Abstract
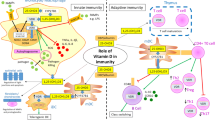
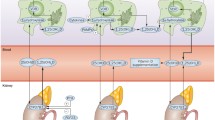
Vitamin D (VitD) is a hormone primarily synthesized in human skin under the stimulation of ultraviolet radiation. Beyond its endocrine role in bone metabolism, VitD is endowed with remarkable immunomodulatory properties. The effects of VitD on the immune system include the enhancement of microbicidal ability of monocytes/macrophages and the down-modulation of inflammatory cytokines produced by T lymphocytes. VitD deficiency is involved in many health problems, including immune-mediated diseases such as autoimmune disorders. Rheumatoid arthritis (RA) is a chronic inflammatory systemic autoimmune disease that compromises the joints, causing cartilage destruction and bone erosion. RA treatment usually consists of combined therapies that generally suppress the entire immune response leading to increased susceptibility to infections. This review describes the main effects of VitD on innate and adaptive immune system and also VitD status in inflammatory rheumatic diseases such as RA. Despite some controversies, the majority of reports reinforce the idea that lower VitD levels correlate with more severe clinical manifestations in RA and other rheumatic diseases. Therefore, supplementation with VitD to achieve normal serum levels is worthwhile as an aforethought. Original data concerning the potential applicability of 1,25-dihydroxyvitamin D3 (VitD3), the active form of vitamin D, as a tolerogenic adjuvant are also included. In this sense, the effect of VitD3 associated with proteoglycan (PG), which is a specific cartilage antigen, was tested in the course of experimental arthritis. This association significantly lowered clinical scores and local histopathological alterations. Even though local analysis of T cell subsets and cytokine production did not reveal any difference between the experimental groups, VitD3+PG association significantly reduced cytokine production by spleen cells. These results suggest that VitD3 played a role as a tolerogenic adjuvant by down-modulating the course of experimental RA. Considering this tolerogenic effect of VitD3+PG association, further investigations will reveal its plausible use in human RA.







Similar content being viewed by others
References
Lamberg-Allardt C (2006) Vitamin D in foods and as supplements. Prog Biophys Mol Biol 92:33–38. doi:10.1016/j.pbiomolbio.2006.02.017
Aranow C (2011) Vitamin D and the immune system. J Investig Med 59:881–886. doi:10.231/JIM.0b013e31821b8755
Holick MF (2003) Vitamin D: a millennium perspective. J Cell Biochem 88:296–307. doi:10.1002/jcb.10338
Dusso AS, Brown AJ, Slatopolsky E (2005) Vitamin D. Am J Physiol Renal Physiol 289:F8–28. doi:10.1152/ajprenal.00336.2004
Zhang J, Habiel DM, Ramadass M, Kew RR (2010) Identification of two distinct cell binding sequences in the vitamin D binding protein. Biochim Biophys Acta 1803:623–639. doi:10.1016/j.bbamcr.2010.02.010
Suzuki Y, Landowski CP, Hediger MA (2008) Mechanisms and regulation of epithelial Ca2+ absorption in health and disease. Annu Rev Physiol 70:257–271. doi:10.1146/annurev.physiol.69.031905.161003
Holick MF (2011) Vitamin D deficiency in 2010: health benefits of vitamin D and sunlight: a D-bate. Nat Rev Endocrinol 7:73–75. doi:10.1038/nrendo.2010.234
Holick MF, Binkley NC, Bischoff-Ferrari HA, Gordon CM, Hanley DA, Heaney RP, Murad MH, Weaver CM, Endocrine Society (2011) Evaluation, treatment, and prevention of vitamin D deficiency: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab 96:1911–1930. doi:10.1210/jc.2011-0385
Adorini L, Penna G (2009) Dendritic cell tolerogenicity: a key mechanism in immunomodulation by vitamin D receptor agonists. Hum Immunol 70:345–352. doi:10.1016/j.humimm.2009.01.016
Norman AW, Okamura WH, Bishop JE, Henry HL (2002) Update on biological actions of 1alpha,25(OH)2-vitamin D3 (rapid effects) and 24R,25(OH)2-vitamin D3. Mol Cell Endocrinol 197:1–13. doi:10.1016/S0303-7207(02)00273-3
Kongsbak M, Levring TB, Geisler C, von Essen MR (2013) The vitamin D receptor and T cell function. Front Immunol 4:148. doi:10.3389/fimmu.2013.0014
Carlberg C, Polly P (1998) Gene regulation by vitamin D3. Crit Rev Eukaryot Gene Expr 8:19–42. doi:10.1615/CritRevEukarGeneExpr.v8.i1.20
Holick MF (2007) Vitamin D deficiency. N Engl J Med 357:266–281. doi:10.1056/NEJMra070553
Agmon-Levin N, Theodor E, Segal RM, Shoenfeld Y (2014) Vitamin D in systemic and organ-specific autoimmune diseases. Clin Rev Allergy Immunol 45:256–266. doi:10.1007/s12016-012-8342-y
Kroner Jde C, Sommer A, Fabri M (2015) Vitamin D every day to keep the infection away? Nutrients 7:4170–4188. doi:10.3390/nu7064170
Nouari W, Ysmail-Dahlouk L, Aribi M (2016) Vitamin D3 enhances bactericidal activity of macrophage against Pseudomonas aeruginosa. Int Immunopharmacol 30:94–101. doi:10.1016/j.intimp.2015.11.033
Di Rosa M, Malaguarnera M, Nicoletti F, Malaguarnera L (2011) Vitamin D3: a helpful immuno-modulator. Immunology 134:123–139. doi:10.1111/j.1365-2567.2011.03482.x
Cantorna MT, Snyder L, Lin YD, Yang L (2015) Vitamin D and 1,25(OH)2D regulation of T cells. Nutrients 7:3011–3021. doi:10.3390/nu7043011
Lemire JM, Archer DC, Beck L, Spiegelberg HL (1995) Immunosuppressive actions of 1,25-dihydroxyvitamin D3: preferential inhibition of Th1 functions. J Nutr 125:1704S–1708S
Boonstra A, Barrat FJ, Crain C, Heath VL, Savelkoul HF, O’Garra A (2001) 1alpha,25-Dihydroxyvitamin d3 has a direct effect on naive CD4(+) T cells to enhance the development of Th2 cells. J Immunol 167:4974–4980. doi:10.4049/jimmunol.167.9.4974
Staeva-Vieira TP, Freedman LP (2002) 1,25-dihydroxyvitamin D3 inhibits IFN-gamma and IL-4 levels during in vitro polarization of primary murine CD4+ T cells. J Immunol 168:1181–1189. doi:10.4049/jimmunol.168.3.1181
Singh RP, Hasan S, Sharma S, Nagra S, Yamaguchi DT, Wong DT, Hahn BH, Hossain A (2014) Th17 cells in inflammation and autoimmunity. Autoimmun Rev 13:1174–1181. doi:10.1016/j.autrev.2014.08.019
Fawaz L, Mrad MF, Kazan JM, Sayegh S, Akika R, Khoury SJ (2016) Comparative effect of 25(OH)D3 and 1,25(OH)2D3 on Th17 cell differentiation. Clin Immunol 166–167:59–71. doi:10.1016/j.clim.2016.02.011
Tang J, Zhou R, Luger D, Zhu W, Silver PB, Grajewski RS, Su SB, Chan CC, Adorini L, Caspi RR (2009) Calcitriol suppresses antiretinal autoimmunity through inhibitory effects on the Th17 effector response. J Immunol 182:4624–4632. doi:10.4049/jimmunol.0801543
Chang JH, Cha HR, Lee DS, Seo KY, Kweon MN (2010) 1,25-Dihydroxyvitamin D3 inhibits the differentiation and migration of T(H)17 cells to protect against experimental autoimmune encephalomyelitis. PLoS One 5:e12925. doi:10.1371/journal.pone.0012925
Berer A, Stöckl J, Majdic O, Wagner T, Kollars M, Lechner K, Geissler K, Oehler L (2000) 1,25-Dihydroxyvitamin D(3) inhibits dendritic cell differentiation and maturation in vitro. Exp Hematol 28:575–583. doi:10.1016/S0301-472X(00)00143-0
Adler HS, Steinbrink K (2007) Tolerogenic dendritic cells in health and disease: friend and foe! Eur J Dermatol 17:476–491. doi:10.1684/ejd.2007.0262
Sakaguchi S, Miyara M, Costantino CM, Hafler DA (2010) FOXP3+ regulatory T cells in the human immune system. Nat Rev Immunol 10:490–500. doi:10.1038/nri2785
Penna G, Roncari A, Amuchastegui S, Daniel KC, Berti E, Colonna M, Adorini L (2005) Expression of the inhibitory receptor ILT3 on dendritic cells is dispensable for induction of CD4+Foxp3+ regulatory T cells by 1,25-dihydroxyvitamin D3. Blood 106:3490–3497. doi:10.1182/blood-2005-05-2044
Gregori S, Giarratana N, Smiroldo S, Uskokovic M, Adorini L (2002) A 1alpha,25-dihydroxyvitamin D(3) analog enhances regulatory T-cells and arrests autoimmune diabetes in NOD mice. Diabetes 51:1367–1374. doi:10.2337/diabetes.51.5.1367
Smolders J, Thewissen M, Peelen E, Menheere P, Tervaert JW, Damoiseaux J, Hupperts R (2009) Vitamin D status is positively correlated with regulatory T cell function in patients with multiple sclerosis. PLoS One 4:e6635. doi:10.1371/journal.pone.0006635
Spach KM, Nashold FE, Dittel BN, Hayes CE (2006) IL-10 signaling is essential for 1,25-dihydroxyvitamin D3-mediated inhibition of experimental autoimmune encephalomyelitis. J Immunol 177:6030–6037. doi:10.4049/jimmunol.177.9.6030
Pelajo CF, Lopez-Benitez JM, Miller LC (2010) Vitamin D and autoimmune rheumatologic disorders. Autoimmun Rev 9:507–510. doi:10.1016/j.autrev.2010.02.011
Ponsonby AL, Lucas RM, van der Mei IA (2005) UVR, vitamin D and three autoimmune diseases—multiple sclerosis, type 1 diabetes, rheumatoid arthritis. Photochem Photobiol 81:1267–1275. doi:10.1562/2005-02-15-IR-441
Grazio S, Naglić ĐB, Anić B, Grubišić F, Bobek D, Bakula M, Kavanagh HS, Kuna AT, Cvijetić S (2015) Vitamin D serum level, disease activity and functional ability in different rheumatic patients. Am J Med Sci 349:46–49. doi:10.1097/MAJ.0000000000000340
Durmus B, Altay Z, Baysal O, Ersoy Y (2012) Does vitamin D affect disease severity in patients with ankylosing spondylitis? Chin Med J (Engl) 125:2511–2515. doi:10.3760/cma.j.issn.0366-6999.2012.14.021
Zold E, Szodoray P, Gaal J, Kappelmayer J, Csathy L, Gyimesi E, Zeher M, Szegedi G, Bodolay E (2008) Vitamin D deficiency in undifferentiated connective tissue disease. Arthritis Res Ther 10:R123. doi:10.1186/ar2533
Touma Z, Eder L, Zisman D, Feld J, Chandran V, Rosen CF, Shen H, Cook RJ, Gladman DD (2011) Seasonal variation in vitamin D levels in psoriatic arthritis patients from different latitudes and its association with clinical outcomes. Arthritis Care Res (Hoboken) 63:1440–1447. doi:10.1002/acr.20530
Gatenby P, Lucas R, Swaminathan A (2013) Vitamin D deficiency and risk for rheumatic diseases: an update. Curr Opin Rheumatol 25:184–191. doi:10.1097/BOR.0b013e32835cfc16
Caramaschi P, Dalla Gassa A, Ruzzenente O, Volpe A, Ravagnani V, Tinazzi I, Barausse G, Bambara LM, Biasi D (2010) Very low levels of vitamin D in systemic sclerosis patients. Clin Rheumatol 29:1419–1425. doi:10.1007/s10067-010-1478-3
Arnson Y, Amital H, Agmon-Levin N, Alon D, Sánchez-Castañón M, López-Hoyos M, Matucci-Cerinic M, Szücs G, Shapira Y, Szekanecz Z, Shoenfeld Y (2011) Serum 25-OH vitamin D concentrations are linked with various clinical aspects in patients with systemic sclerosis: a retrospective cohort study and review of the literature. Autoimmun Rev 10:490–494. doi:10.1016/j.autrev.2011.02.002
Bivona G, Agnello L, Pivetti A, Milano S, Scazzone C, Sasso BL, Ciaccio M (2016) Association between hypovitaminosis D and systemic sclerosis: true or fake? Clin Chim Acta 458:115–119. doi:10.1016/j.cca.2016.04.026
Cutolo M, Otsa K, Paolino S, Yprus M, Veldi T, Seriolo B (2009) Vitamin D involvement in rheumatoid arthritis and systemic lupus erythematosus. Ann Rheum Dis 68:446–447. doi:10.1136/ard.2008.093476
Mok CC, Birmingham DJ, Leung HW, Hebert LA, Song H, Rovin BH (2012) Vitamin D levels in Chinese patients with systemic lupus erythematosus: relationship with disease activity, vascular risk factors and atherosclerosis. Rheumatology (Oxford) 51:644–52. doi:10.1093/rheumatology/ker212
Azrielant S, Shoenfeld Y (2016) Eppur Si Muove: vitamin D is essential in preventing and modulating SLE. Lupus 25:563–572. doi:10.1177/0961203316629001
Gao CC, Liu SY, Wu ZZ, Li TF, Gao GM, Liu ZS, Zheng ZH (2016) Severe vitamin D deficiency increases the risk for moderate to severe disease activity in Chinese patients with SLE. Lupus. doi: 10.1177/0961203316635289
Watad A, Neumann SG, Soriano A, Amital H, Shoenfeld Y (2016) Vitamin D and systemic lupus erythematosus: myth or reality? Isr Med Assoc J 18:177–182
Robinson A, Tangpricha V, Yow E, Gurion R, Schanberg LE, McComsey GA (2014) Vitamin D status is a determinant of atorvastatin effect on carotid intima medial thickening progression rate in children with lupus: an Atherosclerosis Prevention in Pediatric Lupus Erythematosus (APPLE) sub-study. Lupus Sci Med 10:e000037. doi:10.1136/lupus-2014-000037
Schoindre Y, Jallouli M, Tanguy ML, Ghillani P, Galicier L, Aumaître O, Francès C, Le Guern V, Lioté F, Smail A, Limal N, Perard L, Desmurs-Clavel H, Le Thi HD, Asli B, Kahn JE, Sailler L, Ackermann F, Papo T, Sacré K, Fain O, Stirnemann J, Cacoub P, Leroux G, Cohen-Bittan J, Hulot JS, Lechat P, Musset L, Piette JC, Amoura Z, Souberbielle JC, Costedoat-Chalumeau N, Group PLUS (2014) Lower vitamin D levels are associated with higher systemic lupus erythematosus activity, but not predictive of disease flare-up. Lupus Sci Med 1:e000027. doi:10.1136/lupus-2014-000027
Petri M, Bello KJ, Fang H, Magder LS (2013) Vitamin D in systemic lupus erythematosus: modest association with disease activity and the urine protein-to-creatinine ratio. Arthritis Rheum 65:1865–71. doi:10.1002/art.37953
Mao S, Huang S (2014) Association between vitamin D receptor gene BsmI, FokI, ApaI and TaqI polymorphisms and the risk of systemic lupus erythematosus: a meta-analysis. Rheumatol Int 34:381–388. doi:10.1007/s00296-013-2898-6
Kuhn A, Wenzel J, Bijl M (2016) Lupus erythematosus revisited. Semin Immunopathol 38:97–112. doi:10.1007/s00281-015-0550-0
Tabasi N, Rastin M, Mahmoudi M, Ghoryani M, Mirfeizi Z, Rabe SZ, Reihani H (2015) Influence of vitamin D on cell cycle, apoptosis, and some apoptosis-related molecules in systemic lupus erythematosus. Iran J Basic Med Sci 18:1107–1111
Rosen Y, Daich J, Soliman I, Brathwaite E, Shoenfeld Y (2016) Vitamin D and autoimmunity. Scand J Rheumatol 18:1–9. doi:10.3109/03009742.2016.1151072
Quintero OL, Amador-Patarroyo MJ, Montoya-Ortiz G, Rojas-Villarraga A, Anaya JM (2012) Autoimmune disease and gender: plausible mechanisms for the female predominance of autoimmunity. J Autoimmun 38:J109–119. doi:10.1016/j.jaut.2011.10.003
Boissier MC, Semerano L, Challal S, Saidenberg-Kermanac’h N, Falgarone G (2012) Rheumatoid arthritis: from autoimmunity to synovitis and joint destruction. J Autoimmun 39:222–228. doi:10.1016/j.jaut.2012.05.021
Raczkiewicz A, Kisiel B, Kulig M, Tłustochowicz W (2015) Vitamin D status and its association with quality of life, physical activity, and disease activity in rheumatoid arthritis patients. J Clin Rheumatol 21:126–30. doi:10.1097/RHU.0000000000000233
Lin J, Liu J, Davies ML, Chen W (2016) Serum vitamin D level and rheumatoid arthritis disease activity: review and meta-analysis. PLoS One 11(1):e0146351. doi:10.1371/journal.pone.0146351
Gopinath K, Danda D (2011) Supplementation of 1,25-dihydroxyvitamin D3 in patients with treatment naive early rheumatoid arthritis: a randomized controlled trial. Int J Rheum Dis 14:332–339. doi:10.1111/j.1756-185X.2011.01684.x
Salesi M, Farajzadegan Z (2012) Efficacy of vitamin D in patients with active rheumatoid arthritis receiving methotrexate therapy. Rheumatol Int 32:2129–33. doi:10.1007/s00296-011-1944-5
Abrahamsen B, Harvey NC (2013) The role of vitamin D supplementation in patients with rheumatic diseases. Nat Rev Rheumatol 9:411–422. doi:10.1038/nrrheum.2013.71
Rossini M, Bagnato G, Frediani B, Iagnocco A, LA Montagna G, Minisola G, Caminiti M, Varenna M, Adami S (2011) Relationship of focal erosions, bone mineral density, and parathyroid hormone in rheumatoid arthritis. J Rheumatol 38:997–1002. doi:10.3899/jrheum.100829
Firestein GS (2003) Evolving concepts of rheumatoid arthritis. Nature 423:356–361. doi:10.1038/nature01661
Kinne RW, Stuhlmüller B, Burmester GR (2007) Cells of the synovium in rheumatoid arthritis. Macrophages. Arthritis Res Ther 9:224. doi:10.1186/ar2333
Valencia X, Stephens G, Goldbach-Mansky R, Wilson M, Shevach EM, Lipsky PE (2006) TNF down-modulates the function of human CD4+CD25hi T-regulatory cells. Blood 108:253–261. doi:10.1182/blood-2005-11-4567
Brzustewicz E, Bryl E (2015) The role of cytokines in the pathogenesis of rheumatoid arthritis—practical and potential application of cytokines as biomarkers and targets of personalized therapy. Cytokine 76:527–36. doi:10.1016/j.cyto.2015.08.260
Mateen S, Zafar A, Moin S, Khan AQ, Zubair S (2016) Understanding the role of cytokines in the pathogenesis of rheumatoid arthritis. Clin Chim Acta 455:161–171. doi:10.1016/j.cca.2016.02.010
Tanaka Y (2016) Current concepts in the management of rheumatoid arthritis. Korean J Intern Med 31:210–218. doi:10.3904/kjim.2015.137
Hopp AK, Rupp A, Lukacs-Kornek V (2014) Self-antigen presentation by dendritic cells in autoimmunity. Front Immunol 5:55. doi:10.3389/fimmu.2014.00055
Catrina AI, Joshua V, Klareskog L, Malmström V (2016) Mechanisms involved in triggering rheumatoid arthritis. Immunol Rev 269:162–174. doi:10.1111/imr.12379
Lutzky V, Hannawi S, Thomas R (2007) Cells of the synovium in rheumatoid arthritis. Dendritic cells Arthritis Res Ther 9:219. doi:10.1186/ar2200
Martinez-Gamboa L, Brezinschek HP, Burmester GR, Dörner T (2006) Immunopathologic role of B lymphocytes in rheumatoid arthritis: rationale of B cell-directed therapy. Autoimmun Rev 5:437–442. doi:10.1016/j.autrev.2006.02.004
Aletaha D, Blüml S (2016) Therapeutic implications of autoantibodies in rheumatoid arthritis. RMD Open 2:e000009. doi:10.1136/rmdopen-2014-000009
van der Helm-van Mil AH, Verpoort KN, Breedveld FC, Huizinga TW, Toes RE, de Vries RR (2006) The HLA-DRB1 shared epitope alleles are primarily a risk factor for anti-cyclic citrullinated peptide antibodies and are not an independent risk factor for the development of rheumatoid arthritis. Arthritis Rheum 54:1117–1121. doi:10.1002/art.21739
Lundy SK, Sarkar S, Tesmer LA, Fox DA (2007) Cells of the synovium in rheumatoid arthritis. T lymphocytes Arthritis Res Ther 9:202. doi:10.1186/ar2107
Komatsu N, Takayanagi H (2015) Arthritogenic T cells in autoimmune arthritis. Int J Biochem Cell Biol 58:92–96. doi:10.1016/j.biocel.2014.11.008
Prots I, Skapenko A, Wendler J, Mattyasovszky S, Yoné CL, Spriewald B, Burkhardt H, Rau R, Kalden JR, Lipsky PE, Schulze-Koops H (2006) Association of the IL4R single-nucleotide polymorphism I50V with rapidly erosive rheumatoid arthritis. Arthritis Rheum 54:1491–1500. doi:10.1002/art.21832
Kleinewietfeld M, Hafler DA (2013) The plasticity of human Treg and Th17 cells and its role in autoimmunity. Semin Immunol 25:305–312. doi:10.1016/j.smim.2013.10.009
Chabaud M, Durand JM, Buchs N, Fossiez F, Page G, Frappart L, Miossec P (1999) Human interleukin-17: a T cell-derived proinflammatory cytokine produced by the rheumatoid synovium. Arthritis Rheum 42:963–970. doi:10.1002/1529-0131(199905)42:5<963::AID-ANR15>3.0.CO;2-E
Ehrenstein MR, Evans JG, Singh A, Moore S, Warnes G, Isenberg DA, Mauri C (2004) Compromised function of regulatory T cells in rheumatoid arthritis and reversal by anti-TNF-alpha therapy. J Exp Med 200:277–285. doi:10.1084/jem.20040165
van Amelsfort JM, Jacobs KM, Bijlsma JW, Lafeber FP, Taams LS (2004) CD4(+)CD25(+) regulatory T cells in rheumatoid arthritis: differences in the presence, phenotype, and function between peripheral blood and synovial fluid. Arthritis Rheum 50:2775–2785. doi:10.1002/art.20499
Turner JD, Filer A (2015) The role of the synovial fibroblast in rheumatoid arthritis pathogenesis. Curr Opin Rheumatol 27:175–182. doi:10.1097/BOR.0000000000000148
Otero M, Goldring MB (2007) Cells of the synovium in rheumatoid arthritis. Chondrocytes Arthritis Res Ther 9:220. doi:10.1186/ar2292
Schett G (2007) Cells of the synovium in rheumatoid arthritis. Osteoclasts. Arthritis Res Ther 9:203. doi:10.1186/ar2110
Jeffery LE, Raza K, Hewison M (2016) Vitamin D in rheumatoid arthritis—towards clinical application. Nat Rev Rheumatol 12:201–210. doi:10.1038/nrrheum.2015.140
Tengvall S, Eneljung T, Jirholt P, Turesson O, Wing K, Holmdahl R, Kihlberg J, Stern A, Mårtensson IL, Henningsson L, Gustafsson K, Gjertsson I (2016) Gene therapy induces antigen-specific tolerance in experimental collagen-induced arthritis. PLoS One 11:e0154630. doi:10.1371/journal.pone.0154630
Fousteri G, Jofra T, Di Fonte R, Battaglia M (2015) Combination of an antigen-specific therapy and an immunomodulatory treatment to simultaneous block recurrent autoimmunity and alloreactivity in non-obese diabetic mice. PLoS One 10:e0127631. doi:10.1371/journal.pone.0127631
Kleist C, Mohr E, Gaikwad S, Dittmar L, Kuerten S, Platten M, Mier W, Schmitt M, Opelz G, Terness P (2016) Autoantigen-specific immunosuppression with tolerogenic peripheral blood cells prevents relapses in a mouse model of relapsing-remitting multiple sclerosis. J Transl Med 14:99. doi:10.1186/s12967-016-0860-6
Harrison LC, Hafler DA (2000) Antigen-specific therapy for autoimmune disease. Curr Opin Immunol 12:704–711. doi:10.1016/S0952-7915(00)00166-7
Torres-Aguilar H, Blank M, Jara LJ, Shoenfeld Y (2010) Tolerogenic dendritic cells in autoimmune diseases: crucial players in induction and prevention of autoimmunity. Autoimmun Rev 10:8–17. doi:10.1016/j.autrev.2010.07.015
Hackstein H, Thomson AW (2004) Dendritic cells: emerging pharmacological targets of immunosuppressive drugs. Nat Rev Immunol 4:24–34. doi:10.1038/nri1256
Adorini L, Giarratana N, Penna G (2004) Pharmacological induction of tolerogenic dendritic cells and regulatory T cells. Semin Immunol 16:127–134. doi:10.1016/j.smim.2003.12.008
Chiuso-Minicucci F, Ishikawa LL, Mimura LA, Fraga-Silva TF, França TG, Zorzella-Pezavento SF, Marques C, Ikoma MR, Sartori A (2015) Treatment with vitamin D/MOG association suppresses experimental autoimmune encephalomyelitis. PLoS One 10:e0125836. doi:10.1371/journal.pone.0125836
Ishikawa LLW, Colavite PM, da Rosa LC, Balbino B, França TG, Zorzella-Pezavento SF, Chiuso-Minicucci F, Sartori A (2014) Commercial bovine proteoglycan is highly arthritogenic and can be used as an alternative antigen source for PGIA model. Biomed Res Int 2014:148594. doi:10.1155/2014/148594
Muehleisen B, Gallo RL (2013) Vitamin D in allergic disease: shedding light on a complex problem. J Allergy Clin Immunol 131:324–329. doi:10.1016/j.jaci.2012.12.1562
Cantorna MT, Hayes CE, DeLuca HF (1998) 1,25-Dihydroxycholecalciferol inhibits the progression of arthritis in murine models of human arthritis. J Nutr 128:68–72
Haga HJ (2013) Vitamin D in rheumatoid arthritis. Expert Rev Clin Immunol 9:591–593. doi:10.1586/1744666X.2013.811177
Pedersen AW, Claesson MH, Zocca MB (2011) Dendritic cells modified by vitamin D: future immunotherapy for autoimmune diseases. Vitam Horm 86:63–82. doi:10.1016/B978-0-12-386960-9.00003-4
Albert ML, Jegathesan M, Darnell RB (2001) Dendritic cell maturation is required for the cross-tolerization of CD8+ T cells. Nat Immunol 2:1010–1017. doi:10.1038/ni722
Menges M, Rössner S, Voigtländer C, Schindler H, Kukutsch NA, Bogdan C, Erb K, Schuler G, Lutz MB (2002) Repetitive injections of dendritic cells matured with tumor necrosis factor alpha induce antigen-specific protection of mice from autoimmunity. J Exp Med 195:15–21. doi:10.1084/jem.20011341
Parungo CP, Soybel DI, Colson YL, Kim SW, Ohnishi S, DeGrand AM, Laurence RG, Soltesz EG, Chen FY, Cohn LH, Bawendi MG, Frangioni JV (2007) Lymphatic drainage of the peritoneal space: a pattern dependent on bowel lymphatics. Ann Surg Oncol 14:286–298. doi:10.1245/s10434-006-9044-6
Jeffery LE, Wood AM, Qureshi OS, Hou TZ, Gardner D, Briggs Z, Kaur S, Raza K, Sansom DM (2012) Availability of 25-hydroxyvitamin D(3) to APCs controls the balance between regulatory and inflammatory T cell responses. J Immunol 189:5155–5164. doi:10.4049/jimmunol.1200786
Farias AS, Spagnol GS, Bordeaux-Rego P, Oliveira CO, Fontana AG, de Paula RF, Santos MP, Pradella F, Moraes AS, Oliveira EC, Longhini AL, Rezende AC, Vaisberg MW, Santos LM (2013) Vitamin D3 induces IDO+ tolerogenic DCs and enhances Treg, reducing the severity of EAE. CNS Neurosci Ther 19:269–277. doi:10.1111/cns.12071
Han GM, O'Neil-Andersen NJ, Zurier RB, Lawrence DA (2008) CD4+CD25high T cell numbers are enriched in the peripheral blood of patients with rheumatoid arthritis. Cell Immunol 253:92–101. doi:10.1016/j.cellimm.2008.05.007
Agrawal T, Gupta GK, Agrawal DK (2013) Vitamin D supplementation reduces airway hyper-responsiveness and allergic airway inflammation in a murine model. Clin Exp Allergy 43:672–683. doi:10.1111/cea.12102
He CS, Handzlik M, Fraser WD, Muhamad A, Preston H, Richardson A, Gleeson M (2013) Influence of vitamin D status on respiratory infection incidence and immune function during 4 months of winter training in endurance sport athletes. Exerc Immunol Rev 19:86–101
Czifra G, Tóth B, Kovács I, Bíró T, Griger Z, Baráth S, Tarr T, Zeher M, Sipka S (2014) The in vitro treatment with vitamin D3 is ineffective on the expression of PKC isoenzymes, but decreases further the impaired production of IL-2 in the T lymphocytes of SLE patients. Rheumatol Int 34:717–720. doi:10.1007/s00296-013-2751-y
Villaggio B, Soldano S, Cutolo M (2012) 1,25-Dihydroxyvitamin D3 downregulates aromatase expression and inflammatory cytokines in human macrophages. Clin Exp Rheumatol 30:934–938
Bemiss CJ, Mahon BD, Henry A, Weaver V, Cantorna MT (2002) Interleukin-2 is one of the targets of 1,25-dihydroxyvitamin D3 in the immune system. Arch Biochem Biophys 402:249–254. doi:10.1016/S0003-9861(02)00082-6
Niino M, Fukazawa T, Miyazaki Y, Takahashi E, Minami N, Amino I, Fujiki N, Doi S, Kikuchi S (2014) Suppression of IL-10 production by calcitriol in patients with multiple sclerosis. J Neuroimmunol 270:86–94. doi:10.1016/j.jneuroim.2014.03.011
Matilainen JM, Husso T, Toropainen S, Seuter S, Turunen MP, Gynther P, Ylä-Herttuala S, Carlberg C, Väisänen S (2010) Primary effect of 1α,25(OH) 2D3 on IL-10 expression in monocytes is short-term down-regulation. Biochim Biophys Acta 1803:1276–1286. doi:10.1016/j.bbamcr.2010.07.009
Moghaddami M, Mayrhofer G, Anderson PH, Morris HA, Van Der Hoek M, Cleland LG (2012) Efficacy and mechanisms of action of vitamin D in experimental polyarthritis. Immunol Cell Biol 90:168–177. doi:10.1038/icb.2011.22
Bansal AS, Henriquez F, Sumar N, Patel S (2012) T helper cell subsets in arthritis and the benefits of immunomodulation by 1,25(OH)2 vitamin D. Rheumatol Int 32:845-852. doi: 10.1007/s00296-011-2077-6
Kang Y, Xu L, Wang B, Chen A, Zheng G (2008) Cutting edge: immunosuppressant as adjuvant for tolerogenic immunization. J Immunol 180:5172–5176. doi:10.4049/jimmunol.180.8.5172
Kang Y, Zhao J, Liu Y, Chen A, Zheng G, Yu Y, Mi J, Zou Q, Wang B (2009) FK506 as an adjuvant of tolerogenic DNA vaccination for the prevention of experimental autoimmune encephalomyelitis. J Gene Med 11:1064–1070. doi:10.1002/jgm.1387
Mannie MD, Abbott DJ, Blanchfield JL (2009) Experimental autoimmune encephalomyelitis in Lewis rats: IFN-beta acts as a tolerogenic adjuvant for induction of neuroantigen-dependent tolerance. J Immunol 182:5331–5341. doi:10.4049/jimmunol.0803756
Colmegna I, Ohata BR, Menard HA (2012) Current understanding of rheumatoid arthritis therapy. Clin Pharmacol Ther 91:607–620. doi:10.1038/clpt.2011.325
Uysal H, Nandakumar KS, Kessel C, Haag S, Carlsen S, Burkhardt H, Holmdahl R (2010) Antibodies to citrullinated proteins: molecular interactions and arthritogenicity. Immunol Rev 233:9–33. doi:10.1111/j.0105-2896.2009.00853.x
Song YW, Kang EH (2010) Autoantibodies in rheumatoid arthritis: rheumatoid factors and anti-citrullinated protein antibodies. QJM 103:139–46. doi:10.1093/qjmed/hcp165
Bläss S, Engel JM, Burmester GR (2001) The immunologic homunculus in rheumatoid arthritis. A new viewpoint of immunopathogenesis in rheumatoid arthritis and therapeutic consequences. Z Rheumato 60:1–16. doi:10.1007/s003930170093
Penna G, Adorini L (2000) 1 Alpha,25-dihydroxyvitamin D3 inhibits differentiation, maturation, activation, and survival of dendritic cells leading to impaired alloreactive T cell activation. J Immunol 164:2405–2411. doi:10.4049/jimmunol.164.5.2405
Chen S, Sims GP, Chen XX, Gu YY, Chen S, Lipsky PE (2007) Modulatory effects of 1,25-dihydroxyvitamin D3 on human B cell differentiation. J Immunol 179:1634–1647. doi:10.4049/jimmunol.179.3.1634
Heine G, Niesner U, Chang HD, Steinmeyer A, Zügel U, Zuberbier T, Radbruch A, Worm M (2008) 1,25-Dihydroxyvitamin D(3) promotes IL-10 production in human B cells. Eur J Immunol 38:2210–2218. doi:10.1002/eji.200838216
Colin EM, Asmawidjaja PS, van Hamburg JP, Mus AM, van Driel M, Hazes JM, van Leeuwen JP, Lubberts E (2010) 1,25-Dihydroxyvitamin D3 modulates Th17 polarization and interleukin-22 expression by memory T cells from patients with early rheumatoid arthritis. Arthritis Rheum 62:132–142. doi:10.1002/art.25043
Jeffery LE, Qureshi OS, Gardner D, Hou TZ, Briggs Z, Soskic B, Baker J, Raza K, Sansom DM (2015) Vitamin D antagonizes the suppressive effect of inflammatory cytokines on CTLA-4 expression and regulatory function. PLoS One 10:e0131539. doi:10.1371/journal.pone.0131539
Laragione T, Shah A, Gulko PS (2012) The vitamin D receptor regulates rheumatoid arthritis synovial fibroblast invasion and morphology. Mol Med 18:194–200. doi:10.2119/molmed.2011.00410
Feng X, Lv C, Wang F, Gan K, Zhang M, Tan W (2013) Modulatory effect of 1,25-dihydroxyvitamin D3 on IL1 β -induced RANKL, OPG, TNF α, and IL-6 expression in human rheumatoid synoviocyte MH7A. Clin Dev Immunol 2013:160123. doi:10.1155/2013/160123
Tetlow LC, Woolley DE (1999) The effects of 1 alpha,25-dihydroxyvitamin D(3) on matrix metalloproteinase and prostaglandin E(2) production by cells of the rheumatoid lesion. Arthritis Res 1:63–70. doi:10.1186/ar12
Kim TH, Lee B, Kwon E, Choi CH, Sung IH, Kim Y, Sohn J, Ji JD (2013) 1,25-Dihydroxyvitamin D3 inhibits directly human osteoclastogenesis by down-regulation of the c-Fms and RANK expression. Joint Bone Spine 80:307–314. doi:10.1016/j.jbspin.2012.09.011
Acknowledgments
The authors are thankful to São Paulo State Foundation (FAPESP) for the financial support. Special thanks are given to Prof. Yehuda Shoenfeld, Mrs. Anna Varsanyi, and the Federico Fellows from the Zabludowicz Center for Autoimmune Diseases, Israel, for the scientific contribution.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Funding
This study was funded by São Paulo State Foundation (grant no. 2011/17325-4 and grant no. 2010/20470-3).
Ethical Approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. All procedures performed in studies involving animals were in accordance with the ethical standards of the institution or practice at which the studies were conducted.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
ESM 1
Supplementary material (DOC 35 kb)
Rights and permissions
About this article
Cite this article
Ishikawa, L.L.W., Colavite, P.M., Fraga-Silva, T.F.d.C. et al. Vitamin D Deficiency and Rheumatoid Arthritis. Clinic Rev Allerg Immunol 52, 373–388 (2017). https://doi.org/10.1007/s12016-016-8577-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12016-016-8577-0