Abstract
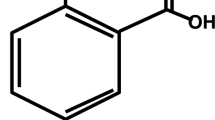
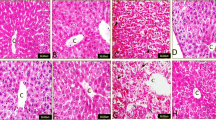
Bioflavonoids are plant compounds touted for their potential to treat or prevent several diseases including cancer caused by various stress conditions. Galangin (4H-1-Benzopyran-4-one, 3, 5, 7-trihydroxy-2-phenyl-), a flavonoid, is a polyphenolic compound found primarily in medicinal herb, Alpinia galanga. This study aims to demonstrate the galangin as a pharmacological lead compound using in vitro, in vivo, and in silico model targeting specific cancer condition and proteins. The proliferation of MCF-7 and Ehrlich ascites carcinoma (EAC) cells was significantly inhibited with an IC50 of 34.11 and 22.29 μg/ml, respectively. In an animal model system, galangin has inhibited the tumor growth by 73.51% ± 4.742 in EAC-induced Swiss Albino mice with no evidences of mortality as compared to standard drug, 5-fluorouracil. The effectiveness of galangin is proven in an animal system suggesting its pharmacokinetics behavior in an animal model which is also complemented by outcome of in silico analysis with more than 88 % of human intestinal absorption and significant Caco-2 cell, MDCK cell, and skin permeability as predicted by in silico methods. Galangin was docked against 19 different proteins involved in tumorogenesis and apoptosis; the energetic analysis indicates that it exhibits higher predicted binding free energy of −12.7 kcal/mol with Bcl-xL protein.





Similar content being viewed by others
References
Rethinking cancer. (2010). Available from http://physics.cancer.gov/global/docs/PWJun10davies.pdf 25 February 2012
American Cancer Society, A biotechnology company dedicated to cancer treatment. (2006). Available from: www.cancervax.com/info/index.htm.25 January 2006
Somkumar, A. P. (2003). Studies on anticancer effects of Ocimum sanctum and Withania somnifera on experimentally induced cancer in mice. Ph D thesis, J. N. K. V. University, Jabalpur, India.
Pandey, G., & Madhuri, S. (2006). Medicinal plants: Better remedy for neoplasm. Indian Drugs, 43, 869–874.
Cseke, L. J., Kaufman, P. B., Podila, G. K., & Tsai, C. J. (2004). Molecular and cellular methods in biology and medicine (2nd ed.). Boca Raton: CRC Press.
Block, G., Patterson, B., & Subar, A. (1992). Fruit, vegetables, and cancer prevention: A review of the epidemiological evidence. Nutrition and Cancer, 18, 1–29.
Messina, M., Descheemaker, K., & Erdman, J. W., Jr. (1998). The role of soy in preventing and treating chronic disease. American Journal of Clinical Nutrition, 68, 1329S (Proceedings of the Second International Symposium on the Role of Soy in Preventing and Treating Chronic Disease held on September 15–18, 1996, and a satellite symposium held on September 19, 1996, in Brussels, Belgium—Preface).
Steinmetz, K. A., & Potter, J. D. (1991). Vegetables, fruit, and cancer. I. Epidemiology. Cancer Causes & Control, 2, 325–357.
Wattenberg, L. W. (1992). Inhibition of carcinogenesis by minor dietary constituents. Cancer Research, 52(suppl), 2085s–2091s.
Wattenberg, L. W. (1992). Chemoprevention of cancer by naturally occurring and synthetic compounds. In L. W. Wattenberg, M. Lipkin, C. W. Kelloff, & G. J. Kelloff (Eds.), Cancer chemoprevention (pp. 19–39). Boca Raton: CRC Press.
Waladkhani, A. R., & Clemens, M. R. (1998). Effect of dietary phytochemicals on cancer development. International Journal of Medical, 1, 747–753.
Steinmetz, K. A., & Potter, J. D. (1991). Vegetables, fruit, and cancer. II. Mechanisms. Cancer Causes & Control, 2, 427–442.
Adlercreutz, H. (1990). Western diet and Western diseases: Some hormonal and biochemical mechanisms and associations. Scandinavian Journal of Clinical & Laboratory Investigation, 50(Suppl. 201), 3–23.
Havsteen, B. (1983). Flavonoids, a class of natural products of high pharmacological potency. Biochemical Pharmacology, 32, 1141–1148.
Middleton, E., Jr., & Chithan, K. (1993). The impact of plant flavonoids on mammalian biology: Implications for immunity, inflammation and cancer. In J. B. Harborne (Ed.), The flavonoids: Advances in research since 1986. London: Chapman and Hall.
Grange, J. M., & Davey, R. W. (1990). Antibacterial properties of propolis (bee glue). Journal of the Royal Society of Medicine, 83, 159–160.
Harborne, J. B., & Baxter, H. (1999). The handbook of natural flavonoids, vols 1 and 2. Chichester: Wiley.
Scalbert, A., & Williamson, G. (2000). Dietary intake and bioavailability of dietary polyphenols. Journal of Nutrition, 130, 2073S–2085S.
Vaouzour, D., Vafeiadou, K., Rodriguez-Mateos, A., Rendeiro, C., & Spencer, J. P. (2008). The neuroprotective potential of flavonoids: A multiplicity of effects. Genetics Nutrition, 3, 115–126.
Rathee, P., Chaudhary, H., Rathee, S., Rathee, D., Kumar, V., & Kohli, K. (2009). Mechanism of action of flavonoids as anti-inflammatory agents: A review. Inflammation & Allergy Drug Targets, 8, 229–235.
Andres, A., Donovan, S. M., & Kuhlenschmidt, M. S. (2009). Soy isoflavones and virus infections. The Journal of Nutritional Biochemistry, 20, 563–569.
Li, B. H., & Tian, W. X. (2003). Presence of fatty acid synthase inhibitors in the rhizome of Alpinia officinarum hance. Journal of Enzyme Inhibition and Medicinal Chemistry, 18, 349–356.
Volpi, N. (2004). Separation of flavonoids and phenolic acids from propolis by capillary zone electrophoresis. Electrophoresis, 25, 1872–1878.
Borrelli, F., Maffia, P., Pinto, L., Ianaro, A., Russo, A., Capasso, F., & Ialenti, A. (2002). Phytochemical compounds involved in the anti-inflammatory effect of propolis extract. Fitoterapia, 73(Suppl 1), S53–S63.
So, F. V., Guthrie, N., Chambers, A. F., & Carroll, K. K. (1997). Inhibition of proliferation of estrogen receptor-positive MCF-7 human breast cancer cells by flavonoids in the presence and absence of excess estrogen. Cancer Letters, 112, 127–133.
Wall, M. E., Wani, M. C., Manikumar, G., Abraham, P., Taylor, H., Hughes, T. J., Warner, J., & McGivney, R. (1988). Plant antimutagenic agents: Flavonoids. Journal of Natural Products, 51(6), 1084–1091.
Cholbi, M. R., Paya, M., & Alcaraz, M. J. (1991). Inhibitory effects of phenolic compounds on CCl4-induced microsomal lipid peroxidation. Experientia, 47(2), 195–199.
Imamura, Y., Migita, T., Uriu, Y., Otagiri, M., & Okawara, T. (2000). Inhibitory effects of flavonoids on rabbit heart carbonyl reductase. Journal of Biochemistry, 127(4), 653–658.
Capasso, R., & Tavares, I. A. (2002). Effect of the flavonoid galangin on urinary bladder rat contractility in vitro. Journal of Pharmacy and Pharmacology, 54(8), 1147–1150.
Heo, M. Y., Yu, K. S., Kim, K. H., Kim, H. P., & Au, W. W. (1992). Anti-clastogenic effect of flavonoids against mutagen-induced micronuclei in mice. Mutation Research, 284(2), 243–249.
Heo, M. Y., Lee, S. J., Kwon, C. H., Kim, S. W., Sohn, D. H., & Au, W. W. (1994). Anticlastogenic effects of galangin against bleomycin-induced chromosomal aberration in mouse spleen lymphocytes. Mutation Research, 311(2), 225–229.
Gabor, M., & Razga, Z. (1991). Effect of benzopyrone derivatives on simultaneously induced croton oil ear oedema and carrageenin paw oedema in rats. Acta Physiologica Hungarica, 77(3–4), 197–207.
Kang, S. S., Kim, J. S., Son, K. M., Kim, H. P., & Chang, H. W. (2000). Isolation of COX-2 inhibitors from Alpinia officinarum. Korean Journal of Pharmacognosy, 31, 57–62.
Shih, H., Pickwell, G. V., & Quattrochi, L. C. (2000). Differential effects of flavonoid compounds on tumor promoter-induced activation of the human CYP1A2 enhancer. Archives of Biochemistry and Biophysics, 373(1), 287–294.
Buening, M. K., Chang, R. L., Huang, M. T., Fortner, J. G., Wood, A. W., & Conney, A. H. (1981). Activation and inhibition of benzo[a]pyrene and aflotoxin B1 metabolism in human liver microsomes by naturally occurring flavonoids. Cancer Research, 41(1), 67–72.
Ciolino, H. P., & Yeh, G. C. (1999). The flavonoid galangin is an inhibitor of CYP1A1 activity and an agonist/antagonist of the arly hydrocarbon receptor. British Journal of Cancer, 79(9–10), 1340–1346.
Sohn, S. J., Huh, I. H., Au, W. W., & Heo, M. Y. (1998). Antigenotoxicity of galangin against N-methyl-N-nitrosourea. Mutation Research, 402(1–2), 231–236.
Hansen, M. B., Nielsen, S. E., & Berg, K. (1989). Re-examination and further development of a precise and rapid dye methods for measuring cell growth/cell kill. Journal of Immunological Methods, 119, 203–210.
Morris, G. M., Goodsell, D. S., Halliday, R. S., Huey, R., Hart, W. E., Belew, R. K., et al. (1998). Automated docking using a Lamarckian genetic algorithm and empirical binding free energy function. Journal of Computational Chemistry, 19, 1639–1662.
Ahmed, H., Chatterjee, B. P., & Debnath, A. K. (1988). Interaction and in vivo growth inhibition of Ehrlich ascites tumor cells by jacalin. Journal of Biosciences, 13(4), 419–424.
Murray, T. J., Yang, X., & Sherr, D. H. (2006). Growth of a human mammary tumor cell line is blocked by galangin, a naturally occurring bioflavonoid, and is accompanied by down-regulation of cyclins D3, E, and A. Breast Cancer Research, 8(2), 2–13.
Brown, D. G., Sun, X. M., & Cohen, G. M. (1993). Dexamethasone-induced apoptosis involves cleavage of DNA to large fragments prior to internucleosomal fragmentation. Journal of Biological Chemistry, 268, 3037–3039.
Oberhammer, F., Wilson, J. W., Dive, C., Morris, I. D., Hickman, J. A., Wakeling, A. E., Walker, P. R., & Sikorska, M. (1993). Apoptotic death in epithelial cells: Cleavage of DNA to 300 and/or 50 kb fragments prior to or in the absence of internucleosomal fragmentation. EMBO Journal, 12, 3679–3684.
Walker, P. R., Smith, C., Youdale, T., Leblanc, J., Whitfield, J. F., & Sikorska, M. (1991). Topoisomerase II reactive chemotherapeutic drugs induce apoptosis in thymocytes. Cancer Research, 51, 1078–1085.
Granville, D. J., Carthy, C. M., Hunt, D. W., & McManus, B. M. (1998). Apoptosis: Molecular aspects of cell death and disease. Laboratory Investigation, 78, 893–913.
Nagata, S. (2000). Apoptotic DNA fragmentation. Experimental Cell Research, 256, 12–18.
Nicholson, D. W., & Thornberry, N. A. (1997). Caspases: Killer proteases. Trends in Biochemical Sciences, 22, 299–306.
Tao, H., Luo, H., Wu, J., Lan, L-bo, Fan, D-hua, Zhu, K-dan, Chen, X-yi, et al. (2010). Galangin induces apoptosis of hepatocellular carcinoma cells via the mitochondrial pathway. Apoptosis, 16(27), 3377–3384.
Heo, M. Y., Sohn, S. J., & Au, W. W. (2001). Anti-genotoxicity of galangin as a cancer chemopreventive agent candidate. Mutation Research: Reviews in Mutation Research, 488, 135–150.
Monasterio, A., Urdaci, M. C., Pinchuk, I. V., Lopez-Moratalla, N., & Martinez-Irujo, J. J. (2004). Flavonoids induce apoptosis in human leukemia U937 cells through caspase- and caspase–calpain- dependent pathways. Nutrition and Cancer, 50, 90–100.
Kennedy, T. (1997). Managing the drug discovery/development interface. Drug Discovery Today, 2, 436–444.
Clark, D. E., & Grootenhuis, P. D. (2002). Progress in computational methods for the prediction of ADMET properties. Current Opinion in Drug Discovery & Development, 5, 382–390.
Modi, S. (2003). Computational approaches to the understanding of ADMET properties and problems. Drug Discovery Today, 8, 621–623.
Waterbeemd, H., & Gifford, E. (2003). ADMET in silico modelling: Towards prediction paradise? Nature Reviews Drug Discovery, 2, 192–204.
Artursson, P., & Karlsson, J. (1991). Correlation between oral drug absorption in humans and apparent drug permeability coefficients in human intestinal epithelial (Caco-2) cells. Biochemical and Biophysical Research Communications, 175, 880–885.
Artursson, P., & Borchardt, R. T. (1997). Intestinal drug absorption and metabolism in cell cultures: Caco-2 and beyond. Pharmaceutical Research, 14, 1655–1658.
Lipin-ski, C. A., Lombardo, F., Dominy, B. W., & Feeny, P. J. (1997). Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Advanced Drug Delivery Reviews, 23, 3–25.
Kola, I., & Landis, J. (2004). Can the pharmaceutical industry reduce attrition rates? Nature Reviews Drug Discovery, 3, 711–716.
Kararli, T. T. (1995). Comparison of the gastrointestinal anatomy, physiology, and biochemistry of humans and commonly used laboratory animals. Biopharmaceutics & Drug Disposition, 16, 351–380.
Hidalgo, I. J., Raub, T. J., & Borchardt, R. T. (1989). Characterization of the human colon carcinoma cell line (Caco-2) as a model system for intestinal epithelial permeability. Gastroenterology, 96, 736–749.
Artursson, P. (1990). Epithelial transport of drugs in cell culture. I: A model for studying the passive diffusion of drugs over intestinal absorptive (Caco-2) cells. Journal of Pharmaceutical Sciences, 79, 476–482.
Irvine, J. D., Takahashi, L. K., Lockhart, J., Cheong, J. W., Tolan, H. E., Selick, J., & Grove, R. (1999). MDCK (Madin–Darby canine kidney) cells: A tool for membrane permeability screening. Journal of Pharmaceutical Sciences, 88, 28–33.
Jelic, D., Rusak, G., Domac, J., Goran, S., Kovarik, Z., & Antolovic, R. (2010). Structural aspects of flavonoids as inhibitors of human butyrylcholinesterase. European Journal of Medicinal Chemistry, 45, 186–192.
Gupta, M., Upal, K., Haldar, P. K., & Kandar, C. C. (2007). Anticancer activity of Indigofera aspalathoides and Wedelia calendulaceae in Swiss Albino mice. Iranian Journal of Pharmaceutical Research, 6(2), 141–145.
Acknowledgments
This study was supported by University Grants Commission, New Delhi under the Major Research Project Scheme. We are also thankful to Department of Veterinary Pharmacology and Toxicology, PG Institute of Veterinary Sciences, Akola, India for their assistance in animal studies experiments. We acknowledge Bioinformatics Infrastructure Facility, Department of Biotechnology, Government of India for providing computer analysis facility.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jaiswal, J.V., Wadegaonkar, P.A. & Hajare, S.W. The Bioflavonoid Galangin Suppresses the Growth of Ehrlich Ascites Carcinoma in Swiss Albino Mice: A Molecular Insight. Appl Biochem Biotechnol 167, 1325–1339 (2012). https://doi.org/10.1007/s12010-012-9646-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-012-9646-3