio (OR) for the general population of LHS enrollees and individuals with DS that were positive for COVID-19 and we found OR = 2.33, 95% CI 1.08–3.45, (p = 0.023). However, we did not find significance when we compared COVID-19 positive cases in the general population with COVID-19 positive cases in individuals with DS, OR = 1.53, 95% CI 0.91–2.58, (p = 0.12) (Table 1). Surprisingly, chronic medical conditions such as dementia, cardiovascular disease and chronic lung disease that are considered to be risk factors for COVID-19 in previous studies, were not found as increasing the rate of infection in the LHS cohort (Table 1). It is possible that this is due to the severe social distancing imposed on all the population, and due to reduced social interaction by patients with chronic medical conditions.
The multiple logistic regression model for all patients tested for SARS-CoV-2 was applied to adjust for possible confounders. An association between DS and the likelihood of being tested positive for SARS-CoV-2 was significant (adjusted OR 1.64 (95% CI 0.98–2.97; p = 0.05) (Table 2). COVID-19 positive patients were mostly males (adjusted OR 1.20 (95% CI 1.14–1.26), with a lower-medium SES OR 2.01 (95% CI 1.88–2.15), and obese OR 1.17 (95% 1.09–1.25) (Table 2).
Individuals with DS who were positive for COVID-19 were younger compared with non-DS individuals who were positive for COVID-19 (18.48 (13.40–23.56) vs. 31.43 (31.00–31.85) years, p-value < 0.001). SES or gender were not a contributing factor to this difference (Table 3). Moreover, chronic lung disease was significantly more prevalent in SARS-CoV-2-positive individuals with DS (Table 3). These data suggest that individuals with DS have an increased risk for SARS-CoV-2 infection, particularly those with higher rates of chronic lung disease (p-value < 0.001, Table 3, and supplementary methods). Indeed, a survey that recently assessed the suceptability to COVID-19 in DS found that the mean age of COVID-19 patients with DS was lower than the general population. In addition, mortality rates were increased in individuals with DS aged 40 and above (Hüls et al. 2021).
Quality of Life in Individuals with DS during Social Distancing
Mention has been made about of the behavioral characteristics of people with DS that may feature in their susceptibility to infection and their physiological responses once infected. People with DS enjoy and are likely to have frequent contact with family members, caregivers, support staff, and people in the community, possibly increasing their risk of contact with infected individuals. Furthermore, they may have difficulty in understanding the importance of social distancing and wearing masks as a means of protecting against virus infection. Another important consideration is the potential impact of the pandemic on the behaviors and stress experienced by those with DS. Individuals with DS use routines and repetition to help them complete daily tasks, which are important to support their mental health. These behaviors include performing tasks at the same time and in the same way. Interrupting with these routines could, therefore, significantly affect their sense of wellbeing. Older adults with DS tend to be physically and cognitively frailer than those in the general population. As a consequence, they often live in sheltered community residences, assisted group living situations, and nursing homes. Social distancing recommendations in the context of the pandemic raise the possibility that people with DS experience increased stress, anxiety and depression and loss of social connectivity, which combine to compromise mental health concerns and induce undesirable changes in behavior. Furthermore, receiving a vaccine, as well as any other invasive medical procedure, may contribute to the stress burden experienced by individuals with DS during the pandemic. In this respect, those with DS may be representative of those in the general population who live in similar settings, especially those with dementia. As one approach to safely mitigating the deleterious effects of social distancing, the use of digital technology may provide a means by which to allow people with DS and those with memory loss and dementia to reengage with caregivers, family, and friends. Activities in a virtual space also enable availability and accessibility to services and information and improve the range of possibilities for communicating in a risk-free environment. These methods may prove useful for the elderly in general and people with DS and other sources of ID for bridging physical, geographical, and financial barriers (Caton & Chapman, 2016).
Concluding Remarks
Here we have provided an overview of the contributing susceptibility factors to COVID-19 infection in DS, with a special focus on adults with DS. The picture that emerges from the immunological and clinical aspects of individuals with DS points to the alarming conclusion that these individuals are at increased risk for contracting specific types of infectious diseases and infection-related deaths.
There is a critical knowledge gap with respect to the underlying immune dysregulation and the specific vulnerabilities of individuals with DS to COVID-19. For example, the influence of human leukocyte antigen (HLA) gene polymorphisms on SARS susceptibility, pathogenesis, and outcome has been investigated in a number of studies. Some HLA alleles have been significantly associated with susceptibility to SARS and/or disease severity in various populations.
In addition, HLA alleles can be computationally predicted for their binding capability to SARS‐CoV‐2 peptides, predicting host susceptibility for COVID-19 disease (Ovsyannikova et al. 2020). Despite this, little is known about HLA polymorphism in the DS population. Therefore, sequencing HLA types in DS patients worldwide is needed to enable a more rapid correlation between infection rate and HLA typing in affected DS patients. This will allow mapping of protective as well as harmful HLA types in DS.
Preparedness for Future Pandemics
Future pandemics could pose a similar or even higher risk for this susceptible population. One of the clear conclusions is that we need to increase the preparedness to future pandemics in individuals with DS in light of their increased and specific susceptibility. Better preparedness with respect to lifestyle, medical practice and basic research can be achieved mainly by forming an international network of experts in various fields, including immunology, epidemiology, neurology, and sociology. With this in mind, the Trisomy 21 Research society has established a COVID-19 task force and a stakeholders group with representatives from all major DS organizations in Europe, the U.S., and further afield.
This calls for a need to formulate steps to increase preparedness for such a pandemic by family members and clinicians. These steps should include:
-
1.
Establishing a DS-specific worldwide database on pandemic-related symptoms and hospitalizations and response to treatment. Such a database will enable a close to a real-time understanding of the specific susceptibility of individuals with DS to pandemics and could be used or linked with existing surveillance efforts.
-
2.
Establishing virtual social network(s) that can be activated during pandemics to support caregivers and the social welfare of individuals with DS. Such networks can partially help mitigate the adverse effects of social isolation during pandemics.
-
3.
Promoting healthy lifestyle changes, for example lowering obesity.
-
4.
Reducing general risk factors in individuals with DS. For example in the COVID-19 relevant medical practices include (i) improving lung health by reducing chronic/regular respiratory infections by offering influenza and pneumococcus vaccines, (ii) screening for and treating sleep apnea as well as other risk factors such as aspiration, (iii) responding early to signs of lung infection to help reduce the risk of COVID-19, and (iv) increasing surveillance and protection of high-risk older individuals, particularly those who also show signs of AD.
-
5.
Confirming the increased risk for infection and presentation in individuals with DS when affected by COVID-19 and future pandemics.
-
6.
Studying the clinical outcomes following COVID-19 and response to treatment in individuals with DS. The Long-term sequelae following recovery from acute COVID-19 need to be considered to understand the need for long-term management in the context of the impaired immune response in individuals with DS.
-
7.
Establishing prior to widespread immunization whether the response to vaccines against SARS-CoV-2 in individuals with DS is similar to the response the general population.
Implementing these steps will help to support an effective response to future pandemics that is supportive not only of individuals with DS but also of family members, caregivers and clinicians, thus providing a much-needed safety net for this unique and vulnerable population.
Data Availability
All the data supporting the findings of this study and MATLAB codes are freely available upon request.
Change history
19 November 2021
A Correction to this paper has been published: https://doi.org/10.1007/s12017-021-08695-7
04 May 2021
A Correction to this paper has been published: https://doi.org/10.1007/s12017-021-08657-z
References
Alsahabi, I., Alobaidi, A., Alahmari, A. S., Almohsen, N., & Alhamoud, A. H. (2021). Clinical Presentation and Successful Management of an Infant With Down Syndrome and COVID-19 in Riyadh Saudi Arabia. Cureus, 13(2), e13188. https://doi.org/10.7759/cureus.13188
Alsaied, T., Aboulhosn, J. A., Cotts, T. B., Daniels, C. J., Etheridge, S. P., Feltes, T. F., et al. (2020). Coronavirus Disease 2019 (COVID-19) Pandemic Implications in Pediatric and Adult Congenital Heart Disease. J Am Heart Assoc, 9(12), e017224. https://doi.org/10.1161/JAHA.120.017224
Altable, M., & de la Serna, J. M. (2021). Down’s syndrome and COVID-19: risk or protection factor against infection? A molecular and genetic approach. Neurological Sciences, 42(2), 407–413. https://doi.org/10.1007/s10072-020-04880-x
Antonarakis, S. E., Skotko, B. G., Rafii, M. S., Strydom, A., Pape, S. E., Bianchi, D. W., et al. (2020). Down syndrome. Nat Rev Dis Primers, 6(1), 9. https://doi.org/10.1038/s41572-019-0143
Avery, D. T., Deenick, E. K., Ma, C. S., Suryani, S., Simpson, N., Chew, G. Y., et al. (2010). B cell-intrinsic signaling through IL-21 receptor and STAT3 is required for establishing long-lived antibody responses in humans. Journal of Experimental Medicine, 207(1), 155–155.
Beckhaus, A. A., & Castro-Rodriguez, J. A. (2018). Down Syndrome and the Risk of Severe RSV Infection: A Meta-analysis. Pediatrics. https://doi.org/10.1542/peds.2018-0225
Berger, R. M. F., Beghetti, M., Humpl, T., Raskob, G. E., Ivy, D. D., Jing, Z. C., et al. (2012). Clinical features of paediatric pulmonary hypertension: a registry study. [Article]. Lancet, 379(9815), 537–546. https://doi.org/10.1016/s0140-6736(11)61621-8
Bittles, A. H., & Glasson, E. J. (2004). Clinical, social, and ethical implications of changing life expectancy in Down syndrome. Developmental Medicine and Child Neurology, 46(4), 282–286. https://doi.org/10.1017/s0012162204000441
Botte, A., & Potier, M. C. (2020). Focusing on cellular biomarkers: The endo-lysosomal pathway in Down syndrome. Progress in Brain Research, 251, 209–243. https://doi.org/10.1016/bs.pbr.2019.10.002
Brenner, E. J., Ungaro, R. C., Gearry, R. B., Kaplan, G. G., Kissous-Hunt, M., Lewis, J. D., et al. (2020). Corticosteroids, but not TNF Antagonists, are Associated with Adverse COVID-19 Outcomes in Patients With Inflammatory Bowel Diseases: Results from an International Registry. Gastroenterology. https://doi.org/10.1053/j.gastro.2020.05.032
Bush, D., Galambos, C., & Ivy, D. D. (2020). Pulmonary hypertension in children with Down syndrome. [Review; Early Access]. Pediatric Pulmonology. https://doi.org/10.1002/ppul.24687
Carsetti, R., Valentini, D., Marcellini, V., Scarsella, M., Marasco, E., Giustini, F., et al. (2015). Reduced numbers of switched memory B cells with high terminal differentiation potential in Down syndrome. European Journal of Immunology, 45(3), 903–914. https://doi.org/10.1002/eji.201445049
Cataldo, A. M., Mathews, P. M., Boiteau, A. B., Hassinger, L. C., Peterhoff, C. M., Jiang, Y., et al. (2008). Down syndrome fibroblast model of Alzheimer-related endosome pathology: accelerated endocytosis promotes late endocytic defects. American Journal of Pathology, 173(2), 370–384. https://doi.org/10.2353/ajpath.2008.071053
Caton, S., & Chapman, M. (2016). The use of social media and people with intellectual disability: A systematic review and thematic analysis. Journal of Intellectual & Developmental Disability, 41(2), 125–139. https://doi.org/10.3109/13668250.2016.1153052
Chan, M., Park, J. J., Shi, T., Martinon-Torres, F., Bont, L., Nair, H., et al. (2017). The burden of respiratory syncytial virus (RSV) associated acute lower respiratory infections in children with Down syndrome: A systematic review and meta-analysis. J Glob Health, 7(2), 020413. https://doi.org/10.7189/jogh.07.020413
Chaushu, S., Yefenof, E., Becker, A., Shapira, J., & Chaushu, G. (2002). Severe impairment of secretory Ig production in parotid saliva of Down Syndrome individuals. Journal of Dental Research, 81(5), 308–312. https://doi.org/10.1177/154405910208100504
Cooney, T. P., & Thurlbeck, W. M. (1982). Pulmonary hypoplasia in downs-syndrome. [Article]. New England Journal of Medicine, 307(19), 1170–1173. https://doi.org/10.1056/nejm198211043071902
Corretger, J. M. (2014). Vaccines and vaccination calendar for people with Down’s syndrome. International Medical Review on Down Syndrome, 18(2), 29–32. https://doi.org/10.1016/S2171-9748(14)70050-9
Costa-Carvalho, B. T., Martinez, R. M., Dias, A. T., Kubo, C. A., Barros-Nunes, P., Leiva, L., et al. (2006). Antibody response to pneumococcal capsular polysaccharide vaccine in Down syndrome patients. Brazilian Journal of Medical and Biological Research, 39(12), 1587–1592. https://doi.org/10.1590/s0100-879x2006001200010
da Rosa Utiyama, S. R., Nisihara, R. M., Nass, F. R., Oliveira, N. P., Fiedler, P. T., & de Messias-Reason, I. T. (2008). Autoantibodies in patients with Down syndrome: early senescence of the immune system or precocious markers for immunological diseases? Journal of Paediatrics and Child Health, 44(4), 182–186. https://doi.org/10.1111/j.1440-1754.2007.01229.x
De Cauwer, H. (2020). The SARS-CoV-2 receptor, ACE-2, is expressed on many different cell types: implications for ACE-inhibitor- and angiotensin II receptor blocker-based cardiovascular therapies: comment. Internal and Emergency Medicine, 15(8), 1581–1582. https://doi.org/10.1007/s11739-020-02406-z
De Cauwer, H., & Spaepen, A. (2020). Are patients with Down syndrome vulnerable to life-threatening COVID-19? Acta Neurologica Belgica. https://doi.org/10.1007/s13760-020-01373-8
Dietz, W., & Santos-Burgoa, C. (2020). Obesity and its Implications for COVID-19 Mortality. Obesity (Silver Spring), 28(6), 1005. https://doi.org/10.1002/oby.22818
Eijsvoogel, N. B., Hollegien, M. I., Bok, V. L. A., Derksen Lubsen, A. G., Dikken, F. P. J., Leenders, S., et al. (2017). Declining antibody levels after hepatitis B vaccination in Down syndrome: A need for booster vaccination? Journal of Medical Virology, 89(9), 1682–1685. https://doi.org/10.1002/jmv.24813
Espinosa, J. M. (2020). Down Syndrome and COVID-19: A Perfect Storm? Cell Rep Med, 1(2), 100019. https://doi.org/10.1016/j.xcrm.2020.100019
Freeman, S. B., Taft, L. F., Dooley, K. J., Allran, K., Sherman, S. L., Hassold, T. J., et al. (1998). Population-based study of congenital heart defects in Down syndrome. American Journal of Medical Genetics, 80(3), 213–217.
George, P. M., Wells, A. U., & Jenkins, R. G. (2020). Pulmonary fibrosis and COVID-19: the potential role for antifibrotic therapy. The Lancet. Respiratory medicine. https://doi.org/10.1016/s2213-2600(20)30225-3
Guffroy, A., Dieudonne, Y., Uring-Lambert, B., Goetz, J., Alembik, Y., & Korganow, A. S. (2019). Infection risk among adults with down syndrome: a two group series of 101 patients in a tertiary center. Orphanet Journal of Rare Diseases, 14(1), 15. https://doi.org/10.1186/s13023-018-0989-x
Hoffmann, M., Kleine-Weber, H., Schroeder, S., Kruger, N., Herrler, T., Erichsen, S., et al. (2020). SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor. Cell. https://doi.org/10.1016/j.cell.2020.02.052
Horne, R. S. C., Wijayaratne, P., Nixon, G. M., & Walter, L. M. (2019). Sleep and sleep disordered breathing in children with down syndrome: Effects on behaviour, neurocognition and the cardiovascular system. [Review]. Sleep Medicine Reviews, 44, 1–11. https://doi.org/10.1016/j.smrv.2018.11.002
Huggard, D., McGrane, F., Lagan, N., Roche, E., Balfe, J., Leahy, T. R., et al. (2018). Altered endotoxin responsiveness in healthy children with Down syndrome. BMC Immunology, 19(1), 31. https://doi.org/10.1186/s12865-018-0270-z
Hüls, A., Costa, A. C. S., Dierssen, M., Asaad Baksh, R., Bargagna, S., Baumer, N. T., et al. (2021). Medical vulnerability of individuals with Down syndrome to severe COVID-19—data from the Trisomy 21 Research Society and the UK ISARIC4C survey. EClinicalMedicine. https://doi.org/10.1016/j.eclinm.2021.100769
Inoue, Y., Tanaka, N., Tanaka, Y., Inoue, S., Morita, K., Zhuang, M., et al. (2007). Clathrin-dependent entry of severe acute respiratory syndrome coronavirus into target cells expressing ACE2 with the cytoplasmic tail deleted. Journal of Virology, 81(16), 8722–8729. https://doi.org/10.1128/JVI.00253-07
Jenkins, E. C., Ye, L., Gu, H., Ni, S. A., Duncan, C. J., Velinov, M., et al. (2008). Increased “absence” of telomeres may indicate Alzheimer’s disease/dementia status in older individuals with Down syndrome. Neuroscience Letters, 440(3), 340–343. https://doi.org/10.1016/j.neulet.2008.05.098
Jenkins, E. C., Ye, L., Gu, H., Ni, S. A., Velinov, M., Pang, D., et al. (2010). Shorter telomeres may indicate dementia status in older individuals with Down syndrome. Neurobiology of Aging, 31(5), 765–771. https://doi.org/10.1016/j.neurobiolaging.2008.06.001
Jenkins, E. C., Ye, L., Velinov, M., Krinsky-McHale, S. J., Zigman, W. B., Schupf, N., et al. (2012). Mild cognitive impairment identified in older individuals with Down syndrome by reduced telomere signal numbers and shorter telomeres measured in microns. American Journal of Medical Genetics. Part B, Neuropsychiatric Genetics, 159B(5), 598–604. https://doi.org/10.1002/ajmg.b.32066
Jensen, H. A., Brown, K. L., Pagel, C., Barron, D. J., & Franklin, R. C. G. (2014). Mortality as a measure of quality of care in infants with congenital cardiovascular malformations following surgery. [Article]. British Medical Bulletin, 111(1), 5–15. https://doi.org/10.1093/bmb/ldu014
Jiang, Y., Rigoglioso, A., Peterhoff, C. M., Pawlik, M., Sato, Y., Bleiwas, C., et al. (2016). Partial BACE1 reduction in a Down syndrome mouse model blocks Alzheimer-related endosomal anomalies and cholinergic neurodegeneration: role of APP-CTF. Neurobiology of Aging, 39, 90–98. https://doi.org/10.1016/j.neurobiolaging.2015.11.013
Kanamori, G., Witter, M., Brown, J., & Williams-Smith, L. (2000). Otolaryngologic manifestations of Down syndrome. [Article]. Otolaryngologic Clinics of North America. https://doi.org/10.1016/s0030-6665(05)70281-4
Kantar, A., Mazza, A., Bonanomi, E., Odoni, M., Seminara, M., Verde, I. D., et al. (2020). COVID-19 and children with Down syndrome: is there any real reason to worry? Two case reports with severe course. BMC Pediatrics, 20(1), 561. https://doi.org/10.1186/s12887-020-02471-5
Kim, S., Sato, Y., Mohan, P. S., Peterhoff, C., Pensalfini, A., Rigoglioso, A., et al. (2016). Evidence that the rab5 effector APPL1 mediates APP-betaCTF-induced dysfunction of endosomes in Down syndrome and Alzheimer’s disease. Molecular Psychiatry, 21(5), 707–716. https://doi.org/10.1038/mp.2015.97
Kusters, M. A., Manders, N. C., de Jong, B. A., van Hout, R. W., Rijkers, G. T., & de Vries, E. (2013). Functionality of the pneumococcal antibody response in Down syndrome subjects. Vaccine, 31(52), 6261–6265. https://doi.org/10.1016/j.vaccine.2013.09.070
MacLean, G. A., McEldoon, J., Huang, J., Allred, J., Canver, M. C., & Orkin, S. H. (2018). Downregulation of Endothelin Receptor B Contributes to Defective B Cell Lymphopoiesis in Trisomy 21 Pluripotent Stem Cells. Sci Rep, 8(1), 8001. https://doi.org/10.1038/s41598-018-26123-y
Malle, L., Gao, C., Hur, C., Truong, H. Q., Bouvier, N. M., Percha, B., et al. (2020). Individuals with Down syndrome hospitalized with COVID-19 have more severe disease. Genetics in Medicine. https://doi.org/10.1038/s41436-020-01004-w
Marcus, C. L., Keens, T. G., Bautista, D. B., Vonpechmann, W. S., & Ward, S. L. D. (1991). Obstructive sleep-apnea in children with down-syndrome. [Article]. Pediatrics, 88(1), 132–139.
Mitra, S., El Azrak, M., McCord, H., & Paes, B. A. (2018). Hospitalization for Respiratory Syncytial Virus in Children with Down Syndrome Less than 2 Years of Age: A Systematic Review and Meta-Analysis. Journal of Pediatrics, 203(92–100), e103. https://doi.org/10.1016/j.jpeds.2018.08.006
Newman, A. M., Jhaveri, R., Patel, A. B., Tan, T. Q., Toia, J. M., & Arshad, M. (2021). Trisomy 21 and Coronavirus Disease 2019 in Pediatric Patients. Journal of Pediatrics, 228, 294–296. https://doi.org/10.1016/j.jpeds.2020.08.067
Ovsyannikova, I. G., Haralambieva, I. H., Crooke, S. N., Poland, G. A., & Kennedy, R. B. (2020). The role of host genetics in the immune response to SARS-CoV-2 and COVID-19 susceptibility and severity. Immunological Reviews. https://doi.org/10.1111/imr.12897
Palaiodimos, L., Kokkinidis, D. G., Li, W., Karamanis, D., Ognibene, J., Arora, S., et al. (2020). Severe obesity is associated with higher in-hospital mortality in a cohort of patients with COVID-19 in the Bronx. New York. Metabolism: clinical and experimental. https://doi.org/10.1016/j.metabol.2020.154262
Paoloni-Giacobino, A., Chen, H., Peitsch, M. C., Rossier, C., & Antonarakis, S. E. (1997). Cloning of the TMPRSS2 gene, which encodes a novel serine protease with transmembrane, LDLRA, and SRCR domains and maps to 21q22.3. Genomics, 44(3), 309–320. https://doi.org/10.1006/geno.1997.4845
Patel, A., Yamashita, N., Ascano, M., Bodmer, D., Boehm, E., Bodkin-Clarke, C., et al. (2015). RCAN1 links impaired neurotrophin trafficking to aberrant development of the sympathetic nervous system in Down syndrome. Nature Communications, 6, 10119. https://doi.org/10.1038/ncomms10119
Penrose, L. S. (1949). The incidence of mongolism in the general population. The Journal of Mental Science, 95(400), 685–688. https://doi.org/10.1192/bjp.95.400.685
Perez-Padilla, R., Fernandez, R., Garcia-Sancho, C., Franco-Marina, F., Aburto, O., Lopez-Gatell, H., et al. (2010). Pandemic (H1N1) 2009 virus and Down syndrome patients. Emerging Infectious Diseases, 16(8), 1312–1314. https://doi.org/10.3201/eid1608.091931
Pontes, G. C. L., Lobato, A. C. R., da Silva, S. M., da Silva, D. F. A., da Silva, A. F., Rocha, R. S. B., et al. (2020). COVID-19 in a Down Syndrome Newborn (2020).
Schoch, J., Rohrer, T. R., Kaestner, M., Abdul-Khaliq, H., Gortner, L., Sester, U., et al. (2017). Quantitative, Phenotypical, and Functional Characterization of Cellular Immunity in Children and Adolescents With Down Syndrome. Journal of Infectious Diseases, 215(10), 1619–1628. https://doi.org/10.1093/infdis/jix168
Shay, J. W. (2018). Telomeres and aging. Current Opinion in Cell Biology, 52, 1–7. https://doi.org/10.1016/j.ceb.2017.12.001
Trotta, M. B., Serro Azul, J. B., Wajngarten, M., Fonseca, S. G., Goldberg, A. C., & Kalil, J. E. (2011). Inflammatory and Immunological parameters in adults with Down syndrome. Immun Ageing, 8(1), 4. https://doi.org/10.1186/1742-4933-8-4
Trucco, F., Chatwin, M., Semple, T., Rosenthal, M., Bush, A., & Tan, H. L. (2018). Sleep disordered breathing and ventilatory support in children with Down syndrome. [Article]. Pediatric Pulmonology, 53(10), 1414–1421. https://doi.org/10.1002/ppul.24122
Turk, M. A., Landes, S. D., Formica, M. K., & Goss, K. D. (2020). Intellectual and developmental disability and COVID-19 case-fatality trends: TriNetX analysis. Disability and Health Journal, 13(3), 100942. https://doi.org/10.1016/j.dhjo.2020.100942
Verstegen, R. H., van Hout, R. W., & de Vries, E. (2014). Epidemiology of respiratory symptoms in children with Down syndrome: a nationwide prospective web-based parent-reported study. BMC Pediatrics, 14, 103. https://doi.org/10.1186/1471-2431-14-103
Weijerman, M. E., van Furth, A. M., van der Mooren, M. D., van Weissenbruch, M. M., Rammeloo, L., Broers, C. J. M., et al. (2010). Prevalence of congenital heart defects and persistent pulmonary hypertension of the neonate with Down syndrome. [Article]. European Journal of Pediatrics, 169(10), 1195–1199. https://doi.org/10.1007/s00431-010-1200-0
Zampieri, B. L., Biselli-Perico, J. M., de Souza, J. E., Burger, M. C., Silva Junior, W. A., Goloni-Bertollo, E. M., et al. (2014). Altered expression of immune-related genes in children with Down syndrome. PLoS ONE, 9(9), e107218. https://doi.org/10.1371/journal.pone.0107218
Zhang, L., Zhang, F., Yu, W., He, T., Yu, J., Yi, C. E., et al. (2006). Antibody responses against SARS coronavirus are correlated with disease outcome of infected individuals. Journal of Medical Virology, 78(1), 1–8. https://doi.org/10.1002/jmv.20499
Zigman, W. B. (2013). Atypical aging in Down syndrome. Developmental Disabilities Research Reviews, 18(1), 51–67. https://doi.org/10.1002/ddrr.1128
Acknowledgement
This research was supported by the Trisomy-21 Research Society (T21RS). This research was supported in part by the Intramural Research Program of the National Institute on Aging. We thank Yael Laure for thorough editing of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Research involving Human Participants
The study protocol was approved by the Shamir Medical Center Review Board and the Research Committee of Leumit Health Services (LHS) as follows:
-
The International Classification of Disease, 9th revision (ICD-9) codes were used for identification of comorbidities.
-
Chronic Lung disease ICD-9 codes for asthma (493.00–493.92) and chronic bronchitis (490–496).
-
Diabetes–ICD-9 codes 250.00- 250.93.
-
Arterial hypertension–ICD-9 codes 401- 405.
-
Obesity was considered as a measurement of BMI > 30 m2/kg.
-
Depression–ICD‐9 codes: 296.20‐296.25, 296.30‐296.35, 300.4, 311, 309.0,309.28.
-
Dementia–ICD‐9 codes: 290.0 -290.4, 331.00, 331.1.
-
Ischemic heart disease–ICD‐9 codes: 410–414.
-
Congestive heart failure- ICD‐9 codes: 428.0–428.9.
-
Socioeconomic status (SES) data was taken from the Israeli Central Bureau of Statistics classification that includes 20 sub-groups, according to the home address. Classifications one to nine were considered low-medium SES, and 10–20 high SES (17).
COVID-19 and DS, AD, cardiovascular diseases, thyroid cancer and leukemia.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this article was revised: The original version of this article unfortunately contained mistakes. The author name Maria del Mar Dierssen Sotos should read as Mara Dierssen and the author's affiliations should appear as follows: Affiliation 11: Center for Genomic Regulation, The Barcelona Institute for Science and Technology, Barcelona, Spain. Affiliation 12: University Pompeu Fabra, Barcelona, Spain. Affiliation 13: Biomedical Research Networking Center for Rare Diseases (CIBERER), Barcelona, Spain. The original article has been corrected.
The original online version of this article was revised: In this article the Acknowledgement section should read as follows: Acknowledgement This research was supported by the Trisomy-21 Research Society (T21RS). This research was supported in part by the Intramural Research Program of the National Institute on Aging. We thank Yael Laure for thorough editing of the manuscript. The original article has been corrected.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Illouz, T., Biragyn, A., Frenkel-Morgenstern, M. et al. Specific Susceptibility to COVID-19 in Adults with Down Syndrome. Neuromol Med 23, 561–571 (2021). https://doi.org/10.1007/s12017-021-08651-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12017-021-08651-5