Abstract
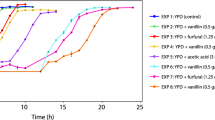
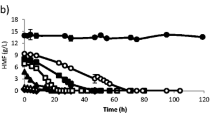
This work described the effect of furfural, a product resulting from the lignocellulosic material pretreatment, on Saccharomyces carlsbergensis growth and ethanol production. Flow cytometry was used to evaluate the yeast membrane potential, membrane integrity, reactive oxygen species production and lipid content. Above 0.3 g/L of furfural, a progressive decrease in the maximal specific growth rate was observed, reaching 53% of the value obtained in the absence of toxic when the cells were grown in the presence of 4 g/L of furfural. In general, the yeast biomass concentration and yield were less affected by the furfural presence than the specific growth rate, and a maximum reduction of 25% was observed for the assay at 4 g/L. The ethanol production was even less affected by the furfural presence than the yeast growth. At 4 g/L of furfural, the maximum ethanol concentration was reduced by only 10% relatively to the maximum ethanol concentration observed in the absence of toxic. At 5 g/L of furfural, the yeast cells were barely able to keep metabolic functions and produced a final ethanol concentration of 0.87 g/L although growth was undetectable. S. carlsbergensis membrane potential was affected by the furfural presence, concomitantly with the ethanol production. However, at 4 g/L, most of the yeast cells (90%) displayed the cytoplasmic membrane depolarized. The proportion of cells with increasing reactive oxygen species (ROS) production levels increased for the experiments at 0–4 g/L. For the experiment at 4.5 g/L of furfural, ROS production was observed for only 11% of the yeast cells. The yeast lipid content was also severely affected by the furfural presence. Both polar and neutral lipids decreased in the presence of furfural, and this reduction was more notorious during the stationary phase.





Similar content being viewed by others
References
Palmqvist, E., Grage, H., Meinander, N. Q., & Hahn-Hägerdal, B. (1999). Main and interaction effects of acetic acid, furfural, and p-hydroxybenzoic acid on growth and ethanol productivity of yeasts. Biotechnology and Bioengineering, 63, 46–55.
Oliva, J. M., Sáez, F., Ballesteros, I., González, A., Negro, M. J., Manzanares, P., & Ballesteros, M. (2003). Effect of lignocellulosic degradation compounds from steam explosion pretreatment on ethanol fermentation by thermotolerant yeast Kluyveromyces marxianus. Applied Biochemistry and Biotechnology, 105-108, 141–153.
Wikandari, R., Millati, R., Syamsiyah, S., Muriana, R., & Ayuningsih, Y. (2010). Effect of furfural, hydroxymethylfurfural and acetic acid on indigenous microbial isolate for bioethanol production. Agricultural Journal, 5, 105–109.
Zhen-yun, W., Yu, D., Li, T., Yue-hong, L., Yi-jie, Z., & Wen-Xue, Z. (2010). Investigating the effects of two lignocellulosic degradation by-products (furfural and acetic acid) on ethanol fermentations by six ethanologenic yeast strains. African Journal of Biotechnology, 9, 8661–8666.
Lee, H., Cho, D. H., Kim, Y. H., Shin, S. J., Kim, S. W., Han, S. O., Lee, J., Kim, S. W., & Park, C. (2011). Tolerance of Saccharomyces cerevisiae K35 to lignocellulose-derived inhibitory compounds. Biotechnology Bioprocess Engineering, 16, 755–760.
Palmqvist, E., & Hahn-Hägerdal, B. (2000). Fermentation of lignocellulosic hydrolisates. I: inhibition and detoxification. Bioresource Technology, 74, 17–24.
Freitas, C., Nunes, E., Passarinho, P. C., Reis, A., & Lopes da Silva, T. (2012). Effect of acetic acid on Saccharomyces carlsbergensis ATCC 6269 batch ethanol production monitored by flow Cytometry (2012). Applied Biochemistry and Biotechnology, 168, 1501–1015.
Freitas, C., Nunes, E., Passarinho, P. C., Reis, A., & Lopes da Silva, T. (2013). Use of multi-parameter flow cytometry as tool to monitor the impact of formic acid on Saccharomyces carlsbergensis batch ethanol fermentations. Applied Biochemistry and Biotechnology, 169, 2038–2048.
Lopes da Silva, T., Baptista, C., Reis, A., Passarinho, P. C. (2016). Using flow cytometry to evaluate the stress physiological response of the yeast Saccharomyces carlsbergensis ATCC 6269 to the presence of 5-Hydroxymethylfurfural during ethanol fermentations. Applied Biochemistry and Biotechnology. doi:10.1007/s12010-016-2271-9.
Liu, Z. L., Slininger, P. J., Dien, B. S., Berhow, M. A., Kurtzman, C. P., & Gorsich, S. W. (2004). Adaptative response of yeasts to furfural and 5-hydroxymethylfurfural and new chemical evidence for HMF conversion to 2, 5-bis-hydroxymethylfuran. Journal of Industrial Microbiology and Biotechnology, 31, 345–352.
Taherzadeh, M. J., Gustafsson, L., Niklasson, C., & Lidén, G. (1999). Conversion of furfural in aerobic and anaerobic batch fermentation of glucose by Saccharomyces cerevisiae. Journal of Bioscience and Bioengineering, 87, 169–174.
Gutiérrez, T., Buszko, M. L., Ingram, L. O., & Preston, J. F. (2002). Reduction of furfural to furfuryl alcohol by ethanologenic strains of bacteria and its effect on ethanol production from xylose. Applied Biochemistry and Biotechnology, 98-100, 327–340.
Modig, T., Lide, G., & Taherzadeh, M. J. (2002). Inhibition effects of furfural on alcohol dehydrogenase, aldehyde dehydrogenase and pyruvate dehydrogenase. Biochemical Journal, 363, 769–776.
Banerjee, N., Bhatnagar, R., & Viswanathan, L. (1981). Inhibition of glycolysis by furfural in Saccharomyces cerevisiae. European Journal of Applied Microbiology and Biotechnology, 11, 226–228.
Almeida, J. R. M., Modig, T., Petersson, A., Hähn-Hägerdal, B., & Gorwa-Grauslund, M. F. (2007). Increased tolerance and conversion of inhibitors in lignocellulosic hydrolysates by Saccharomyces cerevisiae. Journal of Chemical Technology and Biotecnology, 23, 40–349.
Dias, C., Silva, C., Freitas, C., Reis, A., & Lopes da Silva, T. (2016). Effect of medium pH on Rhodosporidium toruloides NCYC 921 carotenoid and lipid production evaluated by flow cytometry. Applied Biochemistry and Biotechnology, 179, 776–787.
Allen, S. A., Clark, W., McCaffery, J. M., Cai, Z., Lanctot, A., Slininger, P. J., Liu, Z. L., & Gorsich, S. W. (2010). Furfural induces reactive oxygen species accumulation and cellular damage in Saccharomyces cerevisiae. Biotechnology for Biofuels, 3, 2–10.
Hewitt, C. J., & Nebe-Von-Caron, G. (2004). The application of multi-parameter flow cytometry to monitor individual microbial cell physiological state. Advances in Biochemical Engineering/Biotechnology, 89, 197–223.
Lopes da Silva, T., Feijão, D., Reis, A., & Roseiro, J. C. (2011). Monitoring Rhodotorula glutinis CCMI 145 physiological response and oil production growing on xylose and glucose using multi-parameter flow cytometry. Bioresource Biotechnology, 102, 2998–3006.
Muller, S., & Losche, A. (2004). Population profiles of a commercial yeast strain in the course of brewing. Journal of Food Engineering, 63, 375–381.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lopes da Silva, T., Santo, R., Reis, A. et al. Effect of Furfural on Saccharomyces carlsbergensis Growth, Physiology and Ethanol Production. Appl Biochem Biotechnol 182, 708–720 (2017). https://doi.org/10.1007/s12010-016-2356-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-016-2356-5