Abstract
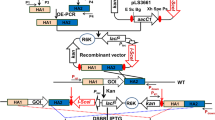
Bacillus amyloliquefaciens LL3 is a glutamate-independent poly-γ-glutamic acid (γ-PGA) producing strain which consists of a circular chromosome (3,995,227 bp) and an endogenous plasmid pMC1 (6,758 bp). The study of the function of native plasmid and the genome-size reduction of the B. amyloliquefaciens LL3 strain requires elimination of the endogenous plasmid. Traditional plasmid-curing procedures using sodium dodecyl sulfate (SDS) or acridine orange combined with heat treatment have been shown to be ineffective in this strain. Plasmid incompatibility is an effective method for curing which has been studied before. In our research, the hypothetical Rep protein gene and the origin of replication of the endogenous plasmid were cloned into the temperature-sensitive vector yielding the incompatible plasmid pKSV7-rep-ori. This plasmid was transformed into LL3 by electroporation. The analysis of the strain bearing incompatible plasmids after incubation at 30 °C for 30 generations showed the production of plasmid cured strains. High frequency of elimination was achieved with more than 93 % of detected strains showing to be plasmid-cured. This is the first report describing plasmid cured in a γ-PGA producing strain using this method. The plasmid-cured strains showed an increase of γ-PGA production by 6 % and led to a yield of 4.159 g/l, compared to 3.918 g/l in control and cell growth increased during the early stages of the exponential phase. Gel permeation chromatography (GPC) characterization revealed that the γ-PGA produced by plasmid-cured strains and the wild strains were identical in terms of molecular weight. What is more, the further study of plasmid function showed that curing of the endogenous plasmid did not affect its sporulation efficiency.




Similar content being viewed by others
Notes
SDS: sodium dodecyl sulfate
References
Ashiuchi, M., & Misono, H. (2002). Biochemistry and molecular genetics of poly-γ-glutamate synthesis. Applied Microbiology and Biotechnology, 59, 9–14.
Shih, I. L., & Van, Y. T. (2001). The production of poly-(γ-glutamic acid) from microorganisms and its various applications. Bioresource Technology, 79, 207–225.
Cao, M. F., Geng, W. T., Liu, L., Song, C. J., Xie, H., Guo, W. B., Jin, Y. H., & Wang, S. F. (2011). Glutamic acid independent production of poly-γ-glutamic acid by Bacillus amyloliquefaciens LL3 and cloning of pgsBCA genes. Bioresource Technology, 102, 4251–4257.
Geng, W. T., Cao, M. F., Song, C. J., Xie, H., Liu, L., Yang, C., Feng, J., Zhang, W., Jin, Y. H., Du, Y., & Wang, S. F. (2011). Complete genome sequence of Bacillus amyloliquefaciens LL3, which exhibits glutamic acid-independent production of poly-γ-glutamic acid. Journal of Bacteriology, 193, 3393–3394.
Nojiri, H., Shintani, M., & Mori, T. (2004). Divergence of mobile genetic elements involved in the distribution of xenobiotic-catabolic capacity. Applied Microbiology and Biotechnology, 64, 154–174.
Rotger, R., & Casadesus, J. (1999). The virulence plasmids of Salmonella. International Microbiology, 2, 177–184.
Perego, M., Hanstein, C., Welsh, K. M., Djavakhishvili, T., Glaser, P., & Hoch, J. A. (1994). Multiple protein-aspartate phosphatases provide a mechanism for the integration of diverse signals in the control of development in B. subtilis. Cell, 79, 1047–1055.
Jiang, M., Grau, R., & Perego, M. (2000). Differential processing of propeptide inhibitors of Rap phosphatases in Bacillus subtilis. Journal of Bacteriology, 182, 303–310.
Smits, W. K., Bongiorni, C., Veening, J. W., Hamoen, L. W., Kuipers, O. P., & Perego, M. (2007). Temporal separation of distinct differentiation pathways by a dual specificity Rap-Phr system in Bacillus subtilis. Molecular Microbiology, 65, 103–120.
Perego, M. (2001). A new family of aspartyl phosphate phosphatases targeting the sporulation transcription factor Spo0A of Bacillus subtilis. Molecular Microbiology, 42, 133–143.
Ghosh, S., Mahapatra, N. R., Ramamurthy, T., & Banerjee, P. C. (2000). Plasmid curing from an acidophilic bacterium of the genus Acidocella. FEMS Microbiology Letters, 183, 271–274.
EI-Mansi, M., Anderson, K. J., Inche, C. A., Knowles, L. K., & Platt, D. J. (2000). Isolation and curing of the Klebsiella pneumonia large indigenous plasmid using sodium dodecyl sulphate. Research in Microbiology, 151, 201–208.
Hara, T., Aumayr, A., Fujio, Y., & Ueda, S. (1982). Elimination of plasmid-linked polyglutamate production by Bacillus subtilis (natto) with acridine orange. Applied and Environment Microbiology, 44, 1456–1458.
Spengler, G., Molnár, A., Schelz, Z., Amaral, L., Sharples, D., & Molnár, J. (2006). The mechanism of plasmid curing in bacteria. Current Drug Targets, 7, 823–841.
Kulkarni, R. S., & Kanekar, P. P. (1998). Effects of some curing agents on phenotypic stability in Pseudomonas putida degrading ε-caprolactam. World Journal of Microbiology and Biotechnology, 14, 255–257.
Imre, A., Olasz, F., Kiss, J., & Nagy, B. (2006). A novel transposon-based method for elimination of large bacterial plasmids. Plasmid, 55, 235–241.
Rajini Rani, D. B., & Mahadevan, A. (1992). Plasmid mediated metal and antibiotic resistance in marine pseudomonas. Biometals, 5, 73–80.
Novick, R. P. (1987). Plasmid incompatibility. Microbiology and Molecualr Biology Reviews, 51, 381–395.
Uraji, M., Suzuki, K., & Yoshida, K. (2002). A novel plasmid curing method using incompatibility of plant pathogenic Ti plasmids in Agrobacterium tumefaciens. Genes & Genetic Systems, 77, 1–9.
Baker, T. A., & Bell, S. P. (1998). Polymerases and the replisome: machines within machines. Cell, 92, 295–305.
Smith, K., & Youngman, P. (1992). Use a new integrational vector to investigate compartment-specific expression of the Bacillus subtilis spoIIM gene. Biochimie, 74, 705–711.
Cliff, J. B., Jarman, K. H., Valentine, N. B., Golledge, S. L., Gaspar, D. J., Wunschel, D. S., & Wahl, K. L. (2005). Differentiation of spores of Bacillus subtilis grown in different media by elemental characterization using Time-of-Flight secondary ion mass spectrometry. Appllied and Environment Microbiology, 71, 6524–6530.
Fay, A., & Dworkin, J. (2009). Bacillus subtilis homologs of MviN (MurJ), the putative Escherichia coli lipid II flippase, are not essential for growth. Journal of Bacteriology, 191, 6020–6028.
Kubota, H., Matsunobu, T., Uotani, K., Takebe, H., Satoh, A., Tanaka, T., & Taniguchi, M. (1993). Production of poly (γ-glutamic acid) by Bacillus subtilis F-2-01. Bioscience, Biotechnology, and Biochemistry, 57, 1212–1213.
Bouanchaud, D. H., Scavizzi, M. R., & Chabbert, Y. A. (1969). Elimination of ethidium bromide of antibiotic resistance in Enterobacteria and Staphylococci. Journal of General Microbiology, 54, 417–425.
Mchuge, G. L., & Swartz, M. N. (1977). Elimination of plasmids from several bacterial species by novobiocin. Antimicrobial Agents and Chemotherapy, 12, 423–426.
Pickett, M. A., Everson, J. S., Pead, P. J., & Clarke, I. N. (2005). The plasmids of Chlamydia trachomatis and Chlamydophila pneumoniae (N16): accurate determination of copy number and the paradoxical effect of plasmid-curing agents. Microbiology, 151, 893–903.
Posno, M., Leer, R. J., van Luijk, N., van Giezen, M. J., Heuvelmans, P. T. H. M., Lokman, B. C., & Pouwels, P. H. (1991). Incompatibility of Lactobacillus vectors with replicons derived from small cryptic Lactobacillus plasmids and segregational instability of the introduced vectors. Applied and Environment Microbiology, 57, 1822–1828.
Meijer, W. J., de Boer, A. J., van Tongeren, S., Venema, G., & Bron, S. (1995). Nucleic Acids Research, 23, 3214–3223.
Mueller, J. P., Bukusoglu, G., & Sonenshein, A. L. (1992). Transcriptional regulation of Bacillus subtilis glucose starvation-inducible genes: control of gsiA by the ComP-ComA signal transduction system. Journal of Bacteriology, 174, 4361–4373.
Mizoguchi, H., Mori, H., & Fujio, T. (2007). Biotechnology and Applied Biochemistry, 46, 157–167.
Mizoguchi, H., Sawano, Y., Kato, J., & Mori, H. (2008). Superpositioning of deletions promotes growth of Escherichia coli with a reduced genome. DNA Research, 15, 277–284.
Morimoto, T., Kadoya, R., Endo, K., Tohata, M., Sawada, K., Liu, S. G., Ozawa, T., Kodama, T., Kakeshida, H., Kageyama, Y., Manabe, K., Kanaya, K., Ara, K., Ozaki, K., & Ogasawara, N. (2008). Enhanced recombinant protein productivity by genome reduction in Bacillus subtilis. DNA Research, 15, 73–81.
Acknowledgments
This study was financially supported by National key 296 Basic Research Program of China (“973”-Program) 2012CB725204, National High Technology Research and Development Program of China (“863”-Program) 2012AA021505, Natural Science Foundation of China Grant Nos. 31070039, 31170030, and 51073081, Project of Tianjin, China (11JCYBJC09500).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Jun Feng and Yanyan Gu contributed equally to this work.
Rights and permissions
About this article
Cite this article
Feng, J., Gu, Y., Wang, J. et al. Curing the Plasmid pMC1 from the Poly (γ-glutamic Acid) Producing Bacillus amyloliquefaciens LL3 Strain Using Plasmid Incompatibility. Appl Biochem Biotechnol 171, 532–542 (2013). https://doi.org/10.1007/s12010-013-0382-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-013-0382-0