Abstract
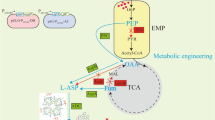
L-Alanine has important applications in food, pharmaceutical and veterinary and is used as a substrate for production of engineered thermoplastics. Microbial fermentation could reduce the production cost and promote the application of L-alanine. However, the presence of L-alanine significantly inhibit cell growth rate and cause a decrease in the ultimate L-alanine productivity. For efficient l-alanine production, a thermo-regulated genetic switch was designed to dynamically control the expression of l-alanine dehydrogenase (alaD) from Geobacillus stearothermophilus on the Escherichia coli B0016-060BC chromosome. The optimal cultivation conditions for the genetically switched alanine production using B0016-060BC were the following: an aerobic growth phase at 33 °C with a 1-h thermo-induction at 42 °C followed by an oxygen-limited phase at 42 °C. In a bioreactor experiment using the scaled-up conditions optimized in a shake flask, B0016-060BC accumulated 50.3 g biomass/100 g glucose during the aerobic growth phase and 96 g alanine/100 g glucose during the oxygen-limited phase, respectively. The L-alanine titer reached 120.8 g/l with higher overall and oxygen-limited volumetric productivities of 3.09 and 4.18 g/l h, respectively, using glucose as the sole carbon source. Efficient cell growth and L-alanine production were reached separately, by switching cultivation temperature. The results revealed the application of a thermo-regulated strategy for heterologous metabolic production and pointed to strategies for improving L-alanine production.



Similar content being viewed by others
References
Hols, P., Kleerebezem, M., Schanck, A. N., Ferain, T., Hugenholtz, J., Delcour, J., & de Vos, W. M. (1999). Conversion of Lactococcus lactis from homolactic to homoalanine fermentation through metabolic engineering. Nature Biotechnology, 17, 588–592.
Lee, M., Smith, G., Eiteman, M., & Altman, E. (2004). Aerobic production of alanine by Escherichia coli aceF ldhA mutants expressing the Bacillus sphaericus alaD gene. Applied Microbiology and Biotechnology, 65, 56–60.
Mallakpour, S., & Dinari, M. (2011). Progress in synthetic polymers based on natural amino acids. Journal of Macromolecular Science-Pure and Applied Chemistry, 48, 644–679.
Zhang, X., Jantama, K., Moore, J. C., Shanmugam, K. T., & Ingram, L. O. (2007). Production of L-alanine by metabolically engineered Escherichia coli. Applied Microbiology and Biotechnology, 77, 355–366.
Smith, G. M., Lee, S. A., Reilly, K. C., Eiteman, M. A., & Altman, E. (2006). Fed-batch two-phase production of alanine by a metabolically engineered Escherichia coli. Biotechnology Letters, 28, 1695–1700.
Wada, M., Narita, K., & Yokota, A. (2007). Alanine production in an H+-ATPase- and lactate dehydrogenase-defective mutant of Escherichia coli expressing alanine dehydrogenase. Applied Microbiology and Biotechnology, 76, 819–825.
Zhou, L., Niu, D., Tian, K., Chen, X., Prior, B. A., Shen, W., Shi, G., Singh, S., & Wang, Z. (2012). Genetically switched D-lactate production in Escherichia coli. Metabolic Engineering, 14, 560–568.
Elvin, C. M., Thompson, P. R., Argall, M. E., Philip Hendr, N., Stamford, P. J., Lilley, P. E., & Dixon, N. E. (1990). Modified bacteriophage lambda promoter vectors for overproduction of proteins in Escherichia coli. Gene, 87, 123–126.
Love, C. A., Lilley, P. E., & Dixon, N. E. (1996). Stable high-copy-number bacteriophage [lambda] promoter vectors for overproduction of proteins in Escherichia coli. Gene, 176, 49–53.
Datsenko, K. A., & Wanner, B. L. (2000). One-step inactivation of chromosomal genes in Escherichia coli K-12 using PCR products. Proceedings of the National Academy of Sciences of the United States of America, 97, 6640–6645.
Zhou, L., Zuo, Z., Chen, X., Niu, D., Tian, K., Prior, B. A., Shen, W., Shi, G., Singh, S., & Wang, Z. (2011). Evaluation of genetic manipulation strategies on D-lactate production by Escherichia coli. Current Microbiology, 62, 981–989.
Kim, S. H., Schneider, B. L., & Reitzer, L. (2010). Genetics and regulation of the major enzymes of alanine synthesis in Escherichia coli. Journal of Bacteriology, 192, 5304–5311.
Villaverde, A., Benito, A., Viaplana, E., & Cubarsi, R. (1993). Fine regulation of cI857-controlled gene expression in continuous culture of recombinant Escherichia coli by temperature. Applied and Environmental Microbiology, 59, 3485–3487.
Hashimoto, S.-I., & Katsumata, R. (1998). L-alanine fermentation by an alanine racemase-deficient mutant of the DL-alanine hyperproducing bacterium Arthrobacter oxydans HAP-1. Journal of Fermention and Bioengineering, 86, 385–390.
Uhlenbusch, I., Sahm, H., & Sprenger, G. A. (1991). Expression of an L-alanine dehydrogenase gene in Zymomonas mobilis and excretion of L-alanine. Applied and Environmental Microbiology, 57, 1360–1366.
Ratkowsky, D. A., Lowry, R. K., McMeekin, T. A., Stokes, A. N., & Chandler, R. E. (1983). Model for bacterial culture growth rate throughout the entire biokinetic temperature range. Journal of Bacteriology, 154, 1222–1226.
Zhou, L., Tian, K., Niu, D., Shen, W., Shi, G. Y., Singh, S., & Wang, Z. (2012). Improvement of D-lactate productivity in recombinant Escherichia coli by coupling production with growth. Biotechnology Letters, 34, 1123–1130.
Yuan, L. Z., Rouvière, P. E., LaRossa, R. A., & Suh, W. (2006). Chromosomal promoter replacement of the isoprenoid pathway for enhancing carotenoid production in E. coli. Metabolic Engineering, 8, 79–90.
Sandoval-Basurto, E. A., Gosset, G., Bolivar, F., & Ramirez, O. T. (2005). Culture of Escherichia coli under dissolved oxygen gradients simulated in a two-compartment scale-down system: metabolic response and production of recombinant protein. Biotechnology and Bioengineering, 89, 453–463.
Lara, A. R., Leal, L., Flores, N., Gosset, G., Bolívar, F., & Ramírez, O. T. (2006). Transcriptional and metabolic response of recombinant Escherichia coli to spatial dissolved oxygen tension gradients simulated in a scale-down system. Biotechnology and Bioengineering, 93, 372–385.
Caspeta, L., Flores, N., Pérez, N. O., Bolívar, F., & Ramírez, O. T. (2009). The effect of heating rate on Escherichia coli metabolism, physiological stress, transcriptional response, and production of temperature-induced recombinant protein: a scale-down study. Biotechnology and Bioengineering, 102, 468–482.
Abdel-Banat, B. M., Hoshida, H., Ano, A., Nonklang, S., & Akada, R. (2010). High-temperature fermentation: how can processes for ethanol production at high temperatures become superior to the traditional process using mesophilic yeast? Applied Microbiology and Biotechnology, 85, 861–867.
Acknowledgments
This work was partly funded by the National Natural Science Foundation of China (31300087, 31400058), the Natural Science Foundation of Jiangsu (BK20130131, BK20130139 and BK20140151), the National High Technology Research and Development Program of China (863 Program, 2014AA021304), the High Foreign Experts Project (GDW20123200114), the Priority Academic Program Development of Jiangsu Higher-Education Institutions, the 111 Project (111-2-06), the Jiangsu Province “Collaborative Innovation Center for Advanced Industrial Fermentation” Industry Development Program, the Public Project for Key Laboratory of Industrial Biotechnology, Ministry of Education, Jiangnan University (KLIB-KF201306) and the Fundamental Research Funds for the Central Universities (JUSRP51411B, JUSRP51504).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhou, L., Deng, C., Cui, WJ. et al. Efficient L-Alanine Production by a Thermo-Regulated Switch in Escherichia coli . Appl Biochem Biotechnol 178, 324–337 (2016). https://doi.org/10.1007/s12010-015-1874-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-015-1874-x