Abstract
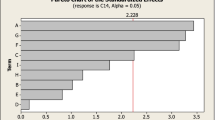
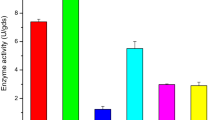
The optimum values of the critical variables determined by the central composite design of response surface methodology (RSM) for maximum phytase production (1881.26 U g−1 dry mouldy residue (DMR)) by Sporotrichum thermophile are 2.5 % Tween 80, 1.0 % yeast extract and 48 h of incubation period. Phytase production in the mixed substrate (sugarcane bagasse and wheat bran) fermentation enhanced 11.6-fold over the initial production as a consequence of optimization. Phytase titres are sustainable in flasks, trays and column bioreactor (1796 to 2095 U g−1 DMR), thus validating the model and the process for large-scale phytase production. When the yeast extract was replaced with corn steep liquor (2 % w/v), a sustained enzyme titre (1890 U g−1 DMR) was attained, making the process cost-effective. Among all the detergents, Tween 80 supported a higher phytase production than others. The enzyme efficiently liberated nutritional components from poultry feed (inorganic phosphate, soluble protein and reducing sugars) in a time-dependent manner.



Similar content being viewed by others
References
Vohra, A., & Satyanarayana, T. (2003). Phytases: microbial sources, production, purification, and potential biotechnological applications. Critical Reviews in Biotechnology, 23, 29–60.
Vats, P., & Banerjee, U. C. (2004). Production studies and catalytic properties of phytases (myoinositolhexakisphosphate phosphohydrolases): an overview. Enzyme and Microbial Technology, 35, 3–14.
Singh, B., & Satyanarayana, T. (2011). Phytases from thermophilic molds: their production, characteristics and multifarious applications. Process Biochemistry, 46(7), 1391–1398.
Singh, B., & Satyanarayana, T. (2015). Fungal phytases: characteristics and amelioration of nutritional quality and growth of non ruminants. Journal of Animal Physiology and Animal Nutrition, 99, 646–660.
Nampoothiri, K. M., Tomes, G. J., Roopesh, K., Szakaces, G., Nagy, Y., Soccol, C. R., & Panday, A. (2004). Thermostable phytase production by Thermoascus aurantiacus in submerged fermentation. Applied Biochemistry and Biotechnology, 118(1–3), 205–214.
Pandey, A., Selvakumar, P., Soccol, C. R., & Nigam, P. (1999). Solid state fermentation for the production of industrial enzymes. Current Science, 77, 149–162.
Thomas, L., Larroche, C., & Pandey, A. (2013). Current developments in solid-state fermentation. Biochemical Engineering Journal, 81, 146–161.
Bogar, B., Szakacs, G., Linden, J. C., Pandey, A., & Tengerdy, R. P. (2003). Optimization of phytase production by solid substrate fermentation. Journal of Industrial Microbiology and Biotechnology, 30(3), 183–189.
Bogar, B., Szakacs, G., Pandey, A., Abdulhameed, S., Linden, J. C., & Tengerdy, R. P. (2003). Production of phytase by Mucor racemosus in solid-state fermentation. Biotechnology Progress, 9(2), 312–319.
Chadha, B. S., Gulati, H., Minhas, M., Saini, H. S., & Singh, N. (2004). Phytase production by the thermophilic fungus Rhizomucor pusillus. World Journal of Microbiology and Biotechnology, 20, 105–109.
Singh, B., & Satyanarayana, T. (2006). Phytase production by thermophilic mold Sporotrichum thermophile in solid-state fermentation and its application in dephytinization of sesame oil cake. Applied Biochemistry and Biotechnology, 133, 239–250.
Singh, B., & Satyanarayana, T. (2008). Phytase production by a thermophilic mould Sporotrichum thermophile in solid state fermentation and its potential applications. Bioresource Technology, 99, 2824–2830.
Maller, A., Vici, C. A., Facchini, A. D. F., Silva, M. T., Kamimura, S. E., Rodrigues, I. M., Jorge, A. J., Terenzi, F. H., & Polizeli, M. T. L. M. (2013). Increase of the phytase production by Aspergillus japonicas and its biocatalyst on chicken feed treatment. Journal of Basic Microbiology, 53, 1–9.
Bala, A., Sapna, Jain, J., Kumari, A., & Singh, B. (2014). Production of an extracellular phtyase from a thermophillic mould Humicola nigrescens in solid state fermentation and its application in dephytinization. Biocatalysis and Agricultural Biotechnology, 3, 259–264.
Sapna, & Singh, B. (2014). Phytase production by Aspergillus oryzae in solid–state fermentation and its applicability in dephyinization of wheat ban. Applied Biochemistry and Biotechnology, 173(7), 1885–1895.
Emerson, R. (1941). An experimental study of life cycle and taxonomies of Allomyces. Lloydia, 4, 77–144.
Sharma, M., Chadha, B. S., Kaur, M., Ghatora, S. K., & Saini, H. S. (2008). Molecular characterization of multiple xylanase producing thermophilic/thermotolerant fungi isolated from composting materials. Letters in Applied Microbiology, 46(5), 526–535.
Fiske, C. H., & Subbarow, Y. (1925). The colorimetic determination of phosphorus. Journal of Biological Chemistry, 66, 376–400.
Miller, G. L. (1959). Use of dinitrosalicyclic reagent for determination of reducing sugars. Analytical Chemistry, 31, 426–428.
Lowry, O. H., Rosebrough, N. J., Farr, A. L., & Randall, R. J. (1951). Protein measurement with the folin phenol reagent. Journal of Biological Chemistry, 193, 265–275.
Plackett, R. L., & Burman, J. P. (1946). The design of optimum multifactor experiments. Biometrika, 33, 305–325.
Sapna, & Singh, B. (2013). Improved production of protease- resistant phytase by Aspergillus oryzae and its applicability in the hydrolysis of insoluble phytates. Journal of Industrial Microbiology and Biotechnology, 40(8), 891–899.
Sharma, K. K., Kapoor, M., & Kuhad, R. C. (2005). In vivo enzymatic digestion, in vitro xylanase digestion, metabolic analogues, surfactants and polyethylene glycol ameliorate laccase production from Ganoderma sp. kk-02. Letters in Applied Microbiology, 41, 24–31.
Lawford, H. G., & Rousseau, J. D. (1997). Corn Steep liquor as a cost-effective nutrition adjunct in high performance Zymomonas ethanol fermentations. Applied Biochemistry and Biotechnology, 63–65, 287–304.
Ghareib, M. (1990). Biosynthesis, purification and some properties of extracellular phytase from Aspergillus carneus. Acta Microbiologica Hungarica, 37(2), 159–164.
Gomes, J. E. G., da Silva Nascimento, T. C. E., de França Queiroz, A. E. S., de Siqueira Silva Júnior, J. I., de Souza-Motta, C. M., de Medeiros, E. V., & Moreira, K. A. (2014). Production, characterization and evaluation of in vitro digestion of phytases, xylanases and cellulases for feed industry. African Journal of Microbiology Research, 8(6), 551–558.
Mandviwala, T. N., & Khire, J. M. (2000). Production of high activity thermostable phytase from thermotolerant Aspergillus niger in solid state fermentation. Journal of Industrial Microbiology and Biotechnology, 24, 237–243.
Bhavsar, K., Kumar, V. R., & Khire, J. M. (2011). High level phytase production by Aspergillus niger NCIM 563 in solid state culture: response surface optimization, up-scaling, and its partial characterization. Journal of Industrial Microbiology and Biotechnology, 38, 1407–1417.
Acknowledgments
The authors acknowledge the financial assistance (BT/PR4771/PID/6/636/2012) from the Department of Biotechnology (DBT), Govt. of India, New Delhi, during the course of this investigation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kumari, A., Satyanarayana, T. & Singh, B. Mixed Substrate Fermentation for Enhanced Phytase Production by Thermophilic Mould Sporotrichum thermophile and Its Application in Beneficiation of Poultry Feed. Appl Biochem Biotechnol 178, 197–210 (2016). https://doi.org/10.1007/s12010-015-1868-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-015-1868-8