Abstract
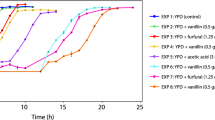
Lignocellulosic materials have been considered low-cost effective substrates for bioethanol production. However, lignocellulosic pretreatment releases toxic compounds such as 5-hydroxymethylfurfural (HMF) that is known to inhibit the yeast growth and ethanol production. In this work, flow cytometry was used to monitor the physiological response of the yeast Saccharomyces carlsbergensis ATCC 6269 in the presence of different initial HMF concentrations within the range of 0–15 g/L, in terms of cell membrane integrity, potential, and intracellular lipids. It was observed that the HMF presence affected more significantly the yeast growth than the ethanol production. At 15 g/L HMF, the yeast growth and fermentation ability were completely inhibited. The cell membrane integrity and potential decreased as the initial HMF concentration increased. At the end of the fermentation process with 10 g/L HMF, the yeast culture contained 45 % of cells with depolarized plasma membrane, 52 % of cells with permeabilized plasma membrane, and 53 % of cells with increasing reactive oxygen species (ROS) levels. Using the Nile Red stain, it was observed that intracellular polar lipids were more affected by the initial HMF concentration than the neutral lipids, probably due to the extensive membrane damage.





Similar content being viewed by others
Abbreviations
- μ :
-
Specific growth rate
- DHR:
-
Dihydrorhodamine 123
- DiOC6(3):
-
3,3-Dihexylocarbocyanine iodide
- FC:
-
Flow cytometry
- HMF:
-
5-Hydroxymethylfurfural
- NR:
-
Nile Red
- PI:
-
Propidium iodide
- ROS:
-
Reactive oxygen species
- X max :
-
Maximum biomass concentration
- Y X/glu :
-
Biomass yield
- YEtOH/glu :
-
Ethanol yield
References
Zheng-yun, W., Yu, D., Li, T., Yue-hing, L., Yi-jie, Z., & Wen-xue, Z. (2010). Investigating the effects of two lignocellulose degradation by-products (furfural and acetic acid) on ethanol fermentations by six ethanologenic yeast strains. African Journal of Biotechnology, 9, 8661–8666.
Almeida, J. R. M., Modig, T., Petersson, A., Hähn-Hägerdal, B., & Gorwa-Grauslund, M. F. (2007). Increased tolerance and conversion of inhibitors in lignocellulosic hydrolysates by Saccharomyces cerevisiae. Journal of Chemical Technology and Biotecnology, 23, 40–349.
Taherzadeh, M. J., Gustafsson, L., Niklasson, C., & Lidé, G. (2000). Physiological effects of 5-hydrpxymethylfurfural on Saccharomyces cerevisiae. Applied Microbiology and Biotechnology, 53, 701–708.
Oliva, J. M., Sáez, F., Ballesteros, I., González, A., Negro, M. J., Manzanares, P., & Ballesteros, M. (2003). Effect of lignocellulosic degradation compounds from steam explosion pretreatment on ethanol fermentation by thermotolerant yeast Kluyveromyces marxianus. Applied Biochemistry Biotechnology, 105-108, 141–153.
Liu, Z. L., Slininger, P. J., Dien, B. S., Berhow, M. A., Kurtzman, C. P., & Gorsich, S. W. (2004). Adaptative response of yeasts to furfural and 5-hydroxymethylfurfural and new chemical evidence for HMF conversion to 2,5-bis-hydroxymethylfuran. Journal of Industrial Microbiology and Biotechnology, 31, 345–352.
Bellido, C., Bolado, S., Coca, M., Lucas, S., González-Benito, G., & García-Cubero, M. T. (2011). Effect of inhibitors formed during wheat straw pretreatment on ethanol fermentation by Pichia stipitis. Bioresource Technology, 102, 10868–10874.
Freitas, C., Nunes, E., Passarinho, P. C., Reis, A., & Lopes da Silva, T. (2013). Use of multi-parameter flow cytometry as tool to monitor the impact of formic acid on Saccharomyces carlsbergensis batch ethanol fermentations. Applied Biochemistry and Biotechnology, 169, 2038–2048.
Freitas, C., Nunes, E., Passarinho, P. C., Reis, A., & Lopes da Silva, T. (2012). Effect of acetic acid on Saccharomyces carlsbergensis ATCC 6269 batch ethanol production monitored by flow cytometry (2012). Applied Biochemistry and Biotechnology, 168, 1501–1015.
Lopes da Silva, T., Feijão, D., & Reis, A. (2010). Using multi-parameter flow cytometry to monitor the yeast Rhodotorula glutinis CCMI 145 batch growth and oil production towards biodiesel. Applied Biochemistry and Biotechnology, 162, 2166–2176.
Modig, T., Liden, G., & Taherzadeh, M. J. (2002). Inhibition effects of furfural on alcohol dehydrogenase, aldehyde dehydrogenase and pyruvate dehydrogenase. Biochemical Journal, 363, 769–776.
Lee, H., Cho, D. H., Kim, Y. H., Shin, S. J., Kim, S. W., Han, S. O., Lee, J., Kim, S. W., & Park, C. (2011). Tolerance of Saccharomyces cerevisiae K35 to lignocellulose-derived inhibitory compounds. Biotechnology and Bioprocess Engineering, 16, 755–760.
Zaldivar, J., Martinez, A., & Ingram, L. O. (1999). Effect of selected aldehydes on the growth and fermentation of ethanologenic Escherichia coli. Biotechnology and Bioengineering, 65, 24–33.
Allen, S. A., Clark, W., McCaffery, J. M., Cai, Z., Lanctot, A., Slininger, P. J., Liu, Z. L., & Gorsich, S. W. (2010). Furfural induces reactive oxygen species accumulation and cellular damage in Saccharomyces cerevisiae. Biotechnology for Biofuels, 3, 2–10.
Muller, S., & Losche, A. (2004). Population profiles of a commercial yeast strain in the course of brewing. Journal of Food Engineering, 63, 375–381.
Chen, X., Li, Z., Zhang, X., Hu, F., Ryu, D. D. Y., & Bao, J. (2009). Screening of oleaginous yeast strains tolerant to lignocellulose degradation compounds. Applied Biochemistry and Biotechnology, 159, 591–604.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lopes da Silva, T., Baptista, C., Reis, A. et al. Using Flow Cytometry to Evaluate the Stress Physiological Response of the Yeast Saccharomyces carlsbergensis ATCC 6269 to the Presence of 5-Hydroxymethylfurfural During Ethanol Fermentations. Appl Biochem Biotechnol 181, 1096–1107 (2017). https://doi.org/10.1007/s12010-016-2271-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-016-2271-9