Abstract
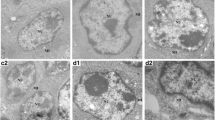
The purpose of this study was to investigate the histopathological lesions, oxidative damage, changes of immunoglobulin G (IgG), immunoglobulin M (IgM), and immunoglobulin A (IgA) contents in the bursa of Fabricius and serum immunoglobulins (IgG, IgM, IgA) induced by dietary nickel chloride (NiCl2). Two hundred and eighty-one-day-old broilers were randomly divided into four groups and fed on a control diet and three experimental diets supplemented with 300, 600, and 900 mg/kg of NiCl2 for 42 days. Lesions were observed in the NiCl2-treated groups. Histopathologically, lymphocytes were decreased in lymphoid follicles with thinner cortices and wider medullae. Concurrently, the activities of superoxide dismutase (SOD), catalase (CAT), glutathione peroxidase (GSH-Px), and the ability to inhibit hydroxyl radical and glutathione (GSH) contents were significantly (p < 0.05 or p < 0.01) decreased, while malondialdehyde (MDA) contents were increased in the NiCl2-treated groups. The serum IgG, IgM, and bursa IgG and IgM contents were significantly (p < 0.05 or p < 0.01) lower in the NiCl2-treated groups than those in the control group. The above-mentioned results show that dietary NiCl2 in excess of 300 mg/kg can cause histopathological lesions via oxidative damage, which finally impairs the function of the bursa of Fabricius and reduces IgG and IgM contents of the serum and the bursa of Fabricius. The study is aimed to provide helpful materials for studies on Ni- or Ni compounds-induced B cell toxicity in both human and other animals in the future.







Similar content being viewed by others
References
Organization WH (1991) GEMS—global fresh water quality. Oxford, UK, Blackwell Reference
Cempel M, Nikel G (2006) Nickel: a review of its sources and environmental toxicology. Pol J Environ Stud 15(3):375–382
Spears J (1984) Nickel as a “newer trace element” in the nutrition of domestic animals. J Anim Sci 59(3):823–835
Samal L, Mishra C (2011) Significance of nickel in livestock health and production. Inter J Agro Vet Med Sci 5(3):349–361
Grandjean P (1983) Human exposure to nickel. IARC Sci Publ 53:469–485
Haber LT, Erdreicht L, Diamond GL, Maier AM, Ratney R, Zhao Q, Dourson ML (2000) Hazard identification and dose response of inhaled nickel-soluble salts. Regul Toxicol Pharmacol 31(2 Pt 1):210–230. doi:10.1006/rtph.2000.1377
Lyon F (2014) IARC monographs on the evaluation of carcinogenic risks to humans
Donskoy E, Donskoy M, Forouhar F, Gillies CG, Marzouk A, Reid MC, Zaharia O, Sunderman Jr FW (1986) Hepatic toxicity of nickel chloride in rats. Ann Clin Lab Sci 16(2):108–117
Doreswamy K, Shrilatha B, Rajeshkumar T, Muralidhara (2004) Nickel-induced oxidative stress in testis of mice: evidence of DNA damage and genotoxic effects. J Androl 25(6):996–1003
Kasprzak KS, Sunderman Jr FW, Salnikow K (2003) Nickel carcinogenesis. Mutat Res 533(1–2):67–97
Haley PJ, Bice DE, Muggenburg BA, Hahn FF, Benjamin SA (1987) Immunopathologic effects of nickel subsulfide on the primate pulmonary immune system. Toxicol Appl Pharmacol 88(1):1–12
Mates JM, Segura JA, Alonso FJ, Marquez J (2012) Oxidative stress in apoptosis and cancer: an update. Arch Toxicol 86(11):1649–1665. doi:10.1007/s00204-012-0906-3
Avery Simon V (2011) Molecular targets of oxidative stress. Biochem J 434(2):201–210. doi:10.1042/bj20101695
Fang YZ, Yang S, Wu G (2002) Free radicals, antioxidants, and nutrition. Nutrition 18(10):872–879
Chakrabarti SK, Bai C (1999) Role of oxidative stress in nickel chloride-induced cell injury in rat renal cortical slices. Biochem Pharmacol 58(9):1501–1510
Misra M, Rodriguez RE, Kasprzak KS (1990) Nickel induced lipid peroxidation in the rat: correlation with nickel effect on antioxidant defense systems. Toxicology 64(1):1–17
Chen CY, Wang YF, Lin YH, Yen SF (2003) Nickel-induced oxidative stress and effect of antioxidants in human lymphocytes. Arch Toxicol 77(3):123–130. doi:10.1007/s00204-002-0427-6
Huang J, Cui H, Peng X, Fang J, Zuo Z, Deng J, Wu B (2013) The association between splenocyte apoptosis and alterations of Bax, Bcl-2 and caspase-3 mRNA expression, and oxidative stress induced by dietary nickel chloride in broilers. Int J Environ Res Public Health 10(12):7310–7326. doi:10.3390/ijerph10127310
Guo H, Wu B, Cui H, Peng X, Fang J, Zuo Z, Deng J, Wang X, Deng J, Yin S, Li J, Tang K (2014) NiCl2-down-regulated antioxidant enzyme mRNA expression causes oxidative damage in the broiler(’)s kidney. Biol Trace Elem Res 162(1–3):288–295. doi:10.1007/s12011-014-0132-3
Tang K, Li J, Yin S, Guo H, Deng J, Cui H (2014) Effects of nickel chloride on histopathological lesions and oxidative damage in the thymus. Health (N Y) 6(21):2875
Wu B, Cui H, Peng X, Fang J, Zuo Z, Deng J, Huang J (2014) Dietary nickel chloride induces oxidative stress, apoptosis and alters Bax/Bcl-2 and caspase-3 mRNA expression in the cecal tonsil of broilers. Food Chem Toxicol 63:18–29
Wu B, Cui H, Peng X, Fang J, Zuo Z, Deng J, Huang J (2013) Dietary nickel chloride induces oxidative intestinal damage in broilers. Int J Environ Res Public Health 10(6):2109–2119
Wu B, Cui H, Peng X, Fang J, Zuo Z, Deng J, Huang J (2013) Investigation of the serum oxidative stress in broilers fed on diets supplemented with nickel chloride. Health 5(3):454–459
Wu B, Cui H, Peng X, Fang J, Cui W, Liu X (2013) Pathology of bursae of Fabricius in methionine-deficient broiler chickens. Nutrients 5(3):877–886. doi:10.3390/nu5030877
Li J, Cao J, Wang Z, Dong Y, Chen Y (2015) Melatonin plays a critical role in inducing B lymphocyte proliferation of the bursa of Fabricius in broilers via monochromatic lights. J Photochem Photobiol B 142:29–34. doi:10.1016/j.jphotobiol.2014.11.002
Reynaud CA, Imhof BA, Anquez V, Weill JC (1992) Emergence of committed B lymphoid progenitors in the developing chicken embryo. Embo j 11(12):4349–4358
Benatar T, Tkalec L, Ratcliffe MJ (1992) Stochastic rearrangement of immunoglobulin variable-region genes in chicken B-cell development. Proc Natl Acad Sci U S A 89(16):7615–7619
Mockett AP (1986) Monoclonal antibodies used to isolate IgM from chicken bile and avian sera and to detect specific IgM in chicken sera. Avian Pathol 15(3):337–348. doi:10.1080/03079458608436297
Schroeder Jr HW, Cavacini L (2010) Structure and function of immunoglobulins. J Allergy Clin Immunol 125(2 Suppl 2):S41–S52. doi:10.1016/j.jaci.2009.09.046
Wieland WH, Orzaez D, Lammers A, Parmentier HK, Verstegen MW, Schots A (2004) A functional polymeric immunoglobulin receptor in chicken (Gallus gallus) indicates ancient role of secretory IgA in mucosal immunity. Biochem J 380(Pt 3):669–676. doi:10.1042/BJ20040200
Perez-Carbajal C, Caldwell D, Farnell M, Stringfellow K, Pohl S, Casco G, Pro-Martinez A, Ruiz-Feria CA (2010) Immune response of broiler chickens fed different levels of arginine and vitamin E to a coccidiosis vaccine and Eimeria challenge. Poult Sci 89(9):1870–1877. doi:10.3382/ps.2010-00753
Boes M (2000) Role of natural and immune IgM antibodies in immune responses. Mol Immunol 37(18):1141–1149
Hirano T, Yasukawa K, Harada H, Taga T, Watanabe Y, Matsuda T, Kashiwamura S, Nakajima K, Koyama K, Iwamatsu A, et al. (1986) Complementary DNA for a novel human interleukin (BSF-2) that induces B lymphocytes to produce immunoglobulin. Nature 324(6092):73–76. doi:10.1038/324073a0
NRC (1994) Nutrient requirements of poultry, 9th edn. Washington, DC, National Academy Press
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. doi:10.1016/0003-2697(76)90527-3
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(−delta delta C(T)) method. Methods 25(4):402–408. doi:10.1006/meth.2001.1262
Gaca MD, Pickering JA, Arthur MJ, Benyon RC (1999) Human and rat hepatic stellate cells produce stem cell factor: a possible mechanism for mast cell recruitment in liver fibrosis. J Hepatol 30(5):850–858
Cooper MD, Peterson RD, South MA, Good RA (2006) The functions of the thymus system and the bursa system in the chicken. 1966. J Immunol 176(11):6370–6404
Kubrak OI, Husak VV, Rovenko BM, Poigner H, Kriews M, Abele D, Lushchak VI (2013) Antioxidant system efficiently protects goldfish gills from Ni(2+)-induced oxidative stress. Chemosphere 90(3):971–976. doi:10.1016/j.chemosphere.2012.06.044
Amudha K, Pari L (2011) Beneficial role of naringin, a flavanoid on nickel induced nephrotoxicity in rats. Chem Biol Interact 193(1):57–64. doi:10.1016/j.cbi.2011.05.003
Wang M, Wang G (2010) Oxidative damage effects in the copepod Tigriopus japonicus Mori experimentally exposed to nickel. Ecotoxicology 19(2):273–284. doi:10.1007/s10646-009-0410-6
Liu CM, Zheng GH, Ming QL, Chao C, Sun JM (2013) Sesamin protects mouse liver against nickel-induced oxidative DNA damage and apoptosis by the PI3K-Akt pathway. J Agr Food Chem 61(5):1146–1154. doi:10.1021/jf304562b
Pari L, Amudha K (2011) Hepatoprotective role of naringin on nickel-induced toxicity in male Wistar rats. Eur J Pharmacol 650(1):364–370. doi:10.1016/j.ejphar.2010.09.068
Zheng GH, Liu CM, Sun JM, Feng ZJ, Cheng C (2014) Nickel-induced oxidative stress and apoptosis in Carassius auratus liver by JNK pathway. Aquat Toxicol 147:105–111. doi:10.1016/j.aquatox.2013.12.015
Ahamed M (2011) Toxic response of nickel nanoparticles in human lung epithelial A549 cells. Toxicol in Vitro 25(4):930–936. doi:10.1016/j.tiv.2011.02.015
Alarifi S, Ali D, Alakhtani S, Al Suhaibani ES, Al-Qahtani AA (2014) Reactive oxygen species-mediated DNA damage and apoptosis in human skin epidermal cells after exposure to nickel nanoparticles. Biol Trace Elem Res 157(1):84–93. doi:10.1007/s12011-013-9871-9
Attig H, Kamel N, Sforzini S, Dagnino A, Jamel J, Boussetta H, Viarengo A, Banni M (2014) Effects of thermal stress and nickel exposure on biomarkers responses in Mytilus galloprovincialis (Lam). Mar Environ Res 94:65–71. doi:10.1016/j.marenvres.2013.12.006
Rao MV, Chawla SL, Sharma SR (2009) Protective role of vitamin E on nickel and/or chromium induced oxidative stress in the mouse ovary. Food Chem Toxicol 47(6):1368–1371. doi:10.1016/j.fct.2009.03.018
Lou J, Jin L, Wu N, Tan Y, Song Y, Gao M, Liu K, Zhang X, He J (2013) DNA damage and oxidative stress in human B lymphoblastoid cells after combined exposure to hexavalent chromium and nickel compounds. Food Chem Toxicol 55:533–540. doi:10.1016/j.fct.2013.01.053
Chen F, Shi X (2002) Intracellular signal transduction of cells in response to carcinogenic metals. Crit Rev Oncol Hematol 42(1):105–121
Misra M, Rodriguez RE, North SL, Kasprzak KS (1991) Nickel-induced renal lipid peroxidation in different strains of mice: concurrence with nickel effect on antioxidant defense systems. Toxicol Lett 58(2):121–133
Chen CY, Sheu JY, Lin TH (1999) Oxidative effects of nickel on bone marrow and blood of rats. J Toxicol Environ Health A 58(8):475–483
Salnikow K, Gao M, Voitkun V, Huang X, Costa M (1994) Altered oxidative stress responses in nickel-resistant mammalian cells. Cancer Res 54(24):6407–6412
Tkeshelashvili LK, Reid TM, McBride TJ, Loeb LA (1993) Nickel induces a signature mutation for oxygen free radical damage. Cancer Res 53(18):4172–4174
Stohs SJ, Bagchi D (1995) Oxidative mechanisms in the toxicity of metal ions. Free Radic Biol Med 18(2):321–336
Hanly WC, Artwohl JE, Bennett BT (1995) Review of polyclonal antibody production procedures in mammals and poultry. Ilar j 37(3):93–118
Wu B, Cui H, Peng X, Fang J, Zuo Z, Deng J, Huang J (2014) Toxicological effects of nickel chloride on IgA+ B cells and sIgA, IgA, IgG, IgM in the intestinal mucosal immunity in broilers. Int J Environ Res Public Health 11(8):8175–8192
Huang J, Cui H, Peng X, Fang J, Zuo Z, Deng J, Wang X, Wu B (2014) Effect of dietary nickel chloride on splenic immune function in broilers. Biol Trace Elem Res 159(1–3):183–191. doi:10.1007/s12011-014-0003-y
Acknowledgments
The study was supported by the program for Changjiang scholars and innovative research team in the university (IRT 0848) and the Shuangzhi Project of Sichuan Agricultural University (03570327; 03571198).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no competing interests.
Rights and permissions
About this article
Cite this article
Yin, S., Guo, H., Cui, H. et al. Nickel Chloride (NiCl2) Induces Histopathological Lesions via Oxidative Damage in the Broiler’s Bursa of Fabricius. Biol Trace Elem Res 171, 214–223 (2016). https://doi.org/10.1007/s12011-015-0528-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-015-0528-8