Abstract
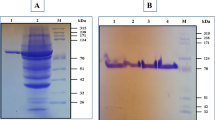
A strain with the power to produce extracellular pullulanase was obtained from the sample taken from a flour mill. By sequencing its 16S rDNA, the isolate was identified as Klebsiella variicola SHN-1. When the gene encoding pullulanase, containing the N-terminal signal sequence, was cloned into Escherichia coli BL21 (DE3), extracellular activity was detected up to 10 U/ml, a higher level compared with the results in published literature. Subsequently, the recombinant pullulanase was purified and characterized. The main end product from pullulan hydrolyzed by recombinant pullulanase was determined as maltotriose with HPLC, and hence, the recombinant pullulanase was identified as type I pullulanase, which could be efficiently employed in starch processing to produce maltotriose with higher purity and even to evaluate the purity of pullulan. To investigate the effect of signal peptide on secretion of the recombinant enzyme, the signal sequence was removed from the constructed vector. However, secretion of pullulanase in E. coli was not influenced, which was seldom reported previously. By localizing the distribution of pullulanase on subcellular fractions, the secretion of recombinant pullulanase in E. coli BL21 (DE3) was confirmed, even from the expression system of nonsecretory type without the assistance of signal peptide.








Similar content being viewed by others
References
Matzke, J., Herrmann, A., Schneider, E., & Bakker, E. (2000). FEMS Microbiology Letters, 183, 55–61.
Albertson, G. D., McHale, R. H., Gibbs, M. D., & Bergquist, P. L. (1997). Biochimica et Biophysica Acta–Gene Structure and Expression, 1354, 35–39.
Kang, J., Park, K.-M., Choi, K.-H., Park, C.-S., Kim, G.-E., Kim, D., et al. (2011). Enzyme and Microbial Technology, 48, 260–266.
Takizawa, N., & Murooka, Y. (1985). Applied and Environmental Microbiology, 49, 294–298.
Gomes, I., Gomes, J., & Steiner, W. (2003). Bioresource Technology, 90, 207–214.
Shim, J. H., Park, J. T., Hong, J. S., Kim, K. W., Kim, M. J., Auh, J. H., et al. (2009). Journal of Bacteriology, 191, 4835–4844.
Hii, S. L., Ling, T. C., Mohamad, R., & Ariff, A. B. (2009). The Open Biotechnology Journal, 3, 1–8.
Singh, R. S., Saini, G. K., & Kennedy, J. F. (2011). Carbohydrate Polymers, 83, 672–675.
Zhang, H., & Jin, Z. (2011). Carbohydrate Polymers, 83, 865–867.
Bender, H., & Wallenfels, K. (1961). Biochemische Zeitschrift, 334, 79–95.
Walker, G. J. (1968). Biochemical Journal, 108, 33–40.
Melasniemi, H. (1988). Biochemical Journal, 250, 813–818.
Ramesh, B., Reddy, P. R. M., Seenayya, G., & Reddy, G. (2001). Bioresource Technology, 76, 169–171.
Kim, C., Choi, H., & Lee, D. (1993). Journal of Industrial Microbiology and Biotechnology, 12, 48–57.
Hii, L., Rosfarizan, M., Ling, T., & Ariff, A. (2012). Food and Bioprocess Technology, 5, 729–737.
Plant, A. R., Clemens, R. M., Daniel, R. M., & Morgan, H. W. (1987). Applied Microbiology and Biotechnology, 26, 427–433.
Odibo, F. J. C., & Obi, S. K. C. (1988). Journal of Industrial Microbiology and Biotechnology, 3, 343–350.
Gantelet, H., & Duchiron, F. (1998). Applied Microbiology and Biotechnology, 49, 770–777.
Ben Messaoud, E., Ben Ammar, Y., Mellouli, L., & Bejar, S. (2002). Enzyme and Microbial Technology, 31, 827–832.
Kunamneni, A., & Singh, S. (2006). Enzyme and Microbial Technology, 39, 1399–1404.
Doman-Pytka, M., & Bardowski, J. (2004). Critical Reviews in Microbiology, 30, 107–121.
Lee, S. Y. (1996). Trends in Biotechnology, 14, 98–105.
Zouari Ayadi, D., Ben Ali, M., Jemli, S., Ben Mabrouk, S., Mezghani, M., Ben Messaoud, E., et al. (2008). Applied Microbiology and Biotechnology, 78, 473–481.
Tomiyasu, K., Yato, K., Yasuda, M., Tonozuka, T., Ibuka, A., & Sakai, H. (2001). Bioscience, Biotechnology, and Biochemistry, 65, 2090–2094.
Bertoldo, C., Armbrecht, M., Becker, F., Schafer, T., Antranikian, G., & Liebl, W. (2004). Applied and Environmental Microbiology, 70, 3407–3416.
Sivashanmugam, A., Murray, V., Cui, C., Zhang, Y., Wang, J., & Li, Q. (2009). Protein Science, 18, 936–948.
Graslund, S., Nordlund, P., Weigelt, J., Hallberg, B. M., Bray, J., Gileadi, O., et al. (2008). Nature Methods, 5, 135–146.
Jana, S., & Deb, J. K. (2005). Applied Microbiology and Biotechnology, 67, 289–298.
Krehenbrink, M., Chami, M., Guilvout, I., Alzari, P. M., Pecorari, F., & Pugsley, A. P. (2008). Journal of Molecular Biology, 383, 1058–1068.
Guilvout, I., Chami, M., Berrier, C., Ghazi, A., Engel, A., Pugsley, A. P., et al. (2008). Journal of Molecular Biology, 382, 13–23.
D’Enfert, C., & Pugsley, A. P. (1989). Journal of Bacteriology, 171, 3673–3679.
Chami, M., Guilvout, I., Gregorini, M., Remigy, H. W., Muller, S. A., Valerio, M., et al. (2005). Journal of Biological Chemistry, 280, 37732–37741.
Buddelmeijer, N., Francetic, O., & Pugsley, A. P. (2006). Journal of Bacteriology, 188, 2928–2935.
Francetic, O., Buddelmeijer, N., Lewenza, S., Kumamoto, C. A., & Pugsley, A. P. (2007). Journal of Bacteriology, 189, 1783–1793.
Buddelmeijer, N., Krehenbrink, M., Pecorari, F., & Pugsley, A. P. (2009). Journal of Bacteriology, 191, 161–168.
D’Enfert, C., Ryter, A., & Pugsley, A. P. (1987). The EMBO Journal, 6, 3531–3538.
Takizawa, N., & Murooka, Y. (1984). Agricultural and Biological Chemistry, 48, 1451–1458.
Michaelis, S., Chapon, C., D’Enfert, C., Pugsley, A. P., & Schwartz, M. (1985). Journal of Bacteriology, 164, 633–638.
Ruijssenaars, H. J., & Hartmans, S. (2001). Applied Microbiology and Biotechnology, 55, 143–149.
Ueda, S., & Ohba, R. (1972). Agricultural and Biological Chemistry, 36, 2381–2391.
Yamashita, M., Kinoshita, T., Ihara, M., Mikawa, T., & Murooka, Y. (1994). Journal of Biochemistry, 116, 1233–1240.
Takizawa, N., Shiro, H., Hatta, T., Nagao, K., & Kiyohara, H. (1991). Agricultural and Biological Chemistry, 55, 1467–1473.
Wu, S. J., Kim, J. M., Zhou, C. A., Jin, Z. Y., & Tong, Q. Y. (2010). Biotechnology Letters, 32, 1143–1145.
Singh, R. S., Saini, G. K., & Kennedy, J. F. (2010). Carbohydrate Polymers, 80, 401–407.
Sauvonnet, N., & Pugsley, A. P. (1996). Molecular Microbiology, 22, 1–7.
Acknowledgments
Financial support from Hi-Tech Research and Development Program of China (863 Program) (no. 2012AA022207). The National Key Basic Research and Development Program of China (973 Program) (no. 2011CB710800 and 2009CB724706) and the Program of Introducing Talents of Discipline to Universities (111 Project) (111-2-06) is greatly appreciated.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chen, WB., Nie, Y. & Xu, Y. Signal Peptide-Independent Secretory Expression and Characterization of Pullulanase from a Newly Isolated Klebsiella variicola SHN-1 in Escherichia coli . Appl Biochem Biotechnol 169, 41–54 (2013). https://doi.org/10.1007/s12010-012-9948-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-012-9948-5