Abstract
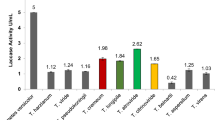
In this study, an easily detectable method was employed for screening laccase-producing microorganisms by using 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonate) as laccase secretion indicator. A novel laccase-producing strain was isolated and identified as Shiraia bambusicola Henn. strain GZ11K2 according to the morphological characteristics and the comparison of internal transcribed spacer ribosomal DNA gene sequences. In further investigation, the production of laccase by S. bambusicola GZ11K2 was greatly enhanced by the nontoxic inducers of copper sulfate and rhodamine B. Copper and rhodamine B were added into the cultivation medium at 24 and 12 h, respectively, and the maximum laccase production was obtained. Under the induction of 2.0 mM copper sulfate and 35 μM rhodamine B, an increment of about 80 times of laccase activity compared with that in the inducer-free medium and about 20 times compared with that in the single copper-supplemented medium was observed. Compared with other species, S. bambusicola GZ11K2 exhibits better laccase-producing characteristics with an activity of 16,400 U/L after 108 h, suggesting its potential ability for industrial application.





Similar content being viewed by others
References
Mayer, A. M., & Staples, R. C. (2002). Laccase: new functions for an old enzyme. Phytochemistry, 60, 551–565.
Kunamneni, A., Camarero, S., Garcia-Burgos, C., Plou, F., Ballesteros, A., & Alcalde, M. (2008). Engineering and applications of fungal laccases for organic synthesis. Microbial Cell Factories, 7, 1–17.
Tinoco, R., Acevedo, A., Galindo, E., & Serrano-Carreon, L. (2011). Increasing Pleurotus ostreatus laccase production by culture medium optimization and copper/lignin synergistic induction. Journal of Industrial Microbiology & Biotechnology, 38, 531–540.
Leonowicz, A., Cho, N. S., Luterek, J., Wilkolazka, A., Wojtas-Wasilewska, M., Matuszewska, A., et al. (2001). Fungal laccase: properties and activity on lignin. Journal of Basic Microbiology, 41, 185–227.
Baldrian, P. (2006). Fungal laccases—occurrence and properties. FEMS Microbiology Reviews, 30, 215–242.
Morozova, O. V., Shumakovich, G. P., Gorbacheva, M. A., Shleev, S. V., & Yaropolov, A. I. (2007). “Blue” laccases. Biochemistry (Mosc), 72, 1136–1150.
Elisashvili, V., Kachlishvili, E., Khardziani, T., & Agathos, S. N. (2010). Effect of aromatic compounds on the production of laccase and manganese peroxidase by white-rot basidiomycetes. Journal of Industrial Microbiology & Biotechnology, 37, 1091–1096.
Hu, Y., Cai, Y., Liao, X., Ma, W., Li, Z., & Zhang, D. (2011). Optimization of Shiraia bambusicola liquid-state fermentation for laccase production. Journal of Food Science and Biotechnology, 05, 773–776.
Liu, Z., Zhang, D., Hua, Z., Li, J., Du, G., & Chen, J. (2009). A newly isolated Paecilomyces sp. WSH-L07 for laccase production: isolation, identification, and production enhancement by complex inducement. Journal of Industrial Microbiology & Biotechnology, 36, 1315–1321.
Tychanowicz, G., Souza, D., Souza, C., Kadowaki, M., & Peralta, R. (2006). Copper improves the production of laccase by the white-rot fungus Pleurotus pulmonarius in solid state fermentation. Brazilian Archives of Biology and Technology, 49, 699–704.
Tavares, P. M., Coelho, A. Z., Coutinho, A. P., & Xavier, R. B. (2005). Laccase improvement in submerged cultivation: induced production and kinetic modeling. Journal of Chemical Technology and Biotechnology, 80, 669–676.
Du, W., Han, Y., Liang, J., & Liang, Z. (2009). Isolation and determination of anamorph of Shiraia bambusicola. China Journal of Chinese Materia Medica, 34, 1640–1642.
Van Oorschot, C. A. N. (1980). A revision of Chrysosporium and allied genera. Studies in Mycology, 20, 1–89.
Liang, J. D., Han, Y. F., Zhang, J. W., Du, W., Liang, Z. Q., & Li, Z. Z. (2011). Optimal culture conditions for keratinase production by a novel thermophilic Myceliophthora thermophila strain GZUIFR-H49-1. Journal of Applied Microbiology, 110, 871–880.
Kumar, S., Tamura, K., & Nei, M. (2004). MEGA3: integrated software for molecular evolutionary genetics analysis and sequence alignment. Briefings in Bioinformatics, 5, 150–163.
Miller, G. L. (1959). Use of dinitrosalicylic acid reagent for determination of reducing sugar. Analytical Chemistry, 31, 426–428.
Cheng, T. F., Jia, X. M., Ma, X. H., Lin, H. P., & Zhao, Y. H. (2004). Phylogenetic study on Shiraia bambusicola by rDNA sequence analyses. Journal of Basic Microbiology, 445, 339–350.
Galhaup, C., Wagner, H., Hinterstoisser, B., & Haltrich, D. (2002). Increased production of laccase by the wood-degrading basidiomycete Trametes pubescens. Enzyme and Microbial Technology, 30, 529–536.
Castermans, D., Somers, I., Kriel, J., Louwet, W., Wera, S., Versele, M., et al. (2012). Glucose-induced posttranslational activation of protein phosphatases PP2A and PP1 in yeast. Cell Research, 22, 1058–1077.
Rigling, D., Heiniger, U., & Hohl, H. R. (1989). Reduction of laccase activity in dsRNA-containing hypovirulent strains of Cryphonectria (Endothia) parasitica. Physiology and Biochemistry, 79, 219–223.
Malhotra, K., Sharma, P., & Capalash, N. (2004). Copper and dyes enhance laccase production in γ-proteobacterium JB. Biotechnology Letters, 26, 1047–1050.
Liu, L., Lin, Z., Zheng, T., Lin, L., Zheng, C., Lin, Z., et al. (2009). Fermentation optimization and characterization of the laccase from Pleurotus ostreatus strain 10969. Enzyme and Microbial Technology, 44, 426–433.
Zhang, Y., Sun, S., Hu, K., & Lin, X. (2012). Improving production of laccase from novel basidiomycete with response surface methodology. African Journal of Biotechnology, 11, 7009–7015.
Karp, S. G., Faraco, V., Amore, A., Birolo, L., Giangrande, C., Soccol, V. T., et al. (2012). Characterization of laccase isoforms produced by Pleurotus ostreatus in solid state fermentation of sugarcane bagasse. Bioresource Technology, 114, 735–739.
Gnanamani, A., Jayaprakashvel, M., Arulmani, M., & Sadulla, S. (2006). Effect of inducers and culturing processes on laccase synthesis in Phanerochaete chrysosporium NCIM 1197 and the constitutive expression of laccase isozymes. Enzyme and Microbial Technology, 38, 1017–1021.
Kittl, R., Gonaus, C., Pillei, C., Haltrich, D., & Ludwig, R. (2012). Constitutive expression of Botrytis aclada laccase in Pichia pastoris. Bioengineered, 3, 1–3.
Solé, M., Müller, I., Pecyna, M. J., Fetzer, I., Harms, H., & Schlosser, D. (2012). Differential regulation by organic compounds and heavy metals of multiple laccase genes in the aquatic hyphomycete clavariopsis aquatica. Applied and Environmental Microbiology, 78, 4732–4739.
Yang, Y., Fan, F., Zhuo, R., Ma, F., Gong, Y., Wan, X., et al. (2012). Expression of laccase gene from white rot fungus in Pichia pastoris can enhance the resistance of yeast to H2O2-mediated oxidative stress by stimulating the glutathione-based antioxidative system. American Society for Microbiology, 15. doi:10.1128/AEM.00218-12.
Acknowledgments
This work was financially supported by the National Natural Science Foundation of China (no. 30960004), Natural Science Foundation of Guizhou Province (2011-3094, 2010-4008, 2009-7012-1, 2011-204), and Natural Science Foundation of Guizhou University (2007-035, 2010-102).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Du, W., Sun, C., Yu, J. et al. Effect of Synergistic Inducement on the Production of Laccase by a Novel Shiraia bambusicola Strain GZ11K2. Appl Biochem Biotechnol 168, 2376–2386 (2012). https://doi.org/10.1007/s12010-012-9943-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-012-9943-x